Abstract
Spinal cord injury (SCI) is one of the major disabilities dealt with in clinical rehabilitation settings and is multifactorial in that the patients suffer from motor and sensory impairments as well as many other complications throughout their lifetimes. Many clinical trials have been documented during the last two decades to restore damaged spinal cords. However, only a few pharmacological therapies used in clinical settings which still have only limited effects on the regeneration, recovery speed, or retraining of the spinal cord. In this paper, we will introduce recent clinical trials, which performed pharmacological treatments and cell transplantations for patients with SCI, and evaluate recent in vivo studies for the regeneration of injured spinal cord, including stem-cell transplantation, application of neurotrophic factors and suppressor of inhibiting factors, development of biomaterial scaffolds and delivery systems, rehabilitation, and the combinations of these therapies to evaluate what can be appropriately applied in the future to the patients with SCI.
Introduction
The incidence of traumatic SCI is about 10–30 new cases per million among the population in Europe and 27–83 per million population in USA. Estimated prevalence is approximately 225,000 to 288,000 cases in the United States [1]. The major cause of traumatic SCI is motor vehicle accidents (45%–47% of all traumatic SCI), sports-related, accidents and falls [2].
Most of the currently used managements for SCI have focused on either the rehabilitation of patients with paraplegia or tetraplegia to maximize the remaining functions of the upper and/or lower extremities, or the prevention and management of complications after spinal cord injury, such as neurogenic bladder and bowel, decubitus ulcer, orthostatic hypotension, deep vein thrombosis, and autonomic dysreflexia. These management focuses have improved the quality of life for patients with SCI, but fundamental treatment to regenerate the damaged spinal cord tissues and neural cells has not been standardized, and no drug has yet to be effective in improving the functional and clinical status.
Many studies have revealed some effective strategies for regenerating injured spinal cord through in vivo and in vitro studies, but there are many steps to reach the clinical application for the patients with SCI, due to the lack of mechanism of treatment, safety for humans, and potential adverse effects. We reviewed recent clinical trials of medications and stem cell transplantation for SCI patients, and advanced treatment strategies in animal studies in order to understand the mechanism of SCI treatment and to find future appropriate clinical applications.
Time Course of Patients with SCI
Time sequence of SCI is divided into three stages: acute (seconds to minutes after SCI), subacute (minutes to weeks after SCI), and chronic (months to years after SCI). The therapeutic target should be set according to these stages. In the acute and subacute stages, the purpose of treatment is neuroprotection whereas neural restoration is the target of chronic stage.
Primary Injury (Acute Stage)
Primary injury is due to the direct compression and contusion of the spinal cord due to bone or disc displacement within the spinal column, as results of fracture-dislocation or burst fracture of the spine [3]. The injured nerve cells usually fail to restore normal neural function and progress to spinal shock, which represents a generalized failure of circuitry in the spinal neural network, about 24 hours after injury [2]. Primary injury usually leads to secondary degenerative processes that further exacerbate SCI.
Secondary Injury (Subacute Stage)
Secondary injury starts with depolarization and voltage-dependent sodium, potassium, and calcium ions channel opening. Following this, calcium ion overload initiates mitochondrial dysfunction and the activation of cytoplasmic nitric oxide synthase and phospholipase A2, which leads to microvascular damage and consequential ischemia, as well as calpain activation which further leads to axonal damage [4].
Chronic Stage
After secondary injury, a dense glial scar accumulates around the lesion of the spinal cord by reactive astrocytes, glial progenitors, microglia and macrophages, fibroblasts, and Schwann cells. A cyst usually develops after contusion SCI, and axons near a cyst can regenerate into trabeculae, but most of the spontaneous regeneration process is incomplete [5].
Treatment Targets to Regenerate Damaged Neural Networks in the Spinal Cord
There are four targets to overcome for the fundamental treatment of a damaged spinal cord. The first target for treatment is the reduction of secondary injury, such as inflammation, edema, and scar formation, all of which interfere in neuronal regeneration. The next treatment should focus on the regeneration of damaged axons and myelin. Third target is the reconnection of efferent and afferent pathways crossing injured axons located in the white matter of spinal cords which is essential for the restoration of motor and sensory functions. The last target for treatment is the injured neurons in the gray matter of spinal cord should be regenerated for the restoration of function in situ. Most therapeutic interventions are effective in acute to subacute stages because the dense scar which would form otherwise could not be easily removed, and degenerated neuronal and glial cells are hard to restore once they have reached the chronic stage.
Clinical Advances in SCI
Thus far, there is no treatment of SCI, but several clinical trials have provided some information on both the regeneration of injured neuronal cells and the protection from additional damage to the remaining neuronal cells. Present treatment option for humans is only pharmacological, which is an expanding potential as there are some new drugs and cells in ongoing clinical trials.
Pharmacological Approaches
Steroid Therapy
Methylprednisolone sodium succinate (MPSS) has been investigated in clinical settings and is known to be effective for improving motor and sensory functions and reducing the amount of cellular damage from secondary injury, if it is applied within 8 hours after SCI [6–8]. MPSS has potencies of antiapoptotic effect on oligodendrocytes [9, 10] as well as anti-inflammatory and antioxidant effects [11, 12] after SCI. But according to the recent studies, MPSS also brings with it many detrimental effects, such as the risk for infections and gastrointestinal complications [13, 14] and insufficient evidence for standard treatment in patients with acute SCI [14, 15]. MPSS treatment is still only as an optional choice for acute SCI patients [16, 17].
Monosialotetrahexosylganglioside (GM-1 Ganglioside)
GM-1 ganglioside is naturally located in the outer membranes of nervous tissue and it has been tested in large scale clinical studies for SCI patients. Some researchers have revealed that administration of GM-1 ganglioside could improve the lower extremity, bladder and bowel function, sacral sensation, and anal contraction even in severe incomplete SCI (ASIA Impairment Scale B) patients [18, 19]. Nevertheless no evidence has supported the use of ganglioside in patients with acute SCI when considering its effect on the reduction of the death rate and improving recovery or quality of life in survivors [20].
Cethrin (Rho Pathway Antagonists)
Cethrin is a Rho antagonist which causes downregulation of growth inhibitor production, but Rho antagonist reportedly block intracellular signaling pathways such as Rho-associated kinase (ROCK) which is essential for vascular endothelial growth factor-mediated angiogenesis [21]. In animal study, Rho antagonist could restore an abnormal increment of Rho in neurons and glial cells and rapid locomotor improvement after acute SCI [22]. A phase I/IIa clinical trial revealed that topical administration of Cethrin was safe following surgical decompression for patients with SCI [23].
Riluzole (Sodium Channel Blocker)
Riluzole is a benzothiazole anticonvulsant sodium channel antagonist which has neuroprotective effect and promotes functional recovery after spinal cord contusion of rats [24]. Recently, one 2-year clinical trial for acute SCI patients was approved by the Food and Drug Administration. The study for patients with multiple sclerosis in the brain and spinal cord revealed that Riluzole reduced the rate of cervical cord atrophy, but with no evidence for clinical improvement [25].
Minocycline
Minocycline is a second-generation tetracycline derivate, and it has some neuroprotective effects, such as inhibition of microglial activation, attenuation of apoptosis, and suppression of free-radical production through blocking mitochondrial cytochrome c release and lastly an improved functional recovery in animal SCI [26–28]. No beneficial effect of Minocycline on animal SCI models was also reported recently [29] and no clinical trial for SCI patients was performed.
Erythropoietin
Erythropoietin is located at high concentration within capillaries of adult human spinal cords. It is known to be neuroprotective to ischemia of animal spinal cord as well as for its hematopoietic effects [30]. Phase II clinical trial showed that the administration of human recombinant erythropoietin intravenously was relatively safe and seemed to be effective in reducing the infarct size in stroke patients [31].
Nimodipine
Nimodipine is a calcium channel blocker and effective to increase cerebral blood flow and restore behavioral and histopathological abnormalities following ischemia reperfusion brain injury in rats [32]. Electrophysiological abnormality was improved after intravenous administration of nimodipine to the rats with partially transected spinal cord [33]. One clinical trial was performed but showed that no difference was found among SCI patients receiving nimodipine, MPSS, both of them, and placebo in acute phase [34].
Cell Therapy (Table 1)
Clinical trial of stem cell transplantation in patients with SCI was reported in 2006 at first [35], and several clinical research projects were performed to detect the safety and efficacy of transplanted cells to the patients in SCI (Table 1). Most of these experiments used autologous bone marrow stromal cells; the results were relatively safe but the effect was limited. Olfactory ensheathing cells, Schwann cells, and macrophages were also attempted to be transplanted to SCI patients. The purpose of these clinical trials was basically to test safety rather than therapeutic improvement. Moreover, these studies have many statistical problems; the sample size of most previous clinical studies was quite small, they were not double-blind randomized and had no placebo controls to compare with which is essential for valid SCI clinical trial [49], and they introduced unclear mechanisms which might lead to functional improvements, thus ultimately requiring further clinical trials.
Clinical trials of cell transplantation to SCI patients. “AIS” indicates ASIA (American Spinal Injury Association) impairment scale. Abbreviations are defined as follows: BMSCs: bone marrow stromal cells; OECs: olfactory ensheathing cells; IT: intrathecal administration; IV: intravenous administration; DI: direct injection surrounding the lesion; IA: intra-arterial administration; CSF: cerebrospinal fluid; SSEP: somatosensory evoked potential.
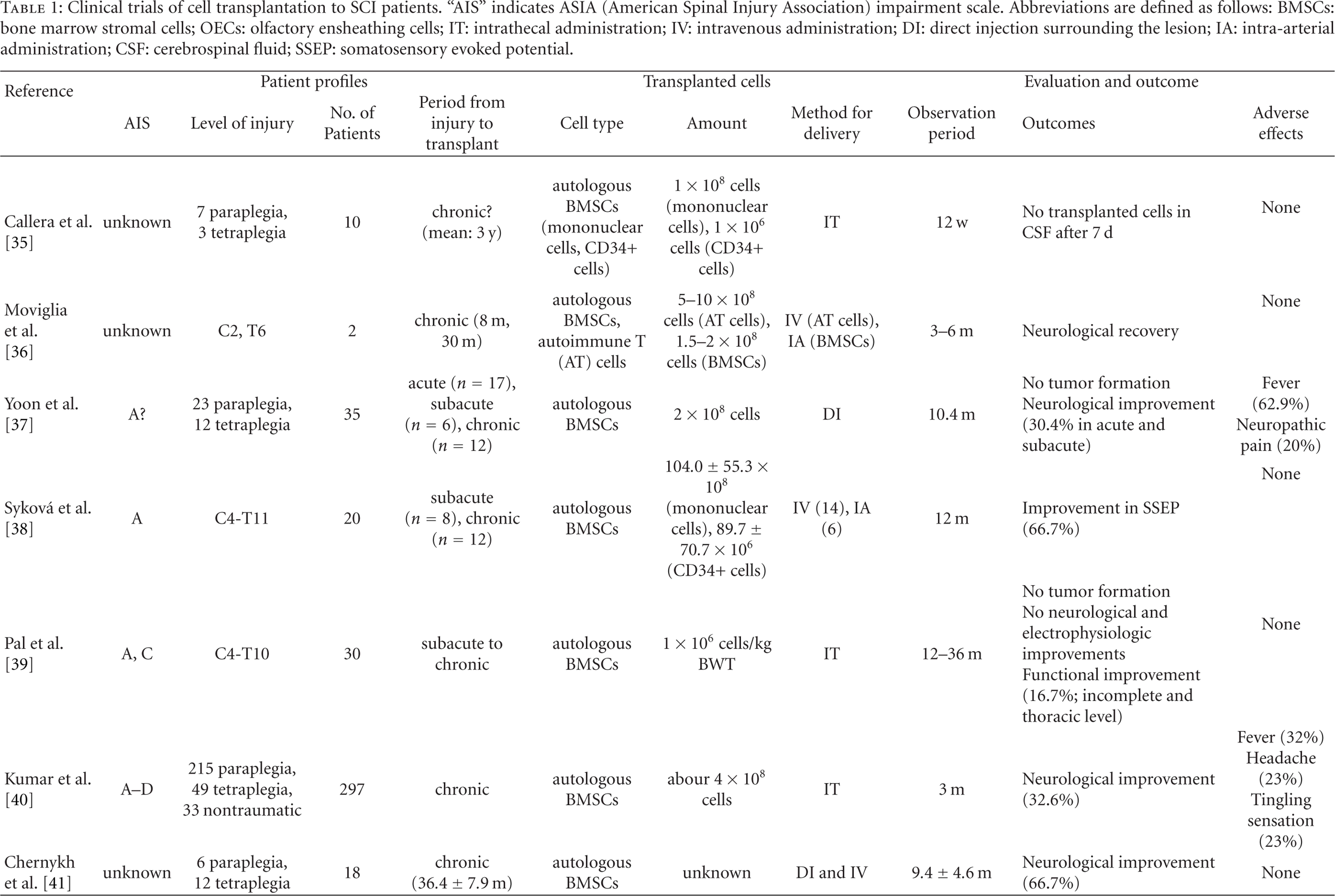

Clinical trials of cell transplantation to SCI patients. “AIS” indicates ASIA (American Spinal Injury Association) impairment scale. Abbreviations are defined as follows: BMSCs: bone marrow stromal cells; OECs: olfactory ensheathing cells; IT: intrathecal administration; IV: intravenous administration; DI: direct injection surrounding the lesion; IA: intra-arterial administration; CSF: cerebrospinal fluid; SSEP: somatosensory evoked potential.


Mesenchymal stem cells (MSCs) can be safely and easily obtained from bone marrow in human for use of autologous transplantation, without any ethical problems. This is very advantageous for clinical application. MSCs secrete cytokines and neurotrophic factors and also have anti-inflammatory effects [50]. Nine studies were performed using autologous BMSCs transplantation into SCI patients from 2006, where the total number of patients was 460. All clinical trials using BMSCs transplantation were performed outside of United States: Brazil [35, 43], India [39, 40], Argentina [36], Czech [38], Russia [41], Turkey [42], and South Korea [37].
Callera and Nascimento first reported BMSCs transplantation in 10 SCI patients intrathecally, and they did not find any transplanted cells in cerebrospinal fluid 7 days after transplantation (Table 1) [35]. Moviglia et al. transplanted autologous bone marrow-driven transdifferentiated neural stem cells and patient's autoimmune T cells into the injured spinal cord, via a feeding artery, in two chronic patients with spinal cord injury, and they found clear clinical improvements were seen during the 3 months after transplantation, without any adverse effect [36]. Yoon et al. reported 35 patients with complete SCI who were transplanted with autologous BMSCs around injured spinal cord in acute, subacute, or chronic stages, and they found that 20% of patients suffered from neuropathic pain after transplantation but the number of affected cases was not significantly more than controls (7.7%). Less than half (30.4%) of patients who were transplanted in acute and subacute stages showed improvements in the ASIA (American Spinal Injury Association) impairment scale (AIS) during the 10-month followup period [37]. Functional status in the acute stage would not be accurate when the patients are in the spinal shock stage; during several days of the spinal shock stage just after SCI the sensory and motor functions are usually disabled even in case of incomplete injury. Therefore, we cannot be convinced of the transplantation effects on acute-stage SCI patients. Sykova et al. also found that one of the 12 chronic patients with complete spinal cord injury, as well as 5 of 8 subacute patients showed sensory and motor improvements after transplantation of autologous BMSCs intravenously or intra-arterialy, and they recommended the therapeutic window of 3 to 4 weeks following injury as being critical for stem-cell treatment [38]. Pal et al. reported 30 patients with subacute or chronic SCI who received autologous BMSCs intrathecally, and only incomplete SCI patients (16.7%) were seen to have improved functionally without neurological or electrophysiological improvements [39]. Kumar et al. performed a large-scale clinical trial in India, consisting of was 297 patients with chronic SCI, who were transplanted with autologous BMSCs intrathecally, but detailed information about the period from injury to transplantation was not clarified and a 3-month followup period is too short to conclude the results [40]. Chernykh et al. in Russia and Deda et al. in Brazil transplanted autologous BMSCs to patients with chronic SCI directly into the lesion and intravenously [41, 42] or direct injection, intravenously and intrathecally [42] at the same time, and they found neurological improvements in many of the transplanted patients (66.7% and 100% resp.) [41, 42]. Critante et al. in Brazil also reported electrophysiological improvement in about 66.7% of chronic SCI patients with paraplegia or tetraplegia at the 30-month followup after BMSCs transplantation intraarterialy [43].
MSCs are thought to act as a neuroprotector by secreting various neurotrophic factors, namely: BDNF, NGF, VEGF, and HGF, rather than by neural regeneration from transdifferentiation into neuronal or glial cells [51, 52]. The exact mechanism of this action is still unknown [53]. Some in vitro and in vivo studies showed the transdifferentiation of MSCs into neuron and glial cells [54–57], but the proportion to neuronal differentiation was very small and the function of transdifferentiated cells as neurons is still doubtful [58]. Therefore, neural stem cells or pluripotent stem cells are appropriate to promote neural restoration or in the replacement of damaged host neurons and glial cells, more than are MSCs.
Olfactory Ensheathing Cells (OECs)
OECs are glial cells that ensheath the olfactory nerve fascicles and continue to support regeneration of olfactory axons throughout life in mammals [59]. Transplanted OECs into injured spinal cord promote axonal regeneration and functional recovery after SCI in animals [59]. OECs can be obtained via biopsy of the olfactory mucosa [44].
Mackay-Sim et al. observed six patients with chronic spinal cord injury for 3 years after transplantation of autologous OECs directly into the injured spinal cord (Phase I/IIa design) [44]. All patients were safe, and there were no significant functional changes or neuropathic pain 3 years after transplantation and one patient showed sensory improvement below the the lesion. Lima et al. found some neurological, functional, electrophysiological and urodynamic improvements in 20 chronic SCI patients after OECs transplantation into the injured spinal cord [45]. But Chhabra et al. reported syrinx formation and the lengthening of myelomalacia seen on MRI after OECs transplantation in some of the five chronic SCI patients, without any neurological or functional improvements [46].
Huang et al. reported clinical trials of OECs transplantation for chronic SCI patients three times to one Chinese journal and they found some functional improvements without significant complication [60–62]. But Dobkin et al. reported that among seven chronic SCI patients who received OECs transplantation from Dr. Huang in China, five patients had complications including meningitis and clinical improvement was neither clear nor proven and the procedures did not meet international standards of clinical trials for safety or efficacy [63].
Schwann Cells (SCs)
SCs are the supporting cells surrounding peripheral nerves and form the myelin sheath. SCs were the first transplanted cells into injured spinal cord in animals [64], and they enhance remyelination of demyelinated axons and promote axonal regeneration in combination with polymer scaffolds through many animal studies [53]. In a clinical trial, Saberi et al. transplanted autologous SCs into the injured spinal cords of 4 patients with chronic SCI, but only one patient with incomplete SCI showed sensory and motor improvement 1 year after transplantation [47].
Macrophages
Peripheral macrophages can synthesize nerve growth factor after peripheral nerve damage and eliminate myelin which inhibits neural regrowth [65]. A phase I study was performed: incubated autologous macrophages were transplanted into patients’ spinal cord within 14 days of injury, and 3 of 8 patients showed improvements of motor and sensory functions without any critical complications [48].
Experimental Advances in SCI (through In Vivo and In Vitro Studies)
Considering Animal Models and Injury Severity for SCI Experiments
The contusion model is the most relevant type for human SCI [66] and appropriate for assessment of acute management strategies [67]. Computer-assisted devices using an impactor such as NYU impactor are the representative tools to regulate the severity of contusion injury and they can simulate human SCI, but it is hard to differentiate the axonal regeneration from the trophic effects on the functional improvement following treatment, because the spared axons and regenerating axons in the injured spinal cord are not easy to distinguish.
Transection model is more appropriate for studies focusing on axonal regeneration. A gap following transection can also be made to implant a device or biopolymer scaffolds. Partial transection models, such as the hemisection or dorsal transection, have generally been used for the transplantation of neurotrophic factor-containing hydrogel [68, 69], genetically modified cells secreting neurotrophic factors [51] or biopolymer scaffolds [70, 71]. The complete transection model is preferred to eliminate the plasticity of remaining host neurons in the injured spinal cord and is appropriate for the study of the recovery of autonomic dysfunctions, such as neurogenic bladder, but the researcher should consider that this model is the most severe type of injury and is hard to regenerate, even after appropriate treatments.
Functional assessment is essential to confirm the treatment effects clinically whether an experiment attempting neuroprotection or neuronal regeneration is successful. Most studies using rodent models measure Basso, Beattie, and Bresnahan (BBB) score as a locomotor function of hindlimb, and the most meaningful BBB score is 9 (plantar weight-support) or more compared with nonachieved (below 9) controls [66]. But in a case of complete transection model, an increment of BBB score is very limited and the locomotor function cannot reach to plantar weight support even after successful treatment. Therefore, other methods such as tracing of spinal tracts or electrophysiology should be added to reveal the reconnection of proximal and distal stumps following spinal cord transection.
Treatment Focus in SCI
Time sequence of SCI is divided into three stages as mentioned earlier: acute, subacute, and chronic, and the treatment strategies differ according to the stages. Treatments in acute and subacute stages after SCI should focus on the neuroprotection, and treatment options in the chronic stage focus on neurorestoration [50].
Treatments for neuroprotection should ideally be started within several hours of injury to prevent secondary injury process. Both anti-inflammatory drugs and some neurotrophic factors such as brain-derived neurotrophic factor (BDNF) and neurotrophin-3 (NT-3) are known to be effective in vivo studies. Cell transplantation is not appropriate in the acute inflammatory stage because various cytokines following inflammation decrease the survival of transplanted cells and change the properties of differentiation of transplanted stem cells [72, 73]. Biomaterials such as poly (epsilon-caprolactone) (PLC) and poly (lactic-co-glycolic acid) (PLGA) have a potency to reduce the inflammatory process and concomitant scar formation in the traumatic brain injury model [74]. Also, functional improvements were seen after the cell-seeded biopolymer was implanted just after spinal cord transection [75, 76].
Neurorestoration strategies are very important in the clinical setting because most populations of SCI patients are in the chronic stage. Neurorestoration is divided into two categories: restoration of host neurons and neural replacement. The method for the restoration of host neurons includes suppression of inhibiting factors (which inhibit to regrowth of host axons) [77, 78], neurotrophic factors for neurite outgrowth and synaptic plasticity [5], and transplantation of glial cells for remyelination of host axons [79]. Pluripotent or multipotent stem cells such as embryonic stem cells, induced pluripotent stem cells, and neural stem cells can be sources of exogenous neurons and glial cells for neural and glial replacement, but the safety and efficiency for human use has not yet been addressed.
Stem Cell Therapy
Most of past and current researches using cell transplantation have been successful in subacute SCI, but researchers should consider the adverse effects of stem cells such as tumor formation or abnormal circuit formation within the injured spinal cord which leads to abnormal function [80]. Stem cells also can be used as vehicles for neurotrophic factors and gene delivery as well as differentiated neurons and glial cells to promote recovery [81].
Embryonic Stem Cells
Embryonic stem cells are pluripotent, being able to differentiate into specific cell lineages of the adult organism, and have an ability to proliferate in long-term cultures while maintaining their pluripotent nature [82]. Some researchers have reported the possibility of chromosomal abnormalities during prolonged culture [83], teratoma formation due to the remaining undifferentiated embryonic stem cells within the graft site, and graft rejection after allogeneic embryonic stem cell transplantation [82]. Many countries restrict clinical trials using embryonic stem cells due to ethical and political issues [84]. Geron Corporation (Menlo park, CA) started the first clinical trials (phase I) of human embryonic stem cells to SCI patients approved by US Food and Drug Administration last year [85].
Neural Stem Cells
Neural stem cells (NSCs) can differentiate into neurons and glial cells with the support of neurotrophic factors in vitro. These cells accelerate restoration of host neurons and remyelination of demyelinated axons as well as neuronal cell replacement [81]. Endogenous NSCs are located in the subventricular zone of the lateral ventricle and the subgranular zone of the hippocampal dentate gyrus in the adult human brain, but the self-renewal capacity following brain or spinal cord injury is not enough to promote recovery of injury [86]. The sources of exogenous NSCs are various, from embryonic stem cells to fetal and adult brain and spinal cord. Some in vivo studies were performed using NSCs and achieved functional recovery following SCI [87–89]. Most sources of NSCs are exogenous; therefore, allograft or xenograft may cause graft rejection, as in ESCs. Expression of various neurotrophic factors from differentiated astrocytes can cause complications such as allodynia after NSCs transplantation [90]. Ethical concerns, safety, and efficiency should also be considered for clinical trials [53]. No clinical trials of NSCs transplantation in SCI patients have been reported until now.
Induced Pluripotent Stem Cells
Induced pluripotent stem cells (iPSCs) were first introduced in 2006 by Takahashi and Yamanaka from gene modified mouse embryonic and adult fibroblasts [91]. iPSCs have pluripotency, a characteristic similar to that of ESCs. The transplantation of iPSCs in human SCI patients can overcome graft rejection after transplantation with the same potency as ESCs and ethical and political problems regarding the use of human embryos [92].
Recently, some investigators use iPSCs to restore impaired functions after focal cerebral ischemia in rats was combined with fibrin glue. They found functional improvement and anti-inflammatory response following transplantation [93]. Undifferentiated iPSCs have tumerigenesis similar to ESCs. Kawai et al. reported a tridermal teratoma after transplantation of iPSCs into the ischemic brain in mice [94]. Viral integration due to the use of viral vectors during the reprogramming process, c-Myc which is one of four transcription factors has oncogenic properties, and incomplete reprogramming due to the slow and relatively inefficient process are other problems which need to be solved before clinical application [95].
The techniques and methods to generate iPSCs continue to be developed rapidly. Recombinant proteins or small molecules also can be used for generating iPSCs [96, 97]. Kaji et al. made iPSCs without viral vectors in mouse and human fibroblasts, eliminating exogenous transcriptions factors with high efficiency [98]. The combination of transcription factors excluding c-Myc and even two of them were enough to regenerate iPSCs by some researchers [97, 99]. More efficient reprogramming was possible using human amnion-derived cells and three transcription factors [100]. We hope for a novel method to generate iPSCs which differentiate into neurons and glial cells effectively and safely, for use in SCI patients in the near future.
Neurotrophic Factors
Neurotrophic factors (NTFs) are known to modulate neuronal survival, axonal outgrowth and synaptic reformation after SCI [5]. NTFs which have therapeutic effects on the regeneration following SCI are brain-derived neurotrophic factor (BDNF) [51], Neurotrophin-3 (NT-3) [101], glial derived neurotrophic factor (GDNF) [102], nerve growth factor (NGF) [103], fibroblast growth factor (FGF) [104], insulin-like growth factor (IGF) [103], ciliary neurotrophic factor (CNTF) [105], and inosine [106].
Recent studies using NTFs and nonviral vector delivery systems are shown in Table 2. Sasaki et al. applied BDNF-secreting human MSCs into the dorsal-transected spinal cord in rats, and they found functional improvement and increments of fiber sprouting in the corticospinal tracts [51]. Han et al. used collagen-binding BDNF to hemisected spinal cords in rats, and they also reported functional improvement [68]. NT-3 could be delivered through genetically modified OECs or a heparin delivery system. Functional improvement and increment of neural fiber density were achieved following transplantation [69, 101]. GDNF-secreting MSCs or cotransplantation with SCs could increase GDNF secretion and regenerated axons and blood vessels [102, 107]. CNTF-secreting oligodendrocyte precursor cells were well survived and promoted remyelination of demyelinated axons and functional improvement after transplantation into contused spinal cord of rats [105].
Recent studies on the application of neurotrophic factors or suppressors of inhibiting factors to animal SCI models. “NTF” indicates neurotrophic factor, and “SIF” indicates suppressor of inhibiting factor. Abbreviations are defined as follows: SD rat: Sprague-Dawley rat; LE rat: Long-Evans rat; BDNF: brain-derived neurotrophic factor; NT-3: neurotrophin-3; GDNF: glial derived neurotrophic factor; CNTF: ciliary neurotrophic factor; ChABC: chondroitinase ABC; MSCs: mesenchymal stem cells; OECs: olfactory ensheathing cells; OPCs: oligodendrocyte precursor cells; DI: direct injection surrounding the lesion; IT: intrathecal administration; SCs: Schwann cells;.HRP: horseradish peroxidase; RSN: rubrospinal neurons.

Recent studies on the application of neurotrophic factors or suppressors of inhibiting factors to animal SCI models. “NTF” indicates neurotrophic factor, and “SIF” indicates suppressor of inhibiting factor. Abbreviations are defined as follows: SD rat: Sprague-Dawley rat; LE rat: Long-Evans rat; BDNF: brain-derived neurotrophic factor; NT-3: neurotrophin-3; GDNF: glial derived neurotrophic factor; CNTF: ciliary neurotrophic factor; ChABC: chondroitinase ABC; MSCs: mesenchymal stem cells; OECs: olfactory ensheathing cells; OPCs: oligodendrocyte precursor cells; DI: direct injection surrounding the lesion; IT: intrathecal administration; SCs: Schwann cells;.HRP: horseradish peroxidase; RSN: rubrospinal neurons.

Inhibiting factors which interfere the recovery of damaged axons and their reconnection after trauma to central nervous system are chondroitin sulfate proteoglycans (CSPG) [108, 109], myelin-associated glycoprotein (MAG) [110], Nogo-A [111] and oligodendrocyte-myelin glycoprotein (OMgp) [112]. Their mechanisms and functions are well understood in vitro studies, and some animal studies were performed for applying the suppressor of these inhibiting factors. Chondroitinase ABC (chABC), which digests CSPG, was administered intrathecally into injured spinal cord in rats, and this treatment upregulated regeneration-associated protein, restored electrophysiological activities, and promoted functional recovery [113]. Ikegama et al. performed chABC treatment combined with NSCs transplantation to the contused spinal cord of rats, and chABC-treated MSCs were well migrated and enhanced the outgrowth of growth associated protein-43 positive axons [109]. A recent in vivo study, however, failed to show improved functions following chABC injection into hemisected cervical cords of rats [108]. As in NTFs, suppressors of inhibiting factors should be released by genetically modified cells or combined with polymer-delivery system, to gain sustained effects and concomitant functional improvement following SCI. Administration of Nogo-66 receptor, which blocks MAG, Nogo, and OMgp, via osmotic pump improved electrophysiologic and locomotor recovery after dorsal transection of thoracic spinal cord in rats [114], and immunization with Nogo-66 receptor also showed axonal regeneration and locomotor recovery after dorsal hemisection and spinal cord contusion in rats [115]. Sialidase, an inhibitor of MAG was effective on axonal sprouting and locomotor function in spinal cord contusion model in rats [116].
Biomaterials
Development of biomaterials that are bioactive, compatible with nerve cells, and even nerve regenerative is also considered as a potential tool. In particular, the biomaterials can be an effective carrier of drugs and neurotrophic factors as well as play a scaffolding role of stem cells. When drug or NTF was loaded in a polymer nanocarrier, the drug delivery systems better reduce inflammation in acute stage [117] and act as a guidance of outgrowing axons or synaptic reconnection in subacute and chronic stages [118]. Some biopolymers such as PLGA and PLA there have also been reported to have anti-inflammatory capacity [119]. Even better is the engineering of the biomaterials to have microchannels or in the form of fibers in terms of improving a directionality of regenerating axons after SCI [120, 121]. Biomaterial-based treatment has some disadvantages; long-term implantation of nonbiodegradable tube type scaffold causes chronic compression of regenerating axons [122], and the transplant site can act as a sink, attracting sprouting axons but not allowing them to escape and reconnect with the host spinal cord.
Delivery Systems
To enhance the efficacy of neurotrophic factors and inhibiting agents, the use of an appropriate carrier can be considered. A range of polymeric materials (natural or synthetic origin) have been developed for targeting either systematic or local delivery of drugs. While liposome, micelles and dendrimers are usually used for the systemic delivery into the central nervous system, degradable polymers such as poly(lactic acid) (PLA), poly(lactic-co-glycolic acid) (PLGA), chitosan, and collagen are newly developed for the local delivery of drugs [123, 124]. Compared to the systematic delivery, local delivery system has many advantages in the SCI applications. Neurotrophic factors, suppressor of inhibiting factors can be delivered into injured spinal cord without any systemic side effects, and sustained release of regenerating factors is possible avoiding the difficulty in penetration into the blood-brain barrier. Degradable polymer-based delivery systems enable the control of drug dosage and release rate after implantation and modulation of the biodegradation rate after therapeutic period. Some recent studies have demonstrated the efficacy of degradable polymeric materials such as fibrin or PLGA for delivering neurotrophic factor [125, 126], and collagen or heparin-binding system for delivering neurotrophic factors into the injured spinal cord in animals [68, 69]. Hydrogel system made of hyaluronan and methylcellulose was also shown to deliver effectively erythropoietin intrathecally as the cavitation after SCI was greatly reduced [127].
Nanoparticulate carriers such as nanospheres and nanocapsules are good candidate for delivering proteins and even genes. A range of polymers have been developed in the form of nanospheres and nanocapsules and the sizes could be tunable to tens to a few hundreds of nanometers. As the compositions are degradable and the degradability is adjustable, the drug release rate can also be effectively modulated [105]. Drugs can be encapsulated either within the polymer nanoparticulates during the processing stage or posttreated (conjugated) on the surface of the particles through chemical reactions. Furthermore, the nanoparticulates can be combined with 3D tissue scaffolds to provide a sustained release system of single or multiple drugs for potential regeneration of nerve tissues. Synthetic polymer PLGA microspheres embedded with neurotrophic factors have been the most widely studied either with or without the combination of a variety of polymeric scaffolds. Wang et al. performed local administration of PLGA with glial cell line derived neurotropic factor (GDNF) and reported effective preservation of neuronal fibers leading to the hindlimb locomotor recovery in rats with SCI [128]. Takenaga et al. has demonstrated the prostaglandin E1 loaded nanoparticles are effective on locomotor recovery and decrement of cavity volume after spinal cord contusion in rats [129]. Moreover, Das et al. applied quercetin-loaded nanoparticles into the brain lesion model and achieved antioxidative effect [130]. While there have been increasing reports on the in vitro performance of the delivery systems, there are still limited animal studies using the nanocarriers for the treatment of SCI.
Scaffolds
Biomaterials can be developed to support and guide cell behaviors by engineering the macro- and micromorphology. To play an effective role as a scaffold, tissue perfusion of the biomaterials is prerequisite. Therefore, hydrogels or porous structured materials are the general form of scaffolds as these can provide large space for cells to grow and migrate and the continuous supply of fluid and nutrients. When scaffolds are implanted in lesions of CNS they can be a supporting matrix of surrounding cells to adhere and migrate to gain regenerative potential. In this stage, the properties of scaffolds, such as surface status, chemical composition and physical stiffness are the possible determinants of the cell behaviors and fate. In other words, one needs to consider significantly the design of scaffolds in terms of physical and chemical properties to gain optimal recovery and regeneration of nerve tissues. In practical issues, method for implantation of scaffolds into injured spinal cord should be considered because most human spinal cord injuries involve contusions of the spinal cord. To minimize adjacent tissue damage during implantation, injectable type biopolymer is preferred.
Among the compositions, degradable polymers, such as PLA and PLGA and natural collagen have been most popularly used as the porous scaffolds for the treatment of SCI. The PLA porous scaffolds were fabricated to have longitudinally aligned pores with incorporation of BDNF and implanted in the transected thoracic spinal, which, however, showed little improvement in the axon numbers along the scaffold [133]. When the same scaffold was seeded with genetically modified Schwann cells which secrete a bi-functional neurotrophin (D15A) with BDNF and NT-3 activities and implanted for 6 weeks, modest axonal regeneration was noticed [134]. In another study of PLGA, when human NSCs were seeded into the porous scaffold and then implanted into hemisected spinal cord of monkey, there was no significant functional improvement [75], which, however, was reported to be more suitable for the brain repair applications [124].
For the in vivo study, those polymeric scaffolds need to be developed into a tubular form, which support axonal guidance of nerve tissues after SCI, and the examples of tubular form scaffolds are summarized in Table 3. The incorporation of neurotrophic factors within the tubular guidance made of synthetic hydrogel (pHEMA-MMA) or fibrin has shown an improvement of specific supraspinal and local axonal regeneration and locomotor function after complete spinal cord transection [131]. When NSCs or SCs were seeded within the PLGA scaffold containing seven small channels inside, lesion-crossing neurons and outgrowing axons within scaffold were increased however, there was no functional improvement [76, 132]. Nomura et al. made chitosan tube and implanted with NSCs into completely transected spinal cord of rats, but no functional improvement was still noticed [120].
Recent studies on the application of tubular form biopolymer scaffolds to animal SCI models. Abbreviations are defined as follows: pHEMA-MMA: poly (2-hydroxyethyl methacrylate-co-methyl methacrylate); PLGA: poly (lactic-co-glycolic acid); SD rat: Sprague-Dawley rat;; SCs: Schwann cells;.NSCs: neural stem cells, NSPCs: neural stem and progenitor cells.

Recent studies on the application of tubular form biopolymer scaffolds to animal SCI models. Abbreviations are defined as follows: pHEMA-MMA: poly (2-hydroxyethyl methacrylate-co-methyl methacrylate); PLGA: poly (lactic-co-glycolic acid); SD rat: Sprague-Dawley rat;; SCs: Schwann cells;.NSCs: neural stem cells, NSPCs: neural stem and progenitor cells.

A fibrous form of scaffolds, such as microfibers and nanofibers, has also been developed as a nerve guiding scaffold after aligning or texturing the morphology to enhance the neurite outgrowth and cell guidance. Some in vitro studies showed the neurite outgrowth along the aligned fibers [135–137]. However, there were few in vivo reports on the fibrous scaffolds. One of the challenges in the fibrous scaffolds is that this structure is hard to directly apply to animal models, therefore, new techniques are needed to develop them into three dimensional scaffolds for the SCI implantation. Currently, there is increasing trend of researches from using only the scaffolds in tubular or fibrous form towards their applications in combining either neurotrophic delivery systems and/or cells.
Since several decades ago, exercises such as locomotor training, strengthening and passive range of motion exercises, and occupational therapies have been commonly performed on SCI patients in rehabilitation units. Various types of exercises and supporting strategies including functional electrical stimulation and robot-assistive devices have been developed to enhance walking capacity for SCI patients, but the effects on the regeneration of injured spinal cord according to the types of exercise remain unknown [138].
The cellular and molecular mechanisms of the effect of exercise training after SCI are not clearly understood. Some researchers have recently tried to reveal theses mechanisms through animal studies. Endogenous NSCs exist around the central canal of the adult spinal cord, and Foret et al. found that treadmill exercise for compressive SCI rats enhanced locomotor recovery and increased the number of NSC proliferation [139]. The supraspinal pathway, including the somatosensory cortex, might be changed after SCI. Kao et al. made a complete transection model of neonatal rats and found that treadmill exercise for 6–8 months could restore the reduced response of the somatosensory cortex to forelimb stimulation, and the percent and magnitude of responding cells in the hindlimb somatosensory cortex was increased after exercise [140]. BDNF level in the spinal cord increases following 4-weeks of treadmill exercise in normal adult rats [141], and selective upregulation of BDNF in the motor nuclei improves functional recovery in complete SCI rats [142]. But early exercise in the acute inflammatory phase (1 week) after contused SCI in rats can induce allodynia with aberrant sprouting of C afferent fibers through BDNF-tropomyosin-related kinase B signaling [143]. Maier et al. performed combination therapy with anti-Nogo-A antibody and treadmill exercise to incomplete SCI rats, but this combination did not show synergistic effects, due to the differential mechanisms between two modalities [144].
Combinations of Therapeutic Strategies
Combination therapies are expected to enhance the regeneration after SCI because each therapeutic strategy targets a different mechanism and result. For example, the combination of strategies which affect different stages, neuroprotection and neurorestoration, will improve the injured spinal cord more than a single strategy [145]. But the mechanisms of many therapeutic strategies are still unknown; therefore, combinations of single useful therapeutic strategies do not always show synergistic result.
Bunge found that the combination of SCs, OECs, and chABC application to spinal cord transection and the bridge model of rats in the acute stage could improve locomotor function and increase myelinated axons in the transected spinal cord. As well, it was found that the combination of SCs and cyclic AMP application in the subacute stage after contusion SCI of rats was the best way to improve locomotor function and increase serotonergic nerve fibers [145]. In Table 4, we reviewed current in vivo studies on combination therapies for SCI repair.
Recent studies on the combination of stem cells, neurotrophic factors, suppressors of inhibiting factors, and drugs to animal SCI models. Abbreviations are defined as follows: SD rat: Sprague-Dawley rat; LE rat: Long-Evans rat; OECs: olfactory ensheathing cells; SIF: suppressor of inhibiting factor; NTF: neurotrophic factor; NSCs: neural stem cells; MSCs: mesenchymal stem cells; NPCs: neural precursor cells; NT-3: neurotrophin-3; PDGF: platelet-derived growth factor; BDNF: brain-derived neurotrophic factor; GDNF: glial derived neurotrophic factor; NGF; nerve growth factor; chABC: chondroitinase ABC; MPSS: Methylprednisolone sodium succinate; DI: direct injection surrounding the lesion; IT: intrathecal injection.


Recent studies on the combination of stem cells, neurotrophic factors, suppressors of inhibiting factors, and drugs to animal SCI models. Abbreviations are defined as follows: SD rat: Sprague-Dawley rat; LE rat: Long-Evans rat; OECs: olfactory ensheathing cells; SIF: suppressor of inhibiting factor; NTF: neurotrophic factor; NSCs: neural stem cells; MSCs: mesenchymal stem cells; NPCs: neural precursor cells; NT-3: neurotrophin-3; PDGF: platelet-derived growth factor; BDNF: brain-derived neurotrophic factor; GDNF: glial derived neurotrophic factor; NGF; nerve growth factor; chABC: chondroitinase ABC; MPSS: Methylprednisolone sodium succinate; DI: direct injection surrounding the lesion; IT: intrathecal injection.


Wang et al. cotransplanted NSCs and OECs into the partially transected spinal cords of rats 7 days after injury, and found functional improvement was significant in the cotransplanted group [146]. However Amemori et al. could not reveal significant functional improvement in the group of cotransplantation of MSCs and OECs into contused spinal cords in rats [147].
Combinations of Stem Cells with NTFs or SIFs
Rho kinase inhibitors, which prevent RhoA activation, were combined with MSCs and transplanted into compressive or contused SCI in rats and functional improvement was better than it was in MSCs or Rho kinase inhibitor transplantation alone groups [148, 149]. Johnson et al. performed combination therapy including fibrin scaffolds containing ESC-derived neural precursor cells (NPCs), NT-3 and platelet-derived growth factor (PDGF) with a heparin-binding delivery system into the dorsal hemisected spinal cords in rats 2 weeks after injury. They found increment of NPCs survival and differentiation into neurons 2 weeks after transplantation [150]. The combination therapy of OECs and BDNF into cervical SCI rats, however, reversely worsened the functional status compared with groups which received OECs, BDNF, or even the vehicle and failed to regenerate supraspinal axons through and beyond the lesion site [151].
Combinations of NTFs with SIFs
The combination treatment of thermostabilized chABC and NT-3 in a hemisection SCI model of rats showed increments of axonal outgrowth and functional improvement [152]. Sharma tried to combine some NTFs, including BDNF, GDNF, NGF, and NT-3, with time intervals for the treatment of transected spinal cords of rats, and they found that the combination of BDNF and GDNF at 60 and 90 minutes after injury was effective in reducing edema formation and cell injury, and achieved concomitant functional improvement [103].
Combinations of SIFs with Neuroprotective Agents
Clenbuterol is the β2-adrenoceptor agonist which acts as a neuroprotective agent inducing expression of neurotrophic factors and anti-inflammatory properties [155]. The combination of chABC and clenbuterol showed the increments of axonal re-growth, lesion-crossing axons and concomitant functional improvement in the complete transection model of rat spinal cords [153]. MPSS is a strong anti-inflammatory agent, as mentioned earlier, and the Nogo 66 receptor can bind MAG, Nogo-A and OMgp, which block neurite outgrowth. The combination of MPSS and Nogo 66 receptor antagonist and intrathecal administration via osmotic pump enabled the increment of the survival of neurons and oligodendrocytes and locomotor improvement [154].
Concluding Remarks
Recent clinical trials of stem cell transplantation for SCI patients were relatively safe and showed functional improvement, to some extent; however, many problems still exist and need to be considered for stem cells to be used clinically. Autologous MSCs and OECs that were used in previous clinical studies are not enough to replace damaged neuronal cells and to reconnect the impaired spinal tracts for the fundamental regeneration of injured spinal cord. Any clinical trials for chronic SCI patients, who constitute the greatest population of SCI patients at present, did not show functional improvement. Clinical trials of stem cell transplantation for the chronic SCI patients still require a progress to phase II studies. There was no evidence as to which method of stem cell delivery to use, such that a better outcome is achieved, and also the amount of transplanted stem cells was quite variable. Some treatment options are developed and have advanced recently, including induced pluripotent stem cells, neurotrophic factors or suppressor of inhibiting factors, biopolymers and exercise training, but the mechanisms of each strategy are not clear enough to progress to clinical translation. The researchers have to reveal the molecular and cellular mechanism of each therapeutic strategy through animal study, and then combine the strategies with different mechanisms of treatment or different stages of SCI to thereby gain a synergistic effect. Clinical trials would ideally follow completion of these animal studies. Recent pioneering researches of regenerative medicine, including induced pluripotent stem cells, biopolymer scaffolds, and delivery systems, are progressing very rapidly and a combination of them, or else the combination with previous effective strategies is, in the near future, expected to create a novel method for the fundamental regeneration of SCI.
Footnotes
Acknowledgment
This work was supported by a Priority Research Centers Program (2009-0093829) and a World Class University (WCU) Project (R31-2008-000-100069-0) through the National Research Foundation (NRF) funded by the Ministry of Education, Science, and Technology.
