Abstract
Members of the bone morphogenetic protein (BMP) family have diverse physiological roles. For instance, BMP-2 stimulates osteogenesis, while BMP-12 induces the formation of tendon/ligament-like tissues. Here, we designed a study to determine whether BMP-12 has bone and/or cartilage regeneration abilities similar to those of BMP-2. We implanted plasmid vectors encoding either BMP-2 or BMP-12 in rats with femur defects, and monitored the bone healing process for 8-weeks. The BMP-12 transgene induced prominent fibrogenesis by 2 weeks, with bone substitution occurring by 8 weeks. BMP-2, however, was associated predominantly with osteogenesis throughout the 8 week period. Thus, we conclude that BMP-12 does not function similarly to BMP-2 during bone healing. Further work is needed to better understand the mechanisms by which it stimulates bony growths to replace the connective tissues formed during the first stages of bone healing.
Introduction
Bone morphogenetic proteins (BMPs) are known to be capable of inducing new bone formation when implanted subcutaneously in vivo [1]. Within the BMP family, BMP-2/4 and BMP-7 (OP-1) are especially well known for their ability to induce bone and cartilage formation, respectively, by influencing the differentiation of mesenchymal progenitor cells [2–4]. On the other hand, bone morphogenetic protein-12 (BMP-12/GDF-7/CDMP-3) is better known for its ability to stimulate the formation of tendon/ligament-like tissues [5]. Many studies, both in vivo and in vitro, have reported that BMP-12 has the potential to form tendon/ligament-like tissues [6–10]. However, no research to date has explored the possible role of this protein on bone or cartilage regeneration. Further, there has been no direct comparison between the activity of BMP-12 and BMP-2/4/7, to determine whether they are equally able to stimulate bone growth, or whether the activity of BMP-12 is centered solely on tendon/ligament-like tissues.
If BMP-12 plays a role in both of these processes—generation of bone as well as connective tissues—it has the potential to be an invaluable tool for specialists working with patients who have suffered joint or periodontal damage. In these cases, it is common for ligaments and tendons to be injured as a result of trauma to the bone, since these structures exist adjacent to each other in the body. Because the healing process involves regeneration of all these tissues simultaneously, BMP-12 could offer a unique way to promote the regrowth of ligaments/tendons, as well as the bone that is needed to anchor them.
The current study was designed to examine whether hBMP-12, which is known to induce tendon/ligament growth, also stimulates bone or cartilage tissue growth when transferred to a rat bone defect model. Additionally, we were interested in directly comparing the activity of hBMP-12 to that of hBMP-2. To this end, we studied bone regeneration among four groups: two groups implanted with a lyophilized atelocollagen pellet containing a plasmid vector encoding BMP-12 and BMP-2, respectively, a sham treatment group receiving only the atelocollagen pellet, and a control treatment group receiving no pellet at all.
Plasmid vectors are stable molecules and easy to handle for chemical modulation [11], and plasmid diffusion from the delivery site avoids systemic toxicity due to the high efficiency of DNA turnover in the bloodstream. Together, these characteristics make plasmid insertion a relatively safe technique. Previously, plasmid DNA combined with atelocollagen (a highly purified type I collagen that has become one of the most useful scaffolds for bone formation) was transplanted into bone defects in rats and dogs and succeeded in inducing bone regeneration [2, 12, 13]. Other collagen-containing formulas—including those containing the rhBMP2 gene—have been used successfully in in vivo gene transfer and bone healing [14, 15]. From the results of this previous work, we expected not only to successfully transfer the BMP genes, but also to observe the dual competence of BMP-12 in inducing growth of both soft and hard tissues. Such a finding could revolutionize treatments in the orthopedic and dental fields, since repairs of joint and periodontal damage would be improved greatly by a protein that could facilitate simultaneous regeneration of multiple types of tissues that comprise these structures.
Materials and Methods
The protocols for our study were approved by the Recombinant DNA Experiment Committee and the Animal Experiment Committee of our institution. All of the experimental processes were performed in accordance with institutional guidelines for the care and manipulation of laboratory animals.
Preparation of Implants
Both hBMP-2 and hBMP-12 cDNA were obtained from teeth extracted from patients at the university hospital. hBMP-2 was cloned from dental pulp using previously published methods [16], while hBMP-12 cDNA was taken from periodontal ligament scraped off from the surface of an extracted tooth (accession #AB158468). The patients approved of these methods and provided informed consent before we proceeded with the extractions.
For hBMP-12 cDNA preparation, total RNA of MG63 cells (human osteosarcoma, JCRB) was extracted with Isogen (Nippon Gene, Japan). Reverse transcription (SuperScript First-Strand Synthesis System, Invitrogen) and PCR (GC-RICH PCR SYSTEM, Roche, USA) were performed with a specific primer set (forward: 5′-CACGGAGCCCATGGACCTGAGCG-3′ reverse: 5′-GTGGCCCGCTGCTGCTCATCAG-3′) constructed using human BAC clone data (accession #AC012065). The 1439-bp PCR product includes complete human BMP-12 cds. Ligation of this product into a vector plasmid, pCRII (Invitrogen, USA), and transformation of JM109 Escherichia coli cells (TAKARA, Japan) with the plasmid were performed according to the manufacturer's instructions. The amplified and purified pCRII plasmids were digested using EcoRI, after which the isolated insertion was subcloned in pEGFP-N1 plasmids (Clontech Laboratories Inc., USA). The final step was transformation of the JM109 cells.
Large culture of E. coli clone 2.2 served as the purified plasmid encoding the human BMP-12 protein (Figure 1). cDNA of hBMP-2 was initially located in the BamHI region of the pBlueBacII plasmid vector multicloning site. This emerged as pBacBH2, after which the cDNA was inserted into the BamHI region of the pEGFP-N1 multicloning site. Then, 100 μg of each plasmid vector was prepared separately in sterile water and mixed with 100 μg of 2% bovine atelocollagen gel (KOKEN Atelocollagen Implant; KOKEN Co., Tokyo, Japan), which is used clinically as a hemostatic agent and to heal wounds [17–19]. Finally, 200 μl of this mixture was placed inside the lids of 1.5 mL Eppendorf tubes and lyophilized. The final product of this step was used for implantation of BMP-2 and BMP-12 into the rat models. We also prepared a sham implant of the vehicle plasmid (pEGFP-N1) only, so that we could gauge the functionality of the transgene system by assaying for a GFP signal.
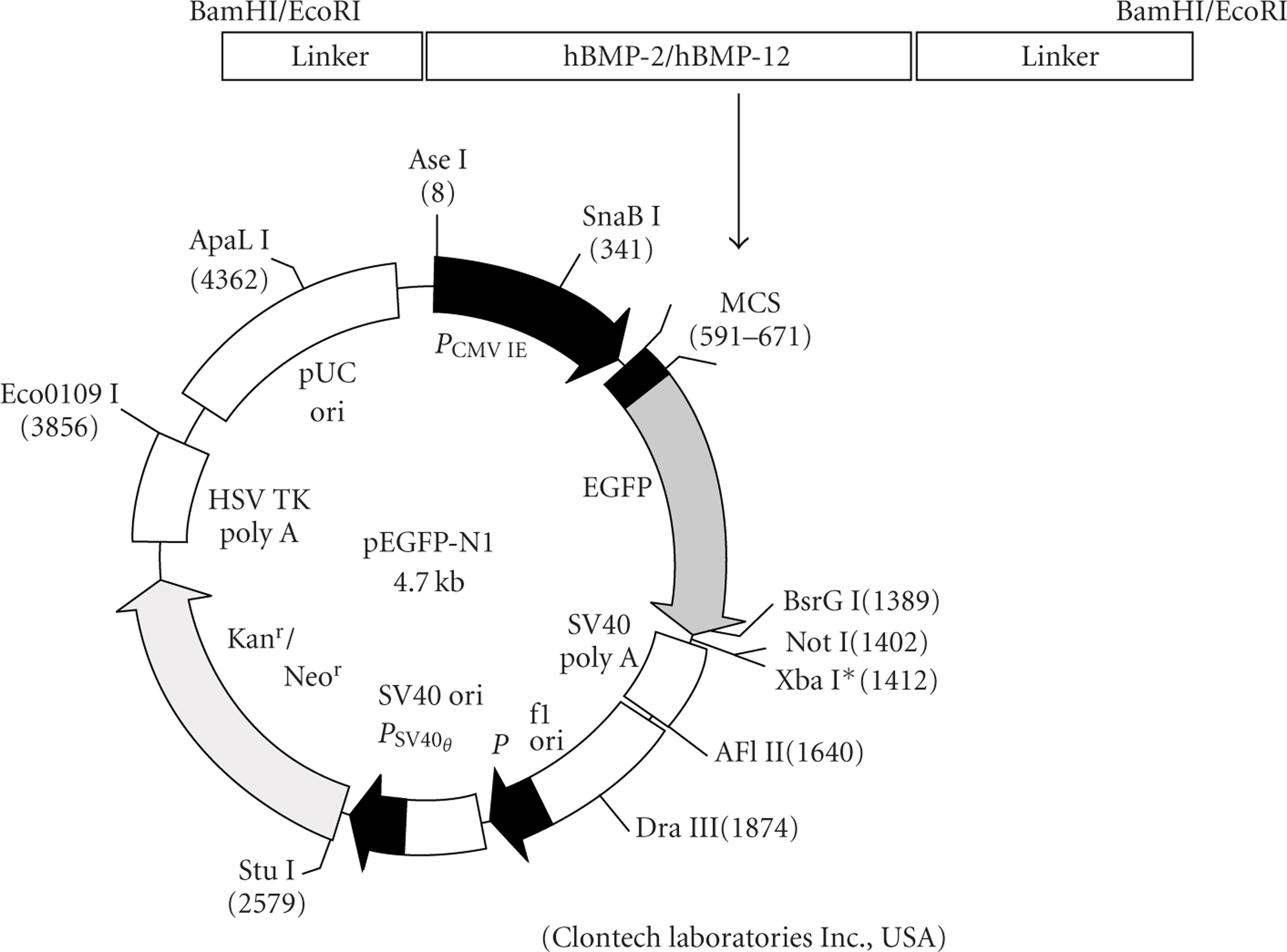
A scheme of the expression plasmid vector and the BMP inserts.
Ninety-six male Wistar rats, eight weeks old, were evenly divided into 4 groups. The rats were anesthetized with a mixture of xylazine, ketamine, and acepromazine (8, 100, and 2 mg/kg, respectively). A round drill bit was used to create a cortical bone defect (3 mm in diameter) in the anterior surface of the right femoral metaphysic of each rat (Figure 2). The pellets were then implanted into the defects; for the two BMP treatment groups, the implant consisted of atelocollagen harboring the plasmid vector encoding rhBMP-12 (100 μg) or rhBMP-2 (100 μg), as appropriate. For the sham group, the implant was an atelocollagen-only pellet. We also had a control group, in which the rats received no implant after undergoing surgery to create the bone defect.

Scheme of the femoral defect model. A defect in the cortex at the distal part of the right femur was prepared without irrigating the marrow.
At 2, 4, and 8 weeks after the operation, animals were sacrificed (via chloroform inhalation in a draft biohazard hood), and their femurs were retrieved. Two specimens per group were used to prepare histological samples, while the other six were used to conduct real-time PCR (RT-PCR) analyses to profile expression of bone- and tendon/ligament-related genes. To confirm successful gene transfer, we amplified specific transgene regions after collecting implants from the sacrificed animals. Additionally, we used a confocal laser scanning microscope (CLSM) (LSM 510, Zeiss) at 488 nm excitation and 509 nm emission at the same time point to detect whether the animal expressed a GFP signal after receiving a subcutaneous implant of the vehicle pEGFP-N1 plasmid vector.
For the histological study, femurs were fixed in 10% neutral formalin. The specimens were decalcified with 10% EDTA; they were then trimmed, washed, dehydrated with gradients of alcohol, and cleared in xylene before being embedded in paraffin. Longitudinal sections (5 μm) that included the bone defects were then prepared and stained with hematoxylin-and-eosin and Masson's trichrome.
Gene Expression
The sites of the bone defects were resected and homogenized immediately using liquid nitrogen. RNA was extracted with an isolation agent (Isogen; Nippon Gene Co., Tokyo, Japan). PCR primers were designed (Table 1) using sequence data obtained from GenBank (National Center of Biotechnology Information, NIH, Bethesda, USA). The chosen mRNAs are known to be expressed in tendons (Six1), adult connective tissues including both tendons and ligaments (elastin, decorin, collagen I, and collagen III), bone or cartilage (aggrecan, biglycan collagen I, alkaline phosphatase (ALP), and osteocalcin), and for housekeeping (glyceraldehyde 3-phosphate dehydrogenase).
Primer sets for RT-PCR. “Fwd” indicates forward primers and “Rvs” indicates reverse primers. All genes sequences are from the rat model. Abbreviations are defined as follows: GAPDH: rat glyceraldehyde 3 phosphate dehydrogenase; Col I: rat collagen I; Col III: rat collagen III; ALP: rat alkaline phosphatase; OCN: rat osteocalcin.

Primer sets for RT-PCR. “Fwd” indicates forward primers and “Rvs” indicates reverse primers. All genes sequences are from the rat model. Abbreviations are defined as follows: GAPDH: rat glyceraldehyde 3 phosphate dehydrogenase; Col I: rat collagen I; Col III: rat collagen III; ALP: rat alkaline phosphatase; OCN: rat osteocalcin.

After the first strand of cDNA was reverse transcribed (SuperScript First-Strand Synthesis System for RT-PCR, Invitrogen), we performed RT-PCR (7300 Real-time PCR machine, Applied Biosystems) with SYBR Green fluorescence dye under an optimal program: 30 s at 95°C, 30 s at 60°C, and 30 s at 72°C for 40 cycles, followed by a dissociation step for the second derivatives with a gradient increase of temperature from 60°C to 95°C. The identities of the PCR products were reconfirmed using 2% agarose gel electrophoresis. RT-PCR results were analyzed with relative quantification software. The expression level of each gene is indicated as the fold change in the number of cDNA copies relative to the amount of expression in the control group after normalization with GAPDH.
Before analyses, the Smirnov-Grubbs test was used for eliminating outliers from the dataset. RT-PCR results were analyzed with nonparametric Kruskal-Wallis tests. Statistical significance was defined as P < .05.
Results
Translation of the Transgenes
Both hBMP-12 (Figure 3(a)) and hBMP-2 (data not shown) gene expressions were detected in bone samples retrieved 2 weeks after implantation. This expression had attenuated by 4 and 8 weeks (data not shown). Images from the CLSM indicated successful uptake and expression of the vehicle (Figure 3(b)).

(a) An example of a specific PCR product for hBMP-12, which was observed in the BMP-12 group at 2 weeks (a product for hBMP-2 is not shown). The atelocollagen group carrying the vehicle vector did not express hBMP-12 at all. (b) Evidence that the transgene system was successful. This GFP signal was produced in a specimen treated only with the vehicle pEGFP-N1 plasmid vector. The image was captured by CLSM after extracting the specimen two weeks after the initial operation.
Two weeks after operation, the two BMP groups clearly differed from each other (Figure 4(a)). While fibrogenesis was observed in rats implanted with BMP-12, osteogenesis was prominent in BMP-2 rats. In the sham group, almost all of the atelocollagen remained where it had initially been deposited. On the other hand, the control group had experienced bone regeneration. Formation of new collagen fibers occurred at a higher rate in the BMP-12 group than in any other treatment group. After 4 weeks, the atelocollagen framework remained in all groups, especially the sham group; at this time, the control group showed new bone formation in the bone defect. On the other hand, dense collagen fibers were observed in the BMP-12 group (Figure 4(b)); these were replaced with bone growth by the end of the experiment at 8 weeks. Complete bone replacement, including invasion of bone marrow cells, was finally observed in all groups by the end of the study (Figure 4(c)).
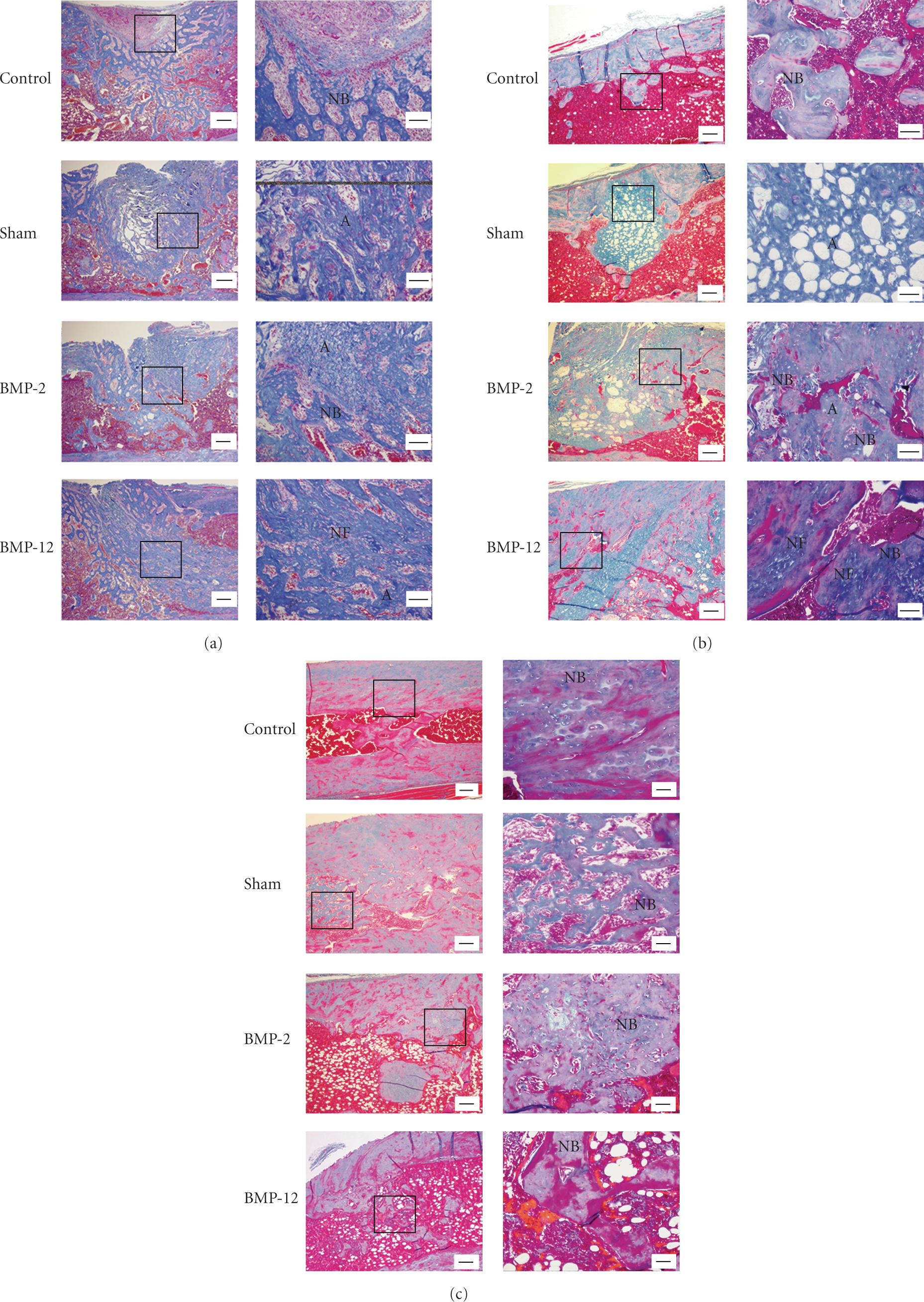
Histological images of defects treated with each of the different implants. Longitudinal sections were prepared at (a) 2 weeks, (b) 4 weeks, and (c) 8 weeks after the operation. The sections were stained with Masson's trichrome. The left-side panels show low-magnification images, and the right-side panels show high-power images. Bars in the left and the right panels show 300 μm and 100 μm, respectively. A: atelocollagen, NB: newly-formed bone, NF: newly-formed fiber.
As early as 2 weeks after the operation, expression of connective tissue genes (specifically, collagen III and decorin) was significantly higher in the BMP groups than in the sham group (Figure 5). Expression of genes associated with connective tissues (collagen I and III), bone and cartilage (ALP, biglycan), and tendons (Six1) were particularly highly upregulated in the BMP-2 group. We noticed two additional interesting patterns: first, expression of decorin did not increase among members of the BMP-2 group; second, ALP gene expression was not elevated in the BMP-12 group despite its prominent increase in the BMP-2 group. In other words, the BMP associated with bone growth (BMP-2) did not increase expression of connective tissue genes, while the BMP associated with connective tissues (BMP-12) did not increase expression of bone/cartilage genes.

Gene expression in three of the study treatments at each of the time steps. Solid lines indicate the BMP-12 group; dashed lines indicate the BMP-2 group; long dashed/double-dot lines indicate the atelocollagen group. Data are expressed as fold change relative to the sham; statistical significance was defined as any P < .05(n = 6). (a) BMP-2 versus sham; (b) BMP-2 versus BMP-12; (c) BMP-2 versus control; (d) BMP-12 versus sham; (e) BMP-12 versus control; (f) sham versus control.
After 4 weeks, the BMP-12 group expressed significantly higher levels of mRNAs associated with bone repair activity than the sham group. These included biglycan, elastin, and, especially, Six1.
At the final time point of 8 weeks, ALP gene expression was higher in both BMP groups than in the control group, and all three of these groups had higher ALP gene expression than the sham group. Notably, osteocalcin gene expression was significantly higher in the sham group than in the BMP-2 group. This was surprising since osteocalcin is responsible for bone/cartilage activity, which should not have been directly stimulated by the sham treatment. Other gene expression related to tendon/ligament regeneration in both BMP groups had decreased by the end of the study.
Most BMP family members induce the differentiation of mesenchymal cells into osteoblastic cells or cartilage cells. Both in vitro and in vivo studies, however, have indicated that BMP-12 is functionally different from other members of the BMP family [5, 8, 9]; in that it stimulates proliferation of tendon fibroblasts [7, 10] and induces tendon and ligament formation [5]. Previously, its effects on bone and cartilage formation have only rarely been reported. Here, we have examined the possibility that BMP-12 might play a role in healing injured bone, as well as connective tissues. If this is the case, BMP-12 could be a valuable new part of bone regeneration treatments, particularly among orthopedic and dental injuries.
Each of the four treatment groups in our study showed different histological appearances at each time point. Generally, changes in bone-specific gene expressions serve as a good indication of the bone formation process. ALP is a marker that intimately associates with early-phase osteoblastic differentiation. Both our histological and RT-PCR results indicated that ALP expression peaked in the BMP-2 group 2 weeks after plasmid insertion. However, ALP gene expression in the BMP-12 group was fairly constant over the experimental period. These results are not surprising, given that ALP is associated with bone and cartilage, structures that are known to be impacted by BMP-2.
At the 2-week time point, the BMP-2 group experienced a strong peak of collagen I similar to the peak seen in ALP. This suggested the initiation of osteogenesis, leading to newly-formed bone by the final time point. On the other hand, gene expression of ALP and collagen I in the BMP-12 group maintained low levels and showed no peak during the study period. This might have resulted in only a partial replacement of the cavity with new bone by 8 weeks. These findings are similar to those from earlier studies, where ALP gene expression and activity were not altered by BMP-12 gene transfer in C3H10T1/2 or tendon cells [8, 9], and, furthermore, where subcutaneous implantation of BMP-12 induced tendon and ligament generation, rather than bone formation [5].
Recent in vivo and in vitro works showed that Achilles tendons produce more collage III after they have ruptured than when they are healthy [20, 21]. This suggests that collagen III has an important role in the healing process of tendons and/or ligaments, perhaps as a provisional matrix [22]. Collagen III is also essential for normal collagen I fibrillogenesis [23, 24]. We observed high levels of collagen III gene expression at the 2-week mark in both BMP groups. This provides additional evidence that BMP-12 plays a vital role in inducing collagen III formation for early tendon/ligament development. The prominent expressions of both ALP and collagen I genes in the BMP-2 group, however, likely masked the function of Collagen III essential for induction of tendon/ligament growth. It is this masking process that may be responsible for making BMP-2 a key part of the osteogenesis process, instead.
Decorin accounts for approximately 90% of the total proteoglycans in fresh ligaments. The other 10% comprises species such as biglycan, versican, and aggrecan [25]. Both decorin and biglycan belong to the small leucine-rich proteoglycan family; members of which are found in tendons and ligaments and have been implicated as regulating agents of fibrillogenesis [25–28]. Biglycan is functionally related to BMPs and seems more likely than decorin to be related to osteogenesis rather than fibrillogenesis [29–31]. Indeed, we found that decorin levels were comparable between BMP-2 and BMP-12 groups at 2 weeks, and, furthermore, that decorin levels had decreased in the BMP-12 group by 4 weeks. Furthermore, we observed higher levels of biglycan gene expression in BMP-2 than in BMP-12 at two weeks, which is consistent with the hypothesis that biglycan is important for osteogenesis. However, we did also find that gene expression of biglycan in the BMP-12 group increased at 4 weeks, suggesting that BMP-12 may play a role not only in early tendon/ligament formation, but also following osteogenesis.
Observation of the control group indicated that the bone defect could heal naturally after 8 weeks. We anticipated that the atelocollagen scaffolding used in the BMP and sham treatments of this study might delay bone healing, since previous clinical trials have shown that it degrades very slowly in the body [17–19]. However, the exogenous BMP-2 and BMP-12, contrarily, brought about the newly formed bone by 2 to 8 weeks, which might be supported by the osteocalcin gene expression profile identical between the BMP-12 and BMP-2 groups. Samples collected 4 weeks after the operation indicated that bone formation in the BMP-2 group occurred even in the presence of the remaining atelocollagen; this may be due to the overriding effects of the BMP-2 gene activity. By 8 weeks after the operation, all groups (even the BMP-12 group) were able to almost completely heal the bony defect.
In addition to differences in bone regeneration, the four treatment groups exhibited differences in the healing process, particularly in the amount and location of newly-formed, collagen-like tissue. Both BMP groups and the sham group were exposed to atelocollagen-containing treatments. However, only the BMP-12 group obtained dense collagen-like tissue prior to osteogenesis (between 2 and 4 weeks after the operation). On the other hand, the other two groups maintained more or less randomly scattered pieces of the implanted atelocollagen, throughout the bone regeneration process, until the end of the study. Thus, the fibrous tissue observed in the BMP-12 group was most likely generated by BMP-12 activity.
Other researchers followed a subcutaneous application of atelocollagen in a rhesus monkey for up to 26 weeks [32]. Their work indicates that, in addition to the uses of atelocollagen detailed in the current study, this delivery system might act as a good space keeper for generating tissues when implanted subcutaneously. Indeed, our research demonstrated a discrepancy in the roles between BMP-12 and BMP-2 when used with materials such as atelocollagen. Our study was not designed to examine whether the regeneration of an injured tendon/ligament adjacent to the bone defect was healed or strengthened by BMP-12. Future work should aim to understand whether BMP-12 synchronizes the activity of genes linked to bones/cartilage and tendons/ligaments during the healing process.
The present study has demonstrated that BMP-12 gene transfer into a rat femoral bone defect induced tendon/ligament-like tissue formation with increased gene expression of tendon/ligament-related genes until 4 weeks after the operation. Further, we have also demonstrated that these tissues were eventually replaced with bone-like tissue by 8 weeks after the operation. However, additional studies are required to further explore the mechanism by which BMP12 initiated the replacement of tendon/ligament-like tissue with bony tissue.
Footnotes
Acknowledgments
This work was made possible by a Grant-in-Aid for Scientific Research (16390579) and a grant from the Center of Excellence Program for Frontier Research on Molecular Destruction and Reconstruction of Tooth and Bone in Tokyo Medical and Dental University.
