Abstract
Background
No published studies have evaluated in pregnant women with HIV weight gain with different antiretroviral drug classes.
Methods
Data from a national cohort study were used. We compared absolute weight gain and occurrence of excessive weight gain in women with HIV who received during pregnancy integrase inhibitors (INSTI), protease inhibitors (PI), or non-nucleoside reverse transcriptase inhibitors (NNRTI). Excessive weight gain was defined according to the Institute of Medicine recommendations. Possible predictors of weight gain were assessed using univariate and multivariate analyses.
Results
Among 273 cases (PI: 191, NNRTI: 43, INSTI: 39), the mean weight increase was 11.3 kg, and 25.4% of the mothers had an excessive weight increase. No significant differences were found among the three treatment groups for absolute weight increase, occurrence of excessive weight gain, infant birthweight, and other pregnancy and laboratory outcomes. The comparisons of individual drugs, although based on a limited number of cases, suggested no major differences. A significant positive correlation was found between weight gain and CD4+ T-cell increase during pregnancy. In multivariate analyses, drug class and nucleoside backbone were not associated with absolute or excessive weight increase. Excessive weight increase was significantly associated with week of delivery (adjusted odds ratio: 1.74, 95% CI 1.15, 2.63), obesity (5.21, 95% CI 1.85, 14.64), overweight (7.95, 95% CI 3.26, 19.39), recent substance use (5.96, 95% CI 1.13, 31.40) and fasting 2nd trimester hyperglycaemia (3.94, 95% CI 1.14, 13.65).
Conclusions
No significant differences in absolute weight change or occurrence of excessive weight gain were found among women with HIV who received during pregnancy different classes of antiretroviral drugs.
Introduction
Recent reports have drawn attention to a possible link between use of integrase inhibitors (or integrase strand transfer inhibitors [INSTI]) and weight gain in individuals with HIV. Some studies have shown a drug-specific effect, with dolutegravir use associated with greater increases in body weight and body mass index (BMI) compared with other antiretroviral drugs [1–5], while others have suggested that this is a class effect common to all integrase inhibitors [6,7]. A recent pooled analysis of eight randomized clinical trials confirmed a greater weight gain with INSTI compared with protease inhibitors (PI) or non-nucleoside reverse transcriptase inhibitors (NNRTI), but also showed for dolutegravir more weight gain compared with elvitegravir [8]. Similar results were shown in a large cohort study conducted in US and Canada [9]. Not all the studies, however, have consistently shown this effect, and some have detected no increased weight gain with dolutegravir [10], or have highlighted that this effect, even if common with INSTI, is not specific for drugs of this class [11], and might represent a component of a multidimensional ‘return to health’, particularly in individuals starting antiretroviral therapy [8,12,13]. In a recent study, switching to elvitegravir-based treatment produced a significantly higher weight gain compared with the weight increase observed during the 48-week interval that preceded the switch, but other factors were also involved, highlighting the possible multifactorial nature of the event [14]. Consistent with this hypothesis, some reports have indicated an additional role for gender, with significant increases in BMI and body weight in women but not in men receiving dolutegravir [15]. Preliminary reports also suggest a potential role of other cofactors, such as ethnicity and nucleoside/nucleotide backbone [13].
Overall, the issue is still debated, and the possible causative mechanisms remain unknown. Importantly, although pregnancy represents an important stress for the metabolic system [16], there are no published data on this topic in pregnant women with HIV. This is an important gap, because gestational weight gain exceeding the current recommendations is associated with adverse outcomes [17] and might therefore further increase the risk of adverse maternal and neonatal outcomes in pregnant women with HIV.
Given the lack of information and the clinical relevance of the issue, we used available data from a national surveillance study of pregnant women with HIV to investigate weight gain with different antiretroviral regimens, with particular reference to integrase inhibitors, evaluating the role of different drug classes and individual drugs involved, and considering an array of possible cofactors.
Methods
Ethics
Patient information sheet and protocol received approval by the competent Ethics Committee (National Institute for Infectious Diseases L. Spallanzani, Rome; ref. deliberation 578/2001, September 28, 2001). The research was conducted in accordance with the Declaration of Helsinki and national and institutional standards.
Study cohort
Data from the Italian National Program on Surveillance on Antiretroviral Treatment in Pregnancy were used. This programme is a national observational study of pregnant women with HIV established in Italy in 2001, structured as an open cohort that includes only pregnant women with HIV followed during pregnancy and postpartum. The programme (currently not funded) was previously supported by public, peer reviewed research grants from the Italian Medicines Agency (AIFA). Prospective enrolment (before pregnancy outcome is known) is recommended, but retrospective reports are also allowed, and represent roughly one-quarter of cases in the programme. The women and infants are followed during routine clinical care, and treatments are decided by the treating physician, usually according to existing guidelines. Laboratory and clinical data are collected from hospital records of Obstetrics, Infectious Diseases, and Pediatrics departments [18].
Population
All the results reported here are based on data extracted from the general database on 2 March 2020, and refer to pregnancies with a date of last menstrual period after 1 January 2008 (that is, after introduction of integrase inhibitors in clinical practice in Italy in December 2007). For the current analysis, inclusion criteria were represented by singleton pregnancy ending in a live birth at term (at least 37 completed weeks of gestational age), antiretroviral treatment ongoing at conception or started during the first trimester of pregnancy, and available information on weight gain during pregnancy. Eligible regimens were those composed of a two-drug nucleoside backbone (any combination of abacavir, lamivudine, emtricitabine and tenofovir, including tenofovir alafenamide [TAF]), plus a third drug represented by dolutegravir, raltegravir or elvitegravir among INSTIs, atazanavir or darunavir among PI (with or without ritonavir or cobicistat), and efavirenz, etravirine or rilpivirine among NNRTIs. Cases with treatment changes during pregnancy were excluded. For women with more than one pregnancy reported (less than 3% in the current analysis), each pregnancy was considered as an individual case.
Covariates
Smoking status and intensity were ascertained at first visit in pregnancy, and classified using a modified version of the criteria by Kotz et al. [19,20]: no smoking was defined by no smoking at all or smoking less than one cigarette per day (CPD), light smoking by one to nine CPD and moderate to heavy smoking by 10 or more CPD. Other covariates (evaluated preconception or at first visit in pregnancy) were CD4+ T-cell count, HIV RNA plasma levels, body mass index (BMI), HIV CDC disease stage, HBV and HCV coinfection, ethnicity/provenance, recent (in the 12 months preceding pregnancy) use of substances (heroin, cocaine, benzodiazepines, methadone, cannabis), alcohol abuse, history of diabetes (as reported by clinicians), of intravenous drug use (IVDU) and of sexually transmitted diseases (STDs: chlamydia, gonorrhoea, genital condylomatosis, genital herpes, syphilis, trichomonas). Alcohol abuse was defined as presence of one or more of the following conditions: an average consumption of more than 35 g per day, inability to perform work, school or family activities because of alcohol consumption, or presence of relational or judicial problems as a consequence of alcohol drinking. According to their BMI at start of pregnancy (calculated on weight at preconception or conception), the women were classified as underweight (BMI <18.5), normal (18.5-24.9), overweight (25.0-29.9) or obese (≥30), and the recommended pregnancy weight gain for each BMI category was defined according the Institute of Medicine (IOM) recommendations for weight gain in pregnancy [21].
Study outcomes
The primary outcome measure was represented by maternal weight gain during pregnancy. Secondary outcomes were represented by: excessive weight gain in pregnancy according to IOM recommendations; weight gain per week of pregnancy (considering entire pregnancy); weight gain greater than 18 kg or greater than 0.400 kg per week; infant birthweight; Z-scores for birthweight adjusted for sex, birth order and gestational age according to national reference standards [22]; non-elective caesarean section (defined by occurrence after the rupture of membranes, the onset of labour or both); fasting glucose levels; macrosomia and low birthweight (birthweight >4,000 g and <2,500 g, respectively). For impaired glucose metabolism, given the variability in screening procedures and diagnostic criteria for gestational diabetes among centres during the long interval considered, we used a restrictive definition that considered among the ADA criteria and those defined in the Italian National guidelines for management of pregnancy [23,24], only fasting plasma glucose levels >92 mg/dl at 20-28 weeks of pregnancy. CD4+ T-cell count change between first and third trimester of pregnancy and third-trimester maternal blood levels of glucose, total cholesterol and triglycerides were also compared among treatment groups.
Statistical analyses
Quantitative variables were summarized as medians with quartiles or means with standard deviations (
In order to control simultaneously the impact of different cofactors potentially involved in determining weight gain, multivariable linear and logistic regression analyses were modelled. The dependent (outcome) variables selected were absolute weight gain in pregnancy for the linear regression model and excessive weight gain in pregnancy according to IOM criteria for the logistic regression model. Some variables were included a priori as independent (predictive) variables in all models: treatment group (INSTI, NNRTI, PI), nucleoside backbone (abacavir/lamivudine and tenofovir/ emtricitabine), BMI category, age, presence of a 2nd trimester fasting glucose >92 mg/dl, antiretroviral status at conception, ethnicity, week of delivery. Additional variables (such as smoking, low CD4+ T-cell count, recent substance use, coinfections, HIV clinical stage) were conditionally included in the models, based on their capacity to increase the fitness of the model, measured through the models’ R 2 score values calculated with and without the variables of interest. Sensitivity analyses were conducted to explore the possible presence of interactions between ARV status at conception and BMI in determining excessive weight gain. For the comparison of birthweight and birthweight Z-scores by nucleoside backbone, a sensitivity analysis was performed that excluded from the tenofovir/emtricitabine group women who received TAF. For all analyses P-values below 0.05 were considered statistically significant. All analyses were performed using the SPSS software, version 25.0 (IBM Corp, 2017, Armonk, NY, USA).
Results
Between January 2008 and March 2020, 1,619 pregnancies with known outcome were reported from 34 centres. Following exclusion of 259 pregnancies not ending in a singleton live birth, 220 pregnancies with preterm delivery, 299 pregnancies with ART started after first trimester of pregnancy, 180 pregnancies with unknown weight increase in pregnancy, and 388 pregnancies not eligible because of antiretroviral treatment criteria, 273 pregnancies were considered for all analyses (Figure 1).

Population flowchart
The general characteristics of the three treatment groups at entry in pregnancy are shown in Table 1. Overall, the three treatment groups had similar demographics, clinical history and immunological status, with only a few differences observed in the pairwise comparisons between groups. Women who received NNRTI were more commonly virally suppressed at entry in pregnancy and more commonly HBV-coinfected. Women on INSTI had better conditions in terms of asymptomatic HIV disease, but the median CD4+ T-cell counts in the three groups were similar. No women in the entire study sample had history of AIDS-defining clinical illnesses, and only a small proportion had less than 200 CD4+ T-cells/mm3, indicating overall good clinical and immunological status, with no relevant differences among treatment groups. Most importantly, BMI values were very similar in the three groups, and the distribution of the women in the different body weight categories according to BMI was also similar. The main significant differences among the three treatment groups were observed in the distribution of the nucleoside backbones, probably refecting the composition of fixed-drug combinations commercially available for treatment. In particular, almost all the women on NNRTI (97.7%) were receiving a backbone of emtricitabine/tenofovir, while in the other two groups this backbone, although still predominant (56.4% in regimens with INSTI and 76.4% in regimens with PIs), was also accompanied by a relatively frequent use of abacavir plus lamivudine (41.0% with INSTI and 22.0% with PIs). Other backbones were markedly less common (less than 3% in all groups).
Clinical and demographic information
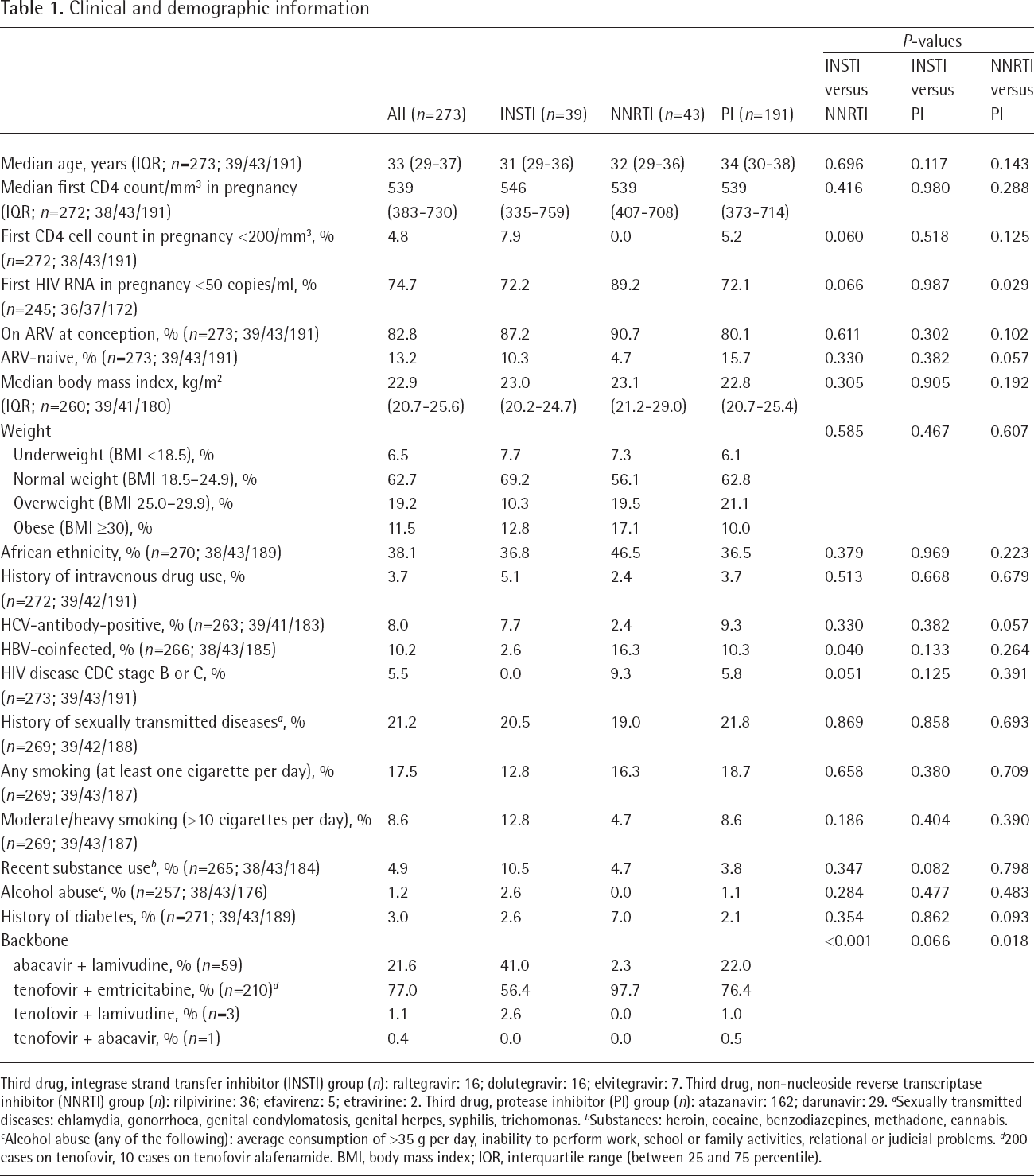
Third drug, integrase strand transfer inhibitor (INSTI) group (n): raltegravir: 16; dolutegravir: 16; elvitegravir: 7. Third drug, non-nucleoside reverse transcriptase inhibitor (NNRTI) group (n): rilpivirine: 36; efavirenz: 5; etravirine: 2. Third drug, protease inhibitor (PI) group (n): atazanavir: 162; darunavir: 29.
Sexually transmitted diseases: chlamydia, gonorrhoea, genital condylomatosis, genital herpes, syphilis, trichomonas.
Substances: heroin, cocaine, benzodiazepines, methadone, cannabis.
Alcohol abuse (any of the following): average consumption of >35 g per day, inability to perform work, school or family activities, relational or judicial problems.
200 cases on tenofovir, 10 cases on tenofovir alafenamide. BMI, body mass index; IQR, interquartile range (between 25 and 75 percentile).
The main study outcomes are reported in Table 2. Overall, the mean increase in weight during pregnancy was 11.3 kg, for a mean increase of 0.294 kg per week; roughly one-quarter of the women (25.4%) had an excessive weight increase during pregnancy according to the IOM recommendations, 21.2% had an increase greater than 0.400 kg per week and 7.7% gained more than 18 kg. No significant differences were observed among the three treatment groups for all the above measures. The rates of non-elective caesarean section were also similar in the three groups. Even if women with preterm delivery were excluded from analysis, the mean duration of pregnancy was slightly but significantly shorter in the PI group (38.2 weeks compared with 38.6 weeks in the other two groups). No significant differences were however observed in infant birthweight and birthweight Z score (adjusted for sex, gestational age at birth and birth order), although low birthweight was more common among infants born to women on NNRTIs compared with the other two groups. The frequency of macrosomia was low, with no differences among treatment groups, and no differences were also observed for the rates of fasting glucose >92 mg/dl and for plasma levels of glucose, cholesterol and triglycerides (Table 2).
Main study outcomes
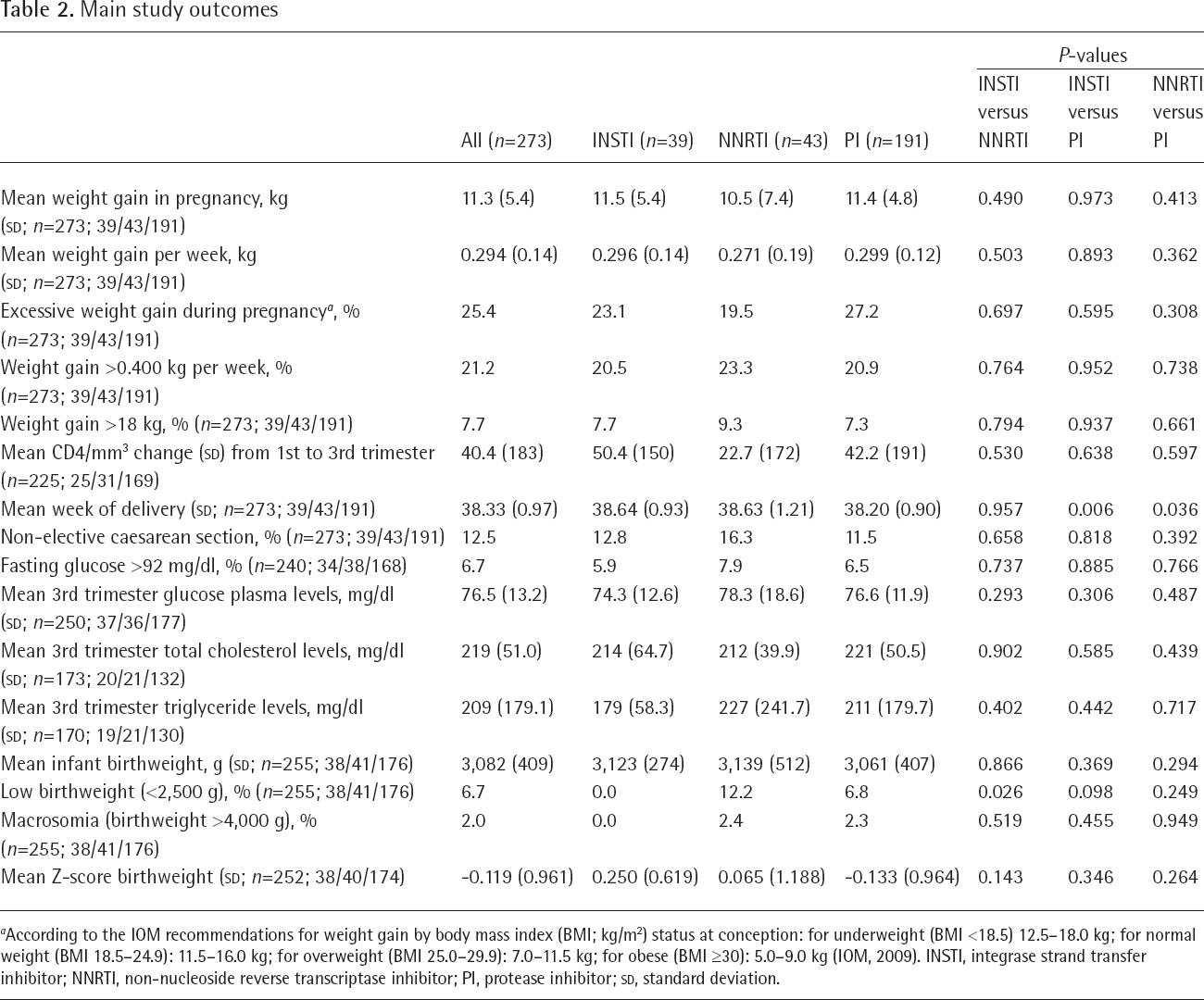
According to the IOM recommendations for weight gain by body mass index (BMI; kg/m2) status at conception: for underweight (BMI <18.5) 12.5–18.0 kg; for normal weight (BMI 18.5–24.9): 11.5–16.0 kg; for overweight (BMI 25.0–29.9): 7.0–11.5 kg; for obese (BMI ≥30): 5.0–9.0 kg (IOM, 2009). INSTI, integrase strand transfer inhibitor; NNRTI, non-nucleoside reverse transcriptase inhibitor; PI, protease inhibitor;
In order to assess with available data the presence of a possible ‘return to health effect’ contributing to weight gain in pregnancy, we first compared the extent of weight increase between women starting treatment in pregnancy (who were more likely to benefit from such effect) and women already on treatment at conception, and then explored in correlational analyses a possible association between weight gain and CD4+ T-cell change during pregnancy. Both absolute and weekly weight increase were not significantly different between women starting treatment in pregnancy and women already on treatment at conception (10.8 versus 11.4 kg, and 0.281 versus 0.297 kg per week, P-values: 0.534 and 0.457, respectively). Weight increase in pregnancy and CD4+ T-cell gain between first and third trimester were however reciprocally correlated, with a low strength of the linear relationship in the entire group (Pearson correlation coefficient 0.213; P=0.001), and a stronger strength in the subgroup of women who started treatment during first trimester (Pearson correlation coefficient 0.427; P=0.004). Similar findings were observed for the association between CD4+ T-cell change and weight gain per week (data not shown).
We then explored the main maternal and infant weight outcomes by individual drugs. No formal testing was performed to compare individual drug groups, because the number of cases in some groups were very low. The data, reported in Table 3, suggest however no major differences among individual drugs. The greatest increases in weight gain (excluding the etravirine group, that had two cases only) were observed with raltegravir and atazanavir (mean increases: 11.8 and 11.6 kg, respectively). The same outcomes were assessed according to the different nucleoside backbones that comprise the regimens. In this case the presence of two main groups (abacavir/lamivudine and tenofovir/emtricitabine) with larger numbers of cases allowed to explore statistical significance. The comparisons showed a trend for lower mean birthweight and birthweight Z-score with abacavir/lamivudine compared with tenofovir/ emtrictabine (weight difference: -113 g; 95% CI of the difference: from -233 to +7; P=0.065; score difference: -0.245; 95% CI of the difference: from -0.52 to +0.04; P=0.089). This trend was confirmed when women on TAF were excluded from the tenofovir group in sensitivity analyses (data not shown). All the other measures appeared to be similar between the two groups (all P-values >0.3, numbers reported in Table 3).
Weight gain in pregnancy and infant birthweight by individual drugs and nucleoside backbones
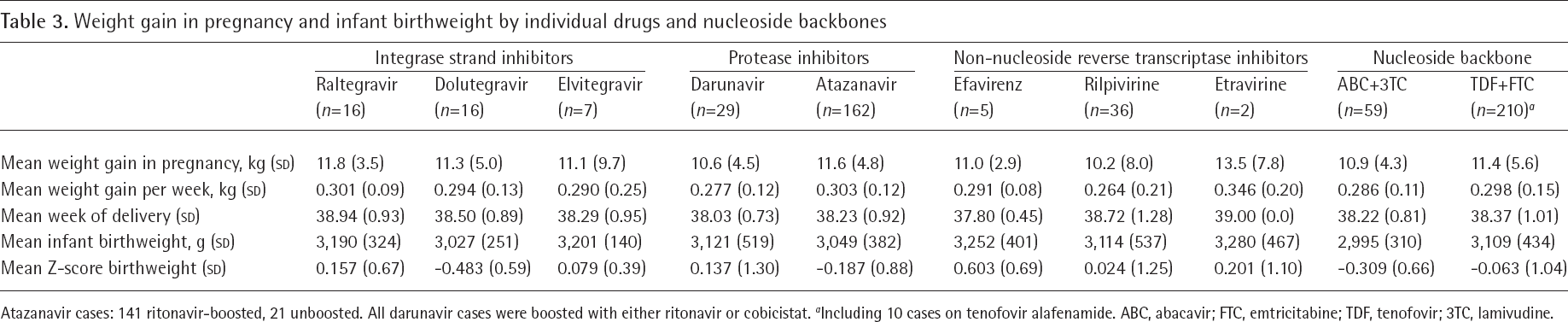
Atazanavir cases: 141 ritonavir-boosted, 21 unboosted. All darunavir cases were boosted with either ritonavir or cobicistat.
Including 10 cases on tenofovir alafenamide. ABC, abacavir; FTC, emtricitabine; TDF, tenofovir; 3TC, lamivudine.
Finally, we assessed the independent role of potential predictors of weight gain in pregnancy using multivariate analyses that adjusted for different cofactors potentially involved. The result of this multivariate analyses are shown in Table 4. In both analyses both treatment group (INSTI, NNRTI and PI) and nucleoside backbone (abacavir/lamivudine and tenofovir/emtricitabine) were not associated with weight increase, confirming the results of univariate analyses. Other factors were however associated with the two outcomes. In the linear regression model, absolute weight increase was significantly associated with week of delivery (positive association), African ethnicity (inverse association), and obesity (negative association). In the logistic regression model, excessive weight increase was significantly and positively associated with week of delivery (adjusted odds ratio [AOR] 1.74, 95% CI 1.15, 2.63), obesity (AOR 5.21, 95% CI 1.85, 14.64), overweight (AOR 7.95, 95% CI 3.26, 19.39), recent substance use (AOR 5.95, 95% CI 1.13, 31.40), and fasting 2nd trimester glucose levels >92 mg/dl (AOR 3.94, 95% CI 1.14, 13.65). The discordant direction of the effect of obesity in the two analyses was further defined. Overall, obese women had a lower weight increase in pregnancy compared with non-obese women (mean values: 7.0 versus 11.8 kg; P<0.001), but this increase was still above the IOM-recommended weight increase in a significantly higher proportion of cases (43.3% of obese women versus 23.0% of non-obese women; P=0.019). In the multivariable analysis of excessive weight gain, sensitivity analyses showed no significant impact of adding the interactions between ARV status at conception and BMI as model covariates, considering BMI both as a continuous variable or as a categorical variable (all P-values >0.35). Their inclusion in the model also did not modify the direction or the level of the other associations found (data not shown).
Multivariable analysis of possible determinants of absolute and excessive weight gain in pregnancy according to the IOM recommendations

AOR, adjusted odds ratio; IOM, Institute of Medicine.
Discussion
We evaluated weight gain in pregnant women with HIV receiving different classes of antiretroviral drugs. Our findings suggest no major differences among the three major drug classes of integrase inhibitors, PIs and NNRTIs, and between the two main nucleoside backbones currently used, represented by tenofovir/emtricit-abine and abacavir/lamivudine. Our data on individual drugs also showed similar weight increases for the main drugs studied, but numbers were more limited, and larger studies are therefore necessary.
An adequate weight control in pregnancy is particularly important in order to prevent adverse outcomes, and therefore is subject to specific recommendations that define optimal weight increase differentially, according to body mass index status at entry in pregnancy. We therefore considered, as study outcomes, not only absolute body weight increase, but also excessive weight increase individually defined according to the baseline BMI status (underweight, normal, overweight and obese). The results of such analyses also showed no differences among drug classes in the proportion of women who had excessive weight gain according to the IOM recommendations.
Finally, in order to consider potential cofactors, we performed multivariable regression analyses that adjusted for an array of covariates, including age, week of delivery, ethnicity, recent substance use, CD4+ T-cell count levels, smoking, hyperglycaemia and timing of starting of ARV in pregnancy. In these analyses, as for univariate analyses, drug class and type of nucleoside backbone, even after adjustment for cofactors, were not related to absolute weight gain or excessive weight gain.
Overall, all the analyses consistently suggest similar weight increase with the three major drug classes and for the two major backbones used in antiretroviral therapy, and may help clinicians who might be reluctant to introduce or maintain drugs that might exacerbate or induce overweight and obesity during pregnancy. Based on these findings, concerns about differential weight increase should not be a primary factor in the selection of optimal treatments in pregnancy.
We cannot conclude with the same confidence on individual drugs, given the small number of cases in each drug group. However, we observed limited differences in weight gain among individual drugs. Large samples are needed to detect as significant small differences. With 200 cases, at standard levels of power (0.8) and significance (0.05), and using as reference a 10.0 kg weight increase, roughly a 2 kg difference in weight increase can be detected as significant in pairwise comparisons. Most likely, multicentre studies are therefore needed to adequately define this issue.
Literature results on a possible drug-specific effect, particularly for dolutegravir, are not consistent, and the issue remains open [1–10]. Possible insights could be provided by more precise evaluations of body composition. Two recent studies in non-pregnant populations evaluated fat mass changes in patients switching to dolutegravir-based regimens. In a first study, patients switching to dual therapy with either dolutegravir/rilpivirine or darunavir/lamivudine had overall similar fat mass changes, with baseline fat mass and baseline CD4+ T-cell counts representing the only predictors of fat mass gain [25]. In another study, patients switched to lamivudine/dolutegravir or lamivudine/atazanavir showed non-significant fat mass and fat percentage increases, and no predictors were found in a multivariable analysis that adjusted for age, sex, HIV risk factor, years of ARV exposure and CD4+ nadir T-cell count [26].
Both the above studies indicated some weight and fat increase irrespective of the regimen used. Despite the very limited sample size, the reported significant role of baseline CD4+ T-cell levels in predicting fat mass change [25] is consistent with the hypothesis that weight change represents a marker of immune recovery and ‘return to health’ and not a drug-specific effect [8,11–13].
We indirectly evaluated this issue examining the correlation between the individual weight gain and the individual CD4+ T-cell gain during pregnancy in the entire population of pregnant women and in the subgroup of women who started antiretroviral treatment in pregnancy, that were most likely to benefit of the possible return to health effect. Our finding of a significant positive association between the above two measures, that had greater strength (despite the smaller numbers) in the subgroup of women who had no antiretroviral experience before pregnancy, significantly supports this hypothesis.
Although different treatments were not significantly associated with absolute and excessive weight gain in pregnancy, our study identified other predictors of these outcomes in multivariable analyses that adjusted for confounders. As expected, any additional week of gestation after week 37 was significantly associated with absolute weight gain and with excessive weight gain, confirming the importance of controlling weight gain also in the last weeks preceding delivery.
The other main determinants of weight gain appeared to be behavioural, possibly driven by social or cultural factors. Aside from duration of pregnancy, risk factors for excessive weight gain were overweight, obesity, recent substance use, hyperglycaemia and (with an inverse association), smoking.
Cultural differences are likely to play an important role in weight gain [27] and could explain the differences that we found related to ethnicity. Prepregnancy BMI, with an increased risk for overweight and obese women, has already been described as a significant predictor of total gestational weight gain [28]. Another potentially relevant factor is represented by smoking. In our study women who smoked appeared to have significantly lower risk of gaining excessive weight during pregnancy. This finding is consistent with the association between smoking and lower maternal weight already described [29], and indirectly confirms that particular attention should be paid to a potential excessive weight increase in women who quit smoking during pregnancy in order to prevent the well-known and important negative consequences of smoking on pregnancy outcomes, intrauterine growth and infant birthweight [30].
Other social and psychological factors may significantly affect weight gain: income level may represent a determinant of weight gain in pregnancy [31], and significant direct associations have been described between psychosocial factors and eating disorders in pregnancy [32]. Although we were not able to perform a full evaluation of socioeconomic and psychological variables, we found that substance use was associated with excessive weight gain. We can only speculate about the potentially involved mechanisms: substance use might represent a marker of associated eating disorders, or a proxy for less frequent access to health services, with lower chances to monitor and control more effectively weight gain in pregnancy. We were unfortunately unable to assess these hypotheses but this is an important issue and further studies should better define the relationships between psychosocial factors, disordered eating and weight gain in pregnancy.
Our data confirms that overweight and obese women are at higher risk of excessive weight gain during pregnancy. However, our findings also indicate some partially effective attempts to control weight gain in pregnancy: obese women, in particular, although more likely to have excessive weight gain with respect to the IOM recommendations, nonetheless had lower absolute weight gain compared with other groups, even after adjusting for confounders. These findings might indicate partial success in pursuing weight control in obese women, that represent the group at higher risk of complications.
High fasting levels of glucose at second trimester were also associated with excessive weight gain. This finding reinforces the need to prevent hyperglycaemia in early pregnancy and the necessity of closely monitoring women with second trimester hyperglycaemia not only for glucose abnormalities but also for weight gain.
A possible limitation of our study is represented by the marked selection of cases from the initial sample. The exclusions performed were however necessary in order to make appropriate comparisons: we had to exclude cases with preterm delivery because of the impact of the shorter duration of pregnancy on weight gain. Our conclusions therefore apply only to term pregnancies. For similar reasons we had to exclude cases with twin or triple pregnancy. A vast group of the cases were finally excluded because of mixed use of multiple classes during pregnancy, drug class change during pregnancy or late start of treatment. The exclusion of women who changed treatment, in particular, has significantly reduced sample size and may have precluded the possibility to evaluate specific effects in particular subgroups (such as weight gain in women switching to INSTI from other drug classes during pregnancy), or particular outcomes (such as changing treatment for weight-related reasons). Although neither obesity nor excessive weight gain were ever reported as a cause for switching treatment in pregnancy in our cohort, we also cannot exclude that this concern may have contributed to switching among classes, together with other reasons. Randomized studies might represent a more appropriate tool to investigate such issues without the multiple potential sources of bias that affect observational studies. Our study also evaluated weight gain and its consequences in a population of women living in Europe. As such, the conclusions might not be entirely applicable to other contexts where rates of obesity and gestational diabetes are significantly different in the general population. Finally, the numbers in the treatment groups were not balanced. This refected the pattern of use in clinical practice, with a predominant use of PIs compared with the other classes, and a predominant use of atazanavir compared with darunavir. The small number of cases for some groups has represented a limitation in the assessment of differences among individual drugs.
In summary, we reported for the first time weight gain in pregnancy in a common setting of clinical practice, finding no significant differences in absolute weight change or excessive weight gain among pregnant women with HIV treated with the currently used anti-retroviral regimens. These findings partially fill a gap of knowledge on this matter and may help clinicians possibly concerned about excessive weight gain in pregnancy with some drug classes. Further studies are still needed in both pregnant and non-pregnant populations.
Footnotes
Acknowledgements
We thank Cosimo Polizzi and Alessandra Mattei, from Istituto Superiore di Sanità, Rome, Italy, for providing technical secretarial for this study. We also thank Ernesto Costabile, from Istituto Superiore di Sanità, Rome, Italy, for providing assistance as documentalist. No compensation was received for these contributions. This work (currently not funded) was supported in the past by public research grants (grant ref.: H85E08000200005) from the Italian Medicines Agency (AIFA). The funder had no role in study design, data collection, data analysis, manuscript preparation and/or publication decision.
None to declare. None of the authors has a commercial or other association, financial interest, activity, relationship or association that might pose a conflict of interest. The corresponding author had full access to all the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis.
