Abstract
Background
Cofactors associated with persistently abnormal CD4+:CD8+ T-cell ratio in people with HIV (PWH) on antiretroviral treatment (ART) might change over time as the population of people with HIV ages or as new ART drugs become available. The main objective of our study was to determine the long-term associations of baseline factors, including the CD4+ T-cell count and ratio, with ratio normalization (≥1). In addition to this, we explored whether the ratio remained associated with the risk of both AIDS and non-AIDS events among individuals on suppressive ART.
Methods
Clinic-based study in a tertiary, university hospital in Madrid. People with HIV starting a first-line ART regimen (January 2006–June 2017) were included in a prospective national multicentre cohort (CoRIS). People with controlled HIV-infection within the first year of ART initiation and complete CD4+ and CD8+ T-cell records were selected. Cox proportional hazard (PH) regression models were used to estimate the cumulative incidence of ratio normalization and to examine associations with socio-demographic and clinical variables. To investigate factors independently associated with the development of AIDS and non-AIDS events we used a time updated Poisson regression model.
Results
The study included 557 subjects. During follow-up (median 5.24 years), 44% of participants achieved a ratio of 1 within a median of 1.49 years. In a multivariate PH model, pre-ART factors negatively associated with ratio normalization were the pre-ART CD4+:CD8+ T-cell ratio and mode of HIV acquisition. For the secondary analysis, 1.3 events/100 person years of follow-up were observed. After adjustment, older age, HIV RNA >200 copies/ml and CD4+:CD8+ T-cell ratios over follow-up, remained significantly associated with the development of AIDS and non-AIDS events. In contrast, pre-ART ratio was not associated with the risk of AIDS and non-AIDS events.
Conclusions
In summary, our study showed that higher pre-ART CD4+:CD8+ T-cell ratio is associated with rates of ratio normalization ≥1. In addition, the risk of AIDS and non-AIDS events seems to be predicted by the time updated CD4+:CD8+ T-cell ratio not by the pre-ART CD4+:CD8+ T-cell ratio. Therefore, CD4+:CD8+ T-cell ratio should be considered as a dynamic marker for translation into clinical practice.
Introduction
The CD4+:CD8+ T-cell ratio is considered to be a useful surrogate marker of immune activation and inflammation. In addition, CD4+:CD8+ T-cell ratio is an important time-dependent prognostic factor for treatment-naive and cART-treated patients [1]. Persistence of a low CD4+:CD8+ T-cell ratio has been reported to be associated with an increased risk of morbidity and mortality [2]. Indeed, it has been reported that the benefits of earlier antiretroviral treatment (ART) initiation and modern regimens are partly due to a shorter time to CD4+:CD8+ T-cell ratio normalization [3].
It is not clear if ART is able to fully restore the immune system even when it is started soon after infection. Important cofactors associated with persistently abnormal CD4+:CD8+ T-cell ratio changes in treated patients might change over time as patients age or as the timing of ART is modified. Methods used in studies that have evaluated the CD4+:CD8+ T-cell ratio were mostly based on cross-sectional designs and only a few longitudinal studies have included virological failure. Additionally, few studies have assessed factors associated with CD4+:CD8+ T-cell ratio restoration or the relationship between baseline CD4+:CD8+ T-cell ratio and the risk of both AIDS and non-AIDS events. Hence, more data about the factors associated with CD4+:CD8+ T-cell ratio changes after initiation of ART, and the association with events in ART-treated people with HIV (PWH) is required. With this information we can help to define a role for the CD4+:CD8+ T-cell ratio in routine clinical care.
The primary objective of our study was to determine the long-term associations of baseline factors, including CD4+ T-cell count and CD4+:CD8+ T-cell ratio before ART initiation, with CD4+:CD8+ T-cell ratio recovery in patients with controlled HIV-infection within 12 months of treatment initiation. Our secondary objective was to determine if the CD4+:CD8+ T-cell ratio remains associated with the risk of AIDS and non-AIDS events among individuals on suppressive ART after adjusting for baseline factors.
Methods
Study Design, Setting and Participants
We conducted a clinic-based study among PWH who were ART-naive at the University Hospital Ramón y Cajal, Madrid. We selected those starting a first-line ART regimen from January 2006 to June 2017 who were included in a prospective national multicentre cohort of HIV-positive subjects (CoRIS). Ethics committee approval was obtained from the institutional review board at the Ramón y Cajal Hospital, and all participants provided written informed consent at enrolment to CoRIS.
We included people with controlled HIV infection defined as at least one viral load (VL) <200 copies/ml (2006–2010) or <50 copies/ml (2011 onwards) within 12 months of treatment initiation. Inclusion criteria included: 6 months of follow-up after initial plasma HIV RNA suppression and at least one CD4+:CD8+ T-cell ratio assessment 6 months after ART initiation.
Variables
The following data were collected: age at ART initiation, sex (male, female), geographical origin, education level (high school or lower, secondary, university, unknown), mode of HIV acquisition (sex between men, injection drug use [IDU], sex between men and women, unknown), ART regimens and dates of ART initiation, CD4+ and CD8+ T-cell counts (from immunology records) and HIV VL from ART initiation onwards, diagnosis of AIDS (defined according to the Centers for Disease Control and Prevention) and the following non-AIDS events [4]: non-AIDS defining malignancies, cardiovascular disease/stroke, decompensated liver disease (variceal bleeding, encephalopathy, ascites, hepatocarcinoma), end-stage renal disease and diabetes mellitus.
Baseline assessments were determined during the 6-month period prior to ART initiation. Follow-up continued until the occurrence of death from any AIDS or non-AIDS cause (as listed above) or from any other cause of death not listed above or the last date of CD4+ and CD8+ T-cell count measurement.
Statistical Analysis
For the primary analysis, participant follow-up started on the date of ART initiation and ended on the date of normalization of the ratio, defined as a CD4+:CD8+ T-cell ratio ≥1. This cutoff was selected based on previous literature [5]. Follow-up on subjects was right-censored on the earliest date of development of a new AIDS-defining event, a non-AIDS defining event, or on dis-enrolment from the hospital (loss to follow-up). Cox proportional hazard (PH) regression models were used to estimate the cumulative incidence of CD4+:CD8+ T-cell ratio recovery and to examine associations of this with socio-demographic and clinical variables. Based on published findings [2,6–8], we examined associations with mode of HIV acquisition, gender, age at treatment initiation, baseline CD4+:CD8+ T-cell ratio, antiretroviral treatment and calendar period of treatment initiation. Calendar time was split into three periods: 2006–2008 (refecting the approval of tenofovir disoproxil fumarate [TDF]/emtricitabine [FTC]/ efavirenz [EFV]); 2009–2014 (refecting the introduction and use of integrase strand transfer inhibitors [INSTIs]); and 2015–2017 (refecting the extended use of INSTI-based single-tablet regimens). To avoid the potential collinearity between HCV, HBV and IDU, hepatitis was excluded from the analysis and IDU was included because it was the variable with the strongest association.
To investigate factors independently associated with the development of AIDS and non-AIDS events, we used Poisson regression models. For these analyses, follow-up started 6 months after ART initiation and ended at the earliest date of a clinical event (date of AIDS event, date of non-AIDS event or date of death), date of last clinical visit or date of loss to follow-up before June 2017. We examined associations with mode of HIV acquisition, baseline T-cell CD4:CD8 ratio, time-updated age, time-updated HCV status and time-updated T-cell CD4:CD8 ratio. Selection of variables for the multivariable model were based on factors significantly associated with the risk of events in our population.
Results
Characteristics of the Patients
From January 2006 to June 2017, 617 ART-naive individuals initiated ART and had at least one undetectable plasma HIV RNA within the first year after ART initiation. Of these, 557 had sufficient CD4+ and CD8+ T-cell counts for inclusion. In total, these individuals contributed 7,560 CD4+ and CD8+ T-cell measurements over follow-up. Median total follow-up time from ART initiation was 5.24 (IQR: 2.50–7.91) years.
The study population comprised 467 men (83.8%), 338 men having reported sex with men (MSM; 60.7%) with 380 individuals from Spain (68.2%). Pre-ART initiation, the median (IQR) age was 37 (29–44) years, the CD4+ T-cell count was 342 (232–468) cells/ml and the CD8+ T-cell count was 960 (720–1,302) cells/ml (Additional file 1). Compared with the excluded sample (n=60), participants included in the study (n=557) had been less frequently diagnosed with CDC classification C (14% versus 25%), were more likely to have started ART in the period between 2009 and 2014 (55% versus 23%), were more commonly treated with an INSTI-based regimen (28% versus 22%) and were less frequently HBsAg positive (3% versus 8%).
CD4+:CD8+ T-cell Ratio Changes
As mentioned, only patients with all CD4% and CD8% or absolute values for each period were included. The median CD4+:CD8+ T-cell ratio at baseline was 0.32 (IQR 0.19–0.50) and minimum-maximum values were 0.01 and 1.45. 28% exhibited a baseline ratio of less than 0.2. 245 (44%) attained a CD4:CD8 ratio of ≥1 within a median time of 1.49 (0.83–3.22) years of initiating ART.
Figure 1 shows the median CD4+:CD8+ T-cell ratio stratified by the baseline CD4+:CD8+ T-cell ratio (Panel A) and CD4+ T-cell count (Panel B). Whilst the CD4+:CD8+ T-cell ratio increased over time in all groups, individuals with a pre-ART ratio below 0.6 or a CD4+ T-cell count lower than 500 cells/ml did not appear to normalize the CD4:CD8 T-cell ratio within the 2-year period.

Ratio dynamics in the first two years after ART initiation by pre-ART CD4+:CD8+ T-cell ratio and pre-ART CD4+ T-cell count
Factors associated with CD4+:CD8+ T-cell Ratio Normalization
The association of normalization was assessed with the following pre-ART variables: age, gender, origin (region), risk factors for HIV acquisition, year of ART initiation, type of ART regimen, pre-ART CD4+:CD8+ T-cell ratio.
In the adjusted Cox regression model pre-ART factors negatively associated with CD4+:CD8+ T-cell ratio normalization were the pre-ART CD4+:CD8+ T-cell ratio and mode of HIV acquisition (Table 1).
Pre-ART associations with CD4+:CD8+ T-cell ratio normalization in 557 individuals with complete data

Cox regression model after backward selection. ART, antiretroviral treatment; HBsAg, hepatitis B surface antigen; INSTI, integrase strand transfer inhibitor; NNRTI, non-nucleoside reverse transcriptase inhibitor; NRTI, nucleoside reverse transcriptase inhibitor; PI, protease inhibitor.
We also explored various models to determine associations of the various immunological markers (pre-ART CD4+:CD8+ T-cell ratio, pre-ART CD4+ and CD8+ T-cell count, or pre-ART CD4+ and CD8+ percentage) with normalization of the ratio (Figure 2). Although similar conclusions were reached with each combination of parameters, the CD4+:CD8+ T-cell ratio has been included in subsequent analyses as we had complete data for CD4+:CD8+ T-cell ratio for each period.

Forest plot summarizing hazard ratios and CIs from multivariable analyses assessing association between different combinations of pre-ART CD4+ T-cell count (and %), CD8+ T-cell count (and %) and CD4+:CD8+ T-cell ratio with ratio normalization
Risk of AIDS and non-AIDS Defining Conditions
Of the 557 included people, 548 were eligible for this analysis (Table 2). Nine individuals were excluded due to insufficient follow-up after ART initiation (six MSM, two with AIDS at baseline, eight starting ART during the period 2012-2014 and eight who were receiving an INSTI). After initial suppression post cART initiation, individuals had undetectable viral load for 91% of their total follow-up time (based on monthly intervals).
Pre-ART characteristics of people with HIV according to event occurrence

ART, antiretroviral treatment.
There were a total of 41 recorded events in 35 eligible participants: 13 AIDS events, 9 non-AIDS-defining malignancies, 6 cardiovascular events, 1 stroke, 7 diabetes and 5 hepatic events. Nine subjects died of causes related to AIDS and non-AIDS-defining conditions, four from other causes, and in one person the cause of the death was unknown. Three people had two or more AIDS events and three had two or more non-AIDS events; the median time to the first event was 1.9 (CI: 0.49, 4.02) years. The 41 events were observed over a total time of 2,726 person-years (1.38 [CI 0.99,1.93] events per 100 person years of follow-up). Event rates varied by CD4+:CD8+ T-cell ratio category, ranging from 6.56 (CI 3.13, 13.77) events per 100 person-years in those with a ratio <0.2 to 0.29 (CI 0.72, 1.15) events per 100 person-years in those with a ratio >1 (Figure 3).

Event rates (per 100 person-years) within each category of the time-updated CD4+:CD8+ T-cell ratio category
Using Poisson regression analysis, we calculated univariable and multivariable incidence rate ratios (IRR) adjusting for mode of HIV acquisition, HCV serology, age (per 5 years), HIV RNA copies/ml (≤200, >200) and CD4+:CD8+ T-cell ratios over follow-up all as time-updated covariates.
An unadjusted time-dependent model did not show a higher risk of events with higher pre-ART CD4+:CD8+ T-cell ratio (0.1 increase; IRR 0.89 [CI 0.75,1.05]). After including significant factors into a multivariable model (Table 3), only older age, HIV RNA >200 copies/ml and CD4+:CD8+ T-cell ratios over follow-up remained significantly associated with the outcome, although the effects of other factors (for example, mode of HIV acquisition) generally remained similar although were attenuated (and failed to reach statistical significance, likely due to lack of power).
Poisson regression analysis: risk of AIDS and non-AIDS events in people with HIV
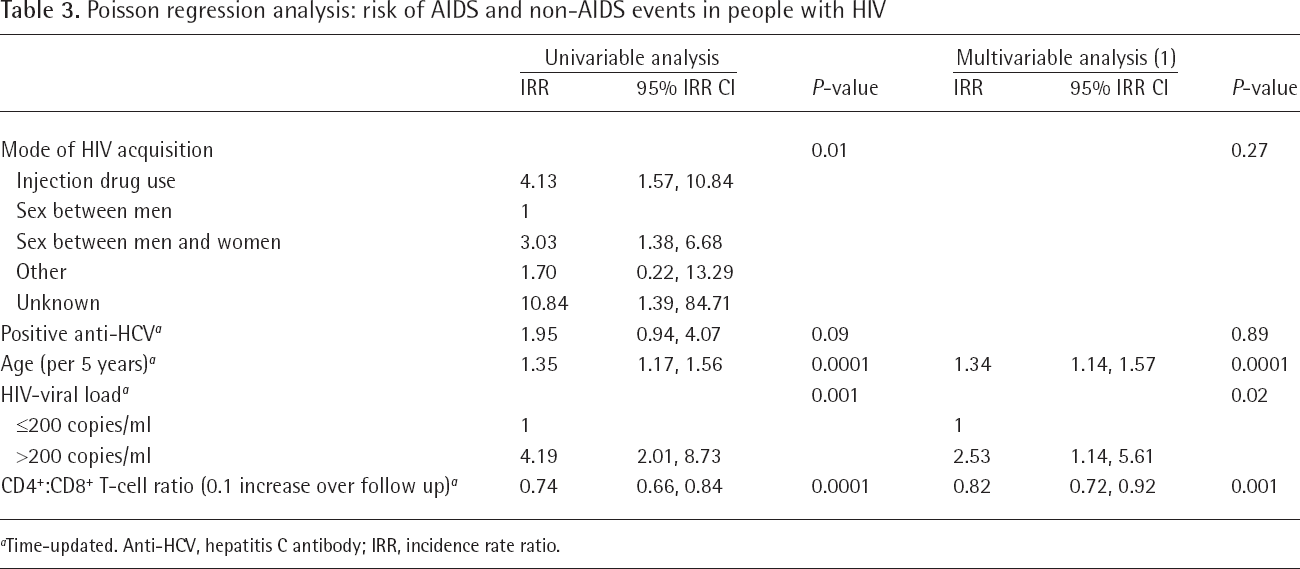
Time-updated. Anti-HCV, hepatitis C antibody; IRR, incidence rate ratio.
Discussion
In this cohort study, we show that pre-ART CD4+:CD8+ T-cell ratio levels are associated with CD4+:CD8+ T-cell ratio normalization during therapy. However, the development of AIDS and non-AIDS events appear to be better related to the changing CD4+:CD8+ T-cell measurement after starting ART compared with the pre-ART CD4+:CD8+ T-cell ratio, as additional factors could influence the ratio evolution during the time. The finding is supported by the fact that the ratio generally increases under ART but can also drop in some individuals. Of note, CD4+:CD8+ T-cell ratios were seen to improve even in people with AIDS and those with low CD4:CD8 ratio before ART, thus reducing the risk of future events even in these people.
This is the first study to show a different association between the pre-ART and on-ART CD4+:CD8+ T-cell ratio and the risk of any event over follow-up. In our cohort, the pre-ART CD4+:CD8+ T-cell ratio did not appear to continue to result in poorer clinical outcomes over time provided HIV was controlled and the ratio was recovered, at least over the median follow-up period included.
Of the 557 individuals included in the study, 245 (44%) achieved a CD4+:CD8+ ratio of 1 within a median of 1.49 (0.83–3.22) years of initiating ART. This contrasts with lower rates of normalization (around 30%) found at 8 years in a French study [9] and the probability of achieving normalization at 5 and 10 years of 19% and 39%, respectively, in a Thai prospective cohort [10].
Consistent with prior studies [6,9], pre-treatment CD4+ T-cell count is described as a predictor of failing to normalize the CD4+:CD8+ T-cell ratio. We were not able to show that other factors at the time of cART introduction or the first-line regimen were also associated with a better restoration of the ratio. Few studies have examined demographic factors associated with CD4+:CD8+ T-cell ratio normalization under ART. Due to our population characteristics, those who acquired HIV through IDU were significantly less likely to experience a normalization of their CD4+:CD8+ T-cell ratio. Whilst some studies reported better normalization in women than men [11,12], we found no association in normalization with female sex. In addition, although age was generally associated with ratio outcomes [11–13], in our study, younger age was not associated with ratio normalization, but was associated with a lower risk of AIDS or non-AIDS events.
Regarding the prognostic value of CD4+:CD8+ T-cell ratio, some previous research has shown that pre-treatment CD4+ T-cell count may be the best marker to predict the immune recovery [3,14], although conflicting information still exists [2,7,15–18]. The large study of Trickey et al. [17] concluded that the magnitude of adjusted associations of CD4+:CD8+ T-cell ratio or CD8+ T-cell count with mortality was too small for them to be useful as independent prognostic markers in virally suppressed patients on ART. According to our results, the pre-ART CD4+:CD8+ T-cell ratio, or the combination of both CD4+ and CD8+ T-cell count or CD4+ and CD8+ percentages were all similarly associated with ratio normalization.
In the general population, ratios of 1.5–2.5 are accepted as normal, although the normal CD4+:CD8+ T-cell ratio is heterogeneous because sex, age, ethnicity, genetics, exposures and infections may also impact the ratio [5]. However, in the HIV-positive population, HIV infection causes a depletion of CD4+ T-cells whereas CD8+ T-cell counts increase. After ART introduction, CD8+ T-cell counts decrease, but tend to stabilize at higher levels than is usual in the general population [19]. Furthermore, whilst patients on ART have a progressive improvement in CD4+:CD8+ T-cell ratio as their CD4+ T-cells increase and CD8+ T-cell counts remain high in most cases, it is also known that HIV-positive individuals who fail to normalize their ratio have an overall increased risk of morbidity and mortality [2]. Most importantly, the immune activation and senescence are seen at a much younger age [18].
Therefore, some studies have suggested that there may be immunological benefits to the initiation of ART prior to an increase in CD8+ T-cell count [17]. According to the START study, the risk of AIDS and non-AIDS events was reduced by 57% in those patients who initiated ART immediately after diagnosis, whilst the CD4+ T-cell count remained >500 cells/ml, compared with those who deferred treatment until their CD4+ T-cell count had fallen below 350 cells/ml [20].
The reason for the discrepancy with pre-ART CD4+:CD8+ T-cell ratio and the risk of AIDS and non-AIDS events remains unclear. Both early initiation, mainly during primary HIV-infection [21,22], and continuous adherence to treatment are important to achieve normalization of CD4+:CD8+ T-cell ratio. For this reason, we adjusted for time-updated viral load in our regression models, as we were not able to include adherence in our analysis.
Some limitations of our study should be considered. We have no information on other factors that can influence the ratio, including smoking, alcohol, cytomegalovirus (CMV) infection, and other comorbidities (for example, obesity, hypertension). It is well known that CMV infection has a significant impact on the CD4+:CD8+ T-cell ratio through the expansion of CD8+ T-cells [23]. One of the limitations of this study had to do with the small number of AIDS and non-AIDS events. Of note, events occurred at different times but more frequently in recent time periods, specifically, from 2011. There are also implications regarding immune recovery depending on the timing of ART initiation from the real date of HIV infection that we were unable to address in our study [3]. On the other hand, the impact of different ART regimens during follow-up has not been analysed. A study from a French cohort reported that initiation of cART with an INSTI regimen was strongly associated with a faster rate of ratio normalization when compared with initiation with non-INSTI containing regimens [24]. INSTIs became available more recently than protease inhibitors (PIs) and non-nucleoside reverse transcriptase inhibitors (NNRTIs), and this coincides with changing views on the timing of initiation of ART, with a consensus toward universal treatment emerging just as INSTIs became more widely used and NNRTIs and PIs became less favoured options.
Unfortunately, after viral suppression, treatment failure, resistance tests and switching regimens could not be addressed clearly in our database. Major strengths of this study are the time update HIV viral load after viral suppression in the analysis, and the prospective study follow-up design.
In conclusion, our study showed that higher pre-ART CD4+:CD8+ T-cell ratios are associated with rates of ratio normalization. In addition, the risk of AIDS and non-AIDS events are more strongly associated with the CD4+:CD8+ T-cell ratio over follow-up than with the pre-ART CD4+:CD8+ T-cell ratio at baseline. Therefore, the CD4+:CD8+ T-cell ratio should be considered as a dynamic marker for translation into clinical practice.
Footnotes
Acknowledgements
This work has been partially funded by the SPANISH AIDS Research Network (RIS) RD16/0025/0001 project as part of the Plan Nacional R + D + I and cofinanced by ISCIII- Subdirección General de Evaluación y el Fondo Europeo de Desarrollo Regional (FEDER). MJVG is funded by grants from the Spanish Ministry of Science and Innovation (Ayudas para Contratos de Formación en Investigación Río Hortega).
The funding bodies did not have a role in the design or conduct of the study, the analysis and interpretation of the results, the writing of the report or the decision to submit the manuscript for publication.
All authors have revised and approved the manuscript and contributed significantly to the work. This manuscript has not been previously published nor has it been considered for publication elsewhere.
We thank all the study participants who contributed to this work, as well as the clinical research staff of the participating institutions who made this research possible.
MJVG: design of the work, analysis, writing and interpretation of the data. MJPE, CGA, AMZ, JLC, CQ, JMS, MSC and FD: collected clinical data. SDC and JCG: collected laboratory data. HO and CAS: statistical analysis, participated in writing and technical editing of the manuscript. SM and SSV: revising manuscript critically for important intellectual content. SM and CAS: final approval of the manuscript. Authors agreed on all aspects of the work.
MJVG reports grants and personal fees from Gilead and ViiV outside the submitted work. MJPE reports grants and personal fees from AbbVie, ViiV Healthcare, Gilead Sciences, Janssen Cilag. MSC reports personal fees from MSD, personal fees from ViiV Healthcare, personal fees from Gilead, outside the submitted work. SSV reports grants and personal fees from Gilead, grants and personal fees from MSD, personal fees from ViiV, outside the submitted work. SM reports grants and personal fees from ViiV Healthcare, grants and personal fees from Gilead Sciences, grants and personal fees from MSD, grants and personal fees from Janssen Pharmaceuticals, outside the submitted work. CAS has received funding for membership of Data Safety and Monitoring Boards, Advisory Boards and for preparation of educational materials from Gilead Sciences and ViiV Healthcare. All other authors report no potential conflicts of interest.
