Abstract
Background
To compare effectiveness and safety of initial antiretroviral therapy (ART) among premenopausal and postmenopausal women living with HIV aged 45–60 years from the cohort of the Spanish HIV/AIDS Research Network (CoRIS) who initiated ART between 2004 and 2015.
Methods
Multivariable regression models were used to compare post- versus premenopausal women regarding viral suppression (≤50 copies/ml), change in CD4+ T-cell count and time to treatment change (TC) at 48 and 96 weeks after ART initiation.
Results
Among 230 women, 154 (67%) were premeno-pausal at ART initiation. The most frequent initial regimen was tenofovir disoproxil fumarate/emtricitabine/efavirenz prescribed in 49 (32%) premenopausal and 22 (29%) postmenopausal women. The proportion of TC was 35.7% and 30.3% at 48 weeks and 51.3% and 47.4% at 96 weeks, for pre- and postmenopausal women, respectively. There were no significant differences in CD4+ T-cell count changes from ART initiation, viral load suppression, time to TC or reason for TC between both groups. The main reason for TC was occurrence of an adverse event, followed by simplification, in both groups.
Conclusions
ART effectiveness and safety did not differ significantly between pre- and postmenopausal women.
Introduction
The use of highly active antiretroviral therapy (ART) has caused a substantial reduction of mortality and increased life expectancy among people living with HIV (PLWH) [1,2]. In Spain, the median age of PLWH increased from 38 years (interquartile range [IQR]: 34–42) in 2002 to 47 years (IQR: 41–54.5) in 2017 [3]. Thus, a substantial proportion of women living with HIV (WLWH) will live to experience menopause, which represents a complex transition in a woman's life [4] and is an indicator of morbi-mortality in HIV-negative women [5].
In addition to these menopause-related problems, WLWH also face the consequences of living longer with HIV infection and a longer exposure to ART. These interactions between HIV infection and menopause increase the complexity of the management of HIV infection in older women.
Levels of endogenous female hormones could affect CD4+ T-cell recovery (TCR) and HIV replication, so estradiol potently inhibits TCR activation of HIV-1 transcription [6] and might have an impact on short-or long-term treatment responses [7,8]. Thus, we could expect the effects of ART to be different among premenopausal and postmenopausal women. At the present time, little is known about the impact of menopause on HIV infection and ART effectiveness and there are conflicting data in the literature [9,10]. The Women Against Viruses in Europe Working Group has identified several priority areas for optimizing the care of women who live with HIV, including the study of pharmacokinetics, toxicity and tolerability of ART [11].
The aim of this study was to compare the effectiveness and safety of initial ART among premenopausal and postmenopausal WLWH aged 45-60 years from a multicentre cohort in Spain who initiated ART between 2004 and 2015.
Methods
Study design and study population
CoRIS is a prospective, open multicentre cohort of individuals newly diagnosed with HIV naive to antiretro-viral treatment at cohort entry [12].
We included women aged 45 to 60 years, who started their first ART between 1 January 2004 and 31 May 2015, and who had information on both CD4+ T-cell count and viral load within 24 weeks prior to ART and at 48 weeks after starting treatment, and information about menopausal status at ART initiation. Menopause was defined as 12 consecutive months without menstruation, which was based on self-report about menstrual cycles as defined in other similar studies [13,14].
Ethics
Every individual provided written informed consent to participate in the cohort, the HIV Biobank Ethics Committee approved the CoRIS cohort and the Ethics Committee of the Carlos III Institute of Health approved the study.
Outcomes
The primary outcomes were viral suppression (VS), defined as achieving an HIV viral load ≤50 copies/ml, and the change in CD4+ T-cell count from ART initiation. The secondary outcome was time from ART initiation to treatment change and the reason for the change.
The primary outcomes were analysed at weeks 48 and 96 (+12 weeks) from ART initiation and time to treatment change during the first 48 weeks and during the first 96 weeks, by menopausal status at ART initiation. Later changes in menopausal status were ignored.
Statistical analysis
Descriptive analyses were carried out using frequency tables for categorical variables and median and interquartile range (IQR) for continuous ones. The χ 2 test for independence was used for comparison of categorical variables and the non-parametric Mann-Whitney test for comparison of continuous variables.
Multivariable regression models were used to compare post- versus premenopausal women regarding viral suppression, change in CD4+ T-cell count and time to treatment change.
Multivariable models were adjusted for the following potential confounders at ART initiation: CD4+ T-cell count (<200; 200–350; >350 cell/μl; unknown), HIV viral load (<10,000; 10,000–100,000; >100,000 copies/ml; unknown), transmission category (heterosexual; people who inject drugs [PWID]; others/unknown), educational level (none or primary; secondary or university), AIDS diagnosis (yes; no; unknown), presence of HCV (yes; no; unknown) and year of ART initiation (2004–2009; 2010–2015).
All statistical analyses were performed using STATA software (version 16.0; Stata Corporation, College Station, TX, USA).
Results
A total of 230 women were included in the study, of which 67.0% (n=154) were classified as premeno-pausal and 33.0% (n=76) as postmenopausal at ART initiation. Median age of pre- and postmenopausal women at diagnosis was 40.3 (IQR 36.9–43.7) and 46.3 (40.8–49.8) years (P<0.001), respectively. The sociodemographic and clinical characteristics according to menopausal status at ART initiation are shown in Table 1. The most frequently prescribed initial regimen was tenofovir disoproxil fumarate (TDF)/emtricitabine (FTC)/efavirenz (EFV) for both groups, and 63.0% of all regimens included TDF/FTC. At ART initiation, there were no significant differences between pre- and postmenopausal women in CD4+ T-cell counts (208 versus 194 cell/μl; P=0.845), but premenopausal women had significantly lower viral loads than the postmenopausal ones (4.7 versus 5.1 log copies/ml; P=0.027).
Sociodemographic and clinical characteristics of 230 women included in the study, according to menopausal status at ART initiation

Data are n (%) unless otherwise indicated.
Other: retired, housewife, student. ART, antiretroviral treatment; ATV/r, atazanavir/ritonavir; AZT, zidovudine; DRV/r, darunavir/ritonavir; EFV, efavirenz; FTC, emtricitabine; II, integrase inhibitor; LPV/r, lopinavir/ritonavir; NNRTI, non-nucleoside reverse transcriptase inhibitor; NRTI, nucleoside reverse transcriptase inhibitor; PI, protease inhibitor; PWID, people who inject drugs; RAL, raltegravir; TDF, tenofovir disoproxil fumarate; 3TC, lamivudine.
Viral Suppression and Immunological Response
At week 48 after ART initiation, 87.7% (95% CI 81.4%, 92.4%) and 86.8% (77.1%, 93.5%) of pre-and postmenopausal women, respectively, achieved VS. At 96 weeks, 87.8% (81.2%, 92.5%) of premeno-pausal and 94.4% (86.2%, 98.4%) of postmenopausal women achieved VS. After adjustment for potential confounders, there were no significant differences on the probability of achieving VS between both groups at 48 or 96 weeks (Table 2).
Viral suppression at week 48 and 96 after ART initiation among premenopausal and postmenopausal women

Adjusted for CD4 count at ART initiation, viral load at ART initiation, mode of transmission, educational level, AIDS defining condition at ART initiation, HCV at ART initiation and calendar period of ART initiation. OR, odds ratio from logistic regression for the association between menopausal status at ART initiation and viral suppression; Post-meno, postmenopausal women; Pre-meno, premenopausal women.
Compared with premenopausal women, the mean increase of CD4+ T-cell count was higher in postmenopausal women at week 48 (207.40 [95% CI 180.32,234.48] versus 232.26 [193.72,270.81] cells/μl) and week 96 (287.68 [253.46,321.89] versus 348.75 [299.35,398.14] cells/μl), but these differences did not reach statistical significance after adjusting for confounding variables (Table 3).
Immunological response at week 48 and 96 after ART initiation among premenopausal and postmenopausal women
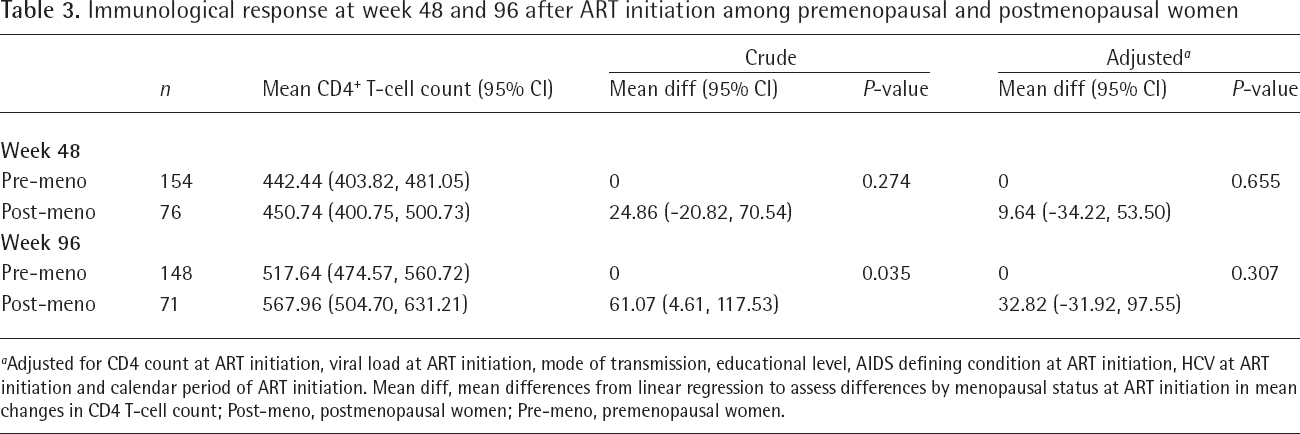
Adjusted for CD4 count at ART initiation, viral load at ART initiation, mode of transmission, educational level, AIDS defining condition at ART initiation, HCV at ART initiation and calendar period of ART initiation. Mean diff, mean differences from linear regression to assess differences by menopausal status at ART initiation in mean changes in CD4 T-cell count; Post-meno, postmenopausal women; Pre-meno, premenopausal women.
Time to treatment change and reasons for it
During the first 48 weeks of ART initiation, 35.7% and 30.3% of pre- and postmenopausal women, respectively, had changed their first ART (P=0.411). These proportions were 51.3% and 47.4% (P=0.575), respectively, before week 96. In multivariable analyses, we failed to find significant differences in time to treatment change between pre- and postmenopausal women neither before 48 nor 96 weeks. Among women who changed treatment, the main reason was occurrence of an adverse event before both 48 (49.1% and 56.5% [P=0.549] in pre- and postmenopausal women) and 96 weeks (41.8% and 52.8% [P=0.271]) followed by treatment simplification (16.4% and 26.0% [P=0.320], before week 48 and 15.2% and 27.8% [P=0.111], before week 96). Virological failure was the reason for treatment change in 3.6% and 4.3% (P=0.883) of the changes in pre- and post-menopausal women before week 48 and in 2.5% and 5.6% (P=0.411) of the changes before week 96. There were no significant differences on reasons for treatment change between both groups (Table 4).
Treatment discontinuation at week 48 and 96 after ART initiation among premenopausal and postmenopausal women

Adjusted for CD4 count at ART initiation, viral load at ART initiation, mode of transmission, educational level, AIDS defining condition at ART initiation, HCV at ART initiation and calendar period of ART initiation. Post-meno, postmenopausal women; Pre-meno, premenopausal women; sHR, sub-distribution hazard ratios from competing-risks survival regression to assess differences by menopausal status at ART in treatment change.
Discussion
In this study, we have evaluated effectiveness and safety of initial ART in pre- and postmenopausal WLWH from a national cohort. Our results show that viral suppression and immunological response at 48 and 96 weeks after treatment initiation were similar between pre- and postmenopausal women. The frequency of treatment changes due to adverse events, simplification or virological failure did not differ between both groups.
Very few studies have previously evaluated the effect of menopause status on the virological or immunological responses to ART. Two studies [9,10] have also found similar effectiveness of ART in pre- and postmenopausal women: these studies were performed in the United States and South America, where ethnicity, socioeconomic status, ART regimens and health-care access are different to our setting. We have found no other studies assessing tolerability to ART after menopause.
The most frequently prescribed ART regimen was the combination of TDF/FTC/EFV as this was a recommended drug combination for treatment initiation by the national guidelines during the study period [15]. The Spanish HIV treatment guidelines recommended the same regimens for HIV-infected women and men, although there are specific guidelines in women who are pregnant or wishing to conceive [16].
Previous studies have found that women are more likely to discontinue ART than men, mainly due to intolerance, toxicity and/or poorer adherence, although there are no differences in treatment effectiveness [17]. In our study, most of the women who changed their initial ART regimens did so because of adverse effects. This could be partly due to the high proportion of women receiving TDF/FTC/EFV, which was found to be the most frequently discontinued regimen in a previous study in the CoRIS cohort, mainly because of side effects [18]. The reasons for treatment change in our study were similar for pre- and postmenopausal women, and changes were due to virological failure only in a minority of women in both groups. During the study period, antiretroviral drugs with better tolerability such as integrase inhibitors became available, but only 11% of women were receiving integrase inhibitors in our study. In this analysis, we were unable to assess adherence to treatment, since this information is not collected routinely in the CoRIS cohort. Presence of menopausal symptoms can impair adherence to ART, and therefore its effectiveness, in WLWH [19].
The proportion of women in our cohort was low, refecting the epidemiology of HIV infection in Spain, where women account for only 15% of the new HIV diagnoses [20]. However, this is a well-established multicentre cohort which is representative of the Spanish general HIV-infected population [20], and which provides long follow-up periods. Another limitation was the lack of hormonal data that could help to better classify women's menopausal status and to assess the potential hormonal effects on response to ART initiation.
In conclusion, in our study there were no significant differences on viral suppression nor CD4+ T-cell count recovery after ART initiation among pre- and post-menopausal women. Treatment change due to adverse effects were low in both groups and did not show significant differences according to menopausal status.
Footnotes
Acknowledgements
This study would not have been possible without the collaboration of all patients, medical and nursery staff and data mangers who have taken part in the project. VH, ISG and IJ were involved in the study design. BA and VH participated in the collection and analysis of the data and wrote the first version draft of the manuscript. ISG and IJ made a critical revision of the manuscript for important intellectual content. All authors contributed to data collection, reviewed the draft and approved the final manuscript.
This work was supported by (i) the Instituto de Salud Carlos III through the Red Temática de Investigación Cooperativa en Sida (RD06/006, RD12/0017/0018 and RD16/0002/0006) as part of the Plan Nacional I+D+i and cofinanced by ISCIII-Subdirección General de Evaluación and the Fondo Europeo de Desarrollo Regional (FEDER) and Accion Estrategica en Salud Intramural (PI15CIII/00027).
This work has been presented as Oral Communication at the 17th European AIDS Conference, 6–9 November 2019, Basel, Switzerland.
BA has received conference grants from ViiV Healthcare. ISG has received conference grants or speaker fees from BMS, ViiV Healthcare and Gilead. IJ has received teaching fees from ViiV Healthcare and fees for advisory boards from Gilead. All other authors declare no competing interests.
