Abstract
Steroid cell tumor not otherwise specified (NOS) is a rare subtype of sex cord stromal tumor of the ovary and contributes less than 0.1% of all ovarian neoplasms. The majority of tumors occur in pre-menopausal women (mean age: 43 years), in which 56-77% of patients present with virilization due to excess testosterone. An 80-year-old woman with worsening alopecia and excessive growth of coarse hair on abdomen and genital area was found to have elevated serum testosterone level (462 ng/mL). Radiologic studies were consistent with bilateral adrenal adenomas. Bilateral adrenal venous sampling ruled out the adrenal gland as origin of hormone secretion. A diagnostic and therapeutic bilateral salpingo-oophorectomy confirmed steroid cell tumor NOS of the left ovary. Post-operatively, the patient had complete resolution of her symptoms and normalization of testosterone level. Our case emphasizes the importance of a clinical suspicion for an occult testosterone secreting ovarian tumor in a symptomatic patient without obvious ovarian mass on imaging.
Case Report
An 80 year old post-menopausal woman was evaluated in our clinic complaining of worsening alopecia and excessive growth of coarse hair on her abdomen and genital area for one year. She also reported an unusual increase in her libido for the past 6 months. There was no history of acne, deepening of voice, blurred vision, weight gain, muscle weakness or striae. Her medical history was significant for essential hypertension which was recently uncontrolled on her home regimen of diltiazem 240 mg. Prior to our evaluation, her physician prescribed oral estrogen and testosterone to alleviate her symptoms. Subsequently she was found to have an elevated testosterone level (446 ng/dL) and was advised to discontinue oral hormonal supplements. On physical examination her BMI was 32.5 kg/m2 with an elevated systolic blood pressure of 165 mmHg. Alopecia, increased facial hair and coarse hair on lower abdomen and thighs were noted. Pelvic examination was remarkable for an enlarged clitoris with no obvious adnexal masses. Elevated serum testosterone level of 462 ng/dL (normal 8-60 ng/dL) and androstenedione 225 ng/dL (normal 30-200 ng/dL) were found. Serum luteinizing hormone (23.3 IU/L), follicle stimulating hormone (38.8 IU/L), estradiol (64 pg/mL), DHEAS (99.3 mcg/dL), CA-125 (13 U/mL) were normal. Renal and liver functions were within normal limits. A negative dexamethasone suppression test ruled out subclinical Cushing syndrome.
Pelvic ultrasound showed normal left (3.0×1.7×2.0 cm) and right (2.0×1.2×1.5 cm) ovaries. No ascites or other abnormalities were present. Computed tomography (CT) abdomen and pelvis demonstrated bilateral adrenal adenomas, right (1.6 cm) and left (1.7 cm). Bilateral adrenal venous sampling ruled out the adrenal gland as the source of excess hormone. With a clinical suspicion of an occult testosterone producing ovarian tumor, patient was advised a diagnostic and possibly therapeutic laparoscopic bilateral salpingo-oophorectomy.
Intra-operatively, the uterus, tubes and ovaries were unremarkable and no free fluid was present. Pelvic washings sent for cytology were negative. Histopathology confirmed a 2.0×1.9×1.4 cm left ovarian benign steroid cell tumor NOS. There was absence of significant atypia and necrosis, though a rare mitotic figure was seen (Figure 1A). Immunoperoxidase studies demonstrated strong and diffuse staining for inhibin and calretinin with weak staining for keratin AE1/AE3 (Figure 1B,C). The left fallopian tube revealed a small paratubal cyst. Right ovary and fallopian tube were normal. Post-operatively, serum testosterone level decreased to 17 ng/dL and patient experienced dramatic relief of symptoms and continues to be without evidence of recurrence.

A) High power Hematoxylin and Eosin view demonstrating nests of polygonal cells with eosinophilic cytoplasm. Focal cytoplasmic vacuolization is present. Nuclei are round with prominent nucleoli. No Reinke's crystals are seen. B) Strong and diffuse cytoplasmic staining for Inhibin. C) Diffuse nuclear and cytoplasmic staining in the tumor cells for Calretinin.
Discussion
The histopathological term steroid cell tumor of the ovary was first described by Scully. 1 Formerly classified as lipid or lipoid cell tumor, this description was later discontinued as the growths had little or no fat content. 2 These rare tumors are now reclassified as sex cord stromal tumors, and account for less than 0.1% of all ovarian neoplasms. 1
Based on the cell of origin, steroid cell tumors are categorized into three subtypes: Stromal luteomas arising from ovarian stromal cells, Leydig cell tumor arising from Leydig cells in the hilum of the ovary, and steroid cell tumor not otherwise specified (NOS) when the lineage of the tumor cell cannot be identified. 1 The last subtype account for the majority of tumors (60%) and tend to affect younger women (mean age; 43 years), as compared to the other two subtypes. 3 The tumors are usually unilateral, though in rare cases (6%), they can involve both ovaries. Steroid cell tumors NOS are usually benign; however, clinically malignant behavior, including peritoneal metastases, occurs in 25-40% of patients. 4
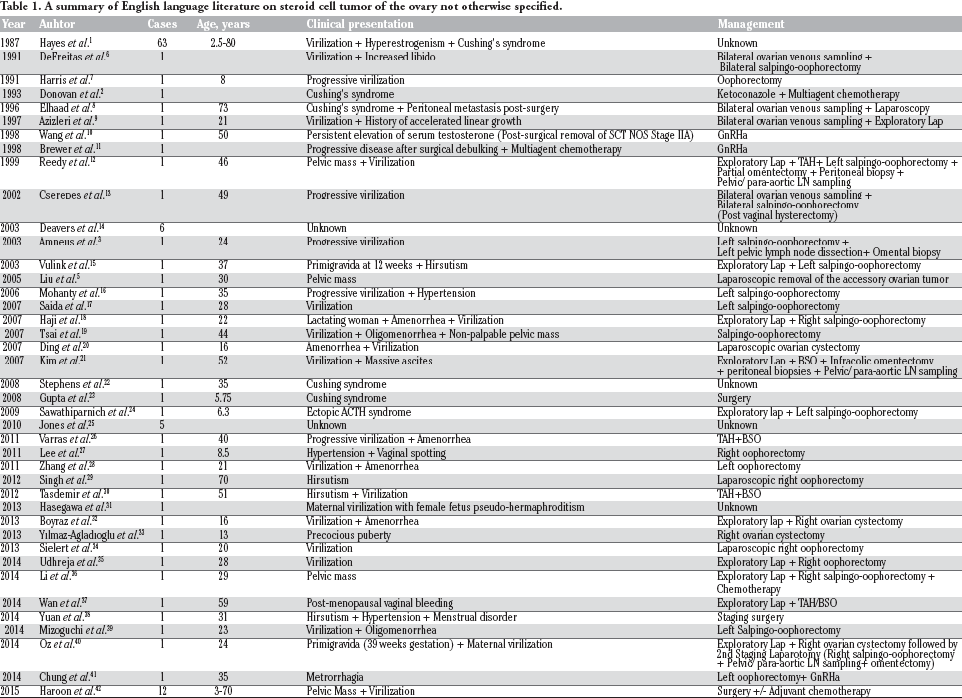
Clinical manifestations are defined by the type of hormone produced by steroid cell tumors, NOS. Most secrete testosterone (56 to 77%) and patients present with virilizing symptoms such as gradually progressive hirsutism, acne, deepening of voice, temporal baldness and amenorrhea (Table 1).5–42 This was the presentation in our patient who had rapidly progressive virilization over the course of one year. Hyperestrogenemia presenting as menorrhagia or irregular uterine bleeding has been reported in 6 to 23% of women. 1 In 6 to 10% of cases, excess cortisol secretion can lead to Cushing syndrome.24,38 However, 25% of patients with steroid cell tumor NOS are asymptomatic. 10
A summary of English language literature on steroid cell tumor of the ovary not otherwise specified.
Histopathology remains the gold standard for diagnosis. Grossly, steroid cell tumor NOS is well circumscribed, solid and non-calcified with a lobulated appearance.10,19,32 Cross section generally shows a yellow-orange surface with occasional cystic changes. 19 On microscopic examination, the tumor cells typically have a nested arrangement but can be organized into columns or cords resembling adrenal zona glomerulosa and zona fasciculate. Cytologically, cells are polygonal or round with distinctive cell borders, central nuclei and prominent nucleoli. The cytoplasm varies from eosinophilic to clear and vacuolated.12,36,38,43 The absence of cytoplasmic Reinke's crystals help differentiate this tumor from the Leydig cell neoplasm. 28
Hayes and Scully have defined five pathological correlates of malignant behavior in these tumors, namely: i) two or more mitoses per 10 high power fields; ii) a tumor diameter of >7 cm; iii) necrosis; iv) hemorrhage; and v) grade 2 to 3 nuclear atypia.1,24 Histologically, our patient's tumor did not have any of the above criteria.
Immunohistochemical markers for inhibin and calretinin are sensitive markers for steroid cell tumors NOS. 44 Calretinin positivity is present in 60 to 90% of tumor cells, whereas inhibin reactivity ranges from 5 to 90%.14,45 Other markers, such as EMA, cytokeratin, CD99 and S100 have been reported to be positive. HMB45, Chromogranin-A, LeuM1, AFP, carcinoembryonic antigen (CEA) and periodic acid Schiff (PAS) are other markers, which have been studied. 25
A unique scenario of maternal virilization in pregnancy with or without fetal female pseudo hermaphroditism has also been reported. Surgery with salpingo-oophorectomy may be safely performed after the first trimester in these patients to avoid torsion or rupture of tumor and protect the developing fetus from harmful maternal androgens (Table 1).27,31 Prepubertal girls presenting with precocious puberty due to excess testosterone from a steroid cell tumor NOS have had a delay in diagnosis and treatment due to confusion with non-classical congenital adrenal hyperplasia (CAH). Thus, failure to respond to treatment for CAH should prompt a consideration for an ovarian etiology in these young patients.32,33
Based on the limited data available, the recommended management of steroid cell tumor NOS is primarily surgical. A benign tumor limited to the ovary in a woman who has completed childbearing should be managed with salpingo-oophorectomy. Conservative surgery with cystectomy has been successfully done in young patients to preserve ovarian function. 33 For patients with a unilateral malignant tumor, but desirous of future fertility, formal staging surgery to include unilateral salpingo-oophorectomy with preservation of the contralateral ovary and uterus is a reasonable option. These patients need careful postoperative surveillance including measurement of sex hormone levels, especially if high levels were present prior to surgery. For tumors found to be malignant after initial conservative surgery, a formal staging procedure at a second surgery should be performed. Brown et al. proposed that surgical staging for a malignant steroid cell tumor does not require formal lymphadenectomy, in contrast to the standard staging procedure for an epithelial ovarian malignancy. 46 Widely metastatic malignant disease should be treated with surgical cytoreduction followed by adjuvant chemotherapy. Although a definitive chemotherapy regimen is not yet defined, Bleomycin, Etoposide and Cisplatin (BEP) is favored and often used. Gonadotropin releasing hormone agonist has been used as therapy for recurrent malignant disease for its suppressive effect on ovarian steroidogenesis.19,38,43,47
Conclusions
Our case emphasizes the importance of clinical suspicion for an occult testosterone secreting ovarian tumor in a symptomatic woman without an obvious ovarian mass on radiologic studies. Prompt surgical management can lead to complete resolution of symptoms as well as normalization of excess testosterone levels within weeks. In post-menopausal women who are candidates for surgery, a diagnostic and therapeutic bilateral salpingo-oophorectomy is a safe option. In women with a small adrenal nodule and no ovarian lesions, bilateral adrenal or ovarian venous sampling is a reasonable choice to evaluate the source of excess hormone production before considering surgery.
