Abstract
Background
Methods
Results
Conclusions
Introduction
Heart disease is the number one cause of death both in the United States and abroad, and an increasingly common cause of death for persons with HIV (PWH) in the era of effective antiretroviral therapy (ART) [1,2]. Current guidelines recommend a risk-based approach to cardiovascular disease prevention including the management of cholesterol with medications such as statins [3]. This begins with baseline risk assessment using the ACC/AHA atherosclerotic Cardiovascular Disease (ASCVD) risk estimator [4]. For those at intermediate risk, the presence of risk enhancing conditions, such as HIV, may favour statin use. However, for patients that remain unsure of the risk/benefit of medication, coronary artery calcium (CAC) scoring is recommended to personalize a patient's risk.
CAC is quantified from a gated, non-contrast computed tomography (CT) scan of the heart [5]. CAC burden has been characterized using Agatston score, calcium mass and calcium volume [5,6].
PWH have a higher risk of atherosclerotic cardiovascular events compared with uninfected controls [7]. This residual risk may be in part mediated by excess inflammation and immune activation despite ART [8]. In this study, we sought to use serial measurements of CAC to assess whether Agatston score, calcium mass or calcium volume were modified by statin use and to assess whether CAC changes were related to biomarkers of inflammation.
Methods
SATURN-HIV (Stopping Atherosclerosis and Treating Unhealthy bone with RosuvastatiN in HIV) was a prospective, randomized, double-blinded, placebo-controlled trial aiming to measure the impact of daily rosuvastatin 10 mg on cardiovascular and bone disease as previously described [9]. Our study is a secondary analysis of longitudinal data from the trial across three time points (0, 48 and 96 weeks). Eligible participants were PWH, ≥18 years of age, fasting LDL ≤130 mg/dl (3.37 mmol/l) and either high sensitivity C-reactive protein of ≥19.05 nmol/l (2 mg/l) and/or ≥19% activated CD8+ T-cells (CD8+CD38+HLD-DR+). Exclusion criteria included active or chronic inflammatory conditions (besides HIV), prior myocardial infarction, active pregnancy or lactation, use of systemic chemotherapy or immunomodulating agents and uncontrolled diabetes mellitus or thyroid disease. Additional eligibility criteria have been published previously [9–11]. The study was approved by University Hospitals Internal Review Board and all participants signed written informed consent.
Clinical information was acquired via self-report and medical records. Clinical laboratory assessments were performed in real time and soluble markers were measured from frozen plasma as previously described [9–11]. Biomarker measurements from 0, 48 and 96 weeks used for this analysis included soluble CD14, soluble CD163, interleukin-6, high-sensitivity C-reactive protein, tumour necrosis factor-α receptors I & II, soluble vascular cell adhesion molecule, d-dimer and fibrinogen.
CAC was assessed at 0, 48 and 96-weeks from 64-slice, ECG-gated, non-contrast CT scans (Somatom Sensation 64, Siemens Medical Solutions USA) with standardized collimation, rotation time and tube voltage. Standard 3 mm slices were obtained from the carina to the diaphragm and reconstructed utilizing the B36f kernel (Siemens Medical Solutions USA). Total CAC Agatston score was verified by an experienced thoracic radiologist. Similarly, CAC mass and volume were determined via a standard calibration factor and was dependent on the reconstruction field of view.
We examined the shape of the distribution of each biomarker and the outcome variables to check normality. When variables were skewed or had outliers, those variables were log transformed to achieve a normal distribution prior to analysis. However, if any transformations did not achieve a normal distribution, we created a binary variable. The CAC outcomes of interest were: mass score, volume score, diffusivity index (1-[CAC in most affected vessel/total CAC]), CAC >0 and number of vessels with CAC [12]. Mixed effects models, ordinal logistic regression and generalized estimating equations were used to examine the statin effect on CAC measures and associations of CAC measures with bio-markers of inflammation and immune activation. To address potential confounding of the biomarker-CAC associations, we first explored whether traditional risk factors (age, systolic blood pressure and smoking) were associated with the CAC outcomes over time. Because time-updated age and systolic blood pressure were associated with CAC outcomes at P<0.05, they were included in the final models. Cholesterol changes were not considered a confounder since this was a statin trial, but all final models adjusted for statin treatment arm. Finally, we performed two sensitivity analyses of the sCD14 findings in which we excluded participants with detectable viral load (HIV-1 RNA >50 copies/ml). Analyses were performed using SAS version 9.4 (SAS Institute, Cary, NC, USA).
Results
The SATURN-HIV trial enrolled 147 total subjects, with baseline characteristics well-balanced between treatment groups (all P>0.05). Of those, 128 completed 48 weeks of follow-up and 118 subjects completed 96 weeks. Reasons for and characteristics of subjects lost to follow-up have been previously described [9–11]. At study entry, median (IQR) age was 46 (40–53) years; 78% were male and 68% were African American. Median (IQR) CD4+ was 613 (425–853) and median (IQR) duration of prior ART was 5.3 (3.2–9.8) years. All were on ART (half on protease inhibitors) and 77% had fully suppressed HIV viral load (78% statin versus 77% placebo; P=0.91). Cardiovascular risk profiles were low with median 10-year Framingham risk score of 3.0% (1.0–7.0%); however, 63% were active smokers. 54 subjects (37% of total subjects) had a CAC score >0 at baseline (Supplemental Tables 1 and 2, Additional file 1), of whom 30 (20% of total subjects) had only a single vessel affected. Among 24 subjects with >1 vessel CAC at baseline, the mean (
Association of biomarkers with total CAC score and number of involved vessels across all three time points
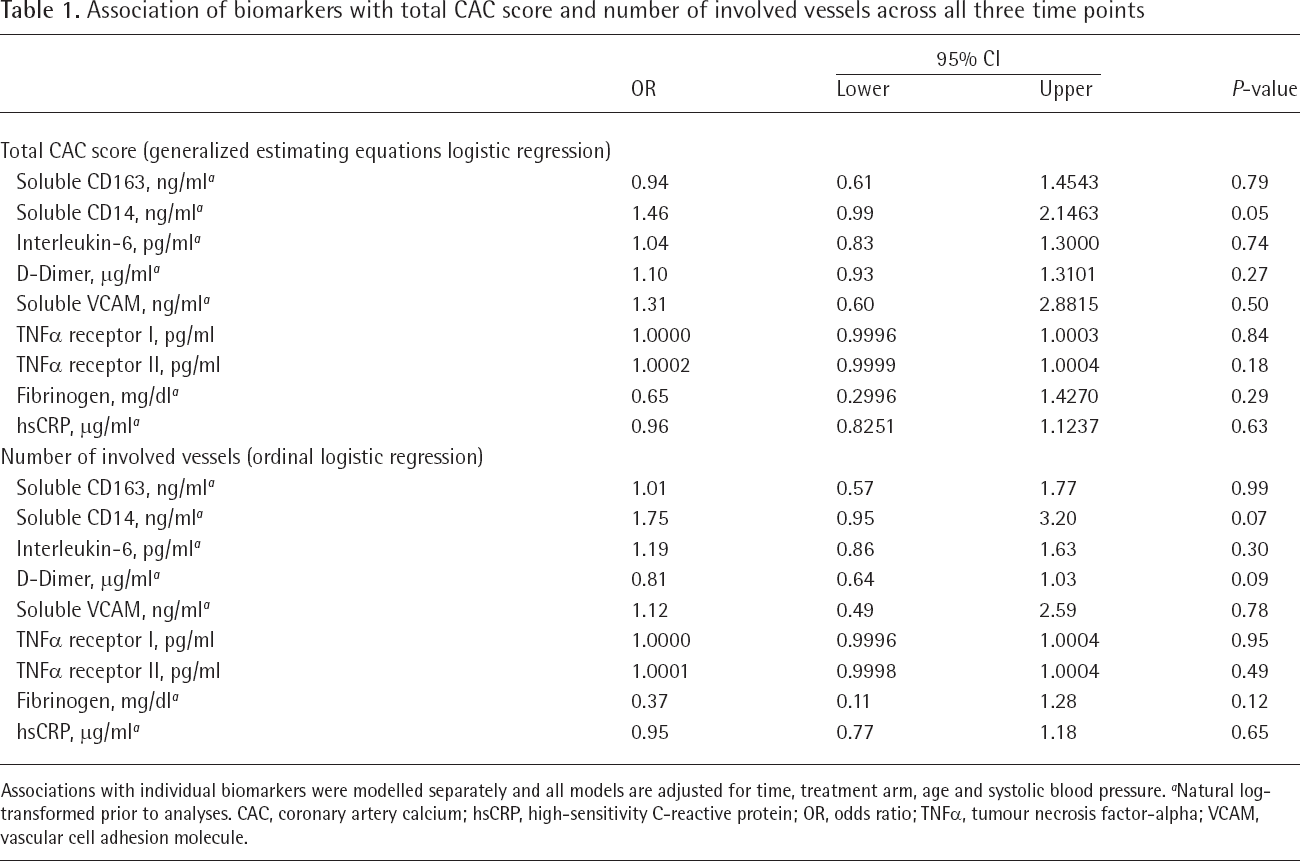
Associations with individual biomarkers were modelled separately and all models are adjusted for time, treatment arm, age and systolic blood pressure.
Natural log-transformed prior to analyses. CAC, coronary artery calcium; hsCRP, high-sensitivity C-reactive protein; OR, odds ratio; TNFα, tumour necrosis factor-α VCAM, vascular cell adhesion molecule.
Among those with detectable CAC >0, randomization to statin therapy was not associated with change in mass score (P=0.65; Figure 1A), volume score (P=0.76; Figure 1B), number of involved vessels (P=0.51; Supplemental Table 3, Additional file 1) or diffusivity index (P=0.13; Supplemental Table 4, Additional file 1) over 96 weeks. Additionally, for vessels without CAC at baseline, statin therapy was not associated with development of incident CAC in that vessel over time (P=0.13). In models that adjusted for treatment group, time, age and time-updated systolic blood pressure, only natural log-transformed soluble CD14 was positively associated with the presence of CAC (Table 1; OR 1.46; P=0.05) and borderline positively associated with number of involved vessels (Table 1; OR 1.75; P=0.07) across all three time points. In a sensitivity analysis limited to those participants with suppressed viral load (n=113 with viral load <50 copies/ml), the association of sCD14 with total CAC was modestly attenuated and not statistically significant (OR 1.33; P=0.11), but the association with number of involved vessels (OR 1.92; P=0.04) was stronger.

Effect of rosuvastatin 10 mg daily on coronary artery calcium mass score and volume score
Discussion
In this study of PWH on ART, we serially assessed the effect of rosuvastatin 10 mg daily on several measures of coronary calcium and explored associations of these measures with markers of inflammation and immune activation. We found no evidence of a differential effect of statin therapy on calcium mass, density or distribution in this population. We also confirmed that soluble CD14 remained independently associated with multiple measures of CAC across all three time points of the clinical trial, as initially described in our cross-sectional analysis of the baseline Agatston scores only [9]. Similar to our initial cross-sectional analysis, there were no other consistent associations of CAC with other biomarkers.
Our findings add to a growing literature on the association of subclinical coronary disease with markers of inflammation and immune activation among PWH [7]. Of all the candidate biomarkers studied, markers of innate immune activation, such as soluble CD14 and CD163, seem to be the most consistently associated with subclinical atherosclerotic disease in this population, whereas associations with other biomarkers are weaker [13]. Our study examined the associations of inflammation with more detailed coronary calcium score measures that have not been studied to date. Our results suggest a clinically relevant association of sCD14 with the presence and diffusivity of CAC that is present to some degree even in analyses limited to PWH with fully suppressed viral load; however, the relative contribution of inflammation to residual CVD risk is likely lower for patients with suppressed viral load. Ultimately, there is much interest in treating inflammation to reduce cardiovascular risk in PWH; however, early data from clinical trials of potent anti-inflammatory drugs evaluating surrogate cardiovascular outcomes have been mixed [14,15].
Statins slow progression of subclinical coronary [16] and carotid disease [17] in PWH, and the statin effect on clinical outcomes is being tested in the ongoing REPRIEVE trial [18]. Although statins reduce the risk of coronary events, they promote calcification of coronary plaque [19]. Limited data exists regarding statin effect on CAC and its measures. Similar to our findings, Lo et al. [16] reported no effect of atorvastatin on total CAC, total mass score or total volume score in a trial of 40 PWH over 12 months despite significant reductions in non-calcified plaque volume. Our findings also suggest that there are no significant differential statin effects on measures of CAC diffusivity.
The SATURN-HIV trial was not powered to detect a change in these exploratory CAC outcomes, and therefore, the lack of statin effect should be viewed with some caution. In addition, we cannot rule out residual confounding as a cause of the association between sCD14 and CAC; however, consistency with prior studies strengthens the validity of this finding [9,20]. Our study was relatively small; therefore, similar analyses of these CAC outcomes should be replicated in the ongoing REPRIEVE sub-study, which is the largest ever placebo-controlled statin trial to evaluate cardiac CT outcomes [18]. The limited follow-up time may explain the lack of a statin effect, since statin effects on plaque may occur more slowly in this relatively young population. Finally, our population was largely male which may limit its generalizability to populations with more equal representation of women such as sub-Saharan Africa.
In conclusion, among PWH on ART, moderate intensity rosuvastatin does not appear to differentially affect measures of coronary calcium mass, volume or diffusivity over 96 weeks. Soluble CD14, a marker of monocyte activation, was associated with measures of coronary calcification over time. Future studies should evaluate how these measures of coronary calcification relate to risk of atherosclerotic cardiovascular events in this high-risk population.
Footnotes
Acknowledgements
Research reported in this publication was supported in part by the National Heart, Lung, and Blood Institute of the National Institute of Health under award numbers R25HL103152 to JL, K23HL123341 to CTL and R01NR012642 to GAM. Technical support was provided by the Center for AIDS Research, Case Western Reserve University (P30 AI36219). Study drugs and matching placebo were donated by AstraZeneca. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health.
CTL has received research grants from Medtronic Philanthropy and Gilead Sciences and served on an advisory board for Esperion Therapeutics. GAM has served as a scientific advisor for Gilead Sciences, ViiV, and Merck, has received research grants from Bristol-Myers Squibb, Merck, Astellas, Tetraphase, Roche and Gilead Sciences. No other authors have any disclosures relevant to the content of this manuscript.
