Abstract
Background
To evaluate the impact of baseline resistance-associated substitutions (RASs) on direct-acting antiviral (DAA) treatment response among pegylated interferon in combination with ribavirin (PR) failing patients in a real-life setting.
Methods
Blood samples and clinical data from 171 patients who failed PR treatment were collected. All of them received rescue DAA regimens. RAS identified in the NS3, NS5A and NS5B regions by Sanger sequencing method were compared by DAA regimen and HCV subtypes. We assessed sustained virological response at 12 weeks (SVR12) and evaluated the impact of baseline RASs on the effectiveness of DAA regimens in clinical practice.
Results
The overall SVR12 rates were: 89.47% (153/171), 92.1% (117/127) in patients without cirrhosis versus 81.8% (36/44) in those with cirrhosis, without significant difference (χ2=3.69, P=0.08); 87.9% in genotype (GT)1b patients (n=116) versus 93.8% in GT2a (n=32) versus 90.5% in GT3 (n=21) versus 100% in GT6 (n=2), without significant difference (χ2=1.02, P=0.84); 66.7% in asunaprevir (ASV) + daclatasvir (DCV) regimen (n=24) versus 94.0% in sofosbuvir (SOF)-based regimen (n=133), with significant difference (χ2=19.7, P=0.001). Our results showed that the prevalence of NS3, NS5A, NS5B RASs was 45.02%, 39.76% and 71.34%, respectively, and higher incidence of RASs in cirrhosis than without cirrhosis (81.8% versus 63.8%), with a statistically significant difference (χ2=4.92, P=0.03). In the ASV/DCV cohort (n=24), 4 of 11 patients (36.4%) with baseline NS3/NS5A RASs achieved SVR12, whereas 12 of 13 patients (92.3%) without RASs achieved SVR12, with significant difference (χ2=8.39, P=0.008). However, this relationship was not seen in the SOF-based subgroup (94.6% versus 92.7%; χ2=0.18, P=0.7). Treatment failure with DAAs occurred in 10.53% (n=18) of our study population, baseline NS5A substitution including L31M or Y93H (n=13) was the most frequently detected RAS, rescue regimen with velpatasvir (VEL)/SOF + ribavirin (RBV) for 12 weeks or 24 weeks was highly effective in patients who failed previous use of NS5A inhibitors, regardless of GT or cirrhosis.
Conclusions
Natural RASs are common in Chinese patients failing with PR treatment. High prevalence of clinically relevant RASs (such as L31M, Y93H) supports the appropriateness of HCV resistance tests to properly guide DAA-based therapy. These findings might be used to select salvage therapies.
Introduction
With the advent of direct-acting antivirals (DAAs), treatment of HCV infection has ushered in a watershed, which achieves sustained virological response (SVR) or cures in a high proportion of patients and with few adverse events [1], especially for the huge number of patients in China who failed with pegylated interferon in combination with ribavirin (PR) and the patients with HCV-related cirrhosis. However, a proportion of patients ranging from 1% to 15% fail to achieve SVR [2]. Virological failure is always associated with the selection of resistance-associated substitutions (RASs) [3]. Our former findings suggested [4] a high frequency of multiclass HCV RASs in patients failing PR antivirals (Additional file 1). The information on the impact of baseline RAS testing among the patients failing with PR antivirals on the outcome of first-line DAA treatment in real life in China is scarce. As a substudy of the Eleventh Five-Year Major Science and Technology projects cohort [5], which was a real-life cohort of patients failing their first PR regimen running in China, we invited the cohort participants to assess the prevalence of baseline RASs according to viral genotype and subtype and to evaluate their impact on first-line DAA efficacy in daily clinical practice.
Methods
Study population
All individuals with chronic HCV infection (including chronic hepatitis C and cirrhotic patients) and failing with PR at Beijing Youan Hospital, Capital Medical University (Beijing, China) from February 2016 to October 2019 were retrospectively analysed (ChiCTR1900020845). The diagnostic criteria of chronic HCV infection were according to the Guideline of Prevention and Treatment of Hepatitis C [6]. Exclusion criteria: patients during pregnancy or lactation, liver damage caused by infectious diseases of HAV and HEV or other causes. Cirrhosis was diagnosed by liver biopsy or transient elastography (Fibroscan; Echosens, Paris, France; ≥12.5 Kpa) combined with related clinical parameters such as ascites, hepatic encephalopathy, upper gastrointestinal bleeding or Child-Turcotte-Pugh score >7, or by any two of the following criteria: radiological imaging showing nodular liver or splenomegaly, platelet count <100,000 in the absence of other explanations, or endoscopic detection of gastroesophageal varices. Decompensated cirrhosis was defined by the presence of ascites, hepatic encephalopathy, upper gastrointestinal bleeding or Child-Turcotte-Pugh score >7 [7–9]. Diagnoses of ascites, splenomegaly and portal hypertension were based on ultrasonography. Patients with PR failure included non-response, virological breakthrough, relapse and intolerance. For this study, baseline serum samples from all individuals treated with DAAs were chosen. Furthermore, samples from patients that once failed DAA treatment were collected again. The study was conducted in accordance with the Declaration of Helsinki, Good Clinical Practice guidelines and local hospital ethics committee approval. All patients provided written informed consent.
Treatment and follow-up
DAA regimen was decided according to the DAA available in the study period, the potential for interactions with concomitant medications, the severity of liver disease and comorbidities, following guidelines [10,11]. Plasma viral load was evaluated at baseline and, at least, at the end of therapy and at week 12 post-treatment. SVR12 was defined as undetectable plasma HCV RNA 12 weeks after the end of therapy. The study flow chart is shown in Figure 1.

Study flow diagram
RASs testing and analysis
Resistance testing of RASs (baseline and emerging) was performed at the Beijing Institute of Hepatology, and was performed on all available samples at baseline, that is, on samples collected prior to treatment initiation and at treatment failure. Total RNA was isolated from plasma samples using the QIAamp viral RNA mini kit (Qiagen, Hilden, Germany). Viral genes from patient samples were amplified by the nested PCR method and then sequenced by the Sanger sequencing method (TAKARA, Tokyo, Japan) with the primers NS3F1/ NS3R1, NS5AF1/NS5AR1 and NS5BF1/NS5BR1. A sensitivity of approximately 15-20% can be assumed for this method. The NS3, NS5A and NS5B sequence results were aligned and analysed using Seq Scape VR Software v2.6 (Applied Biosystems, Thermo Fisher Scientific, Waltham, MA, USA) to generate consensus sequences.
To detect relevant substitutions and evaluate their implications, the consensus sequences were submitted in the web-based mutation detection algorithm, Geno2Pheno [hcv] 0.92 (G2P) [12]. Substitutions scored by G2P were further interpreted as clinically relevant RASs by relating scores with guidelines of the consensus statement [6,8,9], in addition to RASs reported to bear impact on DAA treatment outcome in vitro and/or in vivo in the literature [13–15].
Statistics
Data were expressed as median (with range) or numbers (with percentages), as appropriate. Continuous and categorical variables were compared using Student's t-test (or the Mann-Whitney U test, as appropriate) and the χ 2 test (or Fisher's exact test, as appropriate), respectively. All statistical analyses were performed using Statistical Package for Social Sciences (SPSS) version 20.0 (SPSS, Armonk, NY, USA). P-value <0.05 was considered statistically significant.
Results
Characteristics of the study population
Finally, 171 patients with chronic HCV infection were included for the analysis of DAA efficacy and baseline RASs. Their baseline demographic characteristics are summarized in Table 1. The median patient age was 53 (21-80) years, more than half of them were males (n=98, 57.31%), average HCV RNA viral load was 6.3 +0.7 (log10 IU/ml) and a total of 116 patients (67.84%) were genotype (GT)1b. All of them had prior treatment failure experience with PR (including 99 relapses, 45 non-response and 27 PR intolerance), 25.73% (n=44) with cirrhosis including 12 decompensated cirrhosis, all of which were class B by Child-Pugh.
Baseline characteristics of the patients (n=171)
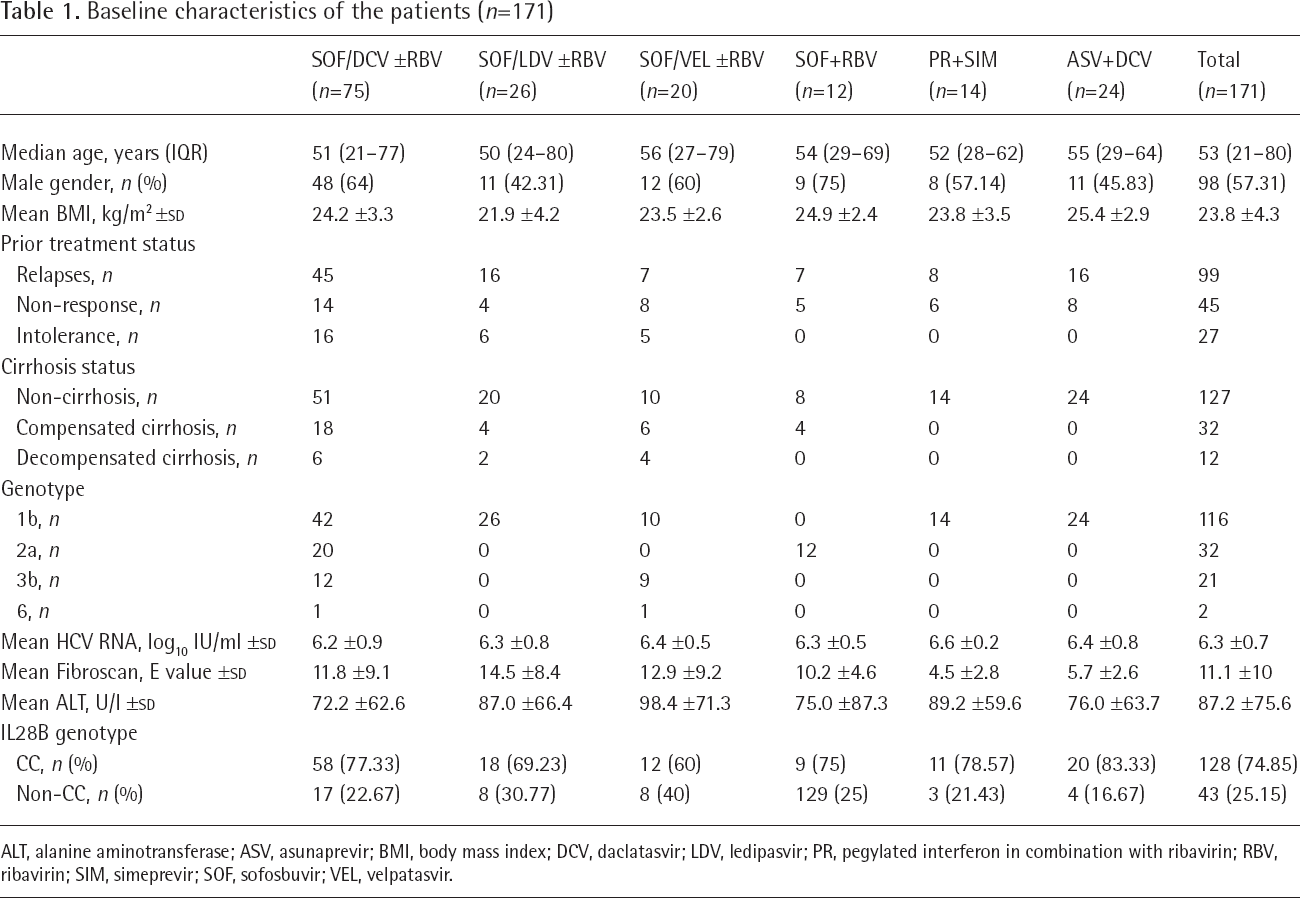
ALT, alanine aminotransferase; ASV, asunaprevir; BMI, body mass index; DCV, daclatasvir; LDV, ledipasvir; PR, pegylated interferon in combination with ribavirin; RBV, ribavirin; SIM, simeprevir; SOF, sofosbuvir; VEL, velpatasvir.
Baseline RASs and DAA efficacy
Among the 171 patients treated with DAAs, the overall SVR rate was 89.47% (153/171), including 92.1% (117/127) in the non-cirrhosis group and 81.8% (36/44) in the cirrhosis group, without significant statistical difference (χ 2 =3.69, P=0.08). The SVR of different genotypes were GT1b: 87.9% (102/116), GT2a: 93.8% (30/32), GT3: 90.5% (19/21) and GT6: 100% (2/2), without significant statistical difference (χ 2 =1.02, P=0.84). The SVR of different treatment regimen was sofosbuvir/daclatasvir (SOF/DCV) ± ribavirin (RBV): 96% (72/75), SOF/ledipasvir (LDV) ±RBV: 88.5% (23/26), SOF/velpatasvir (VEL) ±RBV: 100% (20/20), SOF+RBV: 83.3% (10/12), PR+ simeprevir (SIM): 85.7% (12/14), asunaprevir (ASV)+DCV: 66.7% (16/24), the difference was statistically significant (χ 2 =19.72, P=0.001). These data are represented in Figure 2.

The SVR12 of efficacy of DAA hepatitis C treatment
To determine the prevalence of NS3, NS5A and NS5B RASs in patients prior to initiating DAA therapy, we defined the presence or absence of specific RASs using a set of pre-defined amino acid substitutions. A total of 128 (74.85%) specific RASs (77 in NS3, 68 in NS5A and 122 in NS5B) were included in this analysis. In total, the incidence of RASs was higher in cirrhosis than in patients without cirrhosis (81.8% versus 63.8%), with a statistically significant difference (χ 2 =4.92, P=0.03). For NS3, the most abundant RASs were S122G (36.26%, 62/171), and then V170I (28.01%, n=48), Y56F (8.19%, n=14), R117H (6.43%, n=11/), T54S (5.85%, n=10), V36L (5.26%, n=9), V55A/I (4.09%, n=7), D168Q (2.34%, n=4) and Q80L (1.17%, n=2). Moreover, the largest increases were detected for V36L in HCV GT2a-infected patients than any other GTs (18.75% versus 2.15%; χ 2 =5.17, P=0.02), Q80L only appeared in GT1b, and all of the D168Q were just in GT3.
For mutations occurring in NS5A, Q54H/N/L (31.58%, n=54), Y93H (14.06%, n=24), L31M/V (5.26%, n=9), R30H/A30K (10.53%, n=18), L28M (6.43%, n=11) and P58S (4.68%, n=8) were the most abundant RASs. L28M was found in 3.4% (4/116) of GT1b, 3.1% (1/32) of GT2a, 28.6% (6/21) of GT3 and none of GT6 sequences (χ 2 =12.67, P=0.002). Although R30H was found in only 6.9% (8/116) of HCV-1b, A30K was present in 47.61% (10/21) of GT3 sequences. The most important RASs associated with clinical resistance to NS5A inhibitors were L31V/M and Y93H. L31M was highly represented in GT2 (9.38%, 3/32) and GT3 (14.29%, 3/21) but rare in GT1b (2.59, 3/116). NS5A 93H had a prevalence of 15.52% (18/116) in GT1b, 23.81% (5/21) in GT3.
The prevalence of natural RAS to NS5B nucleoside analogue inhibitor (NI) and non-nucleoside analogue inhibitor (NNI) was also differentially distributed among HCV genotypes. Among those variations affecting NIs, clinical resistance to SOF is associated with mutations in NS5B-S282 and L159F facilitates resistance. S282T was present in just one isolate for GT3 and two isolates for GT1b (frequencies of 4.76%, 1.72%, respectively), and L159F was identified in only 3.45% (4/116) of GT1b, 9.38% (3/32) of GT2a and 14.29% (3/21) of GT3 sequences. In contrast to NIs, natural resistance-associated variants (RAVs) to NNIs were common and varied by virus subtype. Interestingly, C316N was found in 82.76% (92/116) of GT1b, while M414Q (n=17), A421V (n=21) were highly dominant in the other genotypes (31.25%, 34.38% for GT 2 and 21.88%, 47.62% for GT3, respectively).
Overall, 45.61% of patients (n=78) harboured RASs in two or more drug classes. Among them, NS3/NS5B RASs combination was most common (34/78, 43.6%), followed by NS5A/NS5B (21/78, 26.9%) and NS3/ NS5A (16/78, 20.5%), while 9% (7/78) of patients harboured RASs in three drug classes.
Effect of the baseline NS5A/NS5B-associated RASs for SOF-based regimens
To evaluate the impact of baseline NS5A- and NS5B-associated RASs on the effectiveness of SOF-based regimens including DCV/SOF ±RBV or LDV/SOF ±RBV or VEL/SOF ±RBV or SOF+RBV, we analysed a subset of key RASs that were relevant for the DAA regimen that the patients received in clinical practice. In the SOF-based cohort (n=133), overall SVR rate was 93.98% (125/133), 94.6% patients (87/92) with NS5A- or/and NS5B-associated RASs achieved SVR12, and 92.7% subjects (38/41) without RASs (χ 2 =0.18, P=0.7; Figure 3A).

Efficacy of DAA regimens with baseline RASs
The most distinct differences between non-cirrhosis and cirrhosis groups are: an overall SVR rate of 100% (89/89) and 81.8% (36/44), respectively (χ 2 =17.21, P<0.001); the presence of baseline NS5A or/and NS5B RASs were 62.9% (56/89) and 81.8% (36/44), respectively (χ 2 =4.93, P=0.03). In the cirrhosis cohort (n=44) the presence of baseline NS5A or/and NS5B RASs was associated with 6.9% (χ 2 =1.17, P=0.9) lower SVR12 rates (80.6% [(29/36] versus 87.5 [7/8]); while in the non-cirrhosis cohort, both SVR12 were 100%, whether with RASs or not (Figure 3B).
Figure 3C shows the overall differences among various GTs for the presence of baseline NS5A or/ and NS5B RASs and SVR12 rates were not statistically significant (χ 2 =5.29, P=0.18; χ 2 =0.70, P=0.8): GT1: RASs prevalence 73.1% (57/78), SVR12 94.9% (74/78); GT2: 65.6% (21/32), 93.8% (30/32); GT3: 66.7% (14/21), 90.5% (19/21); GT6: 0% (0/2), 100% (2/2). Otherwise, baseline RASs according to different GTs (1b, 2a and 3) were not related to SVR12 (GT1b: 94.7% versus 95.2%, GT2a: 90.5% versus 100%; GT3: 85.7% versus 100%; P>0.05).
Effect of the baseline NS3/NS5A-associated RASs for NS3 PI-based regimens
38 GT1b non-cirrhosis patients received protease inhibitor (PI)-based treatment (Figure 4), including 14 patients with PR+SIM and 24 with ASV+DCV. The overall SVR12 of the former was 85.71% (12/14), the presence of NS3 RASs was associated with a small reduction in SVR12 (83.3% [5/6] versus 87.5% [7/8]), with no significant difference (χ2=0.049, P=0.69). In the ASV/DCV cohort (n=24), 4 of 11 patients (36.4%) with baseline NS3/ NS5A RASs achieved SVR12, whereas 12 of 13 patients (92.3%) without RASs achieved SVR12 (Figure 4), with significant difference (χ2=8.39, P=0.008).

Efficacy of DAA regimens with baseline RASs
Baseline RASs and SVR12 of 18 patients receiving retreatment after previous DAA failure
We analysed the baseline RAS patterns among 18 DAA failure patients who had available information on the failure type (Table 2), and then gave them retreatment with new DAA regimens; all of them achieved SVR12.
Treatment and re-treatment of failing patients and clinically relevant RASs

ASV, asunaprevir; DAA, direct-acting antiviral; DCV, daclatasvir; GT, genotype; LDV, ledipasvir; PR, pegylated interferon in combination with ribavirin; RAS, resistance-associated substitution; RBV, ribavirin; SIM, simeprevir; SOF, sofosbuvir; SVR12, sustained virological response at 12 weeks; VEL, velpatasvir.
The SVR12 for GT1 non-cirrhosis patients previously treated with NS5A and NS3/4 inhibitors (ASV/ DCV) was 66.67% (16/24). All of the eight DAA failure patients had a relapse and exhibited higher prevalences of DAA-specific RASs (87.5%, 7/8) before ASV/ DCV treatment. The NS3/4A resistance profile was Q80L (1/8), S122G (5/8), V170I (2/8), R117H (1/8), while NS5A resistance profile was Y93H (6/8), L31M (1/8), L28M (1/8) and Q54H (1/8) variants. After ASV/ DCV failure, NS5A RASs at Y93 mainly detected and showed increasing RAS prevalence, such as L31M, L28M and R30H. After previous ASV/DCV failure, one patient harbouring baseline S122G+L31M+Y93H RASs received the regimen of SOF+RBV by himself, which HCV RNA decreased but did not below limit of detection at weeks 4, and then rescue regimen was adjusted to SOF/VEL, finally obtained SVR12. The rest of the seven patients were given SOF/DCV or SOF/LDV for 12 weeks, all of them obtained SVR12.
Eight patients with cirrhosis had unsuccessful attempts of achieving SVR12 using previous SOF-based regimens. Interestingly, one decompensated GT1b cirrhosis without baseline RAS had received SOF/DCV without RBV and experienced a viral breakthrough. After DAA failure, S122G+L28M+Y93H+L159F was detected, fortunately, retreatment with SOF/VEL+RBV for 24 weeks (after 6 weeks of combined regimen, RBV was discontinued due to gastrointestinal symptoms and anaemia) was successful in this patient. Three GT1b compensated cirrhotic patients harbouring baseline NS5A and NS5B RAS (L31M, Y93H, L159F, S282T, R30H) had failed SOF/LDV+RBV regimens. Two of them did achieve SVR12 with SOF/VEL+RBV for 24 weeks re-treatment, even if some key RASs were still detected. Another one received the regimen of SOF+RBV by himself, which HCV RNA decreased but did not below limit of detection at week 4, and then rescue regimen was adjusted to SOF/VEL+RBV for 24 weeks, finally obtained SVR12.
For 32 HCV GT2a patients, most of the patients (20/32) were treated with SOF combined with DCV with or without RBV, all of them achieved SVR; notably, among the 12 patients treated with SOF+RBV, 8 patients with non-cirrhosis achieved SVR12, while in 4 patients with compensated cirrhosis, just 50% (2/4) reached SVR12. The total SVR12 was 83.3% (10/12). Both of the two patients with compensated cirrhosis harboured baseline RASs, like L31M+L159F+A421V, L28M+Y93H+L159F, respectively. Fortunately, re-treatment with SOF/VEL+RBV for 12 weeks was successful.
All of the 21 GT3 patients had received SOF-based treatment. Among them, nine were treated with SOF/ VEL with or without RBV, 100% (9/9) did achieve SVR12. For most patients (n=12), SOF was combined with DCV with or without RBV for 12 or 24 weeks, SVR12 was 83.33% (10/12). Two cirrhotic patients who harboured RASs like A30K+L31MS+282T and A30K+Y93H+L159F+C316N, respectively, were treated with SOF/DCV+RBV and experienced a relapse, and then they received a combination with SOF and VEL with RBV for 24 weeks and did achieve SVR.
Discussion
The high error rate of the HCV polymerase coupled with virion production results in a complex mixture of viral genetic populations (termed ‘quasi-species’) that pre-exist within an infected individual before treatment initiation [16]. Almost all patients who fail treatment acquire RASs, for example, NS5A RAS that will persist for years [17]. Even treatment-naive patients could have RASs against currently approved DAAs, that is, resistance at baseline [18,19]. Pre-existence of NS5A RASs with high fold resistance, together with other negative factors, such as high fibrosis stage, GT3, or previous treatment with non-NS5A DAAs, could reduce the efficacy of DAAs. Baseline resistance testing prior to HCV treatment initiation with all oral DAA regimens is still a matter of debate, at least for certain scenarios, especially for Chinese patients who had DAA-based therapy applied for a short time. The prevalence and impact of baseline RASs in post-marketing, diverse, real-world cohorts of patients treated outside of clinical trials remains unclear. Leveraging the data and specimen repository of the prospective, observational cohort study of PR failing patients, we report the prevalence and impact of baseline NS3, NS5A and NS5B RASs in real-world clinical practices in patients treated with DAAs.
In the present study, a total of 89.47% emerging RASs (45.02% in NS3, 39.76% in NS5A and 71.34% in NS5B) were detected among 171 PR-failure patients. High prevalence (81.8% versus 63.8%) of RASs was observed in cirrhotic patients. For NS3, we found that baseline Q80L RAS was uncommon (just two patients) in GT1b patients, and the other clinically important NS3 RASs including R155K, A156S/T/V were not observed in isolates of GT1b, while RAS D168 was only detected in one GT3b sample with compensated hepatic cirrhosis. This was consistent with previous research that NS3 RAS prevalence for treatment-experienced patients who failed therapy with PI drugs was higher compared with non-experienced patients likely due to drug-selective pressure [20]. According to the literature, Q80 is highly associated with resistance to simeprevir [21], one patient who harboured baseline Q80L in our study had failed the PR+SIM regime. However, a rescue therapy with NS5B and NS5A inhibitors was selected and successfully achieved SVR even Q80L+S122G+R117H persisted. Therefore, due to low prevalence of this mutation in GT1b strains reported in previous studies and corroborated here, there is no need to incorporate pretreatment resistance tests for infected patients with subtype-1b of HCV in China. Even with the identification of this variant, the use of NS5A+NS5B is not limited since there is no evidence of resistance [22].
For NS5A, baseline RASs at positions 28, 30, 31, 54, 58, 93 were found in 39.76% of our patient population, which is in agreement with the range of 15–45% in published reports [17,23]. Pre-treatment NS5A RASs have been shown to reduce the SVR rate in GT1b patients who were treated with DCV/ASV (36.4% versus 92.3%), however, none of them were associated with our SOF-based population (92.7% versus 94.6%). Treatment failure with DAAs occurred in 10.53% (n=18) of our study population, baseline NS5A substitution including L31M or Y93H (n=13) was the most frequently detected RAS. More importantly, RASs in NS5A increased after DAA treatment failure (n=16), suggesting that these substitutions selected by DAAs are long-lasting and are likely to affect the results of rescue treatment [17]. Indeed, in our study, two GT1b patients harboured L31M+Y93H/R30H+Y93H, respectively, who failed previous DAA therapy including ASV/ DCV or SOF/LDV obtained viral breakthrough during SOF+RBV re-treatment. Retreatment with VEL/ SOF+RBV for 12 weeks or 24 weeks was highly effective and safe in patients who failed previous use of NS5A inhibitors, regardless of GT or cirrhosis.
In the present study, NS5B RASs were detected in 122 strains (71.34%), notably, due primarily to the high prevalence of C316N (63.16%), which was considered to possibly confer resistance to dasabuvir, a non-nucleoside inhibitor of NS5B (NS5B-NNI). Bertoli et al. [24] reported that C316N was observed at a frequency of 10.9 to 35.6% as a naturally occurring variant. The frequency of C316N was significantly higher in our population, concordant with previous literature data in a Chinese population [25]. The C316N variant may be more fit in Chinese GT1b patients with HCV infection, which should be considered when dasabuvir is used to treat Chinese patients, although it may be a polymorphism commonly seen in GT1b in some parts of the world.
Among the different classes of DAAs, HCV NS5B nucleotide inhibitors (NS5B-NI) appear to be the preferred backbones for interferon-free treatment in terms of potent therapeutic response and a high barrier to resistance. The development of RASs to SOF is rare. It seems only S282T confers resistance in vitro [26] and it was detected in a low number of patients after treatment failure only [15,18,24]. We identified S282T only in a few GT1b (2) and GT3b (1) patients, two of them failed with LDV/SOF or DCV/SOF. At the same time in both individuals who had recurrence after therapy with second-generation DAAs (SOF/VEL) the double mutation S282T and L31M/Y93H was detected simultaneously (100%). Moreover, L159F occurred in association with an SOF treatment failure in GT1b and GT3 without conferring resistance in vitro but the half-maximal effective concentration (EC50) was slightly increased together with S282T [27]. We observed slight increases of L159F, and it was frequently selected in combination with L31/Y93 in GT1b-infected patients who failed SOF-containing regimens, which was reported in another study [28]. Overall, the selection of SOF-associated RASs is rare and the importance of selected variants remains unclear. However, it is important to note that a retreatment with SOF seems to be associated with an increased likelihood for treatment failure and selection of S282T [15,16].
In this study, eight individuals (seven harboured RASs at the baseline) who previously failed treatment with ASV+DCV achieved SVR12 with rescue regimens such as SOF/DCV or SOF/LDV or SOF/VEL, even if all of them had double mutations like L31M, Y93H, Q54H, L28M, R30H before retreatment. At the same time, all eight individuals who did not achieve SVR after an SOF-based regime (SOF/DCV, SOF/LDV, SOF+RBV) had at baseline a combination of ‘difficult-to-treat’ factors that we presumed to impact the efficacy of retreatment: compensated cirrhosis (Child-Pugh B), double NS5A mutation, previous failure of second-generation NS5A inhibitor, GT3b, the mutation S282T or L159F was detected. Nevertheless, only two individuals in our study who had chosen SOF+RBV for their rescue therapy did not achieve SVR, all of them did achieve SVR12 once switched to SOF/VEL+RBV for 12 or 24 weeks.
In conclusion, we considered re-treatment options with broadly approved DAAs based on RAS analyses. In DAA-naive patients moderate RAS frequencies and several treatment alternatives enable a RAS-free treatment for all GTs, except for DCV+ASV regimen which was significantly affected by the RASs in NS5A. For DAA-experienced patients, the retreatment options according to the RAS-free approach are decreasing, especially for those patients who had at baseline a combination of ‘difficult-to-treat’ factors. Moreover, SOF/ VEL+RBV for 12 or 24 weeks was highly effective and well tolerated in Chinese patients who previously failed treatment with a previous DAA regimen. The presence of NS5A or NS5B RASs at baseline, including those associated with virological failure with other DAA regimens, did not impact treatment outcomes.
Footnotes
Acknowledgements
We are grateful to all the subjects who participated in this study. This work was supported by the Beijing Municipal Administration of Hospitals Incubating Program (Code: PX2017022), Open research projects of Key Laboratory in Capital Medical University (Code: 2-03-02-BJYAH2016002PX2017022), Thirteenth Five-Year Major Science and Technology projects (Code: 2017ZX10202201, 2017ZX10201021-001-008); China Primary Health Care Foundation- Youan Foundation of Liver Disease and AIDS:Scientific Research Project of Beijing Youan Hospital, CCMU, 2018 (Code: YNKTQN20180209). The funders had no role in the study design, data collection and analysis, decision to publish or preparation of the manuscript.
The authors declare no conflicts of interest in relation ot this work.
