Abstract
Background
Viral suppression in patients on antiretroviral treatment (ART) is critical to reducing HIV transmission and HIV-related mortality. Although many studies have evaluated factors associated with viral suppression, few have assessed the extent to which missing viral load data may bias results.
Methods
We included data on all patients starting ART from 2005 to 2019 in eight South African cohorts participating in the International epidemiology Databases to Evaluate AIDS (IeDEA) collaboration. Multivariable logistic regression models were used to determine factors associated with having a viral load measurement within 2 months of a scheduled testing date and having a viral load <400 RNA copies/ml (‘viral suppression’). In a sensitivity analysis, missing viral loads were imputed based on patients’ clinical and demographic characteristics and outcomes.
Results
Viral load tests were scheduled in 603,549 and 77,423 intervals in adults and children, respectively, but test results were recorded in only 40.7% and 41.2%, respectively. The proportion of recorded results suppressed was 85.7% in adults and 72.4% in children. After imputation of missing viral load measurements, viral suppression reduced slightly in adults (85.3%) and increased in children (73.2%). Predictors of virological suppression in adults, which included female sex, older age, higher baseline CD4+ T-cell count and recent testing year, were similar in the main analysis and after imputing missing viral loads.
Conclusions
Although viral load information was frequently missing in the South African setting, estimates of viral suppression and predictors of viral suppression did not change substantially after adjusting for missing data.
Introduction
Monitoring of viral suppression in HIV-positive patients on antiretroviral treatment (ART) is important both for optimizing patient health and for reducing risk of HIV transmission. Mortality and morbidity rates in ART patients are critically dependent on viral suppression [1], and viral suppression in ART patients is critical to reducing HIV incidence at a population level [2,3]. The importance of monitoring viral suppression is recognized in the UNAIDS 90-90-90 targets, which aim to achieve a 90% level of diagnosis in people living with HIV by 2020, with 90% of those diagnosed on ART and 90% of those on ART virally suppressed [4]. In 2018, an estimated 86% of all ART patients globally were estimated to have viral loads of less than 1,000 RNA copies/ml [5]. Knowledge of the factors associated with lack of viral suppression is critical to developing interventions to improve suppression, and many previous studies have evaluated these factors [6–9].
However, a major challenge in estimating rates of viral suppression is that data are typically missing for a substantial proportion of patients. Viral load testing coverage (the proportion of ART patients with viral load measurements in a specified period) is often low: the number of countries that have testing coverage of at least 50% increased from only 26 in 2015 to 76 in 2018 (out of 193 countries) [5]. For 35% of all ART patients in 2018, information on viral suppression had to be imputed using regional averages, due to low (<50%) testing coverage [5]. Even in the countries in which reported viral load testing coverage was greater than 50%, substantial bias could exist in reported rates of viral suppression. Although countries are expected to report only on routine viral load testing results, many countries have historically taken a ‘targeted’ approach to viral load testing in patients who appear to be failing treatment, and to the extent that such practices persist, viral suppression might be underestimated [5].
Viral load data could be missing for other reasons. Many viral load tests are done without results being recorded, which can lead to a bias if higher viral loads are more likely to be recorded [10]. Patients with poorer ART adherence may be less likely to attend clinics to receive viral loads at the recommended frequency, or clinicians may delay testing in patients who have recently interrupted ART or have an intercurrent illness, which may introduce a bias towards over-estimating viral suppression. Testing coverage could itself be biased in countries relying on monitoring systems that are not based on a unique patient identifier. For example, guidelines recommend more frequent viral load testing in patients who are failing treatment [11], which could – in settings where there is potential double-counting of tests conducted in the same patient – lead to over-estimation of testing coverage and underestimation of viral suppression.
In view of these uncertainties, it is important to assess not only the factors associated with viral suppression in patients currently on ART, but also the factors associated with the recording of viral suppression, and the potential bias due to missing data. We aim to address these questions using data from South Africa, the country with the largest HIV epidemic in the world. South Africa is one of the few countries in the sub-Saharan African region to have recommended routine viral load testing since the inception of its public ART programme in 2004 [12], and therefore has relatively large amounts of viral suppression data. South Africa also has several well-established ART cohorts, which participate in the International epidemiology Databases to Evaluate AIDS Southern Africa (IeDEA-SA) collaboration. This study makes use of IeDEA-SA data from South African cohorts to analyse viral load testing coverage and viral suppression, both in adults and children.
Methods
The IeDEA-SA collaboration has been described previously [13]. ART cohorts participating in the current analysis included the Harriet Shezi, Hlabisa, Kheth'Impilo, Khayelitsha, Rahima Moosa, Red Cross War Memorial Children's Hospital (RCWMCH), Themba Lethu and Tygerberg cohorts. Participating cohorts submitted anonymized data on patients receiving HIV care to the data centre at the University of Cape Town (UCT), where the data were merged and cleaned. Ethical approval to collect and share the data was obtained by each of the participating cohorts (through their local institutional review boards) and by the UCT data centre (through the UCT Human Research Ethics Committee). With the exception of Themba Lethu, all cohorts provided data on children (aged less than 15 years), and all but three cohorts (Harriet Shezi, Rahima Moosa and RCWMCH) provided data on adults (Table S1 in Additional file 1).
Patients were included in analyses if they had a recorded date of ART initiation in 2005 or later, and a censoring date after the date of ART initiation. The censoring date was defined as the earlier of the last visit date and the date of death. The baseline CD4+ T-cell count was defined as the CD4+ T-cell count measured closest to the date of ART initiation. Patients were excluded if they had missing data on key variables (age, sex, baseline CD4+ T-cell count). Patients were also excluded if they were virally suppressed prior to ART start or (in the case of adults) if they started ART on a regimen that did not contain nevirapine or efavirenz (as such patients were unlikely to be ART-naive at enrolment).
In accordance with national ART protocols [12,14–16], we defined the dates of scheduled viral loads at 6 months and 12 months after ART initiation and at 12-month intervals thereafter. For the Western Cape cohorts (Khayelitsha, RCWMCH and Tygerberg), the first scheduled viral load test date was defined as occurring 4 months after ART start (this is in line with Western Cape guidelines [17], which recommend the same frequency of viral load monitoring as national guidelines at longer durations). Analyses were limited to scheduled testing dates that occurred before the censoring date. Patients were considered to have a missing viral load if there was no viral load recorded in the period between 2 months before and 2 months after the scheduled testing date. If multiple viral load results were recorded in this period, the earliest measurement was taken as the viral load recorded in the testing interval. Patients were considered virally suppressed if their viral load was less than 400 RNA copies/ml. Sensitivity analyses were conducted to assess the effect of using shorter testing intervals (within 1 month before and after the scheduled testing date), longer testing intervals (within 3 months of scheduled testing in the first year of ART, and within 6 months of the scheduled testing dates in later years, in line with South African Department of Health reporting), and a higher viral suppression threshold (1,000 RNA copies/ml, in line with global reporting guidelines [18]).
Multivariable logistic regression models were used to assess predictors of test recording (viral load not missing), separately for adults and children. Multi-variable logistic regression models were used to assess predictors of viral suppression, for those intervals in which a test result was recorded. In a sensitivity analysis, multiple imputation by chained equations was used to impute missing viral load values [19]. Recorded viral loads were log-transformed before imputation, and the imputation model included age, sex, baseline CD4+ T-cell count, cohort, non-nucleoside reverse transcriptase inhibitor (NNRTI), ART duration, year of viral load, indicators of whether the scheduled viral load occurred within 2 years of death or loss to follow-up (LTFU), viral load in the preceding testing interval and viral load in the subsequent testing interval (LTFU was defined as no patient contact for more than 90 days at the time of database closure). The multivariable regression models were applied to five imputed data sets, and results were combined using Rubin's rules [20]. Further details on the multiple imputation are included in section 1 of Additional file 1. Additional sensitivity analyses were conducted to assess the effect of keeping continuous variables (age, ART duration and baseline CD4+ T-cell count) continuous rather than treating these variables as categorical. Generalized estimating equations were used in all regression models to control for multiple observations in the same patient. All statistical analysis was conducted using Stata version 15.0 (StataCorp Inc., College Station, TX, USA).
Results
In the adult analysis, 603,549 viral load tests were scheduled in a total of 117,880 patients (patient characteristics are summarized in Table S2 in Additional file 1). However, viral load results were recorded in only 245,751 (40.7%) of scheduled testing intervals. Factors associated with less frequent recording of viral load included older age (aOR 0.90 [95% CI: 0.88,0.92] and aOR 0.87 [95% CI: 0.84,0.90] in age groups 25–49 and 50+ respectively, compared with 15–24), initiating ART on a nevirapine-containing regimen (aOR 0.96, 95% CI: 0.95, 0.98), higher baseline CD4+ T-cell count (aOR 0.83 [95% CI: 0.81,0.83] at CD4+ T-cell counts 500 and greater compared with less than 200) and longer ART duration (aOR 0.43 [95% CI: 0.41,0.44] at durations of 10 years and longer compared with 0–3 years after ART initiation; Table 1). However, the effects of age, nevirapine use and baseline CD4+ T-cell count ceased to be significant when continuous variables were kept in the model as continuous covariates (Table S5 in Additional file 1). The frequency of recording decreased between 2005–2007 (49.3%) and 2008–2010 (39.5%), but improved substantially in 2017–2019 (47.8%). When the scheduled testing interval was defined as being between 1 month before and after the scheduled testing date, the fraction of testing intervals with a recorded viral load decreased to 31.0%, but the fraction increased to 63.3% when using longer testing intervals. Factors associated with recording of a viral load were similar to those in the main analysis, but lower baseline CD4+ T-cell count ceased to be associated with more frequent recording when using shorter intervals (Table S3 in Additional file 1), and shorter ART durations and younger age ceased to be associated with more frequent recording when using longer intervals (Table S4 in Additional file 1).
Recording of viral loads in adults on ART in South Africa, 2005 to 2019

Adjusted odds ratio (aOR) for a recorded viral load (VL) controlling for antiretroviral treatment (ART) cohort and other explanatory variables shown. NNRTI, non-nucleotide reverse transcriptase inhibitor.
Out of 245,571 recorded viral load measurements in adults, 210,635 (85.7%) were below 400 RNA copies/ml, with rates varying between 78.9% and 89.1% across ART cohorts. Levels of viral suppression declined substantially in the 2010–2011 period, but gradually recovered thereafter (Figure 1A). In multivariable analysis, factors associated with greater rates of viral suppression included female sex (aOR 1.26, 95% CI: 1.22, 1.30), older age (aOR 1.75 [95% CI: 1.65,1.85] and aOR 2.17 [95% CI: 2.03,2.33] in age groups 25–49 and 50+ respectively, compared with 15–24), and higher baseline CD4+ T-cell count (aOR 1.81 [95% CI: 1.70,1.92] at CD4+ T - cell counts 500 and greater compared with less than 200), while factors associated with lower rates of viral suppression included initiating ART on a nevirapine-containing regimen (aOR 0.76, 95% CI: 0.73, 0.79) and longer ART duration (aOR 0.71 [95% CI: 0.65,0.77] at durations of 10 years and longer compared with 0–3 years after ART initiation; Table 2). When considering a viral suppression threshold of 1,000 RNA copies/ml, the fraction of adults who were suppressed increased to 89.6%, but predictors of viral suppression were similar to those in the main analysis (Table S6 in Additional file 1). Defining the testing interval as being between 1 month before and after the scheduled testing date led to only a modest increase in viral suppression (86.2%), and little change in the predictors of viral suppression (Table S7 in Additional file 1). Using a longer testing interval also had little effect on viral suppression, except that the effect of ART duration was attenuated (Table S8 in Additional file 1).
Viral suppression in adults on ART in South Africa, 2005 to 2019
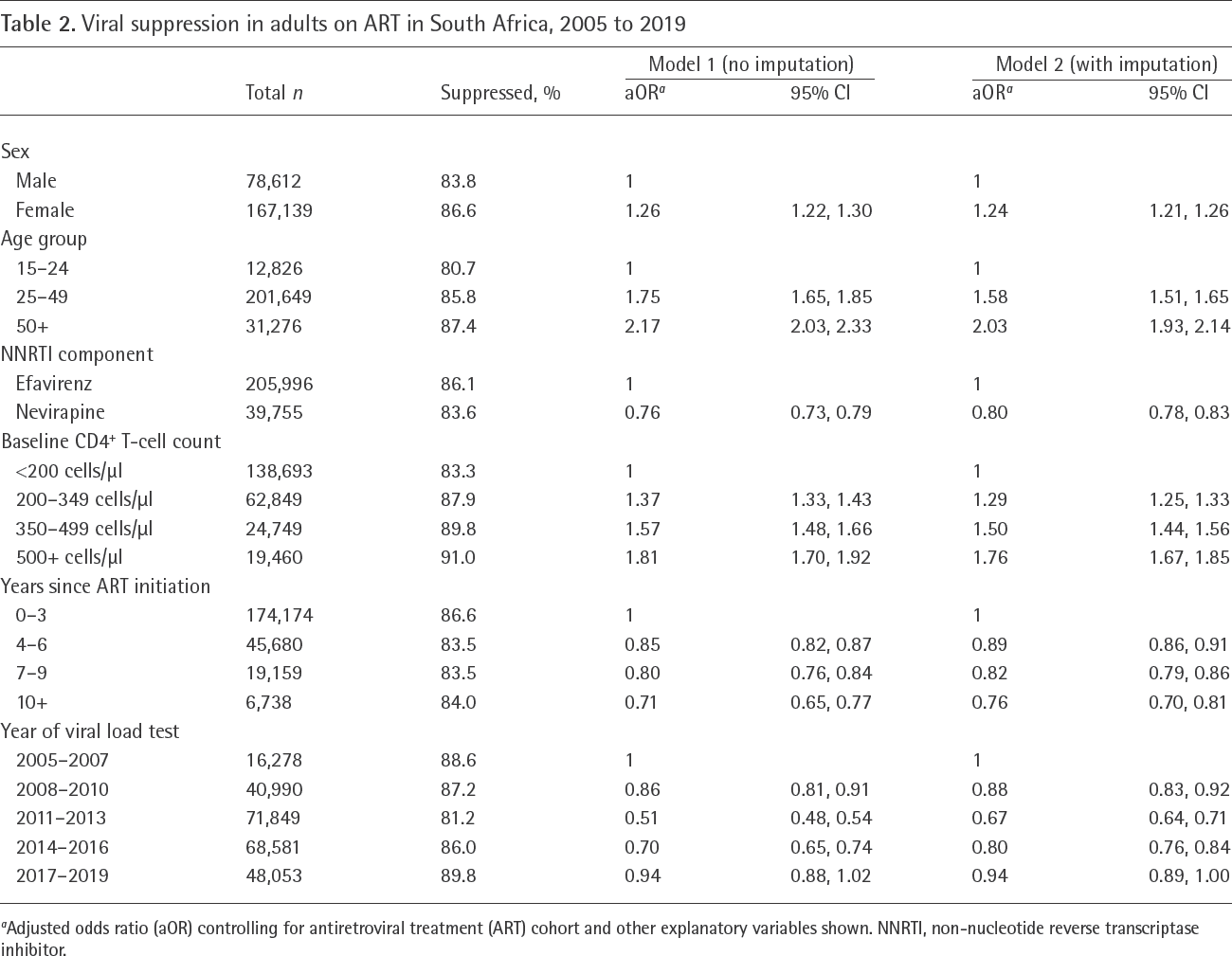
Adjusted odds ratio (aOR) controlling for antiretroviral treatment (ART) cohort and other explanatory variables shown. NNRTI, non-nucleotide reverse transcriptase inhibitor.

Viral suppression trends in antiretroviral treatment patients
Viral loads of <400 RNA copies/ml were less common within 2 years prior to a death (69.5%) and within 2 years prior to transfer/LTFU (82.8%). Imputation, which was based on these outcomes as well as viral load measurements at other testing dates and the other variables listed previously, yielded a slightly lower estimate of viral suppression (85.3% compared with 85.7% before imputation). In the multivariable analysis of the imputed datasets, predictors of viral suppression were similar to those in the main analysis, but adjusted odds ratios were consistently closer to 1 (Table 2). Imputed viral loads were less likely to be below 400 RNA copies/ml than recorded viral loads when controlling for other factors (aOR 0.96, 95% CI: 0.94, 0.98).
In the paediatric analysis, 77,423 tests were scheduled in 14,760 children who started ART (patient characteristics are summarized in Table S10 in Additional file 1). Test results were recorded in 31,932 (41.2%) of scheduled testing intervals, similar to adults. As in adults, less frequent recording of viral load was associated with older age (aOR 0.83 [95% CI: 0.80,0.87] and aOR 0.82 [95% CI: 0.78,0.86] in age groups 5–9 and 10–14 respectively, compared with 0–4 years) and longer ART duration (aOR 0.64 [95% CI: 0.57,0.72] at durations of 10 years and longer compared with 0–3 years after ART initiation), and recording in the 2008–2010 period was significantly less frequent than in the 2005–2007 period (aOR 0.70, 95% CI: 0.66, 0.73; Table 3). When the scheduled testing interval was defined as being between 1 month before and after the scheduled testing date, the fraction of testing intervals with a recorded viral load decreased to 29.9%, but the fraction increased to 63.1% when using longer testing intervals. Factors associated with recording of a viral load were similar to those in the main analysis, except that longer ART durations were associated with higher frequency of viral load recording when using longer testing intervals (Tables S11 and S12 in Additional file 1).
Recording of viral loads in children on ART in South Africa, 2005 to 2019

Adjusted odds ratio (aOR) for a recorded viral load (VL) controlling for antiretroviral treatment (ART) cohort and other explanatory variables shown.
Of the 31,932 recorded viral load results in children, 23,106 (72.4%) were below 400 RNA copies/ml, with rates of suppression varying between 41.3% and 79.3% across cohorts. Viral suppression was highest in the period before 2010, then declined substantially in 2010–2011, with little change thereafter (Figure 1B). In multivariable analysis, rates of viral suppression were highest in children aged 5–9 years (aOR 1.87 [95% CI: 1.73,2.02] compared with ages 0–4), at longer ART durations (aOR 1.40 [95% CI: 1.23,1.58] 7–9 years after ART initiation compared with 0–3 years after ART initiation), and in the period up to 2010 (Table 4). The fraction of children who were virally supressed increased to 77.9% when using a threshold of less than 1,000 RNA copies/ml to define viral suppression, but in all sensitivity analyses, predictors of viral suppression were similar to those in the main analysis (Tables S14–S17 in Additional file 1). When missing viral loads were imputed, the overall fraction virally suppressed increased (73.2% compared with 72.4% before imputation), although imputed values (when controlling for other variables) yielded similar probabilities of viral suppression compared to recorded values (aOR 1.01, 95% CI: 0.97, 1.05). Predictors of viral suppression, when analysed using the imputed data, were similar to those in the main analysis (Table 4).
Viral suppression in children, 2005 to 2019

Adjusted odds ratio (aOR) controlling for antiretroviral treatment (ART) cohort and other explanatory variables shown.
Discussion
In our sample of adult South African ART cohorts, rates of viral suppression were generally high, with the 90% UNAIDS target being met in all years from 2015 to 2018 (Figure 1A). However, viral suppression in earlier years was inconsistent, with a notable dip in 2010– 2011, and a gradual recovery thereafter. Although the rates of viral suppression in adults are encouraging, levels of viral suppression were substantially lower in children.
The reasons for the changes in viral suppression over time are unclear. The dip in 2010–2011 may have been due to a change in the assay used to measure viral load (from the NucliSENS EasyQ assay [21] to the Cobas Ampliprep-Cobas TaqMan [22], which have slightly different performance characteristics [23]). The dip in 2010–2011 could also have been due to changes in test turn-around times, with significant reductions in turnaround times being observed in the Western Cape in 2010, which correlated with reductions in viral suppression [24]. The increase in viral suppression after 2013 might have been driven by the introduction of fixed-dose combination (FDC) ART in 2013, which is generally associated with better adherence [25], and the partial resolution of supply-chain problems, which had resulted in particularly serious drug stock-out problems in 2012–2013 [26–28]. Drug resistance, a major driver of virological failure, appeared to increase in the period up to 2013–2014 and then stabilize/decline [29], which might also partly explain the observed trends in viral suppression.
The low rates of viral suppression in children are consistent with levels of viral load prior to ART initiation being higher in children than in adults [13], especially in the first year of life [30,31]. Although viral suppression in children dropped substantially in the 2010–2011 period, the gradual increase in viral suppression observed in adults after 2011 was not seen in children. This might be because FDCs are not available in children to the same extent as in adults (especially in younger children, in whom dosing is weight-dependent), and thus the FDC introduction in 2013 would not have benefited children to the same extent as adults. The drop in viral suppression in 2010 may also be partly due to the replacement of stavudine with abacavir, which has been associated with poorer viral suppression in South African children [32]. As in a recent analysis of global IeDEA data [6], we found that viral suppression was poorest in children under the age of 5 years, a refection of the higher baseline viral loads in this age group.
Consistent with several previous studies [7,33,34], we also found substantially higher rates of viral suppression in women than in men (aOR 1.24). This may in part be due to biological differences; even in the absence of ART, HIV-positive men tend to have higher levels of viraemia than women [35,36]. However, the differences may also be attributable to behavioural factors; a recent meta-analysis found that the odds of non-adherence were on average 1.73 times greater in men than in women [37]. Further research is required to identify interventions to improve ART adherence in men. Our results are also consistent with previous studies that have shown poor virological suppression to be associated with younger age [6,8,38], use of nevirapine instead of efavirenz [39], and lower baseline CD4+ T-cell count [6,8,9,40]. There is a clear need for enhanced adherence support in younger ART patients and patients starting ART at lower CD4+ T-cell counts.
A novel feature of this analysis is that we have used multiple imputation to address the problem of missing viral load data. Since viral loads are strongly associated with clinical and demographic factors, as well as with outcomes such as death and LTFU, and since successive viral load measurements might be expected to be correlated, imputation is a useful and feasible approach. Our analysis suggests that imputed viral loads differ only slightly from actual viral loads, and thus the change in estimated rates of viral suppression when using imputed data is relatively modest (slightly lower in adults and slightly higher in children). This is reassuring, as it suggests that in settings similar to ours, suppression estimates may be reasonably reliable, despite infrequent recording of viral loads. Previous studies in resource-limited settings have yielded inconsistent findings regarding the bias introduced by missing viral load data, but most of these studies are of limited relevance here because they were conducted prior to routine virological monitoring [34,41]. The only study we are aware of that assessed bias in the context of routine virological monitoring found overall estimates of viral suppression were relatively insensitive to adjustment for incomplete data [10], consistent with our findings.
The testing coverage of around 40% in our study is nevertheless a major concern, and improved recording of test results needs to be prioritized [10]. Multiple imputation may fail if there are unmeasured factors affecting missingness and those factors are also correlated with the outcome of interest. The factors affecting missingness are also likely to differ across settings, and the low levels of bias suggested in our analysis might therefore not be generalizable to other settings. Previous studies have found little consistency in the correlates of viral load recording [10,42,43], and our results suggest that some of this inconsistency may be due to differences in the way ‘missing’ values are defined (the time period in which values are not recorded) as well as differences in the regression model specification (for example, specifying explanatory variables as continuous or categorical). The 1- and 2-month intervals before and after the scheduled testing date may be inappropriate in determining when viral loads are considered ‘recorded’, particularly at longer ART durations, when decisions about when to conduct a test are more likely to be based on factors such as time since last test and desire to ‘batch’ viral load testing, rather than the time since ART initiation.
A limitation of this analysis is that the IeDEA South African cohorts are not representative of the public sector ART programme in South Africa in terms of viral suppression. Although viral load testing coverage in the national programme has been similarly low (around 40–50% over the period up to 2014 [44]), rates of viral suppression have consistently been around 1–5% lower than the rates estimated here [44]. In a national household survey conducted in 2017, the proportion of treated adults who had a viral load of less than 1,000 RNA copies/ml was 87.4% [45], lower than our corresponding estimate in 2017 (92.4%; Figure 1A). This suggests that the IeDEA-SA cohorts may have more resources for patient adherence support or may be catering to a different socio-demographic patient mix, when compared with other public-sector ART services.
In addition, these results may not be generalizable to other African settings, especially those in which routine viral load testing is not well-established. Other limitations include the lack of data on psychosocial factors that might be expected to influence viral suppression, such as alcohol abuse [37], depression [37], migration [46], and exposure to intimate partner violence [47] and other forms of violence [48]. We have also not been able to measure adherence and treatment interruptions, which are also important determinants of viral suppression [38,49].
A strength of the IeDEA data is that they are drawn from well-established research cohorts that have been collecting viral load data since the inception of the ART programme in South Africa. In more recently established monitoring systems, the interpretation of viral load data may be more challenging. In particular, monitoring systems that do not link records in the same patient are at significant risk of under-estimating viral suppression due to the more frequent monitoring of patients who experience virological failure. Unique patient identifiers are critical to reducing this selection bias, and individual-based monitoring systems need to be prioritized in order to evaluate viral suppression more reliably.
In conclusion, the 90% viral suppression target is well within reach in the South African adult ART population, but the target is unlikely to be met in children. Although missing viral load data is a concern, estimates of viral suppression and predictors of viral suppression do not appear to be sensitive to missing data corrections.
Footnotes
Acknowledgements
This research was funded by the National Institutes of Health (grant U01AI069924). We thank Ernest Mokotoane (University of Cape Town, Cape Town, South Africa) for assistance in data preparation and synthesis.
The authors declare no competing interests.
