Abstract
Background
The combination of elbasvir and grazoprevir (EBR/GZR) has been approved for treating HCV infection. This study aimed to evaluate the efficacy of EBR/GZR in terms of sustained virological response (SVR) and improvement of liver fibrosis in Thai patients with HCV genotype-1 (GT1). The utility of serum HCV core antigen (HCVcAg) as an alternative to HCV RNA in assessing SVR was also investigated.
Methods
A total of 101 HCV GT1-infected patients (65 monoinfection and 36 HIV coinfection) who received EBR/GZR for 12–16 weeks were included. Liver stiffness (LS) and controlled attenuation parameter (CAP) were measured by transient elastography. Serum HCVcAg was measured in parallel with HCV RNA.
Results
The overall SVR12 and SVR24 rates in the cohort were 98.0% and 95.0%, respectively. SVR24 rates were consistently high (90.0% to 100%) across all subgroups of patients. A significant LS decline ≥30% was observed more frequently in cirrhotic than non-cirrhotic individuals who achieved SVR (63.3% versus 30.3%; P=0.003). The magnitude of LS decline following HCV eradication was comparable between HCV monoinfection and HCV–HIV coinfection. The reduction of CAP was also observed in responders who had significant steatosis at baseline. Compared with HCV RNA, HCVcAg testing displayed high sensitivity (100%) and specificity (99.0–100%) in determining SVR12 and SVR24.
Conclusions
This study confirms that EBR/GZR is effective for HCV GT1-infected Thai patients with or without HIV infection. HCV eradication is associated with LS and CAP improvement regardless of HIV status. HCVcAg testing could be a potential replacement for HCV RNA for assessing SVR in resource-limited settings.
Introduction
Currently, approximately 180 million people are chronically infected with HCV, with the majority of all cases occuring in low- and middle-income countries [1]. HCV, a single-stranded positive sense RNA virus of the family Flaviviridae, has been classified into seven major genotypes, all of which display diverse patterns of geographic distribution [2]. In Thailand, the prevalence of HCV infection in the general population is approximately 2%, with the most common genotypes (GT) being GT3 (50%), GT1 (30%) and GT6 (20%) [3]. Based on the natural history of chronic HCV infection, it is estimated that 10–20% of patients will develop cirrhosis and these cirrhotic patients will eventually progress to hepatocellular carcinoma (HCC) at an annual rate of 2–7% [4]. The risk of cirrhosis is increased in individuals with older age at infection, heavy alcohol intake, concomitant obesity and coinfection with HIV [1]. HCV–HIV-coinfected patients are at threefold greater risk for progression to cirrhosis or liver decompensation and at tenfold greater risk for liver-related mortality compared with the HCV mono-infected group [5]. Our previous cross-sectional data showed that advanced liver fibrosis was more commonly found in HCV–HIV-coinfected individuals than in HCV monoinfected patients (41% versus 25%) [6]. Previously, a standard of care for chronic HCV infection was the combination of pegylated interferon (PEG-IFN) and ribavirin (RBV). However, the response rate of this regimen was poor, particularly among HCV–HIV-coinfected patients with genotype-1 (GT1) and advanced fibrosis [7].
With the introduction of direct-acting antivirals (DAAs), HCV treatment has been revolutionized as HCV mono- and HCV–HIV-coinfected individuals could achieve sustained virological response (SVR) rates over 90%. It has been shown that SVR is associated with a low risk of HCV reactivation and a reduced risk of the progression to cirrhosis and HCC [8]. The combination of elbasvir (EBR, NS5A inhibitor) and grazoprevir (GZR, NS3/4A protease inhibitor) is a once daily, fixed-dose tablet that has been approved for treating chronic HCV GT1 or HCV GT4 infection. This combination with or without weight-adjusted RBV for 12–16 weeks displays high rates of SVR across various groups of HCV GT1-infected individuals, including treatment-naive or treatment-experienced patients, HCV monoinfection or HCV–HIV coinfection and difficult-to-treat patients, such as those with compensated cirrhosis [9].
It has been demonstrated that HCV eradication with DAA therapy is associated with significant fibrosis regression [8]. Although liver biopsy has been a gold standard to assess liver histopathology, this procedure has some limitations due to its invasive technique and risks of potentially life-threatening complications [10]. Its accuracy might also be compromised because of sampling errors and variations in interpretation [11]. An alternative non-invasive tool to measure the severity of fibrosis is liver stiffness (LS) measurement using transient elastography (FibroScan [TE]), which has been validated in both HCV monoinfected and HCV–HIV-coinfected patients [12]. Beyond cross-sectional observations, TE can also be used to monitor potential regression of liver fibrosis and predict long-term outcome after antiviral treatment [13]. In addition to LS measurement, TE can calculate controlled attenuation parameter (CAP), which is a reliable quantitative marker of liver steatosis [14]. However, it is unclear whether the extent of liver steatosis might improve in HCV-infected patients who achieve SVR after DAA therapy.
The World Health Organization (WHO) recommends nucleic acid testing for HCV RNA as the standard of care to assess treatment response at 12 or 24 weeks after the end of treatment (SVR12 or SVR24) [15]. However, HCV RNA measurement is restricted in developing countries due to high costs and the requirement of equipped laboratories. Recently, serum HCV core antigen (HCVcAg) has been shown to be an alternative assay to conventional HCV RNA testing with the advantages of its rapidity and more feasibility in resource-limited settings. A systematic review and meta-analysis has indicated good correlation between HCVcAg and HCV RNA if viral load is greater than 3,000 IU/ml, the level which is typically found in most untreated HCV-infected individuals [16]. Moreover, a recent study has demonstrated HCVcAg testing is cost saving and can potentially replace HCV RNA for the diagnosis of HCV viraemia and monitoring treatment adherence [17]. Currently, its role in the determination of SVR following DAA therapy is debatable and additional data are needed.
This interventional, non-randomized, open-label study was primarily conducted to evaluate the efficacy of EBR/GZR combination in terms of SVR rates and changes in LS and CAP values in HCV GT1-infected Thai patients with or without HIV infection. Additionally, this study aimed to evaluate the usefulness of HCVcAg as a potential replacement of HCV RNA for the assessment of SVR.
Methods
Patients
Patients treated with EBR/GZR and recruited via clinicaltrials.gov (NCT03037151) at the King Chulalongkorn Memorial Hospital, Bangkok, Thailand between August 2018 and June 2019 were enrolled in this study. Inclusion criteria for EBR/GZR therapy were patients with anti-HCV-positive for at least 6 months, HCV RNA levels >10,000 IU/ml and infected with HCV GT1. For patients with HIV coinfection, each individual received antiretroviral therapy (ART) with undetectable plasma HIV RNA levels. Exclusion criteria were HBV coinfection, previous treatment with DAAs, clinical data suggestive of decompensated cirrhosis or evidence of HCC by imaging studies prior to EBR/GZR therapy. Treatment-naive patients were treated with EBR/GZR for 12 weeks, while treatment-experienced patients, including null responders, partial responders or post-treatment relapsers following PEG-IFN/RBV, were treated with EBR/GZR plus weight-based RBV for 16 weeks. Patients were followed up to assess SVR (defined by HCV RNA level <12 IU/ml) at week 12 and week 24 after the end of treatment (SVR12 and SVR24, respectively).
The study protocol, conducted in compliance with the Helsinki Declaration and Good Clinical Practice guidelines, was approved by the Institutional Review Board of the Faculty of Medicine, Chulalongkorn University, Bangkok, Thailand. Written informed consents were obtained from all patients for the use of their clinical data and sample specimens.
Laboratory assays for HCV
Anti-HCV antibody was tested with a third-generation chemiluminescent microparticle immunoassay (CMIA, ARCHITECT system, Abbott Diagnostics, Wiesbaden, Germany). HCV genotypes and sub-genotypes were determined by nucleotide sequencing of the core and NS5B regions followed by phylogenetic analysis as described previously [3]. HCV RNA quantification was performed using the real-time quantitative reverse transcription polymerase chain reaction (RT-PCR; Abbott Molecular Inc., Des Plaines, IL, USA) in accordance with the manufacturer's instructions. The lower and upper detection limits of the assay were <12 IU/ml and 100,000,000 IU/ml, respectively. HCVcAg quantification was analysed by a two-step CMIA (Abbott Diagnostics, Tokyo, Japan). The test allows the determination of HCVcAg in a linear range 3 to 20,000 fmol/l, with 1:9 automated dilutions that extends the assay linearity to 180,000 fmol/l. Samples with concentrations between 3 and 10 fmol/l were defined as ‘grey zone’ and were re-examined in duplicate as manufacturer recommendations [18]. If at least one duplicate test was positive or in grey zone, the sample was considered as positive test. Serum HCV RNA and HCVcAg were measured in parallel at baseline, week 12 of follow-up (SVR12) and week 24 of follow-up (SVR24).
Measurement of LS and CAP
After fasting for at least 2 h, simultaneously LS and CAP measurement were obtained from each patient before therapy and at 24 weeks after completing therapy using TE (FibroScan 502 Touch, Echosens, Paris, France). Results of LS were recorded in kPa as the median value of all measurements. The procedure was based on at least 10 validated measurements: the success rate was over 60% and interquartile range was less than 30% [19]. The optimal cutoff values for the presence of advanced fibrosis (stages F3 or F4) and cirrhosis (F4) based on the American Gastroenterological Association Institute Technical Review were 9.5 and 12.5 kPa, respectively [20]. Significant declines in LS value were defined as ≥30% drop from baseline. CAP value was provided on the basis of the same criteria and on the same signals to confirm that ultrasound attenuation was obtained from the same volume of liver parenchyma as the assessment of LS. Based on a recent metaanalysis, the cutoff value for significant liver steatosis was 248 dB/m [14].
Statistical analyses
Data were expressed as percentages or mean + standard deviation (
Results
Baseline clinical characteristics
A total of 101 patients with HCV GT1 were recruited. Among these patients, there were 65 individuals with HCV monoinfection and 36 patients with HCV–HIV coinfection. The patients consisted of 79 males and 22 females with a mean age of 47.1 ±11.1 years. Baseline clinical characteristics regarding HIV status are summarized in Table 1. Compared with the coinfected group, patients with HCV monoinfection had significantly higher average age, body mass index (BMI) and the distribution of HCV GT1b. There was no significant difference between groups in terms of gender, biochemical parameters, serum HCV RNA and HCVcAg levels, TE and CAP values and previous PEG-IFN/RBV therapy.
Baseline characteristics of patients in this study
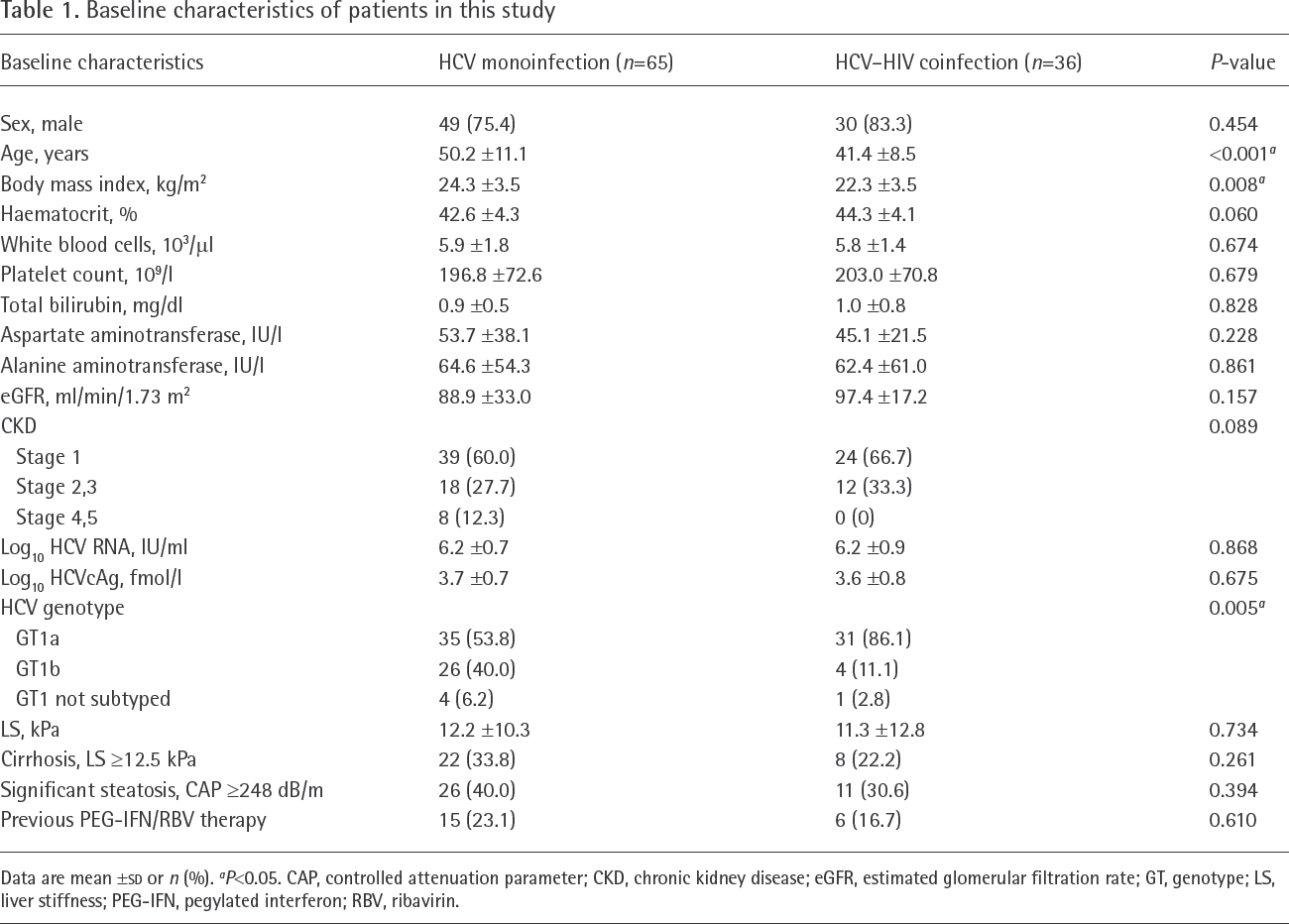
Data are mean ±
P<0.05. CAP, controlled attenuation parameter; CKD, chronic kidney disease; eGFR, estimated glomerular filtration rate; GT, genotype; LS, liver stiffness; PEG-IFN, pegylated interferon; RBV, ribavirin.
Efficacy of EBR/GZR
Regarding treatment outcome, SVR12 rate of the overall cohort was 98.0% (99/101). Of those who achieved SVR12, three patients experienced virological relapse at week 24 during follow-up and thus overall SVR24 was 95.0% (96/101). HCV monoinfected patients and the coinfected individuals achieved SVR12 of 98.5% (64/65) and 97.2% (35/36), respectively (P=1.000). The corresponding figures regarding SVR24 were 93.8% (61/65) and 97.2% (35/36), respectively (P=0.653). Moreover, treatment-naive and treatment-experienced patients achieved SVR12 of 97.5% (78/80) and 100% (21/21), respectively (P=1.000), while the corresponding figures of SVR24 were 93.8% (75/80) and 100% (21/21), respectively (P=0.581; Figure 1).
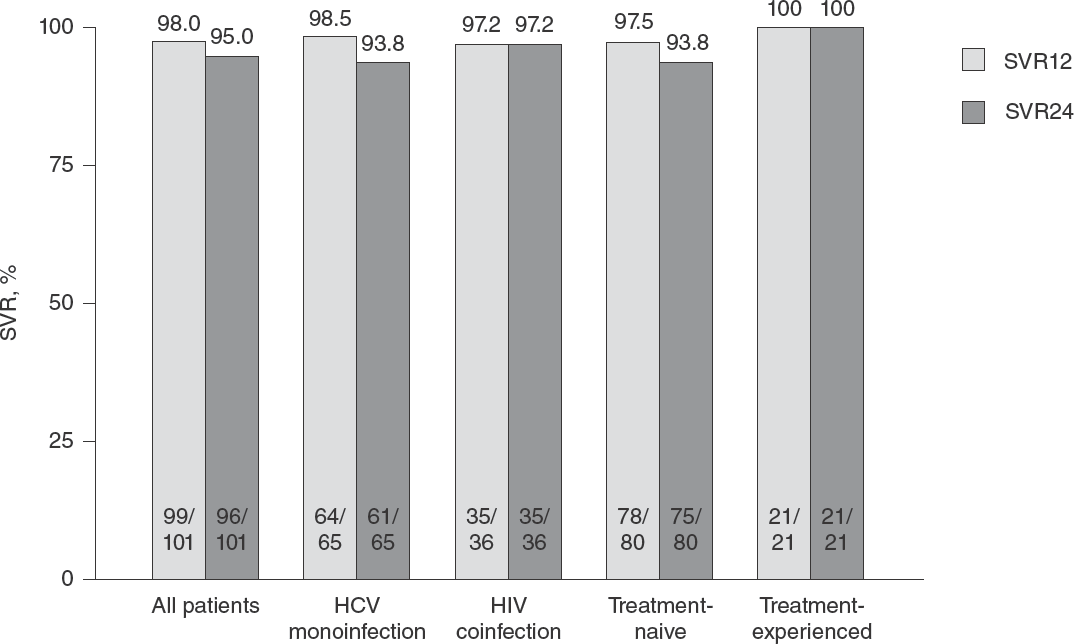
Rates of SVR12 and SVR24 in all patients and subgroups
In this report, SVR24 rates were consistently high across all subgroups of patients treated with EBR/GZR for 12–16 weeks (Additional file 1). There was no impact of age, sex, BMI, prior treatment with PEG-IFN/RBV, HIV status, baseline viral load, presence of cirrhosis or severity of renal impairment. In the GT1a-infected group, 97.0% (95% CI =89.5, 99.6%) of participants had SVR24, and SVR24 rate was slightly lower (90.0%, 95% CI =73.5, 97.9%) in the relatively small number of GT1b-infected patients.
Relationship between baseline LS, CAP and clinical parameters
The relationship between LS values and clinical parameters at baseline was examined. A positive correlation was found between LS and age (r=0.363, P<0.001), BMI (r=0.229, P=0.021), aspartate aminotransferase (AST; r=0.488, P<0.001) and alanine aminotransferase (ALT; r=0.207, P=0.038). A negative correlation was found between LS and platelet counts (r=-0.526, P<0.001) and eGFR (r=-0.201, P=0.044). There was no correlation between LS and other parameters (gender, HIV status, HCV sub-genotype, haematocrit, white blood counts, total bilirubin, CAP, HCV RNA and HCVcAg levels). Regarding CAP at baseline, a positive correlation was only found with BMI (r=0.516, P<0.001). There was no correlation between CAP and other parameters.
Changes of LS and CAP values at SVR24
Overall LS values in this cohort significantly decreased after EBR/GZR treatment, with a mean value prior to treatment of 11.7 ±11.3 kPa to 8.6 ±7.4 kPa at the time of SVR24 (P<0.001). A decrease in LS values of at least 30% was observed in 39.6% (40/101). Both patients with SVR and non-SVR showed a significant reduction in LS values. LS values at baseline and SVR24 in patients achieving SVR were 12.1 ±11.5 and 8.8 ±7.6 kPa, respectively (P<0.001), while the corresponding figures for patients without SVR were 7.1 ±2.4 kPa and 5.6 ±2.4 kPa, respectively (P=0.021). Moreover, a significant LS decline was observed in 40.6% (39/96) of patients with SVR and 20.0% (1/5) of patients without SVR (P=0.646).
In subgroup analysis of individuals achieving SVR24 (96 patients), non-cirrhotic patients displayed mean LS values at baseline and SVR24 of 6.7 ±1.9 and 5.5 ±1.9 kPa, respectively (P<0.001), while the corresponding figures for the cirrhotic group were 23.9 ±14.5 kPa and 15.6 ±9.9 kPa, respectively (P<0.001). Of note, a decrease in LS value of at least 30% was observed more frequently in the cirrhotic group compared with non-cirrhotic individuals (63.3% versus 30.3%; P=0.003).
We further compared the decrease in LS values regarding HIV status in patients who achieved SVR. In the non-cirrhotic group, patients with HCV monoinfection had mean LS values at baseline and SVR24 of 6.7 ±2.0 and 5.7 ±2.1 kPa, respectively (P=0.007), while the corresponding figures for HCV–HIV-coinfected patients were 6.7 ±1.9 kPa and 5.4 ±1.6 kPa, respectively (P=0.001). A decrease in LS value of at least 30% was not signifcantly different between the monoinfection and coinfection groups (23.1% versus 40.7%; P=0.174). Among patients with cirrhosis at baseline, the monoinfection group had mean LS values at baseline and SVR24 of 22.8 ±11.6 and 14.9 ±8.0 kPa, respectively (P<0.001), while the corresponding figures for the coinfected group were 27.0 ±21.1 kPa and 17.7 ±14.1 kPa, respectively (P=0.042). Again, a decrease in LS value of at least 30% was not significantly different between these two groups (68.2% versus 50.0%, respectively; P=0.417).
In the overall cohort, there were no significant changes of CAP values between baseline and the time of SVR24 (229.4 ±52.8 and 221.6 ±50.6 dB/m, respectively; P=0.091). Considering only patients with signifcant liver steatosis at baseline (CAP ≥248 dB/m, n=37), its values at baseline and SVR24 in these patients were 284.4 ±30.7 and 265.6 ±48.9 dB/m, respectively (P=0.018). Among 34 patients achieving SVR24, CAP at baseline and SVR24 were 285.9 ±31.0 and 267.8 ±50.0 dB/m, respectively (P=0.035), while the corresponding figures in the non-SVR group were 267.7 ±27.2 and 240.3 ±10.7 dB/m, respectively (P=0.199). These data indicated that liver steatosis was attenuated after EBR/GZR therapy, particularly in responders who had significant liver steatosis at baseline.
Factors associated with significantly decreased LS after therapy
Univariate and multivariate analyses were performed to identify factors that were associated with a significant decline in LS value. These baseline factors included age, sex, BMI, AST, ALT, platelet counts, HIV status, HCV sub-genotype, HCV RNA level, prior treatment, cirrhosis, significant liver steatosis and treatment outcome. In univariate analysis, parameters associated with reduced LS value were age ≥45 years, history of previous treatment with PEG-IFN/RBV and cirrhosis. In multivariate analysis, previous PEG-IFN/RBV treatment and cirrhosis were independent factors associated with significant LS reduction (Table 2).
Logistic regression analysis of factors associated with significantly decreased liver stiffness

P<0.05. ALT, alanine aminotransferase; AST, aspartate aminotransferase; CAP, controlled attenuation parameter; GT, genotype; LS, liver stiffness; OR, odds ratio; PEG-IFN, pegylated interferon; RBV, ribavirin; SVR, sustained virological response.
Diagnostic performance of HCVcAg assay
To better understanding the role of serum HCVcAg for the assessment of SVR, the antigen was measured in parallel with HCV RNA levels at baseline and after EBR/GZR therapy. Figure 2 demonstrates the correlation between log10HCVcAg and log10HCV RNA levels at several time points. Overall, log10HCVcAg levels at baseline were strongly correlated with corresponding log10HCV RNA levels (r=0.867, P<0.001; Figure 2A). Of note, the concordance of detectable HCVcAg and HCV RNA was observed in all patients before starting therapy (100%). After therapy, log10HCVcAg exhibited consistently strong correlation with log10HCV RNA at SVR12 (r=0.996, P<0.001) and SVR24 (r=0.994, P<0.001; Figure 2B and 2C).
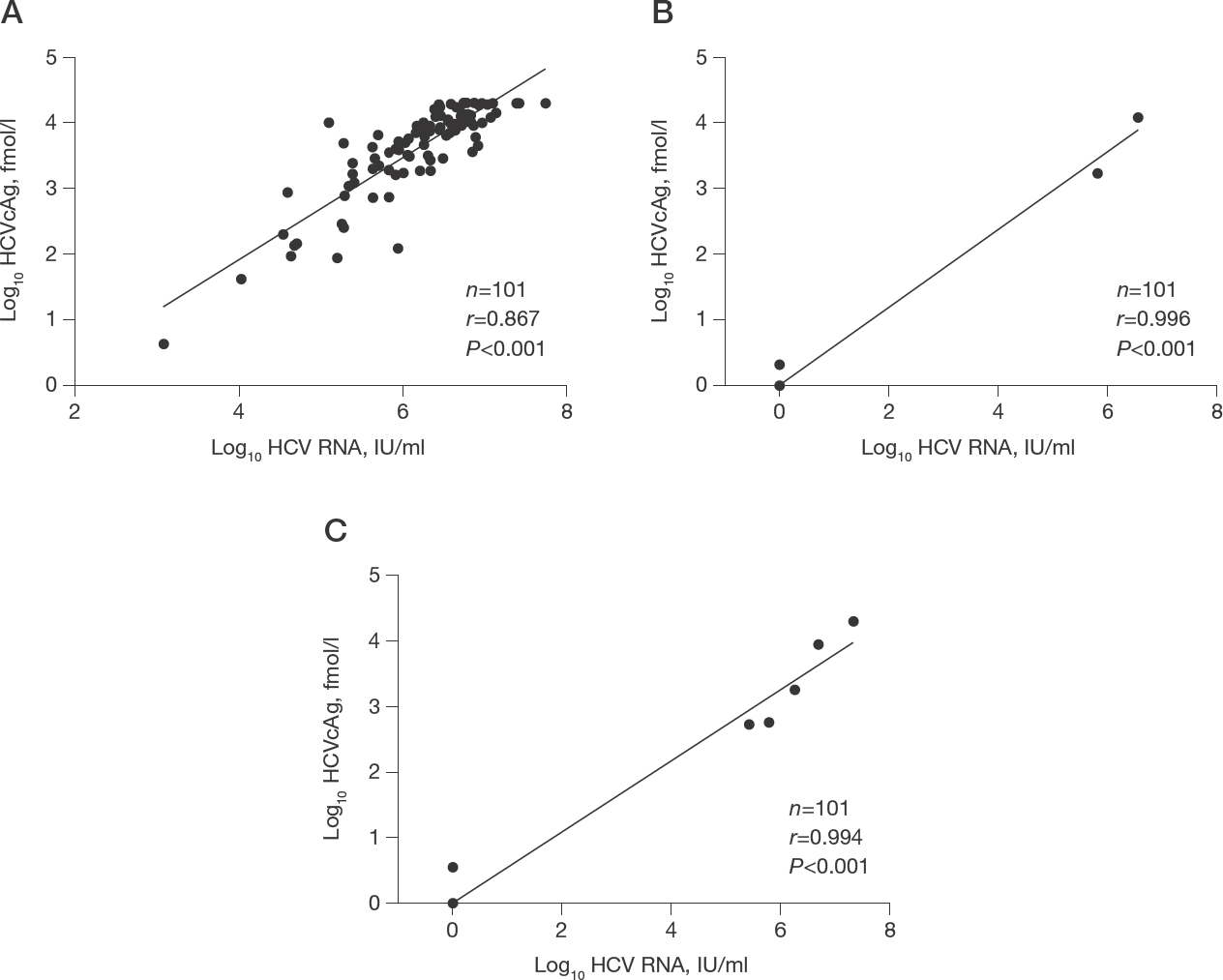
Correlation between HCV RNA and HCVcAg
A subgroup analysis comparing HCVcAg performance between HCV monoinfected and HCV-HIV-coinfected patients demonstrated strong correlation between HCV RNA and HCVcAg at SVR12 and SVR24 for both groups (at SVR12, r=0.995 for HCV monoinfection and r=1.000 for HCV-HIV coinfection, P<0.001 for both; at SVR24, r=0.996 for HCV monoinfection and r=0.992 for HCV-HIV coinfection, P<0.001 for both). However, at baseline, the correlation between HCV RNA and HCVcAg was lower in HCV monoinfection (r=0.798, P<0.001) than in HCV–HIV coinfection (r=0.948, P<0.001).
Using HCV RNA as the gold standard method, HCVcAg demonstrated high sensitivity (100%) and high specificity (99.0–100%) at SVR12 and SVR24 (Table 3). At SVR12, 100% concordance was found between HCV RNA and HCVcAg. At SVR24, the concordance between the two markers was 99%, as one patient with HCV–HIV coinfection had low detectable HCVcAg level (3.5 fmol/l) but undetectable HCV RNA, which was considered as a false positive result.
Diagnostic performance for HCVcAg using HCV RNA as the reference

HCVcAg, HCV core antigen; NPV, negative predictive value; PPV, positive predictive value; SVR, sustained virological response.
Discussion
A new paradigm of HCV therapy has improved substantially after the development of DAAs that are specifically addressed to viral targets in HCV life cycle [8]. Among these new therapies, EBR/GZR combination has been approved for treating chronic HCV GT1 or HCV GT4 infection. This combination with or without weight-adjusted RBV for 12–16 weeks displays high rates of SVR in GT1-infected individuals, including treatment-naive or treatment-experienced patients. In Phase III randomized controlled open label trial, it was shown that a 12-week regimen of EBR/GZR could achieve SVR12 by 92.4% (without RBV) and 94.2% (with RBV). With 16 weeks of treatment, SVR12 was achieved by 92.4% of patients given EBR/GZR and 98.1% of patients given EBR/GZR with RBV [21]. In our cohort, the overall SVR12 rates of 12-week and 16-week regimens were 97.5% and 100%, respectively. The corresponding figures for SVR24 were 93.8% and 100%, respectively. Regarding HIV positivity, our results also showed that EBR/GZR therapy achieved comparable cure rates among patients with HCV monoinfection and HCV–HIV coinfection (93.8% and 97.2%, respectively). The SVR rates among HCV–HIV-coinfected individuals in this cohort were in line with recent reports [22,23]. Notably, adding RBV to EBR/GZR combination therapy was recommended for treatment-experienced patients at the time of the development of this protocol. However, a recent study in Asian populations revealed that the rates of SVR12 in participants received EBR/GZR with or without RBV were comparably high (100% and 96.9%, respectively) [24]. Thus, the addition of RBV to this regimen appears to have limited benefit, which should be considered against the possibility of increased adverse events during RBV-based therapy.
The combination of EBR/GZR has been shown to be highly effective across a diverse range of HCV-infected individuals, including patients with compensated cirrhosis [25], men who have sex with men (MSM) [22], people who inject drugs [26], people with inherited blood disorders [27] and those with chronic kidney disease (CKD) [28]. In addition, EBR/GZR has achieved high SVR rates and has an excellent safety profile among Asian individuals similar to previous reports in Western populations [24,29]. Interestingly, our report showed that patients with cirrhosis achieved a slightly higher SVR24 rate (100%) compared with patients without cirrhosis (93%), although the difference was not statistically significant. Regarding HCV sub-genotype, it was shown in recent Asian cohorts that SVR rates were particularly high in patients with HCV GT1b infection (97.5–98.6%) but were relatively lower (75–89%) among HCV GT1a-infected participants [24,30]. These results might refect the small number of HCV GT1a-infected participants in the mentioned reports. In our study, however, the response rates were slightly higher in HCV GT1a than HCV GT1b, which were comparable to a real-world cohort in Western countries [31]. Moreover, patients with stage 4 or 5 CKD in this report achieved 100% SVR24, which was similar to those with less-advanced CKD stages. Together, our results indicate that EBR/GZR combination is highly effective among Thai individuals infected with HCV GT1, regardless of HCV sub-genotype, prior treatment, advanced liver disease or kidney disease and HIV status.
Remarkably, our study showed that three patients who achieved SVR12 had virological relapse at week 24 during follow-up. These results were consistent with previous data conducted in Asian populations indicating that approximately 2% of participants experienced virological relapse after EBR/GZR therapy [24]. In the aforementioned report, resistance-associated substitutions (RASs) were detected at baseline or at the time of experienced virological relapse. It should be mentioned that distinguishing between virological relapse and reinfection with a new viral strain is essential to determine the true efficacy of DAA regimens [32]. To address such a question, the comparison of RNA sequences before treatment and at the time of HCV RNA reappearance is necessary. Indeed, re-infection after several months of follow-up after HCV eradication is frequently found in specific populations with high-risk behaviours including people who inject drugs (PWID) and men who have sex with men (MSM) [32]. In our patients, however, the possibility of re-infection was presumably low because they did not have any behavioural risks for HCV re-acquisition during a relatively short follow-up post-SVR12.
It has been shown that significant fibrosis regression occurs after HCV eradication following DAA therapy, which is linked to a lower risk of cirrhosis and HCC development [8]. In this study, TE was selected as the reference imaging modality for fibrosis staging due to its high accessibility, reproducibility and diagnostic accuracy [33]. Based on a meta-analysis, TE exhibited AUROCs of 0.84, 0.89 and 0.94 for prediction of ≥F2, ≥F3 and F4 fibrosis stages, respectively [33]. A previous report also showed that TE was correlated with histological collagen proportionate area in patients with chronic viral hepatitis [34]. Moreover, TE is superior to non-invasive fibrosis markers including aspartate aminotransferase to platelet ratio index (APRI) and fibro-sis-4 index (FIB-4) for identifying advanced fibrosis and cirrhosis in patients with chronic viral hepatitis [20]. In this study, we found that LS values in the whole cohort significantly decreased from baseline to the time of SVR24. Among patients achieving SVR, a decrease in LS values of at least 30% was found more frequently in the cirrhotic group than the non-cirrhotic group. These data were confirmed in our multivariate analysis indicating that the presence of baseline cirrhosis was an independent factor associated with LS reduction. Additionally, our longitudinal study demonstrates for the first time that the magnitude of LS decline following HCV eradication is comparable between HCV monoin-fected and HCV–HIV-coinfected patients, irrespective of LS at baseline.
It is well recognized that the occurrence of HCC is one of the most common liver-related complications in patients with advanced fibrosis/cirrhosis. A systematic review has indicated that patients with cirrhosis have a reduction of HCC risk of approximately 70% after achieving SVR with DAA-based therapy [35]. This evidence might suggest that although there is a descending trend in HCC occurrence after curative treatment, the cancer risk remains high to warrant ongoing surveillance using ultrasound with or without serum alfa-feto-protein [36]. Interestingly, recent reports demonstrated that LS at the time of SVR after DAA therapy could predict the clinical outcome of HCV mono- and HCV–HIV-coinfected patients [37,38]. For instance, a Spanish cohort showed that HCV–HIV-coinfected patients with LS less than 14 kPa at SVR time point had very low risk of liver-related complications including HCC development during a median follow-up of approximately 3 years [37]. Thus, LS measurement might also be helpful to select candidates for subsequent HCC surveillance programmes after achieving SVR.
Liver steatosis occurs more frequently in patients infected with HCV GT3 as this genotype could exert direct metabolic effects that lead to the development of steatosis [39]. It was shown that HCV GT3 was linked to a fivefold higher probability of having moderate-to-severe steatosis compared with other HCV genotypes [40]. In this report, approximately one-third of patients had elevated CAP values, indicating that significant liver steatosis was also common in HCV GT1-infected individuals. Previous reports demonstrated that viral eradication by IFN-based therapy could decrease liver steatosis in patients with HCV GT3 infection [39,41]. However, evidence regarding steatosis improvement in HCV GT1-infected patients treated with DAAs are restricted with conflicting results [42–44]. In this study, our data showed that steatosis was significantly attenuated after EBR/ GZR therapy, particularly among responders with significant steatosis at baseline, which was similar to a recent report of Shimizu et al. [44]. However, the mechanism by which steatosis improvement following viral eradication in patients with HCV GT1 is not yet clear.
Detection of serum HCVcAg levels has emerged as a potential marker for active HCV infection and might also be useful for monitoring response to antiviral therapy. However, the limitation of HCVcAg testing is its relatively low sensitivity of 3 fmol/l, which corresponds to approximately 3,000 IU/ml of HCV RNA [16]. At baseline, our data confirmed previous reports that serum HCVcAg had a strong correlation with the corresponding HCV RNA. In fact, 100% concordance of detectable HCV RNA and HCVcAg was observed in this study, which could be explained by the fact that all individuals had high viraemia at baseline. After therapy, our data also showed that HCVcAg testing had very high sensitivity (100%) and specificity (99.0–100%) at SVR12 and SVR24, which are in line with recent reports of IFN-free DAAs [45–48]. Based on their excellent concordance, HCVcAg appears to be a reliable and less expensive test that might replace HCV RNA quantification, particularly at SVR12.
Notably, one patient with HCV–HIV coinfection had HCVcAg positivity in the ‘grey zone’ (3.5 fmol/l) but undetectable HCV RNA at SVR24, suggestive of a false-positive assay that compromised its diagnostic performance. However, from a clinical perspective, one could encourage the use of HCVcAg as a routine testing for assessing SVR, with HCV RNA quantification as the second-line assay in those with detectable HCVcAg, thus certainly excluding a false-positive result. In fact, the European Association for the Study of the Liver (EASL) guidelines also support the clinical utility of HCVcAg as an alternative test when HCV RNA is not accessible [49]. Given high SVR rates achieved with EBR/GZR and other DAAs, HCVcAg testing might be satisfactory for the confirmation of SVR, particularly in resource-limited settings, where a cost-saving strategy is necessary.
This report had some limitations as it recruited a small number of patients, particularly the coinfected group. Moreover, the study had a relatively short follow-up duration after the completion of therapy, thus further large-scale studies with longer follow-up are needed to validate our observations. Finally, follow-up LS measurement in this report was conducted at SVR24, which might not be applicable in a real-world setting. Considering that LS declines significantly over time after 1–6 months of completing DAA therapy as shown in a recent meta-analysis [50], SVR12 may be the best time to determine post-treatment LS in most patients.
In summary, our data have confirmed previous studies that EBR/GZR is highly effective for treating patients with HCV GT1, regardless of various clinical factors, such as sex, age, the status of HIV coinfection, the presence of cirrhosis, prior HCV treatment with PEG-IFN/ RBV, baseline HCV RNA level and HCV sub-genotype. Moreover, our data showed that HCV eradication with EBR/GZR therapy was associated with signifcant improvement of stiffness and steatosis. Finally, our results also confirmed that HCVcAg testing was accurate and could be used as an alternative test for HCV RNA in detecting active HCV infection and determining SVR.
Footnotes
Acknowledgements
This study was funded by Merck & Co., Inc. and Abbott Laboratories Ltd. The study was supported by Center of Excellence in Hepatitis and Liver Cancer, Chulalongkorn University, the Grant for Chula Research Scholar (CU-GRS-61-07-30-02), the Royal Golden Jubilee Ph.D. Program (Grant No. PHD/0061/2559) and the Thailand Research Fund (TRF) Senior Research Scholar (RTA6280004).
The authors declare no competing interests.
