Abstract
Background
Methods
Results
Conclusions
Introduction
Herpes simplex virus (HSV) infections are widespread among human beings globally and are an essential problem in general health care [1]. More than 3.7 billion people under the age of 50 are infected with HSV-1 and more than 400 million people suffer from HSV-2 infection [2].
Both HSV-1 and HSV-2 viruses are transmitted through contact with an infected person's lesions, mucosal surfaces, or genital or oral secretions. HSV infection is characterized by vesicles on different parts of the skin, mainly on the mouth with HSV-1, and on the vaginal surface with HSV-2. The herpes viruses infect the epithelial cells of the skin and mucosa through minor breaks and then travel by retrograde transport to the sensory root ganglia, where they persist throughout life. Classic symptoms are pain, burning, itching, fever, headache. Women can suffer further complications such as urinary tract dysfunction. In rare cases there may be complications such as meningitis, extragenital lesions and, in severe cases, death [3].
The initial diagnosis of herpes infections is often made by a primary care physician, followed by referral to dermatology or genitourinary medicine clinics. Patients with frequent recurrences should be referred to a specialist for further investigation and treatment. If they are started on suppressive therapy, they will often be referred back to the primary care physician for ongoing management [4]. Currently, there is no cure or vaccine for HSV infection [5]. Acyclovir is one of the most used agents for the treatment of HSV infection. However, a severe problem with acyclovir is the emergence of drug resistance in treated patients. Therefore, new antiviral agents exhibiting different mechanisms of action are urgently needed [6].
A large number of natural compounds have been extensively studied for their antiviral activity [7]. A wide variety of those compounds such as phenolics, polyphenols, terpenes, flavonoids have shown promise as anti-herpetic agents [8].
Propolis or ‘bee resin’ is the generic name used to identify a natural and resinous substance that bees (Apis mellifera) collect from buds, exudates and plants such as birch, poplar, pine, alder, willow, palm, Baccharis dracunculifolia and Dalbergia ecastaphyllum [9]. Propolis is a heterogeneous mixture of many substances harvested, processed and used by bees to close hive holes and in general to protect the hive [10]. Several studies have revealed that bee propolis can play an essential role in the colony's immunity as a direct defence against pathogens and intruders [11]. Propolis, along with other beekeeping products such as honey, royal jelly and pollen, has essential nutritional and therapeutic properties in use in integrative medicine [12]. A number of biological properties have been attributed to this natural product such as antioxidant, hepatoprotective, anti-tumour, anti-inflammatory, anti-bacterial and anti-parasitic activities making propolis a natural medicine both for improving general health and preventing diseases [12–14]. Propolis is also used in cough syrups, oral pills, pads, ointments, lotions and with vitamins in products to treat viral diseases, fungal infections, ulcers and surface burns [15].
Propolis is a lipophilic, hard and fragile material that when heated becomes soft, flexible, gummy and sticky [16]. It is poorly soluble in water and partially soluble in alcohol, acetone, ether, chloroform and benzene [16]. Propolis typically consists of resin (approximately 50%) containing flavonoids and poly-phenolic acids, waxes (approximately 30%), essential oils (approximately 10%), pollen (approximately 5%) and other organic molecules (approximately 5%) [17]. More than 300 compounds of different origins have been identified in the various propolis samples [18], and many others have been identified during the chemical characterization of new types of propolis [19,20] belonging to fatty acids and phenols, esters, substituted phenolic esters, flavonoids (flavones, flavanones, flavonols, dihydroflavonols and chalcones), terpenes, aromatic aldehydes and alcohols, sesquiterpenes, derivatives of naphthalene and stilbene [21]. Polar (aromatic acids, esters, and flavonoids) and non-polar (fatty acids, their esters and glycerol) propolis compounds are derived from poplar exudates, bee metabolism, contamination with honey (various sugars) and beeswax [22]. Flavonoids are important bioactive constituents of poplar propolis, mainly pinobanksin, pinocembrin, galangin, chrysin, kaempferol and quercetin. The aromatic acids in poplar brown propolis are derivatives of hydroxybenzoic acid (such as gallic, gentisic, protocatechuic, salicylic and vanillic acids) and derivatives of hydroxycinnamic acid (such as p-coumaric, caffeic and ferulic acids). They are also found as benzyl-, meth-ylbutenyl-, phenylethyl- and cinnamyl-esters [21]. Phenolic glycosides (sugar conjugates) are poorly present in propolis due to the lipophilic character of the resin and to hydrolysis during the propolis collection and processing made by β-glucosidase. For these reasons, most flavonoids found in propolis are aglycones [5].
Specific extraction methods are used to purify the raw propolis from undesired and inert material and to preserve the active components such as the polyphenolic compounds [16]. In the literature, it is possible to find several extraction methods using different solvents such as water, ethanol, methanol, hexane, acetone and chloroform [3,23]. However, the most common technique is ethanol extraction producing final products having a low concentration of wax and high content of bioactive compounds [24].
Chemical variability of propolis has been discussed in the literature with respect to the problem of standardization. Reliable criteria for chemical standardization of different propolis types are needed [12]. Multi Dynamic Extraction (M.E.D.®) method is a patented procedure [25] and the extracts contain no inactive resins and are rich in polyphenols. In particular, the resultant brown propolis extracts are characterized by the presence of a biologically active polyphenol complex identified in six primary polyphenols (M.E.D.® fingerprint) composed of galangin, chrysin, pinocembrin, apigenin, pinobanksin and quercetin having a relative concentration in the extract always higher than 25% w/w [13].
In this study, we aimed to investigate the antiviral activity of different extracts of Standardized Propolis Preparations (M.E.D.®) with glycol, ethanol, glycerol, and soya oil, against herpes simplex type 1 (HSV-1) and type 2 (HSV-2) viruses.
Methods
The cytotoxic effect of each extract was determined in vitro by MTS (3-(4,5-dimethylthiazol-2-yl)- 5- (3-carboxymethoxyphenyl)- 2- (4-sulfophenyl)-2H-tetrazolium) on human immortalized keratinocyte (HaCaT) cell line along with the cytotoxic concentration (CC50). The effective concentration (EC50) that can kill 50% of cells infected with HSV-1 and HSV-2 was evaluated by quantitative real-time PCR, and the antiviral efficacy of the extracts was determined by selective index values (SI: CC50/EC50). Acyclovir was used as a positive control to compare the efficacy of the extracts against the viruses.
Propolis extracts
Standardized propolis preparations as glycolic (98% propylene glycol with the remaining 2% of ethanol), ethanolic (80% ethanol/20% water), glycerol (95% and 5% water) and soya oil (100% oil) extracts were provided by B Natural srl (Milan, Italy). The various samples were analysed by total polyphenols content and HPLC-UV-ESI-MS (Jasco Inc., Tokyo, Japan) for accurate structural characterization and composition [16,19,20]. The phenolic acids and flavonoids from the four propolis preparations were separated by a Discovery-C18 column (from Sigma-Aldrich, Munich, Germany). The eluents were (A) 0.5% acetic acid and (B) acetonitrile. The separation was performed at room temperature by gradient elution from 0 min at 50% A/50% B to 60 min at 100% B at a flow rate of 0.8 ml/min. An Agilent 1100 VL series mass spectrometer (Agilent Technologies, Inc., CA, USA) was used online with HPLC equipment. The electrospray interface was in negative ionization mode with the capillary voltage at 3,500 V and a temperature source of 350°C in full scan spectra (200–2,200 Da, 10 full scans/s). Nitrogen was used as a drying (9 l/min) and nebulizing gas (11 p.s.i.). Software versions were 4.0 LC/MSD trap control 4.2 and Data Analysis 2.2 (Agilent Technologies, Inc.). 10 ml of samples were injected at a standardized concentration expressed as total polyphenols content evaluated by spectrophotometric assay according to Folin-Ciocalteu against a calibration curve performed by pure galangin.
Total polyphenol content was tested on 20 ml of the propolis preparations to which was added 380 ml of ethanol and 100 ml of the Folin-Ciocalteu reagent (Sigma-Aldrich). Then, 200 ml of a saturated sodium carbonate solution and 4.3 ml water was added. After standing at room temperature for 60 min, the absorbance was read at 750 nm against the blank. The concentration of polyphenols in the samples was derived from a standard curve of galangin ranging from 5 to 60 mg (correlation coefficient >0.980), and the total polyphenolic content was expressed as % mg/ml.
Preparation of propolis extracts and acyclovir
Stocks of extracts were sterilized using 0.22 μm Millipore filters, and aliquots of 1 ml were stored at +4°C until used in cell culture. Each extract was warmed for 1 h at room temperature and vortexed just before diluting with media. Ethanol and glycolic samples were prepared at 1 mg/ml, while glyceric and soya oil extracts were diluted to reach 3.5 mg/ml with Dulbecco's Modified Eagle Medium (DMEM).
Each vial of acyclovir (acyclovir sodium, Zovirax) of 250 mg was dissolved in 10 ml sterile saline solution (sodium chloride intravenous infusion BP, 0.9% w/v) to obtain a concentration of 25 mg/ml and further diluted to prepare 20 μM working stock aliquots stored at -20°C until use.
Cell line and viruses
Immortalized human keratinocytes (HaCaT) cell line, herpes simplex type 1 (MacIntyre, #0810005CF, Zeptometrix), and herpes simplex type 2 (MS, #0810006CF, Zeptometrix) viruses were used (cell line and viruses were obtained from the Genetic and Bioengineering Department of Yeditepe University, Istanbul, Turkey).
Cytotoxicity assay
Cytotoxicity assay used to determine the toxic and non-toxic concentration of the compounds was determined on the immortalized HaCaT cell line analysed with MTS (3-(4,5-Dimethylthiazol-2-yl)- 5- (3-carboxymethoxyphenyl)- 2- (4-sulfophenyl)-2H-tetrazolium) method. Briefly, HaCaT cells were seeded as 5x10 3 cells (100 μl/well) in 96-well plate and incubated at 37°C, 5% CO2 for 24 h. After incubation, media were aspirated, and diluted propolis extracts at different concentrations were added onto cells and plates and incubated at 37°C, 5% CO2 for 72 h. After this period, old media were discarded, and 100 μl of MTS solution (10% MTS with DPBS/glucose media) was added onto cells and incubated for 2 h. The absorbance of coloured dye was measured using a microplate reader (Bio-Rad, Tokyo, Japan) at 490 nm, and quantification of the absorbance values was performed by comparison of treated cells with control ones.
Viral titration assay
The most common method for determining viral titres is 50% tissue culture infectious dose (TCID50) by using microscopic observation of cytopathic effect (CPE) or counting viral plaque in a culture plate. However, because the conventional TCID50 method is time-consuming and subject to errors, the colorimetric MTS method used to determine viral titres with small modifications was applied in our study [26]. HaCaT cells were seeded as 3x10 4 cells (200 μl/well) in 96-well plate and incubated at 37°C, 5% CO2 for 24 h. Next day, cells were observed under the microscope (Zeiss Axio Vert.A1, Köln, Germany) to check morphology and confluency, and different virus dilutions were prepared on ice in log scale (log 2) from 10−2 for HSV-1 and 10−1for HSV-2 diluted virus stock to obtain a 50% infectivity point for virus inoculation. After the preparation of virus dilutions, old media were aspirated, and each thoroughly washed three times with DPBS. Diluted virus solutions were inoculated into cells as 50 μl/well and incubated at 37°C, 5% CO2 for 2 h. After incubation, unbound viruses were aspirated and 200 μl of virus media (DMEM with 2% FBS and 1% PSA) were added to each well and incubated at 37°C, 5% CO2 for 72 h. After incubation, media were aspirated, and 200 μl of virus media with 10% MTS was added to cells. After incubation for 3 h, absorbance was measured using a microplate reader (Bio-Rad) at 490 nm, and quantification of the absorbance was performed by comparison of treated cells with controls. This protocol was applied to determine the HSV-1 and HSV-2 viral titration able to induce a 50% virus infection [26–28].
Viral DNA isolation method
For qRT-PCR analysis, viral DNA was isolated using Roche high pure viral DNA isolation Kit (#11858874001, Switzerland) from infected and treated cells. HaCaT cells were seeded at 1x10 5 cells (400 μl/well) in 48-well plates and incubated at 37°C and 5% CO2 for 24 h. The media was aspirated from the well the next day and washed 3 times with DPBS. Cells were inoculated with 150 μl of previously prepared working virus stock and incubated at 37°C and 5% CO2 for 2 h. Meanwhile, the various propolis samples were prepared at different dilutions in DMEM with 2% FBS and 1% PSA under minimum cytotoxic concentration. Acyclovir working stock was also warmed at room temperature to prepare different concentrations from 0.4 up to 5 μM. After incubation, unbound viruses were sucked out, and the cells treated with 400 μl of propolis samples and acyclovir. 20% of DMSO-treated cells were used as control. After incubation at 37°C and 5% CO2 for 72 h, supernatants were collected and centrifuged at 5,000 rpm for 30 min to remove cell debris. Viral DNA was isolated according to specified protocol and stored at -20°C until use. These procedures were applied to different concentrations of each sample and acyclovir for HSV-1 and HSV-2 and repeated 3 times.
Quantitative real-time PCR
The qRT-PCR was performed after DNA isolation using quantitative HSV-1 and an HSV-2 Kit (R-gene HSV-1 #71015, HSV-2 #71016, bioMérieux, Lyon, France) by real-time PCR using TaqMan 5′-nuclease technology. The targeted sequence of HSV-1 is in the US7 gene and amplified fragment size is of 142 base pairs while the targeted sequence of HSV-2 is in the US2 gene and amplified fragment size is of 177 base pairs. EC50 values of the various samples were calculated with the obtained data from qRT-PCR (virus amount expressed as copies/ml). The SI, a widely accepted parameter used to express the in vitro efficacy of a compound in the suspension of virus replication, was calculated based on CC50 values for HaCaT cell lines and EC50 values for HSV-1 and HSV-2, as illustrated: SI = cytotoxic concentration 50 (CC50)/effective concentration 50 (EC50).
Statistical analysis
All experimental data were evaluated using GraphPad Prism (GraphPad Prism version 7.0, GraphPad Software, San Diego, CA, USA). The significance of the groups versus the control group was evaluated with a one-way ANOVA and Dunnett's test. P<0.05 was considered as a significant value.
Results
Evaluation of characteristics and composition of the propolis extracts
Through HPLC separation (Figure 1) and tandem MS analyses [16,20], approximately 30 different biomolecules were identified and quantified in the various M.E.D.® propolis preparations manufactured by B Natural accounting for approximately 60–70% of the total species, mainly polyphenols, phenolic acids and bioflavonoids (Table 1). The remaining unidentified part could be mainly represented by triterpenoids and a small content of glycosylated derivatives.

HPLC separation and MS detection of the four standardized (M.E.D.®) propolis preparations
Molecular bio components of the four propolis samples separated by HPLC and identified by MS and tandem MS

The retention times (RT) determined by HPLC, the maximum wavelengths of absorption in UV, and the mass values of the molecular species and derived fragments through tandem mass are also illustrated.
The four different extracts were found to include flavones (that is, chrysin and apigenin), flavanones (that is, pinocembrin) and flavonols (that is, quercetin, pinobanksin, galangin and their derivatives) as the most common components.
In vitro cytotoxicity of propolis extracts and acyclovir Cytotoxic effects of the four different propolis extracts and acyclovir on HaCaT cells were evaluated by increasing their concentrations. Supplemental viability % of treated cells are illustrated in Additional file 1, and the relative inhibition % values are reported in Additional file 2. The glycolic extract showed a severe cytotoxic effect at 800 μg/ml and a moderate effect at 400 μg/ml on HaCaT cells. The cytotoxic effect was not detected at 200 μg/ml, while the proliferative effect was observed at 100 and 50 μg/ml. Therefore, 200 μg/ml was determined as the minimum cytotoxic concentration (MCC or maximum non-toxic dose). Glycol solvent in the highest concentration of propolis extract was determined as non-toxic for HaCaT cells. The ethanolic propolis sample showed a severe cytotoxic effect at 1,000 μg/ml and a moderate cytotoxic effect at 500 μg/ml. Cytotoxicity was not observed at 200 μg/ml, and proliferation was measured at 150 μg/ml and 100 μg/ml. 200 μg/ml was fixed as MCC and ethanol showed a minimum proliferative effect on HaCaT cells. Propolis in glycerol showed moderate cytotoxic effect at 3,000, 2,500 and 2,000 μg/ml while no effect was determined at 1,300 μg/ml, and proliferation was measured at 1,000 and 900 μg/ml. 1,300 μg/ml was assumed as MCC, and glycerol was found to be non-toxic. The soya oil extract showed cytotoxic effect at 2,500 and 2,000 μg/ml (not shown), and no cytotoxicity was observed below 800 μg/ml (Additional file 2). The proliferative effect was observed for the soya oil solvent tested at the highest concentration of propolis extract, and 800 μg/ml was evaluated as MCC. Acyclovir showed cytotoxic effect producing cell death over 50% at 20 μM and causing limited cytotoxicity between 10 and 1 μM on HaCaT cells. No significant effect was measured at 0.8 μM, and this concentration was determined as MCC.
Cytotoxic concentration 50 (CC50), which is the concentration of a compound capable of killing half of the cells in uninfected cell culture, was calculated from each suspension values graph (Additional file 3). CC50 of glycol extract was calculated as 593 μg/ml, 375 μg/ml for the ethanolic sample, 1,723 μg/ml for the propolis in glycerol and 1,664 μg/ml for soya oil preparation. CC50 of acyclovir was determined to be 15.9 μM for HaCaT cells (Additional file 3).
Antiviral activity determined through quantitative real-time PCR
Antiviral activities of the different propolis extracts and acyclovir were determined for HSV-1 (Figure 2) and HSV-2 (Figure 3) for HaCaT cells based on previously calculated non-toxic concentrations (see Additional file 1). Virus inhibition was expressed as copies/ml in treated cells.

Viral inhibition of non-toxic concentrations of the propolis extracts

Viral inhibition of non-toxic concentrations of the propolis extracts
The EC50, the concentration of a compound capable of producing the 50% suspension of virus replication versus untreated virus-infected control, was also calculated [28]. EC50 values determined for each propolis extract and acyclovir for both viruses are illustrated in Tables 2 and 3.
CC50, EC50 and SI values of each propolis extract and acyclovir determined for HSV-1

Effective concentration that can kill 50% of infected cells (EC50) expressed as μg/mg of polyphenols was calculated by considering the total content of polyphenols determined by the Folin-Ciocalteu assay.
Units are μM. CC50, 50% cytotoxic concentration; HaCaT, human immortalized keratinocyte; SI, selective index.
CC50, EC50 and SI values of each propolis extract and acyclovir calculated for HSV-2
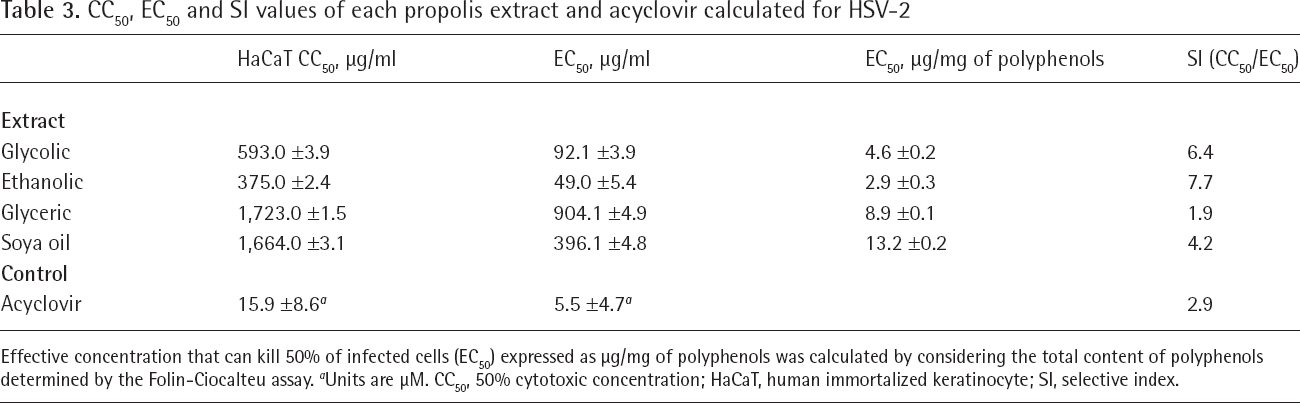
Effective concentration that can kill 50% of infected cells (EC50) expressed as μg/mg of polyphenols was calculated by considering the total content of polyphenols determined by the Folin-Ciocalteu assay.
Units are μM. CC50, 50% cytotoxic concentration; HaCaT, human immortalized keratinocyte; SI, selective index.
The SI values of the various propolis extracts, calculated by CC50 and EC50 data are shown in Tables 2 and 3 for HSV-1 and HSV-2, respectively. SI values of glycol, ethanol, glycerol, soya oil extracts and acyclovir were determined as 6.8, 4.1, 2.2, 3.3 and 6.3, respectively, for HSV-1. SI values of glycol, ethanol, glycerol, soya oil extracts and acyclovir were determined as 6.4, 7.7, 1.9, 4.2 and 2.9, respectively, for HSV-2.
As a result, a glycolic propolis extract possesses a greater antiviral activity than acyclovir for both HSV type 1 and type 2, while glycolic, ethanolic and soya oil preparations were found to have greater activity than acyclovir for HSV-2.
Discussion
In our study, EC50 values for HSV-1 and HSV-2 viruses were determined through a qRT-PCR method that is accepted as the best and most validated quantification method for assessing the antiviral characteristics of a drug [28]. It is known that HSV-1 and HSV-2 show several types of specific differences in interaction with their host cells. In particular, HSV-2 infection of cells is more adequately inhibited by polyanionic substances than HSV-1, while this latter virus infection is particularly inhibited by polycationic substances [29]. These differences between the two viruses and the different types of propolis extracts used in this study can be related to specific differences of viruses. The US Food and Drug Administration (FDA) approved the EC50 range of acyclovir (Zovirax) for HSV-1 between 0.02 to 13.5 μg/ml and HSV-2 between 0.01 to 9.9 μg/ml confirming our results. Previously, it was shown that the antiviral efficacy by propolis in synergy with acyclovir produced higher efficacy than acyclovir alone [27].
When compared with acyclovir, the ethanolic extract shows approximately comparable SI, while glycolic, glyceric and soya oil preparations have lower values. On the other hand, ethanolic, glycolic and soya oil are more efficient than acyclovir against HSV-2, except glyceric extract that shows low antiviral properties. Extracts or compounds with SI value >1 in the initial screen (tested at log dilution) are considered sufficiently active to warrant additional assays and are subjected to further evaluation in the primary screen [30]. Consequently, glyceric extract can be considered an effective product against HSV-2 for further analysis. This is more important when we consider that sensitivity testing results vary greatly depending on several factors, and this also explains differences in our results. Nevertheless, by considering the EC50 values calculated in μg/mg of polyphenols, apart from soya oil preparation, the other extracts show quite comparable values, glycolic and ethanolic. This demonstrates that the total content of polyphenols is a particularly important parameter for the consideration of propolis (and in general, extracts possessing polyphenols as active components) as a product having antiviral capacity. On the other hand, this aspect further draws our attention to accurate and validated analytical procedures that give structural and compositional parameters useful for a better definition of the biological properties of propolis.
Amoros et al. [31] evaluated the antiviral capacities of resin balsam against HSV-1 via different treatment times. Significant virucidal characteristics were observed when HSV-1 was pretreated in the presence of the compound. Moreover, a significant decrease in plaque number was observed by adding propolis during intracellular replication. This data confirms our results showing that viral copy is inhibited after treatment with propolis extracts for both viruses. According to Huleihel and Isanu [32] the antiviral characteristics of propolis were probably due to the prevention of viral adsorption. Schnitzler et al. [33], reported aqueous and ethanolic extracts of propolis had high antiviral activity against HSV-2. They also reported that propolis possesses a higher anti-herpetic effect and SI than isolated components [33].
Therapeutically, antiviral agents can suspend transcription or replicate the viral genome or interrupt viral protein synthesis. In other applications, antiviral agents can avoid virus entry or absorption into the host cells [34]. The capacity of propolis samples to precisely suspend the viral DNA polymerase during the intracellular replication cycle when new viral DNA is synthesized is also confirmed by considering the higher SI values of propolis extracts than acyclovir.
In this study, we also obtained important data for acyclovir (99% HPLC grade) used in cell culture. Acyclovir has poor oral viability and low aqueous solubility properties which delays its absorption [35]. We observed no effect of acyclovir even in 5 mM concentration for HaCaT cells that regularly continued their viability. To solve this problem, acyclovir was used in the form of sodium salt, which can be dissolved and easily absorbed by cells producing a cytotoxic effect at 5 μM.
In conclusion, different extracts of Standardized Propolis Preparations (M.E.D.®) with glycol, ethanol, glycerol and soya oil, were found having potent antiviral activity with different capacities against HSV-1 and HSV-2 viruses. Scientific literature reports data supporting the anti-herpetic effect of propolis extracted with ethanol [36]. However, to date, no comparable data is available regarding the anti-HSV-1 and HSV-2 effect of standardized propolis preparations produced with different solvents. Therefore, high SI values of propolis with standardized and defined composition and properties extracted and solubilized in various solvents against HSV-1 and HSV-2 may be considered an important and novel finding. Finally, future investigations should clarify the biological mechanism of the antiviral activity of propolis preparations both in vitro and in vivo.
Infection with HSV-1 and HSV-2 is commonly seen in general practice worldwide [1,2]. It may be related to a lifelong disease which results in mild to severe symptoms and sometimes death. Viruses occasionally reactivate and can require prolonged antiviral treatment in high doses, especially in immunosuppressed people, often causing the occurrence of drug-resistant strains.
Therefore, new molecules possessing antiviral characteristics should be found to overcome resistant strains. Natural products are one of the best sources of compounds that can be optimized to obtain drug-like molecules. Recently, natural products have attracted attention for antiviral drug discovery studies. Propolis as one such natural product is well known, having rich phenolic content, and possessing anti-inflammatory, antioxidant and anti-microbial capacities reported in vitro and in vivo. There is still a lot of work to be done for propolis usage in the clinic. The standardized propolis may allow researchers to connect a specific chemical propolis type to a particular type of biological activity and formulate recommendations for integrative and mainstream medical practitioners.
Footnotes
Acknowledgements
The authors would like to thank B Natural S.r.l. and Alfredo Fachini (Milan, Italy) for providing standardized propolis samples. They would also like to extend their sincere thanks to James Fearnley, the Director of the Apiceutical Research Centre, North Yorkshire, UK for his constructive criticism and insightful suggestions.
The authors declare no competing interests.
Additional file 1: A figure showing cytotoxic effect of the four propolis samples with a) glycol, b) ethanol, c) glycerol, d) soya oil extracts and e) acyclovir on HaCaT cells viability can be found at ![]()
