Abstract
Background
Methods
Results
Conclusions
Introduction
With the advent of combination antiretroviral therapy (cART), HIV-related morbidity, mortality and new infection rate are continually decreased [1]. To achieve and maintain the full benefits of cART requires lifelong exposure to adequate levels of antiretroviral medications that depends highly on strictly long-term anti-retroviral adherence [2]. Compared with the self-report and other behavioural approaches (for example, pill counts and electronic drug monitoring), measurement of antiretroviral levels in various pharmacokinetic metrics (for example, plasma, dried blood spots and hair) could monitor antiretroviral exposure and provide relatively objective evidence of adherence without the risk of recall bias and social desirability [3–5]. Among all measurements of antiretroviral levels in those phar-macokinetic metrics, hair antiretroviral concentration shows promise due to its various advantages. Hair collection is noninvasive and requires minimal training, and hair specimens can be stored and shipped at room temperature without biohazardous precautions [6,7]. More important, while antiretroviral levels in plasma only measure short-term antiretroviral exposure, and antiretroviral levels in dried blood spots only measure cumulative exposure to antiretroviral medications that are activated intracellularly (for example, tenofovir), hair antiretroviral concentration reflects antiretroviral uptake from the systemic circulation over weeks to months, providing a cumulative record of exposure to all antiretroviral medications. An ever-increasing body of literature has shown that hair anti-retroviral concentration is an independent predictor of virological success as evidenced by a comprehensive review [4], and hair antiretroviral concentration has been associated with antiretroviral exposure measures in other biometrics, such as plasma [8] and dried blood spots [9,10].
Although adequate antiretroviral exposure is mediated predominantly by optimal adherence behaviour, several other factors may also contribute to the variation of antiretroviral levels. For example, previous studies reported that antiretroviral levels in plasma are mediated by factors related to pharmacokinetics (for example, age, weight and co-medication) [11–14]. Given the principal source of drug levels in hair has been proposed to be through active or passive diffusion from the bloodstream [15,16], these factors related to pharmacokinetics may also affect hair antiretroviral concentration. In addition, previous studies also reported that hair-related characteristics (for example, frequency of hair washing and hair cosmetic treatment) might affect hair concentrations of non-HIV drugs (for example, amphetamine) [17,18]. Given drugs in hair may share similar incorporation and ‘wash-out’ mechanisms, these hair-related factors may affect hair anti-retroviral concentration too. However, limited data are available on whether or not those factors affect hair antiretroviral concentration, inducing threats to the validity and reliability of the hair measures as an objective biomarker of adherence. In China, a nationwide free cART programme was initiated in 2002. Lamivudine is one of the commonly used nucleoside reverse transcriptase inhibitors (NRTI) as the basis of both first- and second-line cART in China. Hair lamivudine concentration has been used as a long-term adherence biomarker among people living with HIV (PLHIV) in China [19]. Accordingly, this study aimed to examine the potential factors that may influence the measure of hair lamivudine concentration among PLHIV in China.
Methods
Study participants
Cross-sectional data used in the current study were collected from September 2016 to January 2017 as part of a longitudinal cohort study in Guangxi, China [20,21]. With the assistance and collaboration of Guangxi Zhuang Autonomous Region Center for Disease Prevention and Control (Guangxi CDC), we randomly selected ten clinic sites with the largest number of HIV/AIDS cases from 17 cities and 75 counties in Guangxi. The recruited procedure, including criteria of inclusion and exclusion, were described in previous studies [20]. Briefly, with the referral from medical staff or HIV case managers at the study sites, local team members screened PLHIV for eligibility, discussed the benefits and risks of the study, and invited them to join. Finally, a total of 446 PLHIV completed the survey one-on-one with an interviewer in private offices of local CDC or HIV clinics where they received medical care. The interviewers were local CDC staff or health-care workers in the HIV clinics who had received intensive training on research ethics and interview skills with PLHIV before the field data and hair specimen collection. Appropriate informed consent was obtained before participation, and all procedures received approval by the Institutional Review Boards at the University of South Carolina in the United States and Guangxi CDC in China.
Factors investigated
Based on previous studies, we investigated several factors that might potentially have an impact on hair antiretroviral concentration. Such as demographics (age, sex and ethnicity), anthropometrics (height, weight and body mass index [BMI]), treatment characteristics (duration of HIV diagnosis, duration of current treatment, dosing schedule and concomitant anti-retroviral medications) and hair-related characteristics (frequency of hair washing, hair care products use and hair cosmetic treatment).
Participants provided information on their demographics, anthropometrics, hair-related characteristics and self-reported adherence. The participants’ date of HIV diagnosis, current cART regimen, date of starting current cART regimen and dosing schedule at the time of the survey were extracted from their clinical records. The duration of HIV diagnosis and current cART regimen referred to the time period from the initial date of confirmed HIV diagnosis and the start date of the current cART regimen to the date of the survey, respectively.
Considering the challenges in assessing adherence to cART via self-report, we used a multiple-item approach to measure cART adherence. The participants were asked about adherence behaviour via the three single-item questions. (1) ‘In last month, how often did you take all your HIV medications as your doctor prescribed them?’ with a 5-point response option (none of the time, a few times, some of the time, most of the time, all of the time). (2) ‘In last month, how many days were you able to take your medications exactly as prescribed?’ (3) A modified visual analogue scale (VAS) for missing doses was employed with the following instruction: ‘Place a mark (X) at the point on the line that shows your best guess about how many of you prescribed medications you have missed in last month.’ The 11 possible responses to the scale ranged from ‘0’ representing no missing doses, ‘5’ representing missing half doses, to ‘10’ representing missing all doses.
Given that self-reported data are generally skewed and overestimated, overall adherence was generated according to the following procedure. The responses to each of these self-reported measures were converted into a percentage, and then an overall adherence was the average percentage of the three self-reported measures as the previous study described [21]. The level of overall adherence was analysed either as a continuous measure or categorized into <70%, 70-89%, 90-99% and 100%.
Hair collection and assay
Hair samples of approximately 150 strands cut from the posterior vertex region as close as possible to the scalp following a standard protocol [22,23] after participants finished the survey. The proximal section of the hair sample (about 1 cm, reflecting the last month of ARV exposure) was rinsed with methanol and dried under a blow of pure nitrogen gas. Then hair sample was chopped to 1-2 mm length segments with scissors, and 10 mg weighed, processed and analysed using high-performance liquid chromatography (Agilent 1200 HPLC system; Agilent, Waldbronn, Germany) and tandem mass spectrometry (ABI 3200Qtrap; ABI, Foster City, CA, USA; LC-MS/MS). Briefly, following the 2013 FDA guideline, the lamivudine in the cut hair samples was extracted with methanol and internal standard in a 37°C shaking water bath overnight (>16 h) and then analysed by an LC-MS/MS [24,25]. Standard curves were linear in the range of 6-40,000 mg with good linearity and reproducibility. The relative error (%) and precision (coefficients of variation) for spiked quality control hair samples at low, medium and high concentrations were all <13%. The recoveries at low, medium and high concentrations were all ≥91.1%. The lower limit of quantitation (LLOQ) was 2 pg/mg. Since hair lamivudine concentration was non-normally distributed, hair lamivudine concentration was winsorized (5th/95th percentile) [26], and box-cox transformed [27] for analyses.
Statistical analysis
Descriptive statistics were used to summarize the sample characteristics, self-reported adherence and hair lamivudine concentration. Spearman's correlation and Kruskal-Wallis test were used to test the association of hair lamivudine concentration with self-reported adherence. Linear regression was used to evaluate the associations of hair lamivudine concentration with those potential factors and self-reported adherence. All data analyses were performed using SPSS 26.0 (SPSS Inc., Chicago, IL, USA).
Results
Participant characteristics
Of 446 PLHIV participating in this study, 74 were excluded from the final analysis because they had not yet started ART (n=38) or stopped ART (n=2) or insufficient quantity of hair samples for assaying (less than 10 mg, n=30), or lack of self-reported measures (n=4) leaving an effective sample of 372 participants (Table 1). The median age of participants was 42 years, 68% were men and 63.4% were of Han ethnicity. The median height, weight and BMI were 152 cm, 58 kg and 21.5 kg/m2, respectively. The median duration of HIV diagnosis was 1.50 years, and the median duration of lamivudine-based treatment was 1.42 years. The percentages of concomitant antiretroviral medications were 72.3%, 27.2%, 79.6%, 11.1% and 9.4% for tenofovir, zidovudine, efavirenz, nevirapine and lopinavir/ritonavir, respectively. The percentage of once per day dosing schedule was 60.5%. The median frequency of hair washing was 3x per week, and the percentages of the participant receiving cosmetic treatment and using hair care products were 20.9% and 4.3%, respectively.
Characteristics of the 372 participants

BMI, body mass index; NNRTI, non-nucleoside reverse transcriptase inhibitor; NRTI, nucleoside reverse transcriptase inhibitor; PI, protease inhibitor.
The median hair lamivudine concentration was 212 pg/mg and the median percentage of self-reproted adhernce was 100% (Table 1). Figure 1 shows a scatterplot of hair lamivudine concentration versus self-reproted adherence. Hair lamivudine concentration was positively associated with self-reproted adherence (r=0.143, P=0.006). Hair lamivudine concentrations for four categories of self-reproted adherence: <70% (median: 65.94 pg/mg), 70–89% (median: 216.89 pg/mg), 90–99% (median: 222.93 pg/mg) and 100% (median: 214.28 pg/mg) are displayed in Figure 2. Significant difference in hair lamivudine concentration was found among the four categories of self-reproted adherence (Z=9.813, P=0.020). PLHIV in <70% category had significantly lower hair lamivudine concentration than PLHIV in categories of 70–89% (P=0.004), 90–99% (P=0.018) and 100% (P=0.003) did. No significant difference in hair lamivudine concentration was found among the categories of 70–89%, 90–99% or 100% (all P≥0.291).
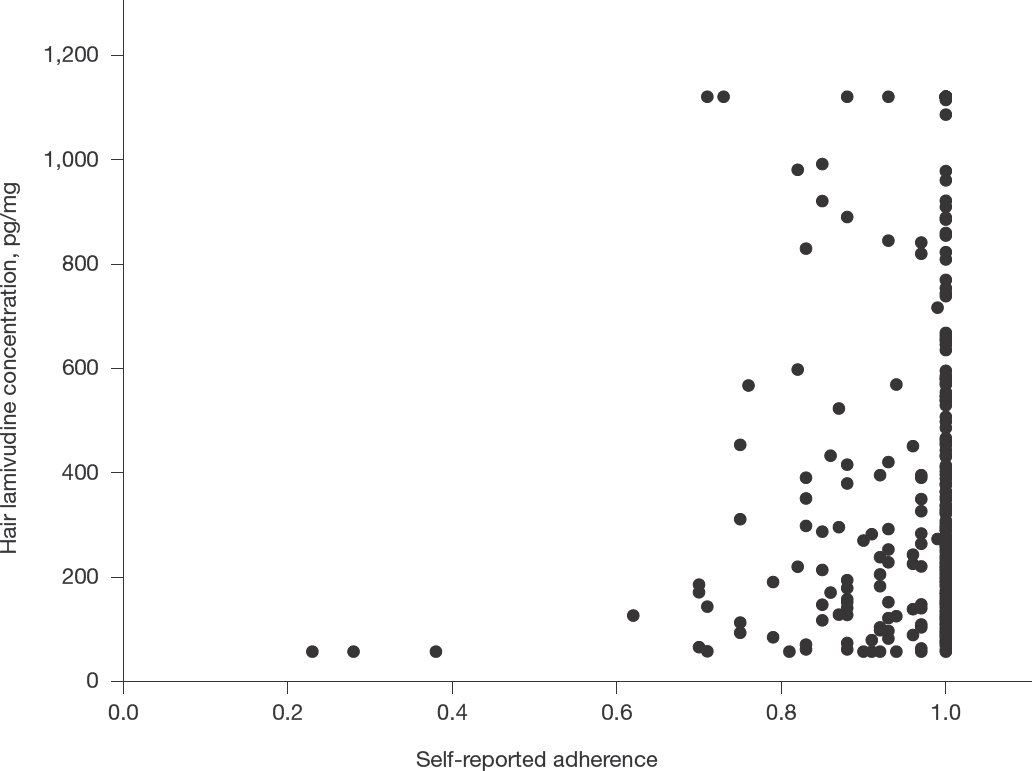
Scatter plot illustrating relationships between hair lamivudine concentration and self-reported adherence (n=372)

Hair lamivudine concentration (pg/mg) in four self-reported adherence categories
The results of univariate and multivariate analysis were shown in Table 2. In univariate regression, hair lamivudine concentration was significantly influenced by ethnicity (β=0.125, P=0.016), frequency of hair washing (β=-0.149, P=0.004), dosing schedule (β=0.194, P<0.001), concomitant tenofovir (β=-0.144, P=0.005), concomitant zidovudine (β=0.149, P=0.004), concomitant efavirenz (β=-0.124, P=0.017) and concomitant lopinavir/ritonavir (β=0.101, P=0.052). Self-reported adherence was significantly associated with hair lamivudine concentration (β=0.143, P=0.006). Including all potential factors and self-reported adherence, hair lamivudine concentration was significantly influenced by frequency of hair washing (β=-0.221, P=0.001) and dosing schedule (β=0.141, P=0.036). Self-reported adherence was significantly associated with hair lamivudine concentration (β=0.160, P=0.002).
Univariate and multivariate regressions of hair lamivudine concentration

B, unstandardized coefficient; β, standardized coefficient; BMI, body mass index.
Discussion
The present study investigated the potential factors influencing hair lamivudine concentration among PLHIV in Guangxi, China. We found that hair lamivudine concentration was influenced by frequency of hair washing and dosing schedule, but age, sex, ethnicity, height, weight, BMI, duration of HIV diagnosis, duration of current regimen, concomitant antiretroviral medications, hair care products use or hair cosmetic treatment.
The findings regarding the lack of association of most potential factors with hair lamivudine concentration were generally in line with the results from previous studies by using hair concentrations of other antiretroviral medications. For example, major previous studies also reported that some of those factors had no significant influence on hair concentrations of tenofovir (for example, age, gender, ethnicity, weight, hair colour, hair treatment) [9,28–33], zidovudine (for example, gender) [24], efavirenz (for example, age, ethnicity) [8,34] and atazanavir (for example, age, gender, ethnicity, BMI, duration of treatment) [35,36]. Taken together, hair antiretroviral concentration was less affected by some of those factors than plasma anti-retroviral concentration.
Although previous studies reported that total oral tenofovir dose was positively associated with hair tenofovir concentration [29,30], we further found that total oral lamivudine dose of 300 mg for all participants, compared with once-daily dosing (300 mg), twice-daily dosing (150 mg) was associated with higher hair lamivudine concentration. The characteristics of hair measure could be one of the potential explanations. Previous steady-state pharmacokinetics studies found that twice-daily dosing had a higher 24 h total lamivudine exposure (AUC24) in plasma than once-daily dosing did, but the difference did not reach significance [37–40]. However, lamivudine concentration in 1 cm hair represents 1-month lamivudine exposure. Therefore, the difference might become significant in the present study. In addition, only one previous laboratory study reported that nevirapine concentration was not significantly influenced by the hair washing process including washing with shampoo (3 min), followed by warm water (around 40–45°C) rinse, then adding hair conditioner (2 min) and rinsing in warm water again. However, we found that the frequency of hair washing in the real-world was negatively associated with hair lamivudine concentration. Therefore, dosing schedule and frequency of hair washing should be considered as important confounders for hair lamivudine concentration in future research.
Several study limitations need to be acknowledged. First, the current study was based on cross-sectional data, which prevents making causal inferences. Second, the current study only investigated potential factors influencing hair lamivudine concentration. Given there are differences in the physicochemical properties of antiretroviral medications [41,42], the factors (for example, frequency of hair washing) influencing hair lamivudine concentration may not be generalizable to other antiretroviral medications. Future studies ought to examine whether or not those factors influence hair concentrations of other antiretroviral medications (for example, tenofovir, zidovudine, efavirenz and lopinavir/ritonavir). Third, because all participants are from China, these findings may not be generalizable to other countries. Fourth, data were not available in the current study on some potential factors (for example, genetic factors) that might influence hair antiretroviral concentration [2,5,8]. Those factors should be considered in future research.
In summary, our findings indicated that hair lamivudine concentration was influenced by frequency of hair washing and dosing schedule. Therefore, frequency of hair washing and dosing schedule should be considered in future research using hair lamivudine concentration as a measure of lamivudine exposure and biomarker of adherence.
Footnotes
Acknowledgements
This study was funded by the National Institutes of Health (NIH) Research Grant (grant numbers R01HD074221, R21AI122919).
The authors declare no competing interests.
