Abstract
Background
Researchers must often rely on creatinine measurements to assess kidney function because direct glomerular filtration rates (GFR) and cystatin-c are rarely measured in routine clinical settings. However, HIV treatments often include dolutegravir, raltegravir, rilpivirine or cobicistat, which inhibit the proximal tubular secretion of creatinine without impairing kidney function, thus leading to measurement bias when using creatinine-based estimated GFR (eGFR). We developed eGFR correction factors to account for this potential bias.
Methods
11,359 treatment-naive HIV-positive individuals in OPERA were included if they initiated dolutegravir, elvitegravir/cobicistat, darunavir/cobicistat, raltegravir, rilpivirine or efavirenz (control) with an eGFR >60 ml/min/1.73 m2. The eGFR was corrected by adding the median decrease reported in the literature to the calculated eGFR; correction factors were not validated. Incidence rates of eGFR <60 ml/min/1.73 m2 (Poisson regression) and the relationship between regimens and eGFR <60 ml/min/1.73 m2 (multivariate Cox proportional hazards models) were estimated with and without eGFR correction.
Results
Without eGFR correction, dolutegravir, elvite-gravir/cobicistat, darunavir/cobicistat, raltegravir and rilpivirine based regimens were statistically significantly associated with a higher likelihood of eGFR <60 ml/min/1.73 m2 than efavirenz. With eGFR correction, each of these regimens was associated with a statistically significantly lower likelihood of eGFR <60 ml/min/1.73 m2 compared with efavirenz.
Conclusions
With increasing use of agents that inhibit tubular creatinine secretion, artificially low eGFR values could lead to erroneous conclusions in studies of HIV treatment and kidney outcomes measured with creatinine-based eGFR equations. Sensitivity analyses assessing the potential magnitude of bias arising from creatinine secretion inhibition should be performed.
Introduction
Researchers must often rely on creatinine measurements to assess kidney function because direct glomerular filtration rate (GFR) measures (for example, urinary clearance of inulin, plasma clearance of iohexol) and cystatin-c-based estimated GFR (eGFR) are rarely available in routine clinical settings. However, dolutegravir (DTG), raltegravir (RAL), rilpivirine (RPV) and cobicistat (COBI, c) are commonly used in HIV antiretroviral therapy (ART) and have been shown to inhibit proximal tubular secretion of creatinine in early-phase clinical trials [1–13] or in vitro [14].
Renal elimination of creatinine mainly occurs passively by glomerular filtration, but 10–15% of creatinine is actively secreted by proximal tubular cells [15]. The inhibition of creatinine secretion results in rapid, non-progressive changes in serum creatinine that do not refect functional kidney injury, leading to artificially low eGFR [6,15,16]. In the absence of kidney injury, a 10–20% decrease in eGFR can be expected within 4 weeks of treatment initiation due to tubular secretion inhibition, followed by a stabilization of eGFR [17]. This may lead to measurement bias when assessing kidney outcomes using the eGFR. We therefore aimed to develop eGFR correction factors to quantify the potential bias introduced when using creatinine-based eGFR equations to assess kidney function.
Methods
The Observational Pharmaco-Epidemiology Research and Analysis (OPERA) cohort is a database of electronic medical records from 85 clinics across the US. ART-naive people living with HIV (PLWH) were included if they initiated ART with either DTG, elvite-gravir (EVG)/c, darunavir (DRV)/c, RAL, RPV, or efavirenz (EFV, control) with an eGFR >60 ml/min/1.73 m2 within 3 months before ART initiation and at least one eGFR during follow-up. The initial ART regimen, initiated between 1 January 2009 and 31 December 2018, included no more than one of these core agents. Person-time was censored at core agent/pharmacoenhancer change, 12 months after the last clinical contact, death or 31 August 2019. DTG, EVG/c, DRV/c, RAL and RPV were selected because they are known to inhibit creatinine secretion [1–14]; EFV was selected because it has no known impact on creatinine secretion [18].
GFR was estimated with the CKD-EPI equation, which is recommended as the most accurate creatinine-based eGFR equation for PLWH [19]. eGFR correction was performed by adding the median eGFR decrease reported in the literature (correction factor) to the calculated eGFR (Table 1) [1–9]. Correction factors have yet to be validated.
eGFR correction factors

ARV, antiretroviral; eGFR, estimated glomerular filtration rate.
Incidence rates (IR) of eGFR <60 ml/min/1.73 m2 after ART initiation were estimated for each regimen with and without eGFR correction using univariate Poisson regression. The relationship between regimens and eGFR <60 ml/min/1.73 m2 was assessed using multivariate Cox proportional hazards models adjusted for baseline age, sex, race, HIV viral load, tenofovir disoproxil fumarate (TDF) use, non-steroidal anti-inflammatory drug (NSAID) use, sulfamethoxazole/trimethoprim use, hypertension, diabetes, hepatitis C (HCV) coinfection and uncorrected baseline eGFR. Age, viral load and eGFR were modelled with restricted cubic splines.
The OPERA database complies with all HIPAA and HITECH requirements, which expand upon the ethical principles detailed in the 1964 Declaration of Helsinki. The OPERA database received annual institutional review board (IRB) approval by Advarra IRB including a waiver of informed consent and authorization for use of protected health information.
Results
A total of 11,359 PLWH initiating DTG (27%), EVG/c (36%), DRV/c (4%), RAL (4%), RPV (12%) or EFV (16%) were identified (Table 2). TDF and tenofovir alafenamide (TAF) were included in 53% and 27% of regimens, respectively, and 19% were prescribed sulfamethoxazole/trimethoprim. Traditional kidney disease risk factors were infrequent. The median baseline eGFR was 113.4 ml/min/1.73 m2 (IQR: 99.2-125.4).
Baseline demographic and clinical characteristics (n=11,359)

ART, antiretroviral therapy; eGFR, estimated glomerular filtration rate; NSAID, non-steroidal anti-inflammatory drug.
As expected, fewer cases of eGFR <60 ml/min/1.73 m2 were identified with the corrected eGFR, and the difference between the uncorrected and corrected IRs was greater for agents known to have a bigger impact on tubular creatinine secretion (Figure 1A). For example, the IR of eGFR <60 ml/min/1.73 m2 with DTG went from 28.6 events per 1,000 person-years (95% CI: 24.8, 33.1) without correction to 5.1 per 1,000 person-years (95% CI: 3.6, 7.1) with correction (eGFR +17). With RAL, the IR went from 34.5 events per 1,000 person-years (95% CI: 24.7, 48.3) without correction to 14.9 per 1,000 person-years (95% CI: 9.0, 24.6) with correction (eGFR +9).
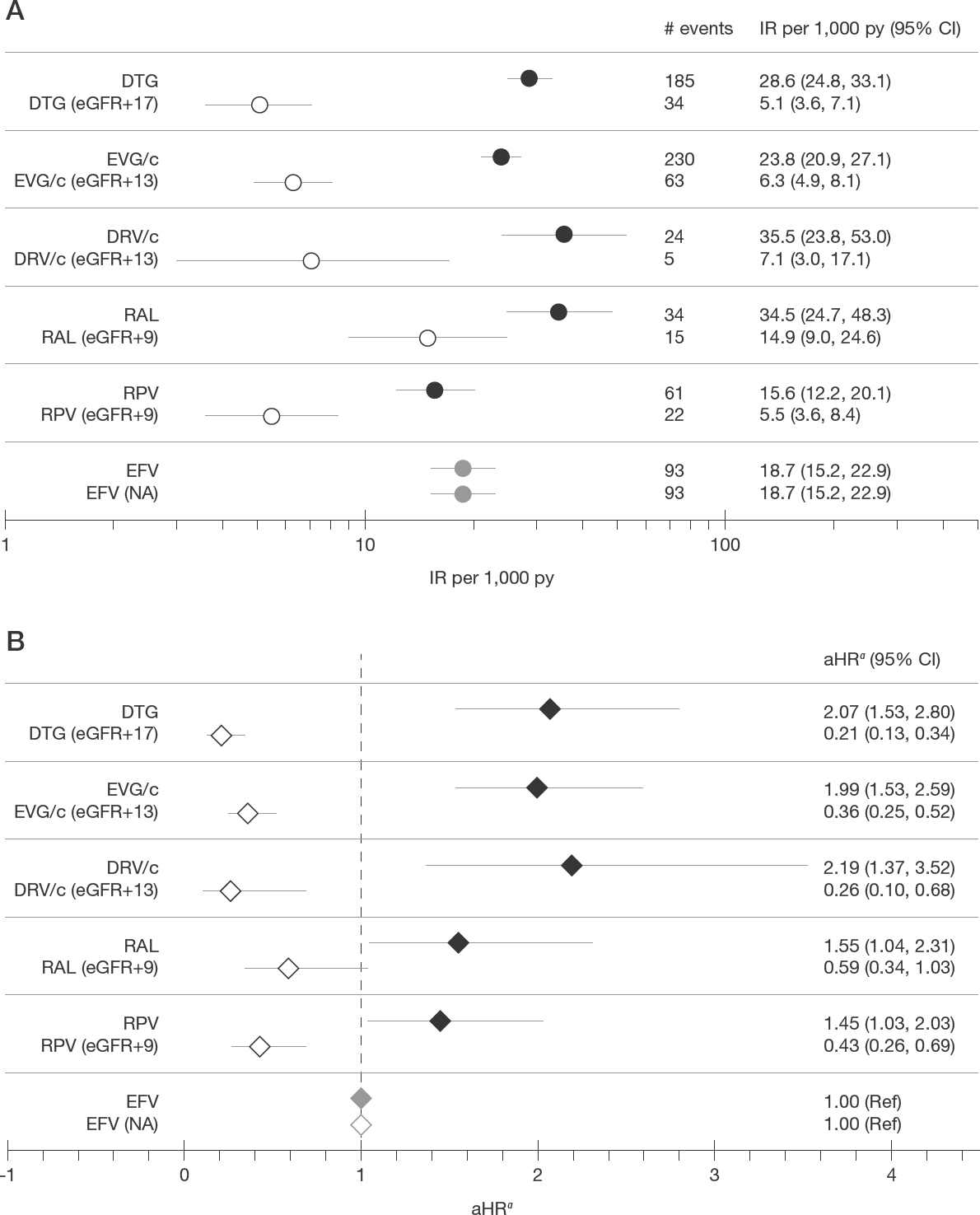
Impact of eGFR correction on incidence rates of eGFR <60 ml/min/1.73 m2 and the association between regimens and eGFR <60 ml/min/1.73 m2
eGFR correction had both a quantitative and qualitative impact on the association between regimen and eGFR <60 ml/min/1.73 m2 (Figure 1B). Without eGFR correction, all regimens were associated with a statistically significant increased likelihood of eGFR <60 ml/min/1.73 m2 compared with EFV; after eGFR correction, DTG, EVG/c, DRV/c and RPV all became statistically significantly associated with a lower likelihood of eGFR <60 ml/min/1.73 m2 compared with EFV. For example, before correction, the likelihood of eGFR <60 ml/min/1.73 m2 with DTG, EVG/c and DRV/c was about twice that of EFV. After eGFR correction, they were associated with a 64–79% lower likelihood of eGFR <60 ml/min/1.73 m2 compared with EFV.
Discussion
The inhibition of proximal tubular secretion of creatinine with common ART agents (DTG, COBI, RAL, RPV) can falsely reduce eGFR levels below clinically important cutoffs, thus leading to misclassification of kidney disease outcomes or stage when using creatinine-based eGFR equations. Correcting for the artificial lowering of eGFR had a substantial impact on the incidence of eGFR <60 ml/min/1.73 m2 and on the association with ART regimen. This suggests that many of the events observed without eGFR correction could have been driven by the inhibition of creatinine secretion and therefore might not be of clinical concern. This is important because direct GFR measures are cumbersome and are rarely performed in the routine clinical setting. Cystatin-c measurements, which are not affected by tubular secretion, are also uncommon. Thus, GFR must often be estimated using serum creatinine, both in clinical settings and in observational studies.
Of note, a greater reduction in the incidence of eGFR <60 ml/min/1.73 m2 was observed after eGFR correction for RPV than for RAL, despite using the same correction factor (eGFR +9). This is likely due to the use of a dichotomous outcome: twice as many PLWH who developed eGFR <60 ml/min/1.73 m2 had uncorrected eGFRs ≥51 ml/min/1.73 m2 in the RPV group than in the RAL group. Therefore, applying the same eGFR correction factor had a bigger impact for RPV than RAL.
Inhibition of the tubular secretion of creatinine with DTG, RPV, COBI and RAL has been demonstrated in clinical trials and in vitro [1–14]. One observational study has also quantified the magnitude of the tubular secretion of creatinine among treatment-experienced PLWH switching to a two-drug regimen containing either DTG, COBI, DTG+RPV, DTG+COBI or a control group (no known inhibitors of creatinine tubular secretion, although 73% included RAL). Despite incomplete statistical adjustments, statistically significant decreases in creatinine-based eGFR were observed with all DTG-containing regimens (-8.4 to -8.6 ml/min/1.73 m2), but not with COBI alone. No proteinuria or cystatin c-based eGFR alterations were observed, confirming that such decreases in creatinine-based eGFR did not refect kidney damage [20].
eGFR correction is an important tool to assess the potential bias introduced by differential inhibition of creatinine secretion when assessing the relationship between ART regimens and kidney outcomes using clinical data. However, the correction factors proposed here should only be considered as a sensitivity analysis, as they are formulated from estimates published in clinical trials [1–9] and have not been validated. Over-or under-correction cannot be excluded. Despite the absence of nephrotoxicity, EFV was associated with an increased risk of corrected eGFR <60 ml/min/1.73 m2 compared with other regimens, which may suggest overcorrection or residual confounding. While statistical adjustments were performed for important con-founders including TDF, hypertension and diabetes, this study did not control for the use of probenecid or creatine supplements.
Moreover, this correction method does not account for the uncertainty around the exact level of inhibition experienced by patients on different regimens, which would have required estimates of sensitivity and specificity currently unavailable in the literature [21]. Such validation was impossible here, as OPERA is based on electronic medical records and direct GFR measurement is rarely performed in routine clinical care. Finally, correction factors were not developed or applied to other common drugs inhibiting the proximal tubular secretion of creatinine (for example, cimetidine, corticosteroids, salicylates, trimethoprim). While the proposed eGFR correction method does not refect all the complexity of tubular creatinine secretion inhibition processes, the results do illustrate the potential extent of bias associated with uncorrected eGFR. More research is required to improve eGFR measurement correction methods when agents inhibiting creatinine tubular secretion are used.
To the best of our knowledge, this is the first attempt to account for the impact of ART agents inhibiting tubular secretion of creatinine using eGFR correction factors. This study was performed using data from a large cohort representative of PLWH in care in the US and included a large number of ART-naive PLWH initiating each ARV known to inhibit creatinine secretion (DTG, COBI, RAL, RPV), as well as a control group initiating an ARV without a known impact on creatinine secretion (EFV). This study illustrates the potential impact of tubular creatinine secretion inhibition on unadjusted IRs and adjusted measures of association.
In conclusion, we demonstrated that using creatinine-based eGFR equations among PLWH taking agents associated with the inhibition of creatinine secretion can lead to incorrect assumptions regarding incident kidney impairment. In the absence of direct GFR or cystatin-c measurement in routine clinical care, sensitivity analyses assessing the potential magnitude of bias arising from creatinine secretion inhibition should always be performed to avoid drawing erroneous conclusions. Cohorts with both measured and estimated GFR data are encouraged to validate and refine the correction method employed here.
Footnotes
Acknowledgements
This research wouldn't be possible without the participation of people living with HIV and their caregivers. Additionally, we are grateful for the following contributions: Robin Beckerman (SAS programming, Epividian, Durham, NC, USA), Jeff Briney (QA/QC, Epividian, Durham, NC, USA), Rodney Mood (site selection and support, Epividian, Durham, NC, USA), Bernie Stooks (database support, Epividian, Durham, NC, USA), Judy Johnson (categorization of clinical terminology, Epividian, Durham, NC, USA) and Jill Poliskey (categorization of clinical terminology, Epividian, Durham, NC, USA). Support for this study was provided by Epividian, Inc. Parts of the data were presented at the 23rd International Workshop on HIV Observational Databases (IWHOD), Athens, Greece, 28–30 March 2019.
LB, JF and GF are employed by Epividian, Inc.; Epividian has had research funded by ViiV Healthcare, Merck & Co. and Janssen Pharmaceutica. CW is a consultant for Epividian, Inc. RH has received a research grant from Gilead, speaker honoraria and advisory boards from ViiV Healthcare, BMS, Merck, Gilead Sciences and Janssen, and advisory board participation of Epividian. KM has received research grants from Gilead Sciences, Merck, Janssen and GSK/ViiV Healthcare, and honoraria for speakers bureau and advisory boards from Gilead Sciences, Merck, Janssen and GSK/ViiV Healthcare; and advisory board participation with Epividian.
