Abstract
Background
Daclatasvir has potent antiviral activity against HCV infection when used in combination with sofosbuvir, however, its pharmacokinetics have not been described in adolescents. The aim is to determine the pharmacokinetic parameters of daclatasvir in adolescents, and to develop a population pharmacokinetic (PopPK) model.
Methods
Seventeen adolescent patients with genotype-4 chronic HCV infection received once daily oral daclatasvir 60 mg in combination with 400 mg sofosbuvir for 12 weeks. Steady state concentrations were determined. Non-compartmental and population PK were determined.
Results
The average PK parameters calculated by non-compartmental analysis (NCA): maximum plasma concentration (Cmax), area under the curve (AUC), apparent oral volume of distribution (V/F), apparent oral clearance (CL/F) and half-life (T1/2) were 1,092 ng/ml, 11,178 ng/ml•h, 55 l, 4.5 l/h and 8.5 h, respectively. Daclatasvir was best described by one compartment structural PK model with zero order absorption and first-order elimination. The absorption rate constant (K0), V/F, and CL/F of the final PopPK model of daclatasvir were 1.5/h, 52 l and 4.7 l/h, respectively. Body weight and serum albumin had significant effect on the V/F parameter.
Conclusions
Body weight and serum albumin were the major determinants of daclatasvir V/F in this population. PK parameters were comparable to those reported in adult HCV patients, demonstrating that 60 mg daclatasvir is an appropriate dose for adolescents. ClinicalTrials.gov NCT03540212.
Introduction
The World Health Organization (WHO) estimated that 71 million people were living with HCV infection in 2015 [1]. Treatment of chronic hepatitis C (CHC) has changed over the years. Until 2011, a combination of weekly pegylated interferon-α (PEG-IFN-α) and daily doses of ribavirin (RBV) in a 24- or 48-week course was the standard of care treatment for CHC [2]. However, this treatment was often discontinued due to adverse events [3]. In 2015, the United States Food and Drug Administration (FDA) approved daclatasvir, a new non-structural protein inhibitor of pangenotypic activity, for the treatment of CHC [4]. Daclatasvir, an oral antiviral drug for CHC, is an HCV non-structural protein 5A NS5A inhibitor. NS5A is an RNA-binding multifunctional viral protein and is essential for viral proliferation by interacting with other HCV non-structural proteins and cellular proteins [5]. The combination of daclatasvir and sofosbuvir has shown high rates of sustained virological response (SVR) in adults when administered for 12 weeks in those with HCV genotype-1, 3 or 4 infection [5,6].
Daclatasvir pharmacokinetics (PK) were evaluated in healthy adult volunteers and HCV patients. The absolute oral bioavailability of daclatasvir in humans was 67% under fasted conditions [7]. Daclatasvir has a median time at peak concentration (Tmax) ranging from 1.0 to 2.0 h. Generally, the steady state was achieved between days 4 and 5 [8]. Primarily daclatasvir is excreted via faeces (88%) in an unchanged form, with slight renal elimination (7%). Following the last dose on day 14 of treatment, the mean half-life (t1/2) values ranged from 13 to 15 h and the area under the plasma concentration–time curve (AUC) was proportional to the administered dose [7]. Daclatasvir is a very weak inducer of cytochrome P450 (CYP) 3A4 [6]. The population PK of daclatasvir have been previously studied in adult patients with HCV but not in children or adolescents [9,10].
In 2017, the combination of 400 mg sofosbuvir with 90 mg ledipasvir was approved by the FDA for the treatment of HCV, genotypes-1, 4, 5 or 6 infections in adolescents (ages 12–17 or weight >35 kg) [11]. Several pangenotypic drug combinations are approved for adults, but none are available for children and adolescents. The sofosbuvir/daclatasvir combination is available and affordable in resource-limited settings in addition to being a pan-genotypic regimen [12].
Therefore, the objectives of this study were to determine the noncompartmental PK parameters of daclatasvir and to develop a population PK model to better understand the factors affecting the PK of daclatasvir in adolescents.
Methods
Study Design and Patients
The study was conducted between December 2017 and March 2019 at the Faculty of Medicine Ain Shams University Clinical Research Centre (MASRI-CRC). It was a prospective, open-label clinical study including adolescents with genotype-4 CHC infection. Enrolled patients were aged between 12 and 17 years or weighing ≥35 kg, HCV treatment-naive, non-cirrhotic with fibrosis stage: F0 to F3, had confirmed HCV infection (positive anti-HCV and HCV RNA polymerase chain reaction [PCR]) and had normal values of haemoglobin, platelets, white blood cells, serum albumin, bilirubin, glucose and creatinine. Patients were excluded if they received previous treatment for HCV, had history of a clinically significant illness or any other medical condition that could interfere with the antiviral treatment, acute hepatitis A or B virus infections, hepatic decompensation, history of immunologically mediated disease, blood/blood product transfusion within 4 weeks prior to the study, psychiatric hospitalization or receiving drugs known to interact with daclatasvir. All patients received the adult dose of daclatasvir 60 mg tablets plus sofosbuvir 400 mg tablets once daily for 12 weeks, then were followed up at 12 weeks post-treatment for SVR. The investigators had delivered the medications to patients every 28 days for 3 consecutive months at MASRI-CRC.
Ethics
The study protocol was approved by the Research Ethics Committees of the Faculty of Medicine, Ain Shams University (FMASU P69a/2017), and the Faculty of Pharmacy, Cairo University (CL 1685) and registered on ClinicalTrials.gov (NCT03540212). The study was conducted according to the principles outlined in the declaration of Helsinki [13]. Written assent and consent forms were obtained from all patients, patients’ parents or legal guardians prior to conducting study-related procedures.
Blood Sampling and Bioanalysis
Blood samples for PK analysis were obtained at steady state, at least 1 week after the dosing had started. Patients were instructed to fast except for water at least 8 h prior to the visit and not to have taken their daily dose of the study drug. A pre-dose blood sample was collected up to 30 min prior to the intended dose, after which patients were administered daclatasvir 60 mg and sofosbuvir 400 mg. Post-dose blood samples were collected at 0.5, 1, 1.5, 2, 4, 8, 12- and 24-h post-dose. Immediately after blood collection, the blood samples were centrifuged at 3,000 rpm for 10 min and 2 ml of plasma samples were stored at -80°C. Before analysis, plasma samples were thawed at room temperature. An aliquot of 0.5 ml plasma was spiked with 50 ml of 100 μg/ml internal standard then vortex mixed for 30 s. Liquid–liquid extraction was carried out by adding 3.5 ml methyl tert-butyl ether, and mixed for 4 min, then centrifuged at 5,000 rpm at room temperature for another 5 min. The clear upper layer was separated and evaporated at 45°C under a vacuum stream, then reconstituted with 1 ml of acetonitrile and 7 ml was injected into the LC-MS/MS system. The plasma concentration of daclatasvir was measured by liquid chromatography-tandem mass spectrometry (LC-MS/MS). The mass spectrometer was an AB Sciex Model API 4000. The mobile phase consisted of acetonitrile: 0.02% ammonium formate, pH 4.0 (40:60 v/v) at a flow rate of 0.7 ml/min. The column temperature was maintained at 30°C. The ion-spray potential was set at 3,000 V and the source temperature was 100°C. The lower limit of quantification for daclatasvir in plasma was 5.0 ng/ml, while the higher limit of quantification was 2,400 ng/ml. The calibration plots were found to be linear over the range of 3.0–3,000 ng/ml with a correlation coefficient of 0.9984. Intra-and inter-day precision had an average coefficient of variation (CV%) of 6.49 and 7.2% respectively with no single value exceeding 15%.
Non-Compartmental Pharmacokinetics
Daclatasvir PK were analysed using non-compartmental pharmacokinetic analyses (NCA). The PK parameters were calculated using Excel add-in programme, PK solver [14]. The following parameters were obtained: area under the plasma concentration versus time curve over the dosing interval (AUCtau), maximum observed concentration of drug in plasma (Cmax), minimum observed concentration of drug in plasma (Cmin), time to reach Cmax (Tmax), plasma half-life (T1/2), apparent total oral body clearance (CL/F) and apparent oral volume of distribution (Vd/F). Non-compartmental parameter values were used as initial estimates for PK modelling and were compared with parameter values estimated by the final model and were compared to the integrated adult data from Phase II and III clinical studies.
Population Pharmacokinetics Analysis
Analysis Platform
The PopPK model of daclatasvir was built using nonlinear mixed effect modelling software, Monolix, Version 2018R1 (Lixoft, S.A.S, Orsay, France). The parameters estimated in the PopPK were apparent volume of distribution (V/F), the absorption rate constant (K0) and apparent oral clearance (CL/F). Fixed effects (8) are the values for parameters in the population while random effects (ω) represent population model estimate of the standard deviation in between-subject variability (BSV) η. PopPK parameters were estimated via the maximum likelihood estimator without any approximation of the model (no linearization). The Monolix estimation method used was stochastic approximation expectation maximization (SAEM) algorithm combined to a Markov Chain Monte Carlo (MCMC) procedure. The number of MCMC chains was fixed to 10 for all estimations. Model selection was guided by minimum objective function value (OFV) of twice the negative of the log-likelihood function (- 2LL), Akaike information criterion (AIC), Bayesian information criterion (BIC), goodness-of-fit (GOF) diagnostic plots and the precisions (relative standard error, %RSE).
Structural and Base Models
Different structural models, including one, two and three compartment models with zero or first order absorption (with or without lag time absorption) and elimination, were evaluated and the model that best described the observed data was selected.
The base model is the model that represents the best description of the data without considering the effect of covariates. The BSV was modelled in terms of η variables assuming loglinear distribution. Individual parameters in terms of clearances and volumes of distribution were calculated using conditional modes.
Variability (error) Model
Constant, proportional and combined error models were evaluated to describe the residual variability and the BSV. The inter-subject variability of the PK parameters was modelled by lognormal distribution. Shrinkage of the parameters were calculated by the following equation:
Shrinkage = 1 - SD(η)/ω
where ηi are the BSV terms and ω is the population model estimate of the standard deviation in η [15].
Covariate Analysis (Full Model Analysis)
After the identification of the base model and error model, the effects of the covariates on the constant PopPK parameters (Cl/F, V/F and K0) were investigated. More than twenty covariates were individually examined for their effect on the PK parameters: age, sex, body weight, body mass index (BMI), body surface area (BSA), serum albumin, serum bilirubin, aspartate aminotransferase (AST), alanine aminotransferase (ALT), blood glucose, serum creatinine, total bilirubin, direct bilirubin, alfa feto-protein (AFP), prothrombin time (PT), international normalized ratio (INR), haemoglobin and HCV RNA viral load. These covariates were added simultaneously to the base model to obtain the full model. The selection of covariates was performed using forward inclusion and backward elimination to identify all statistically signif cant covariate–parameter relationships. This process was continued until no further reduction in the OFV resulted. A reduction in -2LL ≥3.84 (P≤0.05 based on χ 2 distribution) was required for inclusion of a structural parameter, whereas a reduction in -2LL ≥6.63 (P≤0.01) following backward deletion was required for covariate inclusion.
Final Model Evaluation
The final PopPK model was obtained by removing each covariate one at a time from the full model. Testing the final model was done using the OFV (-2x log-likelihood), AIC and BIC. The normalized prediction distribution error (NPDE), individual weighted residuals (IWRES), population weighted residuals (PWRES) and the visual predictive check (VPC) were also used to evaluate the final model. Diagnostic plots: observed and predicted concentrations versus time, observed concentrations versus population predictions, weighted residuals versus time and weighted residuals versus predictions were used for evaluation of the GOF in the final model. A VPC was performed by simulating 1,000 datasets with identical design to the original data using the parameter estimates from the final model, including the inter-subject and residual variability. The 10th, 50th and 90th percentiles of the concentrations predicted were plotted against time with the observed daclatasvir concentrations. The 95% CIs of estimated parameters were calculated by the bootstrap method using bootstrapping (n=500) in R (R: A Language and Environment for Statistical Computing, R Foundation for Statistical Computing, Vienna, Austria, version x64 3.6.3) using Rsmlx (Xpop Inria Team, France).
Results
Demographic Data
A total of 26 HCV adolescent patients were screened for the inclusion and exclusion criteria, eight patients were excluded from the study due to genotype difference, undetected HCV RNA or their lower weight, so the rest of 18 patients were selected to participate in the treatment study. After 6 months, 17 patients completed the study while 1 patient dropped out due to missing follow-up visits. A total of 151 plasma samples from the 17 Egyptian genotype-4 HCV-infected adolescents were included for PopPK evaluation and statistical analysis. Out of the 17 patients only 4 were females. The mean age of the studied population was 14 years and mean body weight was 50 kg; 82% were in the normal weight range (BMI: 18–25 kg/m2) whereas 12% were overweight (BMI: 25–30 kg/m2). Patients’ characteristics are summarized in Table 1. SVR at 12 weeks post-treatment was achieved in all 17 patients.
Demographic and laboratory characteristics of recruited adolescents with chronic HCV infection
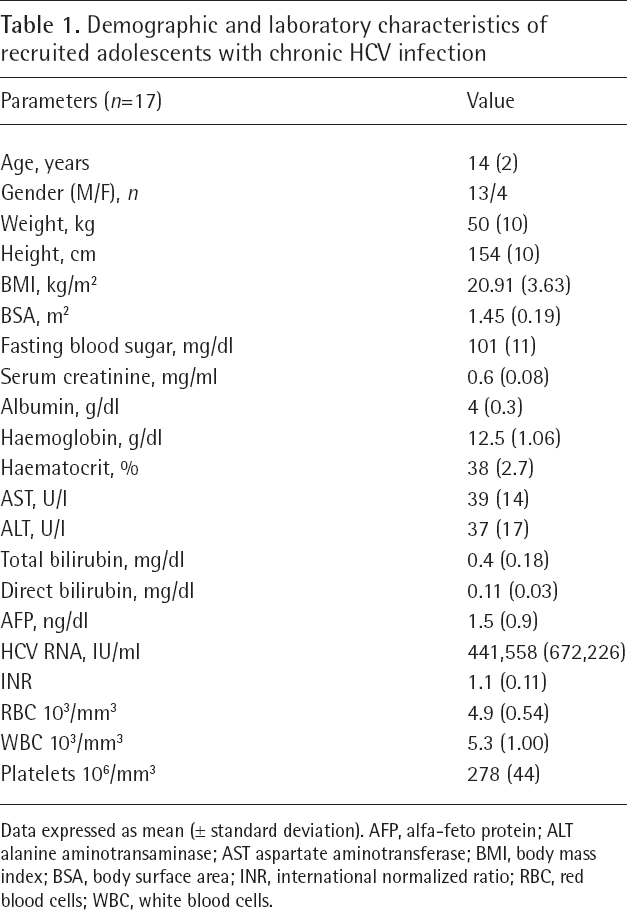
Data expressed as mean (± standard deviation). AFP, alfa-feto protein; ALT alanine aminotransaminase; AST aspartate aminotransferase; BMI, body mass index; BSA, body surface area; INR, international normalized ratio; RBC, red blood cells; WBC, white blood cells.
Non-Compartmental Pharmacokinetics
Daclatasvir demonstrated rapid absorption after oral administration, with all patients reaching Cmax at 2 h or less. The results from the NCA were comparable to those seen with compartmental modelling regarding the CL/F and V/F parameters (Table 2).
Daclatasvir non-compartmental PK parameters

AUC0–24 area under the concentration–time curve from 0 to 24 h; Cl/F, apparent clearance; Cmax, maximum plasma concentration; Cmin, minimum observed plasma concentration; CV, coefficient of variation; GM, geometric mean; Tmax, time of maximum observed concentration; T1/2, half-life; V/F, apparent volume of distribution.
Population Pharmacokinetics Analysis
PK Model Development
There were no measurements below the lower limit of quantification (LLOQ). A one-compartment PK structural model with zero order absorption was identified as an optimal model to describe the PK of daclatasvir in adolescents because this model revealed a smaller OFV (2,034) than other models. The parameters of the model were CL/F, V/F and K0. Residual variability was best described by a constant error model. The AIC and BIC of the model were 2,048 and 2,053, respectively. These results were supported by visual improvements between the observed and the predicted plasma concentrations, and the reductions of the weighted residuals for the predicted concentrations.
Only BW and baseline serum albumin resulted in a decrease in the OFV, decreased the AIC/BIC criteria and improved the GOF for V/F. According to the individual parameters versus covariates plots, serum albumin versus V/F showed a correlation of 0.55% and after adding the albumin covariate to the final model, there was a significant correlation (P-value =0.02). Other covariates did not lead to considerable improvement in the model. The relationship between V/F, body weight and serum albumin was described using the following model: V/Fi=52x(weight i /50)112x(albumin/4)2.71
Where V/F i is the apparent volume of distribution of the ith individual; weight i , is the weight of ith individual; 1.12 is the covariate coefficient of weight on the volume of distribution, albumin i , is the serum albumin of ith individual; 2.71 is the covariate coefficient of serum albumin on the volume of distribution.
Final Model Evaluation
The final PopPK parameter estimates and 95% CIs are summarized in Table 3. All the fixed-effect parameters (8) were well estimated with reasonable precision relative standard error (RSE%). The final model consisted of a one-compartment PK mammillary model with weight and baseline serum albumin as covariates on V/F using non-linear constant residual error model. Inter-individual variances were estimated with reasonable precision (RSE ≤30%). The η shrinkage for all PK parameters did not exceed 30% (16-25%), ruling out shrinkage of individual PK parameters towards population ones [15].
Daclatasvir population PK parameters
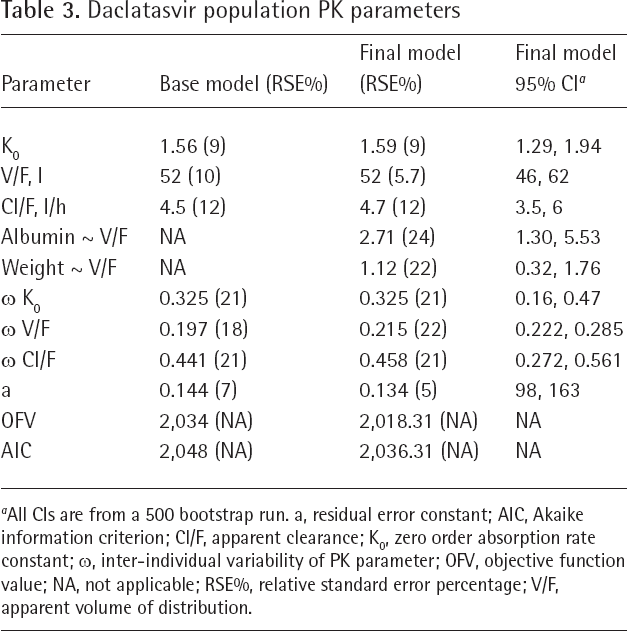
All CIs are from a 500 bootstrap run. a, residual error constant; AIC, Akaike information criterion; Cl/F, apparent clearance; K0, zero order absorption rate constant; w, inter-individual variability of PK parameter; OFV, objective function value; NA, not applicable; RSE%, relative standard error percentage; V/F, apparent volume of distribution.
Goodness-of-fit
The following graphs were plotted to evaluate the GOF: observed and predicted concentrations versus concentrations in the base and final models (Figure 1), weighted residuals versus time and weighted residuals versus predictions (Figure 2). Overall, the plots reasonably supported the final model and demonstrated no bias over model-predicted value and time. No systematic bias was found in PWRES and IWRES and most values were randomly distributed between -2 and 2 indicating proper model prediction (Figure 2). Regarding NPDE, it also showed random distribution within the values of -2 to 2, nearly normal distribution with a mean of zero and standard deviation approximately one, no systematic deviation with time indicating appropriate model fitting (Figure 2). The VPC for the final model showed that most of the observed data were within the prediction interval that encompasses 90% of the population as indicated by the 10th, 50th and 90th percentiles and the average prediction matches the observed concentration–time courses (Figure 3).
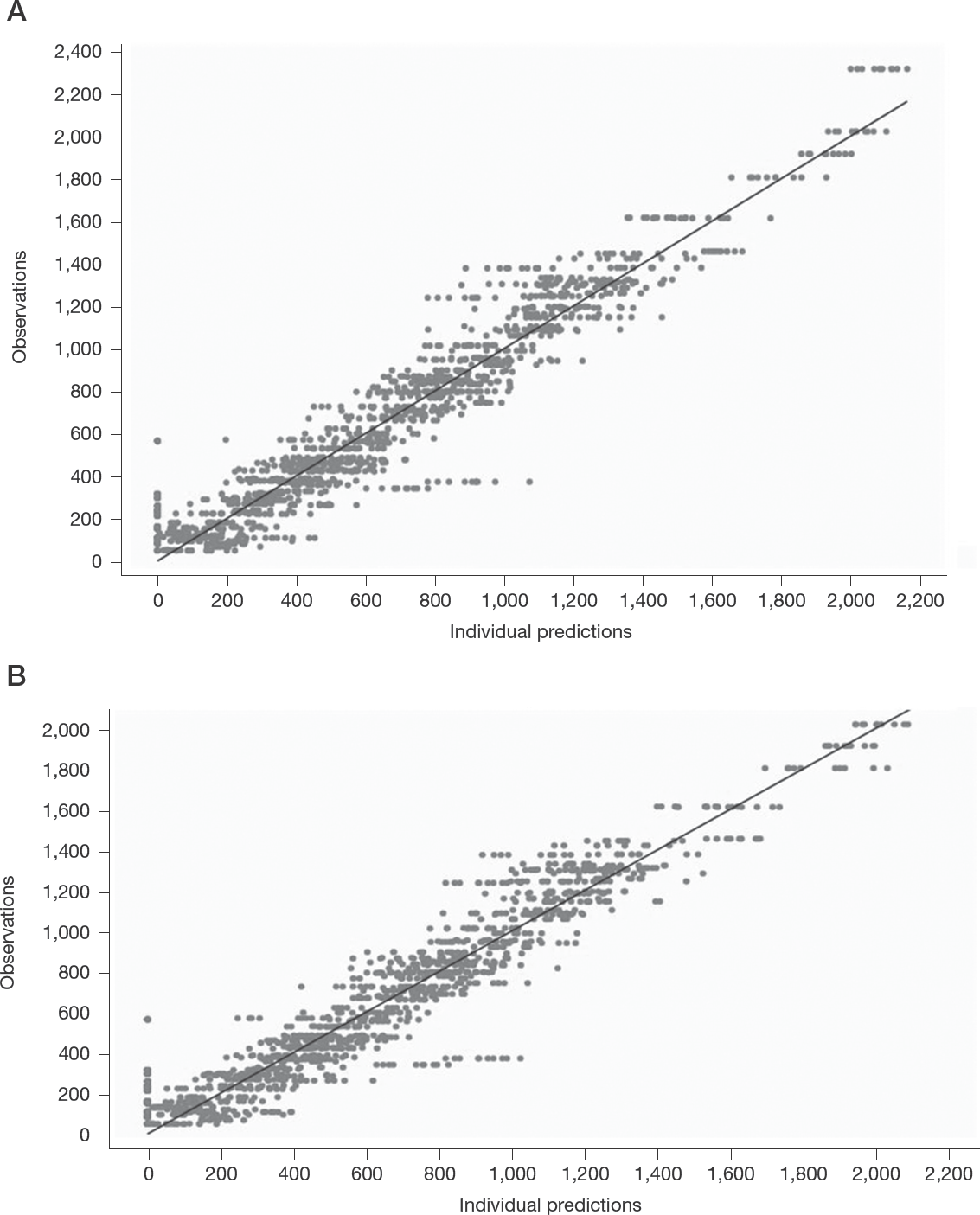
Predicted daclatasvir concentrations (ng/ml) versus observed concentrations
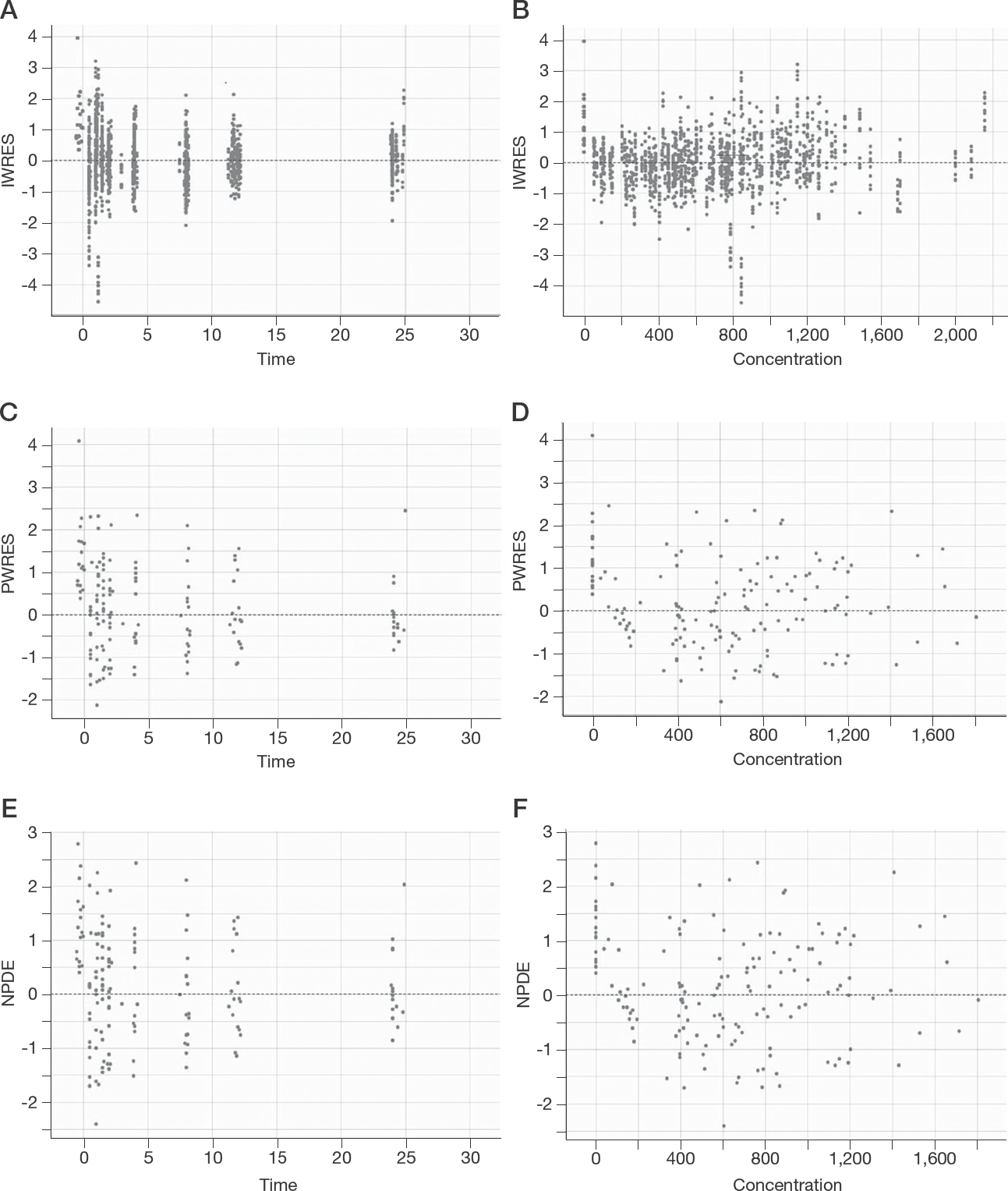
Goodness-of-fit plots

Visual predictive checks of daclatasvir for 1,000 simulations of concentration (ng/ml) versus time (h)
Discussion
To the best of our knowledge, this is the first reported study describing daclatasvir PK in adolescents infected with HCV and assessing the effect of multiple covariates on its PK using nonlinear mixed effects modelling. The concentrations of analysed samples were in the range of 49–2,319 ng/ml. The sample size was the maximum number of patients available.
The results of this study demonstrated that the non-compartmental PK profile of 60 mg daclatasvir was comparable to adults and consistent with the profile of daclatasvir in combination with sofosbuvir [7,16]. Similarly, daclatasvir was readily absorbed following oral administration with a Tmax of 1 to 2 h after dosing. In combination with sofosbuvir, adult daclatasvir Cmax, Cmin and AUC were found to be 1,089 ng/ml, 222 ng/ml and 11,702 ng/ml•h, respectively [14]. Concordant results were obtained in combination with asunaprevir [17]. Half-life at steady-state in adolescents was 8.5 h while in adults it was 12–15 h, so it was still rational to start sampling for the study after 1 week [18]. The reported volume of distribution of daclatasvir at steady state in adults was 58 l which is comparable with our results in adolescents [9], and Cl/F in the current study was 4.5 l/h, consistent with the previous PK analysis of daclatasvir in adults with a value of 4.4 l/h [19].
In the current study, we found that the PopPK of daclatasvir was characterized by a one-compartment model with zero-order absorption and first-order elimination. The final population PK model provided a good description of the PK data. Moreover, GOF criteria revealed that the final model was consistent with the observed data and no systematic bias remained. All PK parameters were estimated with reasonable precision (RSE <22%).
The results from our population model were concordant with the results obtained from two large population PK studies in adults [9,10], however, the population PK models were not similar. In the study by Osawa et al. [10] in Japanese subjects, daclatasvir PK was characterized by a 1-compartment model with first-order absorption. The population estimates of V/F and Cl/F were 64.2 l (95% CI: 60.1, 68.2) and 5.29 l/h (95% CI: 4.98, 5.59), respectively. Baseline body weight was the only single significant covariate on V/F while sex, treatment and baseline serum creatinine were all significant covariates on Cl/F [10]. While Chan et al. [9] reported a 2-compartment model with linear elimination and zero-order release of the drug followed by first-order absorption. Central V/F and Cl/F were 58.6 l (95% CI: 56.3, 60.9) and 5.7 l/h (95% CI: 5.52, 5.88), respectively [9]. In addition to body weight, both sex and race were found to be significant covariates on central V/F since several races were included. Significant covariates on Cl/F were baseline creatinine clearance, genotype, ALT, race and sex.
In our final model, V/F and Cl/F were 52 l (95% CI: 46, 62) and 4.7 l/h (95% CI: 3.5, 6). These CIs overlapped with CIs obtained from adult data. It is to be noted that the CIs obtained from the adolescent data were relatively wide due to the limited number of patients. Moreover, body weight was a significant covariate on V/F in adolescents similar to the two studies in adults. In this study and in the adult studies, weight was not identified as a covariate on Cl/F, even though the Japanese study had adults with weights ranging from 36 to 93 kg [10]. Two adolescent patients had body weights below 40 kg, the clearance seemed lower than the rest of the patients, with higher Cmax and AUC0–tau (data not shown). This effect could not be verified due to the limited number of patients, however the effect of weight on clearance might be more significant in younger patients with lower body weights.
Albumin was also a significant covariate on V/F, however, its effect on PK was not tested in the adult models [9,10]. Albumin is well documented to vary with age, peaking at 20 years [20], as well as in different disease states such as kidney and liver dysfunction [21]. Since daclatasvir is >99% bound to albumin [8], a decrease in albumin will increase the fraction unbound of daclatasvir and thus impact V/F. When daclatasvir PK was studied in patients with kidney dysfunction [19] and hepatic dysfunction [22] V/F, half-life and unbound daclatasvir fraction increased, even though these changes were concluded to be clinically insignificant and not warranting dose adjustment in these populations. Bifano et al. [22] found an inverse relationship between albumin concentration and unbound daclatasvir fraction, thus affecting V/F while clearance was relatively stable. Similarly, in this study albumin was not a significant covariate on Cl/F.
In adult patients, cirrhosis had no impact on the daclatasvir PK parameters [9,10], however, in our study subjects were non-cirrhotic, therefore, it is unclear whether cirrhosis affects the adolescent daclatasvir PK or not. In this study, serum creatinine as well as ALT were not significant covariates on Cl/F, since all patients had normal renal function and normal ALT levels, while they were significant in adults [9,10]. Moreover, all patients came from the same race and were infected with HCV genotype-4; thus, race and HCV genotype were not included in the covariate analysis. Sex was not found to be a significant covariate, as was the case with adults, probably due to the limited number of patients in our study.
Among the studied group of adolescents, age did not exhibit an influence on the PK parameters; this is consistent with adult PK results [9,10] where the adult population had an age range of 18–79 [17].
Since the observed PK in adolescents was comparable to adults using both NCA and PopPK analyses and that all 17 patients achieved SVR at 12 weeks post-treatment, it can be concluded that the 60 mg dose is appropriate for adolescents. Two previous studies demonstrated the efficacy of this dose in Egyptian adolescents but without reporting any PK [23,24].
The study had some limitations, the sample size was small, therefore, some covariates could not be included in the final model of daclatasvir. All patients in the study had genotype-4 so further studies with other genotypes would be needed. Further studies coupling daclatasvir PK to its pharmacodynamics are needed to show whether PK changes may lead to pharmacodynamic changes particularly in the younger age groups.
In conclusion, daclatasvir PK in adolescents was characterized by a one-compartment model with zero-order absorption and first-order elimination. Body weight and serum albumin were the major determinants of daclatasvir volume of distribution in this population. PK parameters were comparable to those reported in adults, indicating that 60 mg daclatasvir combined with 400 mg sofosbuvir is an appropriate dose for adolescents.
Footnotes
Acknowledgements
The authors acknowledge the Faculty of Medicine Ain Shams University Research Institute Clinical Research Centre (MASRI-CRC) and its staff members for conducting the clinical trial and the Ain Shams University Paediatric Hospital staff members for sample collection; Mamaly laboratory, Cairo, Egypt for analysis of blood samples, Zi Diligence laboratory, Cairo, Egypt for daclatasvir analysis, and The National Hepatology and Tropical Medicine Research Institute, Cairo, Egypt for the FibroScan and ultrasound patient investigations.
Laboratory work for patient screening and follow-up (except PK analysis) and medication provision was funded by Pharco Pharmaceuticals, Alexandria, Egypt.
The authors declare no competing interests other than the grant funding this research from Pharco Pharmaceuticals awarded to MHES.
