Abstract
Background
Methods
Results
Conclusions
Introduction
Patients with current or prior exposure to HBV infection have a risk of HBV reactivation (HBVr) related to immunosuppressive treatments (IST) [1]. The risk of reactivation depends upon host-related factors (young age, male sex, underlying disease, presence of cirrhosis), virological factors (hepatitis B surface antigen [HBsAg] positivity, HBe positivity, high baseline HBV DNA) and the IST protocol [2]. The consequences of HBVr can range from subclinical increases in HBV DNA to hepatitis and liver failure. HBVr can also disrupt the IST and hence delay the treatment of underlying disease [3,4].
Current guidelines recommend screening for HBV in all patients who will undergo IST, followed by starting antiviral drugs with a high barrier to resistance, entecavir (ETV) or tenofovir disoproxil fumarate (TDF), to those who have a moderate or high risk of reactivation, as soon as possible [5–8]. Despite these strong recommendations, the real-world data revealed inadequate HBV screening rates. For example, a study among 10,729 cancer patients in the United States reported that only 16.7% were screened for HBV infection before starting chemotherapy [9]. Therefore, HBVr and its associated clinical consequences are likely to be encountered after receiving IST in the near future.
Antiviral treatment should be started as soon as possible to prevent the development of liver decompensation after HBVr [5–8]. Third-generation antivirals (ETV, TDF) are potent drugs and have a higher genetic barrier to resistance, therefore they are recommended over lamivudine for established HBVr. In addition, there is limited data about the efficacy of third-generation antivirals in reducing the mortality and morbidity of patients with HBV flare. To our knowledge, there are only three studies (n=2–32) which reported a mortality rate of 0-13.6% after ETV treatment [10–12] and a small study (n=7) which reported no mortality after TDF treatment [13]. Another small study in patients with severe acute hepatitis (alanine aminotransferase [ALT] >10x upper limit of normal [ULN]) reported a mortality rate of 50% among patients treated with ETV (n=4) and no mortality in one patient treated with TDF [14].
Herein, we performed a multi-centric study in patients who developed HBV flare related to IST and evaluated their clinical course on treatment with third-generation antiviral drugs, mainly focusing on mortality and its predictive factors.
Methods
Study population
The study was conducted at 8 University Hospitals in Turkey. At each department, gastroenterologists or infectious disease specialists searched the medical records and identified the patients who were referred because of HBV flare related to IST between June 2014 and 2019. Patients who were already receiving an antiviral treatment for HBV at the onset of flare, those who had cirrhosis or biliary obstruction on abdominal ultrasonography, coinfection with hepatitis A, C, D or E virus or HIV, associated alcoholic, autoimmune or toxic hepatitis, inadequate follow-up, and age <18 years were excluded from the study.
Definitions
IST included cancer chemotherapeutic agents and other agents such as rituximab, corticosteroids and anti-tumour necrosis factor (TNF). HBVr was defined as a ≥1 log increase in HBV DNA levels from baseline or reappearance of HBV DNA in an individual who previously had an undetectable HBV DNA. HBV flare was defined as HBVr associated with at least a threefold elevation in ALT above the patient's baseline values or upper limit of the reference range, with or without clinical signs and symptoms of hepatitis. Severe HBV flare was defined as a serum ALT increase beyond 10xULN. Acute liver failure was defined as acute liver injury, hepatic encephalopathy, and an elevated international normalized ratio (>1.5). The risk of HBVr was classified according to the AGA guideline into high (>10%), moderate (1-10%) and low-risk (<1%) groups [5]. Time to starting antivirals was defined as the time elapsed from the detection of ALT elevation and initiation of antiviral treatment. Delta-MELD score was calculated by subtracting the MELD score at the diagnosis of HBV flare from the MELD score at the end of first week. Interruption of IST was defined as delaying IST for more than 7 days and discontinuation of IST was defined as failure to restart IST after interruption.
Study protocol and aims of the study
Initially, clinicians recorded the demographic data, primary disease, IST protocol, virological markers including HBsAg, anti-HBc immunoglobulin (Ig)G, hepatitis B e antigen (HBeAg) and HBV DNA, liver and kidney tests, INR, MELD and MELD-Na scores at baseline and at the onset of HBV flare. The decision to start ETV or TDF was at the treating clinician's discretion. The timing of starting antiviral treatment was at the clinician's discretion which was at HBsAg and/or HBV DNA positivity. Hospitalization and other treatments such as intravenous hydration, replacement of electrolytes, albumin and other blood products, antibiotics, lactulose, l-ornithine-l-aspartate and proton pump inhibitors were at the discretion of clinicians. The clinical course of the patients were evaluated by using the following laboratory and clinical parameters; liver tests, INR, HBV DNA, MELD, MELD-Na and delta-MELD scores, development of acute liver failure, duration of hospitalization, interruption of IST and liver-related mortality at 12 weeks. End points of the study were either mortality and liver transplantation or normalization of ALT, bilirubin and HBV DNA undetectability.
The primary aim of the study was to determine the liver-related mortality and/or liver transplantation rates at 12 weeks and its predictive factors. Secondary aims were to compare ETV and TDF with respect to mortality and time to ALT, bilirubin normalization and HBV DNA undetectability. The study protocol was approved by the local ethical committee of Sakarya University School of Medicine.
Statistical analysis
Categorical variables were compared by Fisher's exact test. Continuous variables were compared by Mann– Whitney U test. Kaplan–Meier survival curves of the two treatment groups were compared by using the log-rank test. Factors associated with liver-related mortality were identified with multivariate cox proportional hazards regression test, using variables which were significant in the univariate analysis, for both the reactivation and in the first week. The receiver-operating characteristic (ROC) curve was used to determine the sensitivity, specificity and cutoff values of the prognostic factors. P-values <0.05 were considered to be statistically significant. Statistical tests were performed by using the Statistical Package for the Social Sciences (version 25.0; SPSS, Inc., Chicago, IL, USA).
Results
Baseline clinical findings of the study group
Fifty patients were referred to gastroenterology or infectious disease departments for HBV flare related to IST. Out of them, 10 patients were excluded from the study because of various reasons and the remaining 40 patients (29 male, 11 female, mean age: 59 years) constituted the study group (Figure 1). Lymphoma was the most common primary disease (n=19). Rituximab-CHOP was the most common IST protocol (n=20). Five patients received combined chemotherapies for bone marrow transplantation. Thirty-three patients (82.5%) were screened for HBV in accordance with the guidelines (minimum HBsAg and anti-HBc IgG). Four patients (10%) were screened for only HBsAg. Neither HBsAg nor anti-HBc IgG was screened for in three (7.5%) patients. Screening revealed HBsAg positivity in 23 patients (62.1%), isolated anti-HBc IgG positivity in 9 (24.3%) and anti-HBc IgG/anti-HBs positivity in 5 (13.5%) patients. According to the AGA guideline, 25 patients (62.5%) were at a high risk of HBVr and 15 (37.5%) at a moderate risk. None of the subjects were at low risk of HBVr and prophylaxis should have been commenced in all. Serum transaminase levels were either within the normal range (95%) or <1.5xULN (5%; Table 1). Serum bilirubin levels were all within the normal range. None of the patients were receiving an antiviral drug for prophylaxis at the onset of HBV flare.

Flow chart of the study population
Baseline laboratory and clinical characteristics of the study group
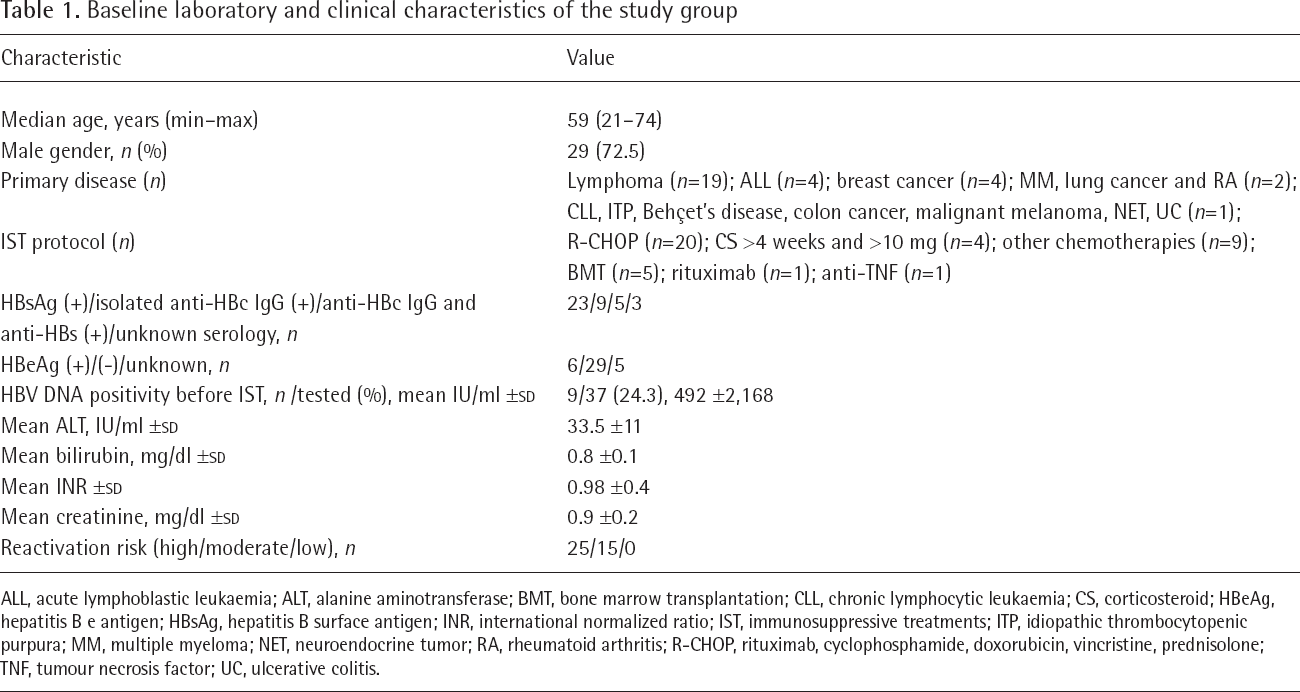
ALL, acute lymphoblastic leukaemia; ALT, alanine aminotransferase; BMT, bone marrow transplantation; CLL, chronic lymphocytic leukaemia; CS, corticosteroid; HBeAg, hepatitis B e antigen; HBsAg, hepatitis B surface antigen; INR, international normalized ratio; IST, immunosuppressive treatments; ITP, idiopathic thrombocytopenic purpura; MM, multiple myeloma; NET, neuroendocrine tumor; RA, rheumatoid arthritis; R-CHOP, rituximab, cyclophosphamide, doxorubicin, vincristine, prednisolone; TNF, tumour necrosis factor; UC, ulcerative colitis.
Among the nine HBsAg negative/anti-HBc-positive/anti-HBs-negative patients, seven patients received R-CHOP for lymphoma, and one patient received R-CHOP for multiple myeloma and fluorouracil, folinic acid, irinotecan and bevacizumab for colon carcinoma. Among the five HBsAg negative/anti-HBc-positive/anti-HBs-positive patients, three patients received combined chemotherapeutic agents for bone marrow transplation and one patient received R-CHOP for lymphoma and rituximab for chronic lymphocytic leukaemia, each. All of the 14 HBsAg-negative/anti-HBc-positive patients were HBsAg-positive at the time of the reactivation.
Clinical and laboratory findings at the onset of HBV flare
HBV flare developed during IST in 24 (60%) patients and 5 (1-9) months after IST in 16 (40%) patients. Mean serum ALT was 598 (135-3,013) IU/ml and 24 patients (60%) had a serum ALT >10xULN. Mean serum HBV DNA was 1.4x10 8 IU/ml (24,000-1,287,509,500). There was no significant difference in HBV DNA levels of the patients developing flare during (1.2 ±2.6x10 8 IU/ml) and after IST (1.6 ±3.3x10 8 IU/ml; P=0.64). Thirty patients (75%) had an elevated bilirubin level (1.3-21.7 mg/dl) and INR levels were elevated in 18 patients (1.2-3.2). MELD and MELD-Na scores were 14 (6-39) and 14 (6-42), respectively. 22.5% (n=9) of the patients had a MELD >20 and 27.5% (n=11) had a MELD-Na >20. None of the patients had acute liver failure at the onset of HBV flare (Table 2).
Laboratory findings at the onset of HBV flare

ALT, alanine aminotransferase; INR, international normalized ratio; ULN, upper limit of normal.
Treatment and prognosis
TDF was started in 26 patients and 14 patients received ETV for the treatment of HBV flare. Median time to starting antivirals was 5 (range: 3–16) days. Antiviral treatment was started at HBsAg positivity in 19 cases and at HBV DNA positivity in 21 cases. Eight patients (20%) developed acute liver failure during antiviral treatment. One patient with ulcerative colitis underwent living donor liver transplantation (LDLT) on the 10th day of flare and was well after 2 years of follow-up. Seven patients (17.5%) died due to liver-related complications after a mean hospitalization stay of 30 (range: 10–46) days and the remaining 32 patients were alive 12 weeks after the onset of HBV flare. Of the 32 surviving patients, 16 (50%) were hospitalized for a mean duration of 17 (6–32) days. Median time to ALT and bilirubin normalization was 5 (1–18) and 3 (1–14) weeks, respectively. HBV DNA became undetectable 9 (2–24) months after antiviral treatment. IST was interrupted in 11 (27.5%) patients for a mean duration of 23 days (7–90) and discontinued in 7 (17.5%) patients (Table 3).
The clinical course of the patients

ALT, alanine aminotransferase; AV, antiviral; ETV, entecavir; IST, immunosuppressive treatments; LT, liver transplantation; TDF, tenofovir disoproxil fumarate.
Factors predicting mortality
Patients were divided into surviving (n=32) and non-surviving (including the case with LDLT; n=8) groups. There was no significant difference between the groups with respect to age, gender, primary disease, viral serology, HBV load, IST protocol, the timing of flare and serum transaminase levels at the onset of flare. However, non-surviving patients had a significantly higher total and direct bilirubin, INR, MELD, MELD-Na scores and lower albumin levels at the onset of flare (P<0.05). Significantly more patients in the non-surviving group developed ascites (87.5% versus 3.1%) and encephalopathy during follow-up (87.5% versus 3.1%; P<0.001). Non-surviving patients had a significantly higher ALT, total and direct bilirubin, INR, MELD, MELD-Na, delta-MELD scores and lower albumin at the first week of flare (Table 4).
Comparison of the surviving and non-surviving patients with respect to laboratory and clinical parameters at the onset and first week of flare

ALT, alanine aminotransferase; AVT, antiviral therapy; ETV, entecavir; INR, international normalized ratio; NS, non-surviving (including the case with liver transplantation); TDF, tenofovir disoproxil fumarate; ULN, upper limit of normal.
We performed logistic regression analysis and ROC analysis to evaluate factors predicting mortality at the onset and first week of HBV flare. On univariate regression analysis, total and direct bilirubin, albumin, INR, MELD and MELD-Na scores at onset and ALT, albumin, INR, MELD, MELD-Na, delta-MELD scores at the first week were significantly associated with mortality. On multivariate analysis, total bilirubin at the onset (HR: 1.22, CI: 1.096, 1.362; P<0.001), ALT (HR: 1.003, CI: 1.001, 1.004; P=0.006) and delta-MELD (HR: 1.251, CI: 1.065, 1.470; P=0.007) at the first week were independent risk factors for liver-related mortality (Table 5). ROC analysis for evaluating prognostic performances are presented in Table 6 and Figure 2.

Receiver operating characteristic curves
Factors predicting liver-related mortality on univariate and multivariate analysis

Statistically significant at the onset of reactivation.
Statistically significant at the first week of flare. ALT, alanine aminotransferase; HR, hazard ratio; INR, international normalized ratio.
Receiver operating characteristic analysis of total and direct bilirubin, albumin levels, INR, MELD, MELD-Na score at the onset of flare, and ALT, total and direct bilirubin, albumin, INR levels, MELD, MELD-Na scores and delta-MELD at the first week to predict mortality

ALT, alanine aminotransferase; AUC, area under the curve; INR, international normalized ratio.
ROC analysis revealed that time to initiate antiviral treatment has no statistically significant ability to predict mortality (cutoff >4, AUC: 0.549, CI: 0.384, 0.706, sensitivity: 75%, specificity: 43.7%, P=0.7). In the surviving patients, there was no significant difference between the time to ALT normalization (5.4 ±3.6 versus 6.3 ±3.9 weeks; P=0.41), bilirubin normalization (4.6 ±3.9 versus 1.9 ±1.1 weeks; P=0.1) and HBV undetectability (9 ±4.7 versus 9.7 ±5.9 months; P=0.92, respectively) among groups who received antiviral treatment within the first 4 days (n=14) or ≥5 days (n=18) after HBV flare.
Comparison of the efficacy of antiviral treatments
Patients receiving ETV and TDF were similar with respect to age, gender, viral serology, serum transaminase and HBV levels at the onset of HBV flare. There were no significant differences between the antivirals with respect to time to starting treatment, time to normalize serum transaminases, bilirubin and undetectability of HBV DNA, hospitalization period, development of acute liver failure (ETV: 28.6% versus TDF: 15.4%; P=0.33) and mortality (ETV: 21.4% versus TDF: 16%; P=0.67).
Discussion
In this study, we found that the mortality due to immunosuppressive therapy related HBV flare is still high (20%), even after treatment with third-generation antivirals, which have a strong potency and high genetic barrier to resistance.
HBVr is a well-known and serious complication of IST [15,16]. Severe hepatitis and/or hepatic failure can develop in up to 25 to 50% of patients with HBVr and lead to death in approximately 25% of patients [17,18]. In addition, antiviral treatments are less effective when hepatitis develops [19]. Therefore, major society guidelines and national guidelines recommend HBV screening in all patients who will undergo IST and start antiviral prophylaxis according to the predicted risk of HBVr based on viral serology and potency of the IST [5–8,20]. Antiviral therapy initiated concurrently or prior to IST can decrease the risk of HBV reactivation (approximately 90%), HBV-related hepatitis and acute liver failure [21]. For these reasons, HBV screening before IST and antiviral prophylaxis when necessary is of great importance. In a retrospective study including 105 HBV carriers with lymphoma, 22 (21%) developed HBVr and 6 (27.2%) died due to hepatic failure following cessation of cytotoxic treatment [22]. In another study conducted in 45 HBV carriers receiving IST for lymphoma, 17 (37.8%) developed hepatitis and 7 (41.2%) died due to hepatic failure without antiviral therapy [23].
There are a limited number of studies evaluating the efficacy of antiviral drugs in the treatment of IST-related HBV flare [10–13,24–27]. Lamivudine (LAM) is the most frequently used AV drug in these studies and three small studies (n=5–15) revealed high mortality rates (12.5–25%) in patients with HBV flare [23–25]. Concerning third-generation antiviral drugs, a study with ETV revealed no mortality in 10 NHL patients who developed HBV flare (7 with severe hepatitis) [11]. Later on, a prospective study randomized 64 patients with IST-related HBV flare to LAM (n=32) and ETV (n=32) groups and found that the 6-month liver-related mortality rates (7.1% versus 13.6%; respectively) were not significantly different between the groups (P=0.41) [12]. Finally, there is only one small study evaluating the efficacy of TDF treatment in 13 patients with HBV reactivation (7 with flare) which revealed no mortality [13].
HBV flare needs to be treated as early as possible since the effects of antiviral therapy take time and may not prevent progression to acute liver failure if treatment is delayed until the patient shows evidence of severe liver disease (for example, jaundice or marked ALT increase) [11,14,25,28]. In our study, although, AV treatment was initiated earlier in the surviving group compared with the non-surviving group (5.2 ±2.2 versus 7 ±3.5 days, respectively), the difference was not statistically significant (P=0.619). A study done by US acute liver failure study group reported that patients with HBV associated acute liver failure after receiving IST (n=28) had an overall 21 day survival of 42.9% and only 12 of them were using a nucleoside/nucleotide therapy on admission [29]. Another study revealed that PT prolongation (HR: 1.21 per 1 s) and HBV viral load (HR: 2.51 per 1 log IU/ml) at the time of diagnosis were independent prognostic factors for liver-related mortality in patients with chemotherapy induced HBV flare [12]. Similarly, a study in patients with acute on chronic liver failure due to HBVr revealed that the use of antiviral agents was associated with viral suppression, disease progression, decrease in mortality, but the beneficial effects were most prominent in patients with a MELD score between 20 and 30 [30]. In our study, MELD >19, MELD-Na score >19.2, total bilirubin >6.2 mg/dl, INR >1.2 and albumin <3.3 g/dl were significantly associated with liver-related mortality at the onset of HBV flare and these findings showed that early diagnosis and initiation of antiviral treatments is of vital importance. In addition, our study showed that total bilirubin levels at the onset, ALT levels and delta-MELD score at the first week were independent predictors for mortality, therefore these patients should be evaluated for possible liver transplantation.
There is no study comparing the efficacy of ETV and TDF in the treatment of IST-related HBV flare. Our study revealed that there was no significant difference between ETV and TDF groups with respect to undetectability of HBV DNA, time to starting AVT, duration of hospitalization, time to normalize ALT and bilirubin levels, and mortality. However, seven patients (17.5%) died due to liver-related complications and one patient (2.5%) underwent liver transplantation, which indicated that mortality was still high despite the use of third-generation antiviral agents.
Our study had several limitations. It was a retrospective study and had a relatively small sample size, especially in the non-surviving group. HBV genotype, HBsAg and anti-HBc IgG quantification could have been analysed to see whether there was an association with outcomes of flares. Unfortunately, they couldn't be evaluated due to the retrospective nature of the study. However, it was a multicentric study, included a considerable number of patients (n=40) and was the first one comparing the efficacy of ETV and TDF in patients with IST-related HBV flare.
In conclusion, IST-related HBV flare still has a high mortality in the third-generation antiviral treatment era. High INR at the first week of flare was an independent risk factor for mortality. Every effort should be spent to increase HBV screening rates and thereby evaluate the patients for prophylaxis before starting immunosuppressive treatments and immediately starting antivirals once HBV flare develops.
Footnotes
Acknowledgements
Conception or design of the work: BT, ASK, GŞ, ATE, SA, SH. Data collection: BT, ASK, Gİ, EÇ, OÜ, MS, GŞ, AEY, SF, ŞA, ATE and MIU. Data analysis and interpretation: BT, ASK, Gİ, EÇ, OÜ, MS, GŞ, AEY, SF, ŞA, ATE and MIU. Drafting the article: BT, ASK, Gİ, ATE and SH. Critical revision of the article: BT and ASK. Final approval of the version to be published: BT, ASK, Gİ, EÇ, OÜ, MS, GŞ, AEY, SF, ŞA, ATE, MIU and SH.
All authors approved the final manuscript as submitted.
All co-authors take full responsibility of the integrity of the study and all parts of the manuscript. The study was funded by the authors.
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. Informed consent was obtained from all individual participants included in the study.
The authors declare no competing interests.
