Abstract
Background
Methods
Results
Conclusions
Introduction
The expansion of combination antiretroviral therapy (ART) has substantially improved treatment outcomes of people living with HIV (PLHIV) [1]. For instance, life expectancy in PLHIV has improved dramatically over the years. According to a recent meta-analysis, PLHIV starting ART at 20 years of age are estimated to live an additional 37 years [2]. However, the additional years of life were estimated to be almost 10 years less in those living in low- and middle-income countries. Gains in survival outcomes are still to be made in regions with mixed country incomes, such as Asia Pacific.
Historically, studies on survival have primarily focussed on survival after ART initiation [3–7]. A previous study on the TREAT Asia HIV Observational Database – Low Intensity Transfer (TAHOD-LITE) showed a mortality rate of 1.6 per 100 person-years (pys) in Asian PLHIV [3]. Country- and region-specific studies have reported somewhat higher mortality rates. In India, one study found 5-year mortality rates ranging from 6.8 to 7.2 per 100 pys [4], whereas another study found a mortality rate of 8.1 per 100 pys over a median follow-up of 1.3 years [5]. A study from China reported a mortality rate of 3.1 per 100 pys over a median follow-up of 4.3 years [6]. Few studies investigated survival based on longer follow-up data, including a Chinese study reporting a mortality rate of 3.4 per 100 pys, during follow-up from ART initiation until 10 years thereafter [7], and a Korean study describing a mortality rate of 4.6 per 100 pys, with a median survival of 16.7 years after HIV diagnosis [8].
Studies investigating survival have shown that predictors of survival are related to adherence to treatment and patient health at the start of ART, such as CD4+ T-cell count, AIDS-defining illnesses, poor functional status, low body weight and tuberculosis coinfection [4–6]. Another finding survival studies have in common is that the overwhelming majority of mortality occurs in the early stages after HIV diagnosis or ART initiation [3–5,7,8]. Therefore, predictors of mortality are likely to be skewed towards the part of the population that enters care in poor health and has difficulties with adherence. Those who have survived and remained in care beyond those early stages are reflective of a more stable group of PLHIV, who have overcome the first challenging few years after diagnosis and ART initiation.
Current guidelines advocate the test-and-treat approach, in which people start ART immediately after HIV diagnosis [9], when – ideally – CD4+ T-cell counts are still high and patients do not show signs of advanced HIV disease. With the implementation of these guidelines, mortality rates in the early stages of ART can be expected to decrease as well, leading to a growing population of PLHIV who have had multiple years of ART experience. Although the currently recommended ART regimens are more tolerable than those introduced in earlier years [10], newer and less toxic antiretroviral drugs are not widely available in low-and middle-income countries, including some countries in Asia. Many PLHIV in the region are still exposed to ART that have been associated with altered metabolic functioning [11,12], thereby predisposing them to non-communicable diseases such as diabetes, cardiovascular disease and kidney disease which can result in fatal outcomes [13–16]. Taken together, this calls for studies focussing their attention on survival after long-term ART use which incorporates factors related to longer duration of ART exposure.
The latest TAHOD-LITE transfer provides over a decade of clinical data of PLHIV and is one of the most sizable multi-country cohorts in the Asia-Pacific region to date. This provided us with the unique opportunity to investigate survival after long-term ART exposure in a large population of PLHIV. The aim of this study was to assess factors associated with mortality from 5 and from 10 years after ART initiation.
Methods
Study design and patients
TAHOD-LITE is a sub-study of the TREAT Asia HIV Observational Database (TAHOD), a prospective observational cohort of the International Epidemiology Databases to Evaluate AIDS. Both studies have been detailed previously [3,17–19]. The 2017 TAHOD-LITE data transfer collected data on all patients receiving care at ten clinical sites in eight Asian countries and territories. Collected data was limited to demographics, hepatitis serology, ART history, HIV-related laboratory results, and fasting plasma glucose (FPG) and creatinine levels. All data, including information on deaths and clinic transfers, were acquired from the medical records. Institutional Review Board approvals were obtained at all participating sites, the data management and analysis centre (The Kirby Institute, UNSW Sydney, Australia), and the coordinating centre (TREAT Asia/ amfAR, Bangkok, Thailand). We included adults (aged ≥18 years at first clinic visit) who started triple ART between January 1992–June 2012, were alive and in active follow-up at 5 years after ART initiation and had at least one clinic visit thereafter.
Variables and definitions
Variables included in our study were sex, age group, HIV exposure category, year of ART initiation, HIV viral load, CD4+ T-cell count, regimen, treatment interruptions, HBV coinfection (based on hepatitis B surface antigen [HBsAg] test result), HCV coinfection (based on anti-HCV test result), FPG and estimated glomeru-lar filtration rate (eGFR; as calculated according to the CKD-EPI equation [20]). We also included pre-ART CD4+ T-cell count, defined as the most recent available CD4+ T-cell count taken within 6 months prior to ART initiation. ART regimen was based on changes within the regimen, regardless of the reason for any changes as these were not available in TAHOD-LITE. First-line ART regimen was defined as the first combination regimen with three or more antiretroviral drugs. Second-line ART regimen was defined as subsequent to the first regimen, undertaken for at least 14 days, which either had one antiretroviral drug class change or two or more antiretroviral drug changes within a class, and third-line ART regimen was similarly defined as subsequent to the second regimen. In line with other literature [21,22], each break from ART (that is, not taking any antiretroviral drugs) of at least 14 days was considered a treatment interruption.
Statistical analysis
Patient characteristics at ART initiation were provided for all patients who had retained in care beyond 5 years post-ART initiation and compared with patients who had started ART between January 1992–June 2012, but had been excluded because they had died, transferred out or become lost to follow-up (LTFU) within 5 years after ART initiation (that is, patients who theoretically could have been on ART for more than 5 years). Furthermore, for those retained in care beyond 5 years post-ART initiation we provided the clinical profile at 5 years on ART taken as the latest available clinical data prior to or at that time point.
Since the focus of our study was on survival after long-term exposure to ART, follow-up time started from 5 years after ART initiation and ended on the date of death or censor date. Patients who remained in active follow-up, had transferred to another clinic or became LTFU (defined as not seen at clinic in the last 12 months) were censored on the date of last visit. Mortality rates were calculated per 100 pys of follow-up and the cumulative incidence of mortality and LTFU were plotted using the competing risk framework in which each outcome was a competing risk for the other outcome [23].
Fine and Gray competing risk regression [24] was used to calculate sub-hazard ratios (sHR) with 95% CIs for factors associated with mortality from 5 years after ART initiation, with LTFU treated as a competing risk. All variables were time updated. Regimen and treatment interruptions were counted starting at ART initiation. If for any variable, data was unavailable throughout follow-up, it was categorized as missing. A multivariable model was built using a stepwise backwards selection procedure in which variables were considered that were univariable associated with mortality at Wald's test P<0.10. In an additional analysis conducted similarly to the main analysis, we investigated survival from 10 years after ART initiation. We thus excluded all patients who were not retained in care beyond 10 years after ART initiation and follow-up time started from 10 years post-ART initiation.
Data management and statistical analyses were performed using SAS software version 9.4 (SAS Institute Inc., Cary, NC, USA) and Stata software version 14.2 (StataCorp, College Station, TX, USA).
Results
Figure 1 shows a flow chart of the patient selection process. Among the 40,921 patients who were included in TAHOD-LITE, 23,461 had started ART at least 5 years prior to the transfer date. Of these, we excluded 9,966 patients who did not attain 5 years of follow-up because they had died (9%), transferred out (29%) became LTFU (55%) or did not have a clinic visit after 5 years on ART but were not yet considered LTFU at 5 years post-ART initiation under the 12-month LTFU definition (7%). The final population eligible for our analysis comprised 13,495 patients who were in active follow-up 5 years from ART initiation.
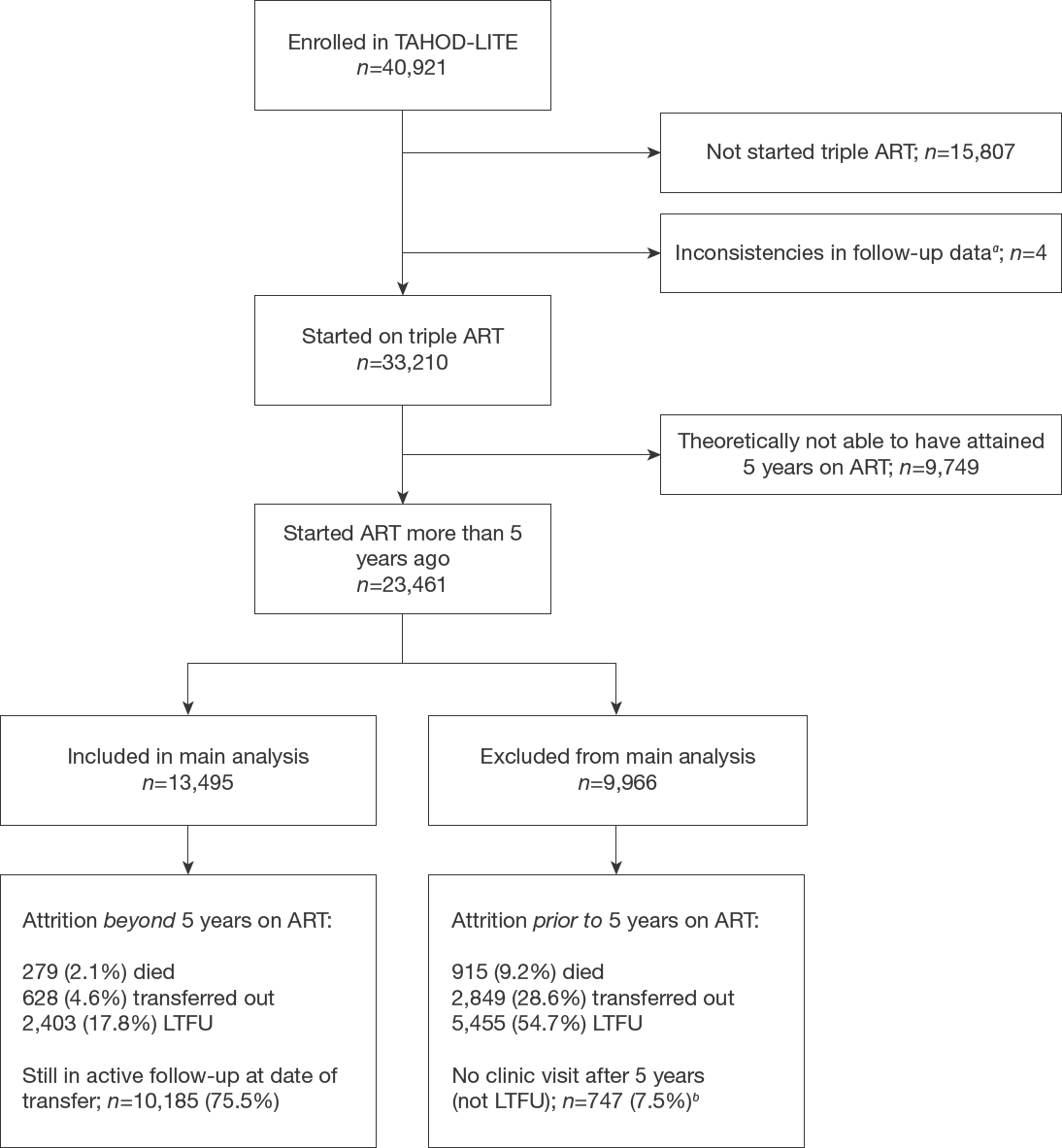
Flow chart study selection
Table 1 compares characteristics at ART initiation of eligible patients who retained in care beyond 5 years on ART versus excluded patients who started ART at least 5 years prior to the transfer date but did not retain in care beyond 5 years on ART. Of the 13,495 eligible patients, 9,463 (70%) were male and 8,087 (60%) had heterosexual HIV exposure category. Median ART initiation year was 2007 (IQR 2005–2010) and median CD4+ T-cell count at ART initiation was 147 cells/μl (IQR 55–243) among the 8,964 (66%) patients who had available data on pre-ART CD4+ T-cell count. Positive HBsAg and anti-HCV were seen in 733 (5%) and 820 (6%) of patients, respectively, whereas 5,523 (41%) and 7,304 (54%), respectively, had never been tested for these serological markers. Excluded patients were largely similar to the eligible patients in terms of sex and age, but a smaller proportion was tested on HBV and HCV coinfection. Furthermore, a lower proportion of the excluded patients had men who have sex with men (MSM) HIV exposure category compared with the included patients. Median ART initiation year was more recent in the excluded patients (2009, IQR 2006–2011). Median pre-ART CD4+ T-cell count was comparable between eligible and excluded patients, however, in the excluded patient who died before 5 years on ART, 70% had a pre-ART CD4+ T-cell count of <200 copies/μl compared with 60% in those who died after 5 years of ART exposure.
Characteristics at ART initiation of patients who were and were not eligible for analysis of survival from 5 years after ART initiation

Only included patients considered lost to follow-up (LTFU) at 5 years post antiretroviral therapy (ART) initiation under the 12-month definition for LTFU.
Ever tested positive on hepatitis B surface antigen (HBsAg) or anti-HCV. IDU, injecting drug use; MSM, men who have sex with men.
Table 2 displays the clinical characteristics at 5 years on ART of the included patients. At 5 years on ART, median CD4+ T-cell count was 457 cells/μl (IQR 310– 627), median HIV viral load was 47 copies/ml (IQR 19–399), median age was 40 years (IQR 35–47) and 839 (62%) patients were still on their first regimen. Treatment interruptions were observed in 2,336 (17%) of patients by 5 years on ART. A large proportion of patients had unknown FPG (54%) or eGFR (48%) up to 5 years after ART initiation, but in those who had these data available median FPG was 89 mg/dl (IQR 80–99) and median eGFR was 99 ml/min/1.73 m2 (IQR 85–110). Furthermore, the majority of included patients were from India (55%), followed by Cambodia (13%), Vietnam (10%), Singapore (9%), Hong Kong (4%), Indonesia (3%), South Korea (3%) and Taiwan (3%).
Clinical profile at 5 years on ART of patients included in the main analysis

Counted from antiretroviral therapy (ART) initiation. eGFR, estimated glomerular filtration rate.
Survival after 5 years on ART
Among the 13,495 patients who were retained in care from 5 years after ART initiation, 279 (2%) subsequently died (incidence rate [IR] 0.56 per 100 pys), 628 (5%) transferred out (IR 1.27 per 100 pys), and 2,403 (18%) became LTFU (IR 4.85 per 100 pys) over 49,533 person-years. Median follow-up time from 5 years after ART initiation was 3.1 years (IQR 1.4–5.5), translating into a total ART exposure time of 8.1 years (IQR 5.4–10.5). Figure 2 shows the cumulative incidence of mortality and LTFU from 5 years after ART initiation. Mortality rates were higher for men, older age, higher HIV viral load, lower CD4+ T-cell count (pre-ART and beyond 5 years on ART), second- and third-line regimens, more treatment interruptions, HBV coinfection, HCV coinfection, higher FPG levels and decreased eGFR. In multivariable analysis (Table 3), age >50 years (sHR 2.24, 95% CI 1.58, 3.15, compared with ≤40 years), HIV exposure through injecting drug use (sHR 2.17, 95% CI 1.32, 3.56), higher HIV viral load ≥1,000 copies/ml: sHR 1.52, 95% CI 1.05, 2.21, compared with <400), regimen (second-line regimen: sHR 2.11, 95% CI 1.52, 2.94, and third-line regimen: sHR 2.82, 95% CI 2.00, 3.98, compared with first-line regimen), HBV coinfection (sHR 2.23, 95% CI 1.49, 3.33), FPG ≥126 mg/dl (sHR 1.98, 95% CI 1.22, 3.21, compared with <100 mg/dl) and eGFR <60 ml/min/1.73 m2 (sHR 2.57, 95% CI 1.56, 4.22) were associated with increased mortality after 5 years on ART, whereas HIV exposure through male-to-male sexual contact (sHR 0.44, 95% CI 0.22, 0.88) and higher CD4+ T-cell count (200–349 cells/μl: sHR 0.27, 95% CI 0.20, 0.38, 350– 499 cells/μl: sHR 0.10, 95% CI 0.07, 0.16 and ≥500 cells/μl: sHR 0.09, 95% CI 0.06, 0.13, compared with <200 cells/μl) were associated with decreased mortality. Borderline associations with increased mortality were found for HIV viral load 400–999 copies/ml (sHR 2.02, 95% CI 0.99, 4.10, compared with <400) and ≥3 treatment interruptions (sHR 1.52 95% CI 0.97, 2.38, compared with no interruptions). No associations were found between mortality and sex, pre-ART CD4+ T-cell count, period of ART initiation or HCV coinfection.

Cumulative incidence of mortality and lost to follow-up beyond 5 years on ART
Factors associated with survival beyond 5 years on ART

All other factors in the model were time-updated. P-values for test for heterogeneity excluded missing or not-tested values. P-values in bold represent significant covariates in the final model. Non-significant factors were presented in the multivariate model adjusted for significant predictors.
Adjusted for site.
Counted from antiretroviral therapy (ART) initiation.
Ever tested positive on hepatitis B surface antigen (HBsAg) or anti-HCV. eGFR, estimated glomerular filtration rate; FPG, fasting plasma glucose; IDU, injecting drug use; MSM, men who have sex with men; pys, person-years; sHR, sub-hazard ratio.
Survival after 10 years on ART
A total of 3,996 patients were still in care 10 years after ART initiation. Of these, 64 (2%) subsequently died (IR 0.72 per 100 pys), 115 (3%) transferred out (IR 1.29 per 100 pys) and 434 (11%) became LTFU (IR 4.86 per 100 pys) over 8,921 person-years. In univariable analysis, HIV exposure category, HIV viral load, CD4+ T-cell count (pre-ART and beyond 5 years on ART), regimen, treatment interruptions, HBV coinfection and HCV coinfection were associated with increased mortality from 10 years after ART initiation. In multivariable analysis, only HIV exposure through injecting drug use (sHR 6.33 95% CI 2.21, 18.14, compared with heterosexual), CD4+ T-cell count beyond 10 years on ART (200–349 cells/μl: sHR 0.30, 95% CI 0.16, 0.57; 350– 499 cells/μl: sHR 0.05, 95% CI 0.04, 0.15, and ≥500 cells/μl: sHR 0.08, 95% CI 0.04, 0.15, compared with <200 cells/μl), and regimen (second-line regimen: sHR 2.81, 95% CI 1.14, 7.05, and third-line regimen: sHR 3.59, 95% CI 1.47, 8.77, compared with first regimen) were associated with increased mortality (Additional file 1). Other factors largely showed similar trends to main analysis, but with weaker associations.
Discussion
The mortality rate in the 13,495 PLHIV who were retained in care beyond 5 years after ART initiation was 0.56 per 100 pys. About 30% of these patients were still alive and in active follow-up beyond 10 years after they started ART and had a mortality rate of 0.72 per 100 pys. Factors associated with increased mortality rates beyond 5 years after ART initiation were older age, HIV exposure through injecting drug use, later regimen, more treatment interruptions and HBV coinfection, as were higher viral load, high FPG, and decreased eGFR measured beyond 5 years on ART. HIV exposure through male-to-male sexual contact and higher CD4+ T-cell count measured beyond 5 years on ART were associated with decreased mortality rates. Results related to mortality after 10 years on ART were roughly similar, although the evidence for most associations was weaker due to limited power.
The current study showed a mortality rate of 0.56 per 100 pys from 5 years after ART initiation. When the study population was restricted to those who retained in care an additional 5 years, the mortality rate increased slightly, to 0.72 per 100 pys from 10 years after ART initiation, most likely due to further ageing of the population. In comparison, the Antiretroviral Therapy Cohort Collaboration (ART-CC) conducted in high-income Western settings found a somewhat higher mortality rate of 1.29 per 100 pys in PLHIV who had retained in care beyond 10 years on ART [25]. While in a previous TAHOD-LITE study male sex, lower pre-ART CD4+ T-cell count and ART initiation in earlier calendar year were indicated as important predictors of survival in the first years on ART [3], these factors were not confirmed as important for survival after long-term exposure to ART. Similarly, the ART-CC study did not substantiate a relation between mortality beyond 10 years on ART and sex or pre-ART CD4+ T-cell count [25].
CD4+ T-cell count at the start of ART can be considered one of the most critical predictors of survival in the first years after ART initiation [26]. In line with this, a pre-ART CD4+ T-cell count of <200 cells/μl was observed in a substantially higher proportion of TAHOD-LITE patients who died within 5 years after ART initiation (70%) compared with those who died beyond 5 years on ART (60%) and those who remained alive (46%). In many South East Asian countries, PLHIV still commonly present to care with low CD4+ T-cell counts. These patients have considerably lower chance of survival beyond the next few years than those who start ART in better health [3,7,8,27], which further underlines the importance of ART treatment guidelines which recommended immediate start of ART, with priority being given to those with lower CD4+ T-cell counts [9]. Evidence shows that as time passes by and patients remain in care, the association between pre-ART CD4+ T-cell count and survival dissipates when accounted for CD4+ T-cell counts at later time points [28]. Consistent with this evidence, our study showed that pre-ART CD4+ T-cell count was not associated with mortality after long-term ART exposure, when controlling for CD4+ T-cell count from 5 years on ART onwards and other confounders.
Corroborating findings from other studies [21,25,29], high viral load and low CD4+ T-cell count from 5 years after ART initiation onwards contributed to poorer survival. Furthermore, mortality rates were higher among those with treatment interruptions, although this association became weaker in multivariable analysis. Our findings thus confirm the importance of continuity in HIV treatment and management. In case of virological, immunological or clinical failure, it is warranted to check patients’ adherence and offer adherence support where appropriate [10]. Ongoing treatment failure despite optimal adherence commonly necessitates a switch in regimen to improve HIV outcomes [10,30,31]. According to a previous TAHOD study, about half of those with treatment failure did not switch regimens in the 12 months after which a regimen was determined as failing in terms of clinical progression, virological failure or immunological failure [32]. As delaying progression from a first-line regimen to a second-line regimen has been associated with poorer survival [33], the delay in switching regimens potentially explains the higher mortality we observed in those who were on a second-or third-line regimen in our cohort.
We found a twofold increased mortality hazard for viral loads between 400–999 copies/ml, compared with virological suppression at <400 copies/ml. The evidence we found for this association was weak, possibly because it was based on a small number of observations (9 deaths over 606 pys) and we did not differentiate between multiple assessments of low-level viraemia within a patient or one-off assessments of HIV viral load 400–999 copies/ml. Previous studies have shown inconsistent results regarding associations of persistent low-level viraemia with all-cause mortality and AIDS events [34–36]. However, one cohort study demonstrated increased levels of markers related to cardiovascular disease, among those with low-level viraemia [36]. Collectively, these findings hint towards a potential pathway from low-level viraemia to cardiovascular disease and mortality, which may only become apparent after many years on ART. Further studies are needed to investigate the importance of low-level viraemia and clinical outcomes, especially in the stable population of PLHIV who have been exposed to long-term ART.
Cardiovascular disease and other chronic non-communicable diseases such as diabetes, kidney disease and cancer are highly prevalent in PLHIV compared with the general population [37–39]. Several studies with a median follow-up of less than 5 years after ART initiation have reported increased mortality among PLHIV with poor kidney function [15,40,41] and high glucose levels or diabetes [13,14]. Our study showed that beyond 5 years after ART initiation, there was about a twofold increased mortality hazard in those with eGFR <60 ml/min/1.73 m2 and a more than twofold increased hazard for those with FPG ≥126 mg/dl. A rise in prevalence of kidney disease and diabetes has been observed in those with longer exposure to ART [11,42] and prolonged presence of these conditions might be increasingly important for survival in the long run. Taken together with evidence that AIDS-related mortality has been decreasing in PLHIV in Asia Pacific [43], this underlines the importance of allocating resources to monitoring glucose levels, kidney function and presumably other markers of non-communicable comorbidities, and intervene where appropriate to further advance survival in PLHIV.
In our study, being HBV coinfected was associated with increased mortality. Similar findings have been reported for study populations in Thailand and South Korea [44,45]. There have also been reports of associations between mortality and both HCV and HBV coinfection in Cambodia [46] or mortality and HCV, but not HBV coinfection in China [7,47]. Most likely the association we found had to do with availability of treatment for HBV, such as the antiretroviral drug tenofovir which is used as treatment for HIV as well as HBV. This is exemplified by a Taiwanese study that con-firmed increased mortality among PLHIV with HBV before the introduction of tenofovir in 2011 but did not find a difference in mortality among those with or without HBV after 2011 [48].
Our findings indicated differences in survival by HIV exposure category. Compared with those with heterosexual HIV exposure, survival was better in MSM, whereas it was worse in people who injected drugs. Previous literature has shown that MSM are more likely to have optimal levels of adherence [49]. Suboptimal adherence is closely related to treatment failure and this may explain why MSM have superior survival outcomes as well. Increased mortality in injecting drug use was also observed in the ART-CC cohort [25]. Findings from this cohort further showed that non-AIDS-related mortality is over 3x more likely in people who inject drugs compared with those who do not inject drugs. The high mortality rate in people who inject drugs in our study population might also be explained by non-AIDS causes of death such as drug overdosing, cardiovascular disease, liver disease and accidents, which are commonly seen in people using illicit drugs [50].
This study had several limitations. As with all observational studies, we cannot rule out the possibility of unmeasured confounding. The TAHOD-LITE cohort includes all adult patients enrolled in care from selected clinical sites that participate in TAHOD, and data of over 50,000 patients are transferred from – in some clinics paper-based – medical files. Due to the tremendous size of the cohort it is outside the scope of TAHOD-LITE to collect detailed data on patients’ laboratory results beyond CD4+ T-cell count, HIV viral load, FPG and serum creatinine. As a result, we could not assess the effect of other markers for comorbid non-communicable conditions, such as cardiovascular disease, liver disease and certain cancers. Our findings may be biased to some extent due to missing data and infrequently performed laboratory testing of variables that were collected in this study. For example, HBV coinfection was based on presence of one positive test result and it is possible that we misclassified patients if the virus had cleared prior to testing or infection occurred after testing. It should be noted that active tracing of patients LTFU or linkage to death registries was not part of the TAHOD-LITE study protocol and an uncertain proportion of patients considered LTFU may have died or self-transferred to other clinics [51,52]. In addition, data on cause of death were not routinely collected, so we were unable to distinguish between deaths related to HIV and other medical causes, which could have been a valuable addition to our analysis considering our findings regarding kidney function and glucose levels. Furthermore, our findings are not necessarily representative of all Asian PLHIV with long-term ART exposure, partly due to differences between the included patients and those we had to exclude due to insufficient follow-up time or attrition prior to 5 years on ART.
In summary, our findings suggest low mortality rates among PLHIV in Asia-Pacific who were retained in care beyond 5 years after ART initiation compared with reported short-term rates. Our study confirmed that maintaining high CD4+ T-cell count, undetectable HIV viral load and not interrupting treatment benefit survival outcomes. Additionally, we found improved survival in those with favourable levels of FPG and eGFR. Thus, to optimize long-term survival outcomes in PLHIV, it is crucial to carefully monitor and manage comorbidities, such as hepatitis, diabetes and kidney disease, next to preventing treatment failure.
Footnotes
Acknowledgements
The TREAT Asia HIV Observational Database Low-Intensity TransfEr study is an initiative of TREAT Asia, a programme of amfAR, The Foundation for AIDS Research, with support from the US National Institutes of Health's National Institute of Allergy and Infectious Diseases, the Eunice Kennedy Shriver National Institute of Child Health and Human Development, the National Cancer Institute, the National Institute of Mental Health, the National Institute on Drug Abuse, the National Heart, Lung, and Blood Institute, the National Institute on Alcohol Abuse and Alcoholism, the National Institute of Diabetes and Digestive and Kidney Diseases, and the Fogarty International Center, as part of the International Epidemiology Databases to Evaluate AIDS (IeDEA; U01AI069907). The Kirby Institute is funded by the Australian Government Department of Health and Ageing, and is affiliated with the Faculty of Medicine, UNSW Sydney. The PhD of RB has been supported through an Australian Government Research Training Program Scholarship. The content of this publication is solely the responsibility of the authors and does not necessarily represent the official views of any of the governments or institutions mentioned above. Additional acknowledgements can be found in Additional file 2.
The authors declare no competing interests.
