Abstract
Introduction
Currently, an estimated 250 million people are chronically infected with HBV in spite of the availability of an effective vaccine that prevents transmission. Mortality related to chronic HBV infection remains high and may even be increasing: approximately 800,000 people die from the consequences of the infection annually, which exceeds the yearly death toll from diseases like malaria and HIV [1]. Nucleotide or nucleoside analogues (NUCs) are currently the most frequently applied treatment option for chronic HBV [2]. Antiviral agents like tenofovir and entecavir effectively inhibit replication of HBV in almost all chronic patients [3]. Nucleotide analogues, such as tenofovir, contain one to three extra phosphate groups compared with nucleoside analogues, such as entecavir. Both nucleotide and nucleoside analogues act as chain terminators when incorporated into newly created DNA strands by viral DNA polymerase. HBV DNA is undetectable in serum within months following the start of treatment and, importantly, NUC treatment greatly reduces the incidence of deadly complications, like end-stage liver disease and hepatocellular carcinoma [4]. Current guidelines [2] advise to start treatment when high serum levels are detected of HBV DNA or alanine aminotransferase (ALT), an enzyme abundant in the cytosol of hepatocytes, released upon lysis. NUC treatment specifically suppresses replication of the viral genome but does not lead to complete viral eradication as evidenced by the persistence of a viral mini-chromosome, called covalently closed circular DNA (cccDNA), and the retention of integrated viral sequence in host DNA in hepatocytes [5]. Both cccDNA and integrated viral DNA can act as templates for the transcription of viral proteins, like HBV surface antigen (HBsAg), which is continuously produced and secreted in serum at all stages of the infection, even during effective treatment with NUCs [6]. Only cccDNA can act as the source of infectious virions while integrated viral DNA cannot [5]. Resolution of a chronic HBV infection is defined as the loss of HBsAg from the blood. Unfortunately, NUC treatment only results in HBsAg loss in a minority of patients (0–3%) with a virological response [7–9]. Although pegylated interferon (IFN) infections result in a durable loss of HBsAg in 7–17% percent of the individuals [10,11], NUCs are generally preferred, because they are much better tolerated than weekly IFN injections. With the aim to improve low curative treatment rates for chronic HBV, a multitude of novel compounds are currently under development [12]. The mechanisms of action of these novel experimental antivirals vary greatly, ranging from activation of the host immune system by Toll-like receptor (TLR) ligation to small molecules that block translation of viral proteins, like silencing RNA. The goal of these new compounds is to clear HBsAg from the host serum, as this event is associated with a clear improvement of the prognosis. HBsAg loss is also called a functional cure, because even after HBsAg has become undetectable in serum, HBV reactivation can still occur during periods of immune suppression by renewed transcription of the remaining cccDNA. It is generally accepted that a combined activity of compounds with distinct mechanisms of action are needed to achieve a functional cure. Since NUC treatment is effective in blocking viral replication and is able to limit the spread of HBV, it will likely remain the backbone of these novel therapeutic regimens [13]. To select novel candidates for the combined treatment strategy, it is important to have a detailed understanding of the effects on the host immune system associated with NUC treatment. This goal is particularly relevant with regard to immune cell populations capable of producing antiviral cytokines or lysing virally infected cells, like virus-specific T-cells and natural killer (NK) cells, as they are a likely target for future immune therapeutic intervention. In this review, we summarize and discuss the literature regarding effects of NUCs on the cellular immune system, with the aim to guide the development of treatment strategies for chronic HBV that combine antiviral and immunological compounds.
The effect of NUCs on HBV-specific T-cells
The adaptive immune response plays an important role in viral clearance and disease pathogenesis of HBV infection. This is supported by several key observations. In acute HBV infections, HBV-specific T-cell responses appear directly after an increase in serum HBV DNA levels [14,15] and, as in many other viral infections like HCV and HIV, acute HBV infections are characterized by multi-specific and polyfunctional HBV-specific T-cell responses, while these responses are not observed when viral replication persists [16,17]. During the chronic phase, the frequency of HBV-specific T-cells in the blood is usually very low [18], and in early studies of NUC treatment it was observed that these frequencies improved in the majority of the treated individuals [19]. In order to reliably detect and study HBV-specific T-cells obtained from chronic HBV patients, these cells generally need to be expanded in vitro prior to flow cytometric or functional evaluation by, for example, ELISPOT assays. In addition to their relatively low frequencies in the blood, other host and viral factors have been proposed to explain the weak T-cell responses to HBV. These factors include the occurrence of HBV escape mutants [20], the dysfunctional interaction of T-cells with NK cells or dendritic cells (DC) together with a weak type I IFN induction [20,21], exhaustion of T-cells as a consequence of continuous high exposure to high viral antigen load observed in mouse models [22] and active suppression by regulatory T-cells or immunosuppressive cytokines like interleukin (IL)-10 or tumour growth factor (TGF)-β [23]. The observation that virus-specific T-cells are activated during acute HBV [14,15] and the abovementioned mechanisms of immune impairment during chronic infection have been followed up by numerous studies that focus on HBV-specific CD4+ and CD8+ T-cells. The majority of these studies used blood leukocytes from chronic HBV patients who are not on NUC treatment, and therefore display variable levels of serum HBV DNA and ALT, characteristic for the natural history of chronic HBV. However, NUC-treated chronic HBV patients are on treatment for many years or even lifelong, and generally have undetectable serum HBV DNA and normalized ALT levels in the absence of other liver-related comorbidities, yet viral eradication is rarely complete since HBsAg is readily detected in serum of NUC-treated patients. Therefore, data on virus-specific T-cells derived from treated patients, preferably in a longitudinal setting, is of particular relevance.
A number of studies have examined the functionality of CD4+ T-cells in chronic HBV patients on NUC therapy [23–30]. As presented in Table 1, in the majority of studies involving CD4+ T-cells, peripheral blood mononuclear cells (PBMC) were stimulated for 1–6 days using viral antigen (HBcAg or HBsAg) and IFN-γ production was subsequently quantified using ELIS-POT [30,31]. NUC treatment significantly increased the production of IFN-γ by HBV-specific CD4+ T-cells in seven longitudinal studies that performed this assay, which suggests that some degree of immune reconstitution does occur. Importantly, three of these studies showed a slight increase in IFN-γ producing HBV-specific CD4+ T-cells at start of treatment, only to see it decline to pre-treatment levels during [29] or after [25] treatment. One cross-sectional study reported unaltered IFN-γ production by specific CD4+ T-cells using a comparable assay [17]. Using a tritium-thymidine based assay, two studies [27,32] reported that NUC treatment improved the proliferative capacity of HBV-specific CD4+ T-cells, while this effect was transient in one study [25] (Table 1). Nonetheless, there is still a possibility that T-cells chronically exposed to high antigen load are long-lasting or permanently damaged and unable to undergo a sustained functional restoration. This possibility is supported by the data in lymphocytic choriomeningitis virus (LCMV)-infected mouse models that transferred impaired CD8+ T-cells from chronically infected mice to a naive uninfected mice which, surprisingly, did not restore T-cell differentiation [33]. In addition, although NUC treatment can control viral replication, HBV e antigen (HBeAg), but especially high serum levels of HBsAg remain present for a long time. To date, the effect of continuous high antigen load on T-cell function is unclear, but it is likely to be a contributing factor in the incomplete recovery of T-cells as observed during NUC treatment.
Longitudinal studies on the effect of NUCs on HBV-specific CD4+ T-cells in the peripheral blood

Tritiated thymidine (3H-TdR) assay. ADV, adefovir; ELISPOT, enzyme-linked immunosorbent spot; FTC, emtricitabine; HBcAg, hepatitis B core antigen; HBeAg, hepatitis B e antigen; HBsAg, hepatitis B surface antigen; ICS, intracellular cytokine staining; IFN-γ, interferon-gamma; LAM, lamivudine; LDT, telbivudine; NA, not applicable; NUCs, nucleotide or nucleoside analogues; ↑, increase.
Current literature provides strong evidence that virus-specific CD8+ T-cells are important effectors during spontaneous clearance of HBV from the liver. In this regard, it has been shown that HBV infected chimpanzees display a delayed HBV clearance after CD8+ T-cell depletion [34,35], and that acutely infected patients show an increase of virus-specific CD8+ T-cells visualized using HLA-A2/HBV tetramers during viral load decline [14]. Comparable to observations for virus-specific CD4+ T-cells, the frequencies of dextramer-positive CD8+ T-cells are very low during chronic HBV infection [17,18], irrespective of whether patients are on-treatment or not [17]. Importantly, in patients who experienced loss of serum HBsAg during NUC treatment, the frequency of HBV-specific CD8+ T-cells was higher, which was an early indication that the frequency of HBV-specific CD8+ T-cells can improve during treatment [17]. It is also worth noting that the low CD8+ T-cell frequency correlates with the activation status directly ex vivo. This could contribute to activation-induced apoptosis during the assay which might lower the measured frequency even more [18]. The frequency of HBV-specific CD8+ T-cells might also be affected by the targeted epitope: HBsAg-specific CD8+ T-cells are generally low or undetectable during chronic HBV, while core and polymerase-specific CD8+ T-cells are more frequent [35]. It has been well established by numerous studies in the last decade that during untreated chronic infection, HBV-specific CD8+ T-cells are functionally impaired, in contrast to CD8+ T-cells with other specificities, and that exhaustion markers, such as PD-1 and CTLA-4, are highly expressed [36–38]. Importantly, only few longitudinal studies investigated the effects of NUC treatment on these parameters [24–26,29,39,40] (Table 2). Three studies using ELISPOT assays on CD8+ T-cells were conducted, of which only one study reported significant recovery [24]. These studies used different HLA-A2-specific peptides for the stimulatory assays, which is important as the specific targets of HBV-specific CD8+ T-cells affect their function [41]. One study by Boni et al. [17] showed the results of an ELIS-POT assay using a broad range of HLA-A2 restricted HBV epitopes in a cross-sectional set up. The authors showed that in those patients who cleared HBsAg following NUC treatment, the production by CD8+ T-cells of IFN-γ, IL-2 and tumour necrosis factor (TNF) was higher than in patients where HBsAg persisted, which suggests restoration of CD8+ T-cells in the absence of HBsAg. In addition, the capacity of CD8+ T-cells to lyse target cells in an antigen-dependent manner was evaluated in three studies [26,39,40] (Table 2). Virus-specific lysis rates improved during treatment in all three studies, but this positive effect was not durable during the follow-up of several months of treatment. With regard to IFN-γ producing HBV-specific CD8+ T-cells (Table 2), an increase at week 14 and 48 was observed in one study [24], but no differences were observed during NUC treatment in two other studies [25,29]. A study by Rigopoulou et al. [25] included five individual patients and detected no increase in IFN-γ production and only a transient improvement of CD8+ T-cell proliferation. One study included the analysis of five paired liver biopsies [42]. The authors reported a treatment-induced increase of intrahepatic CD8+ T-cells by examining formalin fixed and paraffin embedded (FFPE) tissue, and of those patients that cleared serum HBeAg on treatment, the increase was more pronounced, which suggests that CD8+ T-cells may contribute to this important clinical event, which is associated with an improved long-term prognosis. Unfortunately, no additional functional tests on intrahepatic CD8+ T-cells directly ex vivo were performed. Importantly, in several studies discussed in this review, the improvement of CD4+, but especially CD8+ T-cell function was only transient and returned gradually to pre-treatment levels while on NUC-induced viral suppression [25,32]. It is currently unclear what causes this decline, especially since the viral load reduction, the normalization of liver inflammation and cytokine concentrations in the serum are durable. The restoration of weakened functionality of T-cells during therapy for chronic viral infection is in line with data in HCV-infected patients who received direct-acting antivirals [43] and the combined results of the studies on HBV suggest that T-cells could be a target for immune restoration.
Publications on the effect of NUCs on CD8+ HBV-specific T-cells from patients with chronic HBV

Chromium-51 ( 51 Cr) release assays.
Tritiated thymidine (3H-TdR) assay. ADV, adefovir; c, core; ELISPOT, enzyme-linked immunosorbent spot; HBeAg, hepatitis B e antigen; ICS, intracellular cytokine staining; IFN, interferon; LAM, lamivudine; LDT, telbivudine; NUCs, nucleotide or nucleoside analogues; p, polymerase; s, surface; ↑, increase.
The effects of NUCs on regulatory T-cells
The importance of regulatory T-cells in regulating HBV-specific T-cell responses is well recognized [44]. Regulatory T-cells are CD4+ T-cells that express the cell membrane marker CD25 in combination with the transcription factor forkhead box protein P3 (FOXP3). FOXP3 is located in the nucleus of CD4+(CD25+) T-cells, and fixation and permeabilization is necessary to definitively identify these cells. Early studies have convincingly shown that depletion of regulatory T-cells in vitro results in improved HBV-specific CD8+ T-cell function [23,45,46]. In vivo, relatively high frequencies of regulatory T-cells are inversely correlated with liver inflammation during acute HBV infection [23], suggesting that these can mediate liver inflammation during viral fares. In line with this observation, higher frequencies of intrahepatic regulatory T-cells are detected in patients with high viral load [47]. One possible mechanism of this increase is that during liver inflammation and fibrosis progression mouse liver derived stellate cells produce more TGF-beta [48]; this cytokine has been shown to induce differentiation of CD4+ T-cells towards a FOXP3+ regulatory phenotype in vitro [49]. Functional tests to determine the capacity of regulatory T-cells to suppress the functionality of other T-cells are relatively difficult to perform, especially when investigating liver-derived cells. Numerous studies have determined the relative frequency of regulatory T-cells in the peripheral blood during NUC treatment, albeit not always using the same molecules (CD4+/CD25+/FOXP3+/CTLA+/ CD127low) to define regulatory T-cells. As presented in Table 3, the great majority of studies published today demonstrated that NUC-mediated viral load reduction leads to a decrease of regulatory T-cell frequency within the CD4+ T-cell population [32,50–59]. This finding is robust and is observed even when using variable definitions of regulatory T-cells during cytometric analysis, and is irrespective of percentages of HBeAg-positive patients, the degree of viral suppression or treatment duration. It is important to mention that, to the best of our knowledge, no functional assay-based results before and on NUC treatment have been published to date. In addition, as it is proposed that regulatory T-cells may exert local immunosuppressive effects on other lymphocyte populations residing in the liver, like HBV-specific T-cells and NK cells, assessment of liver-derived regulatory T-cells before and on NUC treatment is currently needed to create a complete picture of effects related to antiviral treatment. It is possible that regulatory T-cells contribute to the improved activity of HBV specific (CD4+) T-cells during NUC therapy. Simultaneous assessment of their phenotype/function during NUC treatment would greatly improve the current knowledge on this subject, as well as provide clues to the mechanism of fibrosis regression that has been observed during NUC trials for HBV.
Longitudinal studies on the effect of NUCs on regulatory T-cells in the peripheral blood

Decrease only observed in hepatitis B e antigen (HBeAg)+ patients. ADV, adefovir; ETV, entecavir; FOXP3, forkhead box P3; LDT, telbivudine; NUCs, nucleotide or nucleoside analogues; TDF, tenofovir.
The effect of NUC treatment on NK cells, MAIT cells and NKT cells
Interacting at several stages with antiviral T-cells as described above [60], CD56+CD3− NK cells represent an important antiviral effector cell. Unlike HBV-specific CD8+ T-cells, NK cells have the capacity to lyse infected target cells without prior sensitization and induce non-cytolytic clearance of HBV through IFN-γ release [61]. Their activity depends on the balance of stimulatory and inhibitory receptor expression and ligation, as well as by exposure to inflammatory cytokines (like IFN-α, IL-2, IL-12 and IL-18). Corresponding with observations of acutely infected chimpanzees [62], early research on NK cells in human subjects with acute HBV infection in humans suggested that NK cells have a decline in frequency 1 to 3 weeks before HBV DNA starts to decline [14]. However, during chronic HBV, NK cell frequency or activation status does not seem to correlate strongly with considerable shifts in HBV DNA and ALT, although it should be noted here that longitudinal data is limited. Described as an NK cell ‘functional dichotomy’ during chronic HBV, NK cells appear to retain their in vitro cytotoxic potential, but have marked dysfunctional TNF and IFN-γ production [63–65]. During NUC treatment, the concentration in blood of several cytokines that affect NK cell phenotype is altered [66,67] and it seems likely that NK cell phenotype and function is affected likewise. However, when the HBV DNA declines during the natural history of chronic HBV among untreated HBV patients, NK cell frequencies and phenotypes are only minimally affected by fluctuations in viral load and liver inflammation that characterize the natural history of chronic HBV infection [68]. Here, we summarize the existing information of NK cells before and on NUC treatment [57,65,69–73], including reports on frequencies, expression of activation markers and functional tests (Table 4).
Longitudinal studies on the effect of NUCs on CD56+CD3− NK cells in the peripheral blood
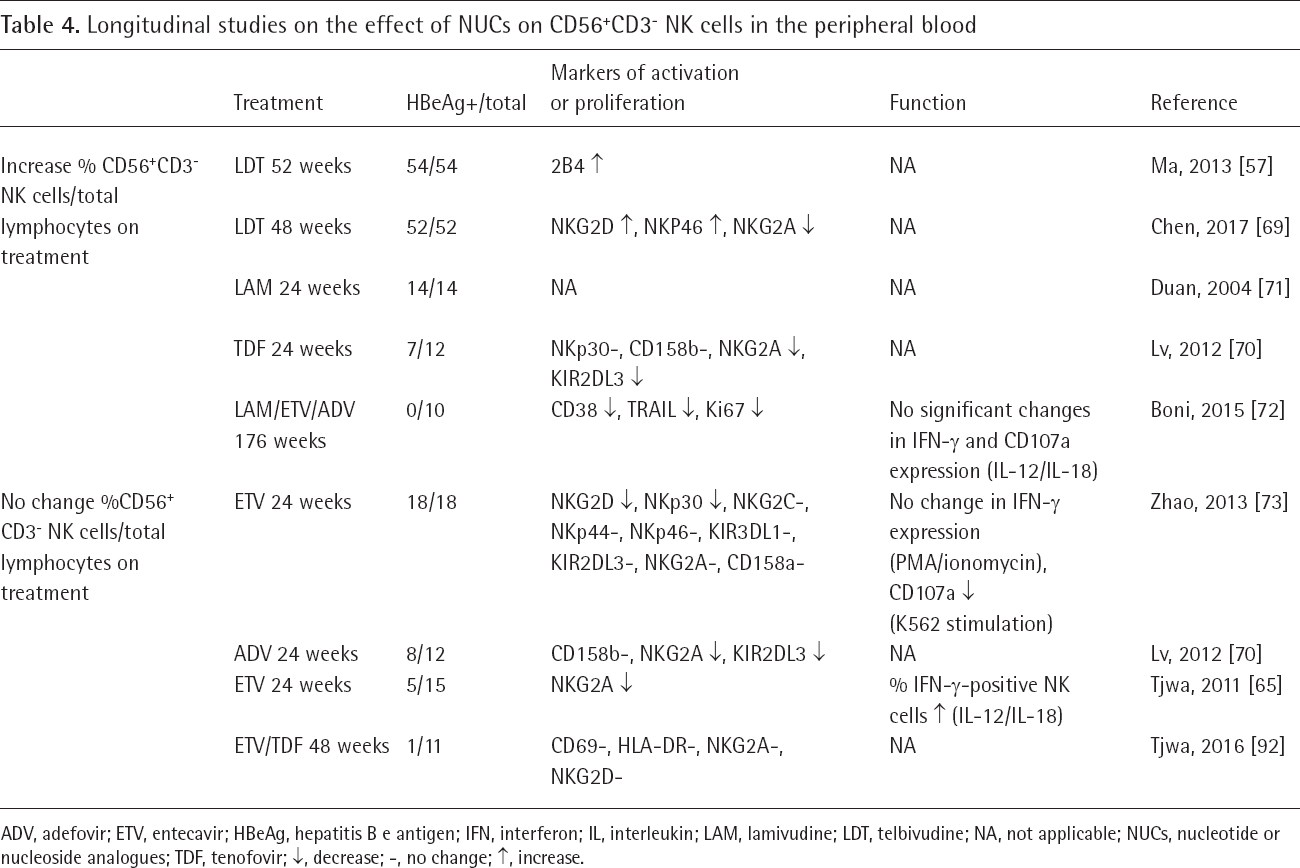
ADV, adefovir; ETV, entecavir; HBeAg, hepatitis B e antigen; IFN, interferon; IL, interleukin; LAM, lamivudine; LDT, telbivudine; NA, not applicable; NUCs, nucleotide or nucleoside analogues; TDF, tenofovir; ↑, decrease; -, no change; ↑, increase.
In four out of nine studies on NUC treatment of chronic HBV patients, the NK cell frequencies in blood were not affected. Interestingly, the relative NK cell frequencies did increase in studies that included high numbers of HBeAg-positive patients [69–71]. Zheng et al. [28] studied five paired FFPE liver biopsies before and on treatment and reported that NK cells increased in the hepatic lobule on treatment. The consequences of the migration of NK cells into the liver or blood following NUC treatment remain unclear, as NK cells have been implicated to play a role in negative regulation of immune responses to HBV, as well as in the (beneficial) mitigation of liver damage possibly by the production of anti-fibrotic IFN-γ [74] or direct lysis of hepatic stellate cells (shown in a mouse model for liver fibrosis [75]). In general, the selection of NK cell activation markers varied greatly among studies. Three studies investigated the inhibitory lectin type receptor NKG2A, and showed that its expression was down-regulated upon treatment (Table 4). In one study by Tjwa et al. [65], treatment resulted in a downregulation of NKG2A expression, as well as in an increase of the frequency of IFN-γ positive NK cells upon IL-12/IL-18 stimulation. In line with this observation, the expression of the activating lectin type receptor NKG2D is reported to be upregulated upon treatment in two studies. These observations suggest that in those patients where NUCs affect NK cell phenotype, the effect is activating rather than a return to ‘resting state’ in the complete absence of virus in the blood. Boni et al. [72] reported that among 10 HBeAg-negative patients, the percentages of NK cells positive for NKp46, CD38 and Ki67 were downregulated, indicating less proliferation and activation of NK cells during treatment. In contrast, among the studies investigating Asian patient cohorts, Zhao et al. [73] and Lv et al. [70] did not detect modulation of natural cytotoxicity receptor expression on blood NK cells (Table 4). The capacity of NK cells to produce cytokines upon NUC treatment was measured longitudinally by only three studies. Two studies [72,73] showed that NUCs did not influence the percentage of IFN-γ-positive NK cells (following IL-12/IL-18 or PMA/ionomycin), while one study [65] reported an increase (following IL-12/IL-18). The interaction of NK cells with DC or monocytes in the context of HBV treatment has not been investigated extensively. In one study by Tjwa et al. [76], NK cells were collected before and on antiviral treatment, and co-cultured with BDCA-1+ DC for 48 h. In vivo viral suppression resulted in higher percentages IFN-γ-positive NK cells following incubation with DC ex vivo, suggesting NK cell cytokine production is supported, not inhibited, by antiviral treatment. With regard to NK cells, as is the case for many immune parameters measured ex vivo, it is not certain that an increased activation or relative frequency would necessarily improve the clinical course of HBV. As mentioned before, NK cells produce immune stimulatory cytokines which activate other antiviral effector cells, but NK cells might also eliminate immune cells, like CD8+ T-cells, that are thought to deplete infected host cells thereby hampering HBV clearance [60]. Conversely, increased numbers of IFN-γ producing NK cells in the liver may be beneficial, since IFN-γ has been shown to attenuate liver fibrosis progression [74] as well as clear the virus in a non-cytolytic manner. Among the studies included in this review, the frequency of NK cells decreased only in studies that included a high number of HBeAg-positive patients. During the natural history of HBV, HBeAg-negative infection has been reported to result in comparable frequencies, and only slightly higher percentages of IFN-γ-positive CD56bright NK cells [68]. This was a study with a cross-sectional design, and it seems unlikely that this difference is caused by a direct effect of the viral HBeAg on NK cell differentiation. It is more likely that HBeAg positivity selects for patients that harbour more active HBV infection, which may indirectly affect NK cell function.
Supplementing the work done on NK cells, in recent years, attention has been directed at other innate immune cell populations, like NKT cells, that depend on the major histocompatibility complex class I-like CD1 for development and recognize lipid antigen presented by CD1. Data from a transgenic HBV mouse model indicated that NKT cells, enriched in human liver, support local T-cell-mediated immune responses, which likely contributed to HBV elimination in these animals [61]. Complementing the literature discussed in this review, several studies investigated NKT cells in the context of NUC treatment. Three studies noted an increase in frequency of NKT cells upon treatment [77–79], one study noted a decrease [80], and two studies reported no change [81,82]. Like the peripheral blood NK cell frequency, NKT cell frequency is decreased predominantly in the studies that included HBeAg-positive patients, which may be related to the higher baseline in vivo HBV replication. Another invariant T-cell population, the mucosal-associated invariant T-cell population, or MAIT cell, is also of interest. Although their role in the context of chronic viral infections like hepatitis B has not been frequently investigated, these cells can produce high amounts of antiviral cytokines and are enriched in the human liver [77,83–85]. Despite the fact that these cells have an invariant T-cell receptor specific for (bacterial) vitamin B metabolites, several indications exist that their frequency and function may alter during chronic viral infection [86]. Unfortunately, MAIT cells have been investigated during NUC treatment in only one study [83], which showed that the MAIT frequency in blood of patients was not different than in healthy controls, but CD38 expression was clearly downregulated during viral suppression by treatment, which may indicate that HBV activates MAIT cells during chronic infection.
Discussion and conclusion
From the existing literature on NUC-associated immune effects, we summarize that HBV-specific CD4+ T-cells recover in number and function in blood, but in some patients only transiently, and that treatment improves their proliferative capacity in vitro (Figure 1). HBV-specific CD8+ T-cells recover their capacity to lyse target cells, albeit transiently, but not their ability to produce antiviral cytokines. In most studies, the most pronounced increase of the specific immune responses to HBV occurs 12–16 weeks after the start of NUCs. Regulatory T-cell frequencies consistently and durably decrease on treatment, and NK cell frequencies increase in most studies.
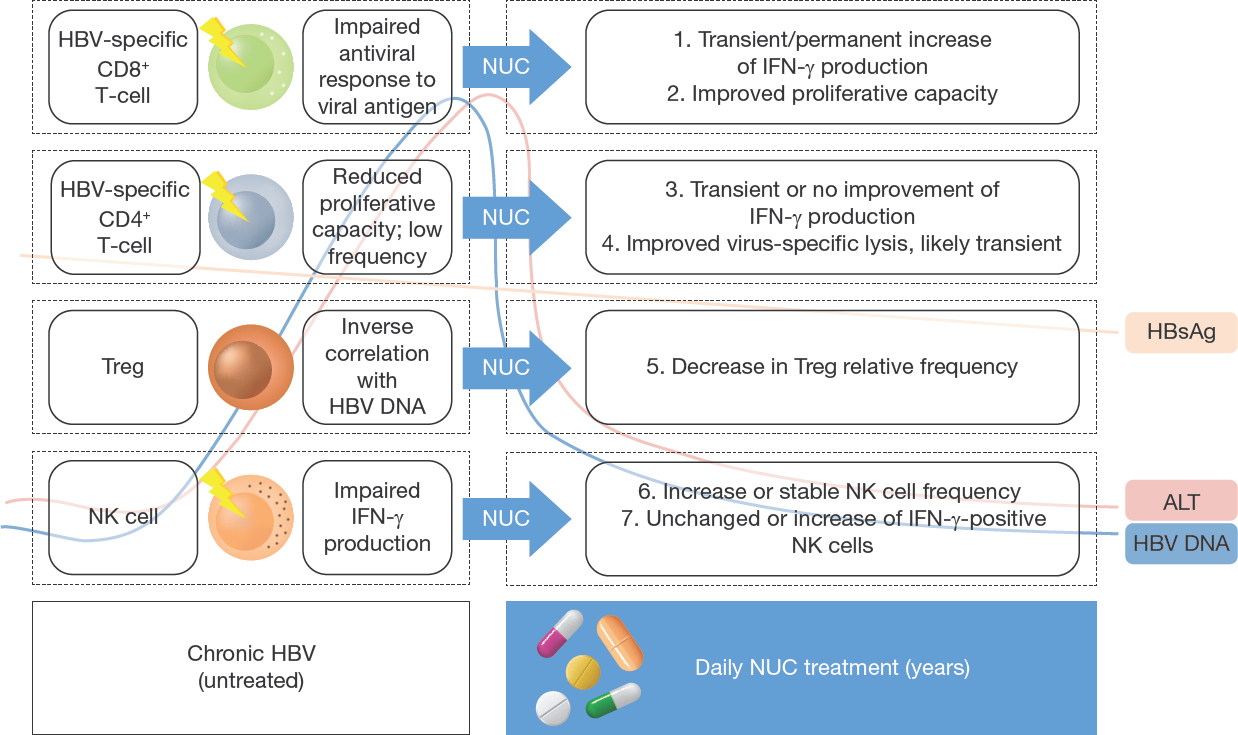
Graphical summary of published longitudinal studies on NUC effects for chronic HBV
In several studies, the detected enhancement of the virus-specific immune response decreased to pre-treatment levels during follow-up. Importantly, the specific T-cell mediated immune response during long-term NUCs does not compare to the polyclonal and strong responses observed in patients who spontaneously clear acute HBV infection [16]. Regulatory T-cell frequencies in blood decreased in almost all NUC treatment studies, along with the sharp decline of HBV DNA in the serum, but functional tests have not been performed longitudinally on these cells. Blood NK cell frequencies recover in most studies included in this review, in particular among HBeAg-positive cohorts. Inhibitory markers like NKG2A on NK cells are downregulated but not measured frequently or consistently in a longitudinal setup. NK cell cytokine production upon stimulation in vitro was unaltered by treatment or only slightly improved, which suggests that from an immunological perspective, the innate immune response is minimally altered by NUC treatment, despite clear decreases of viral load and normalization of liver inflammation. However, very few functional assays have been performed on these cells and on other cell types like B-cells, MAIT cells and liver-derived lymphocytes.
This review describes the effects on the cellular immune system associated with NUC treatment for HBV. The studies included in this summary of the existing literature differ greatly and display considerable variation of important parameters like the type of NUC used, the duration of treatment, and HBeAg status of the included subjects. Therefore, for this review, given the striking heterogeneity of the studies, we consider a systematic review or meta-analysis more prone to inaccurate conclusions than the current more narrative approach. The type of NUC used might be of particular relevance, as nucleotide analogues (adefovir, tenofovir), but not nucleoside analogues (lamivudine, entecavir), have been shown to induce IFN-λ, which might directly activate the host immune cells in vivo [87,88]. Although the differences in the HBV-infected cohorts included do not favour stratification by the type of NUC used, future work may need to include comparisons of nucleotide and nucleoside analogues. The studies included in this review report mainly on relative lymphocyte frequencies, and information on the absolute number of T-cells during NUC treatment are currently not available. Furthermore, several immune cells that have been previously proposed to play a role during viral control of HBV were not included in this review as little or no published studies evaluated their phenotype before and on NUC treatment. Peripheral blood B-cells for example, shown to associate with fluctuation of viral load [89] and relevant for sustained control of HBV replication, have not been investigated in the context of NUC treatment for HBV. Similarly, few reports have been published on treatment effects on other lymphocyte populations like MAIT cells [83] and DC [90]. Therefore, to complement the abovementioned conclusions, future work should include evaluation of these populations. In addition, despite several publications on liver-derived immune cells [28,29,91,92], both from fresh and FFPE biopsies, the possibility to improve our knowledge on immune responses in the liver is a promising one. This is particularly interesting in the context of NUC treatment for HBV, as liver inflammation is reduced in almost all treated patients upon treatment, and subtle changes to the host immune response not detectable in the blood might become detectable during suppression of the virus. Still, as the procedure to obtain liver biopsies is invasive and biopsies not regularly available, it may be desirable to evaluate the resolution of liver inflammation using biomarkers in the serum [93], especially in NUC treatment studies [94].
This review has included findings on studies with patients on NUC treatment for HBV, while also several studies have been published on the effect of NUC discontinuation. In these studies, patients were sampled on treatment and after stopping NUCs. In some countries, stopping NUC therapy is the standard of HBV care [95], as the reimbursement ends after a set period of antiviral suppression. It has been demonstrated that cessation of NUC treatment enhances the HBV specific T-cell response, and that stronger T-cell responses associate with the absence of clinical fares [42]. This suggests that, although the effects on HBV viral load are oppositional, both might boost virus-specific immune responses, at least in the short term.
Overall, the available data suggests that the NUC-induced functional improvement of CD8+ T-cells in the blood might be temporary with a peak between 12 and 16 weeks after the start of NUCs. These findings are important, but – since a large number of patients take NUCs for many years without interruption – are not easy to exploit when further boosting the responses with immunotherapeutic agents. In addition to this information, knowledge of the memory or effector T-cell phenotype of the target cell could further guide the interpretation of future trials. For HBV treatment, however, it is not clear which subsets of CD4+ or CD8+ T-cells display the most pronounced reconstitution during the suppression of viral replication. This is in contrast to the curative treatment with direct-acting antivirals for HCV during which a phenotypical shift towards functional memory CD8+ T-cell phenotype was observed in successfully treated patients, which corresponds to the changes after spontaneous resolution of acute HCV infections [96]. This information aids current HBV research, as it can aid the design of studies investigating the new immunotherapeutic agents. Given the short window of opportunity for optimal combination of antiviral and immune therapy, it may prove beneficial to apply experimental treatment within months following the start of NUCs, as the immunological benefits are not always lifelong, while for many patients, treatment is.
Footnotes
The authors declare no competing interests.
