Abstract
Background
Methods
Results
Conclusions
Introduction
In 2014 there were 37 million people infected with HIV and about 115 million people with antibodies to HCV [1]. Coinfection by HIV and HCV is common worldwide, due to the shared routes of parenteral transmission (especially intravenous drug use and homosexual sex). Parenteral transmission is the primary means of HIV and HCV acquisition in persons who injected drugs (PWID); however, recent studies have reported that, among HIV-infected men who have sex with men (MSM), HCV infection rates and re-infection rates are high [2–6]. In fact, up to 6% of people living with HIV (PLWH) in the United States (US) are coinfected with HCV, 59% of whom are PWID [1], followed MSM, and pregnant or heterosexually exposed populations.
HIV–HCV coinfection has been associated with more rapid HCV-related fibrosis progression, hepatocellular carcinoma (HCC) and liver failure [7]. In addition to liver disease, HCV may be associated with symptomatic vasculitis due to cryoglobulinaemia, renal disease (membranoproliferative glomerulonephritis), lymphoproliferative diseases and porphyria cutanea tarda [8–12]. Since antiretroviral therapy (ART) improves the outcome of HIV infection, HCV in coinfected patients is becoming a major cause of morbidity and mortality [1,13–17].
Recently, the treatment against chronic HCV infection has been revolutionized. Historically, the combination of pegylated interferon (PEG-IFN) and ribavirin showed a low rate of sustained virological response (SVR) and a very high rate of adverse events, and therefore it was rarely used in HIV–HCV-coinfected patients because of the higher rate of adverse events and lower rates of SVR than those observed in HCV-monoinfected patients [18–20]. In the last few years, regimens without IFN, which combine several classes of directly acting antiviral agents (DAAs), have improved the response rate and tolerability of anti-HCV treatment [21–25]. In fact, these IFN-free regimens yield SVR rates of approximately 95%, even in difficult-to-treat patients, such as patients with an advanced liver disease, cirrhosis or HIV coinfection [23,25–28]. However, few data from real-world studies are available on the efficacy and tolerability of DAA regimens in HIV-infected patients [23,26,27]. Moreover, the management of DAA treatment of HCV–HIV-coinfected patients may be complex, especially due to the presence of other co-pathologies and the drug–drug interactions with ART or other comedications, such as oral opioids in PWID [29]. However, switching ART regimens to avoid DAA–ARV interaction was associated with a loss of HIV control and with non-SVR [30].
Therefore, to address this, we evaluated a cohort of HIV-infected patients treated with a DAA regimen in order to identify the variables associated with SVR and the trend in biochemical parameters and clinical events during and after DAA regimen.
Methods
Study design
We performed a multicenter retrospective cohort study, enrolling all the HIV–HCV-coinfected adult patients treated with DAAs from January 2015 to December 2018 in one of the nine participating Infectious Disease Centers in southern Italy, eight in Campania and one in Apulia. These centres have cooperated in several clinical investigations using the same clinical approach and the same laboratory methods [31].
The inclusion criteria were the presence of chronic HCV infection (anti-HCV/HCV RNA positivity for at least 6 months), the presence of HIV infection (anti-HIV positivity), an age of 18 years or older and having received treatment with an IFN-free regimen.
Exclusion criteria were the presence of Child-Pugh C cirrhosis, as required by Italian regulatory agency, a diagnosis of active HCC at baseline, of active disease defining AIDS or the refusal to consent to participate in the present cohort.
Patients who had previously received IFN-based anti-HCV treatment were defined as treatment-experienced; patients who had never received previous treatment against HCV were defined as treatment-naive. The severity of liver disease was graded according to the degree of liver fibrosis by Metavir stage or clinical signs. The Metavir score was estimated with a FibroScan© exam performed within 6 months before the beginning of antiviral treatment. Moreover, clinical cirrhosis was identified on the basis of the presence of unequivocal clinical, biochemical and ultrasound signs including a blood platelet count lower than 100,000/mm3, hypertrophy of the caudate lobe, nodularity of the liver surfaces, altered straightness of hepatic veins, ascites, porto-systemic encephalopathy, oesophageal varices and ultrasound evidence characterizing liver cirrhosis [32].
The indication for antiviral therapy and the choice of the IFN-free regimen and ribavirin was made according to the international guidelines and local availability. The dose of the different DAAs and the duration of the regimen were chosen following the international guidelines at the time of enrolment [33,34].
All patients underwent clinical exam and laboratory tests at enrolment, at month 1 after beginning DAA treatment, at the end of treatment (EOT) and at week 12 after the end of treatment. The patients who were HCV-RNA-negative at month 3 after stopping treatment were defined as having an SVR. Patients were defined as relapsers when, after HCV RNA negativity during treatment, they showed a virological reactivation once treatment was discontinued. The patients who did not clear HCV RNA during treatment were defined as non-responders.
HIV infection was classified according to the revised CDC classification system for HIV-infected adolescents and adults of 1993 [35]. Antiretroviral therapy was received by 100% of patients at enrolment according to the current national and international guidelines [16,17] and modified if required due to drug interaction.
Study outcomes
The primary outcome was sustained HCV viral response (HCV RNA ≤15 IU/ml) 12 weeks after treatment completion (SVR12) and treatment failure as the absence of SVR12, the epidemiological, clinical and virological characteristics associated with SVR. Secondary outcome was to analyse the trend in viro-immunological, clinical and biochemical parameters from baseline to 12 weeks after stopping DAA regimen. Liver events were defined as the development of liver decompensation, HIV events as the development of an AIDS-defining disease.
Serological analysis
The anti-HCV antibody was sought using a 3rd generation commercial immunoenzymatic assay (Ortho Diagnostic Systems, Neckargemund, Germany). Serum hepatitis B surface antigen (HBsAg) was sought using a commercial immunoenzymatic assay (Abbott Laboratories, North Chicago, IL, USA). Antibodies to HIV-1 and 2 were sought using a commercial ELISA (Abbott Laboratories) and positive results were confirmed by western blot (Genelabs Diagnostics, Singapore).
HCV RNA was quantified by performing a real-time polymerase chain reaction, with the detection limit in plasma samples estimated at around 15 IU/ml. HCV genotyping was performed by a Line-Probe assay (INNO-LIPA HCV; Innogenetics, Zwigndrecht, Belgium). The HIV viral load was assessed by real-time PCR with the lowest detection limit of 20 copies/ml. Lymphocyte subsets (CD4+, CD8+) were evaluated by flow cytofluorimetry using monoclonal antibodies and a fluorescence-activated cell sorter scan (Becton Dickinson, Mountain View, CA, USA). Liver function tests, serum triglycerides, cholesterol, creatinine and routine analyses were performed applying standard procedures. Chronic Kidney Disease Epidemiology Collaboration (CKD-EPI) formula was used to estimate glomerular filtration rate [36].
Statistical analysis
Continuous variables were summarized as mean and standard deviation or median and range, and categorical variables as absolute and relative frequencies. For continuous variables, the differences were evaluated by the Student's t-test for comparison between two groups and by the one-way analysis of variance (one-way ANOVA) for comparison including 3 or more groups; in case of non-normally distributed variables the Mann–Whitney U test and the Kruskal–Wallis one-way ANOVA were used for comparison between 2 or more groups, respectively; categorical variables were compared using the Fisher's exact test. A P<0.05 was considered to be statistically significant. Odds ratios (OR) with 95% CIs were estimated using a logistic regression model using the Firth method based on penalized likelihood to identify possible independent associations between the demographic and clinical characteristics of the patients who were significant or with a P<0.15 at univariate analysis and non-achievement of SVR. A P-value below 0.05 was considered statistically significant.
Results
Patient characteristics
The characteristics of the patients at enrolment are summarized in Table 1. Overall, 243 HCV-RNA-positive PLWH treated with DAAs with a median post-DAA treatment follow-up of 48 weeks (range 12–144) were enrolled. The median age of patients was 52 years (range 25–84), 77.8% were males and 64.2% were injection drug users. At enrolment, all patients were treated with ARV therapy and 91.3% showed HIV RNA <50 copies/ml. HCV genotypes-1a and 3 were the most frequent genotypes identified (39.5% and 32.5%, respectively); a diagnosis of cirrhosis and a history of IFN-based treatment was identified in 36.6% and 31.4% of subjects enrolled, respectively (Table 1).
Demographic, virological and clinical characteristics of the patients enrolled
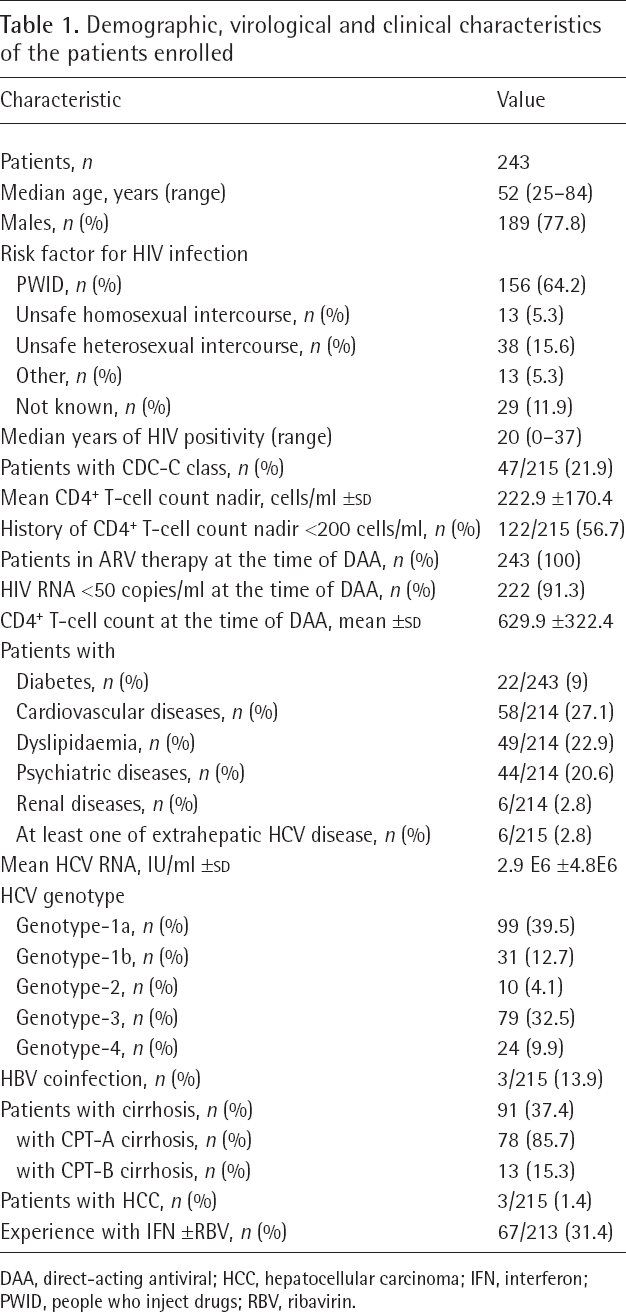
DAA, direct-acting antiviral; HCC, hepatocellular carcinoma; IFN, interferon; PWID, people who inject drugs; RBV, ribavirin.
Additional file 1 shows the DAA regimens used for the 243 patients enrolled according to HCV genotype; the majority were treated with the DAA regimens of the third (46.9%) and second (45.3%) generation; only 19 patients (7.8%) were treated with first-generation DAA regimens (sofosbuvir + ribavirin or sofosbuvir + simeprevir; Additional file 1).
In Additional file 2, the epidemiological, clinical and virological characteristics of the patients according to HCV genotype are reported. The patients with HCV genotype-1a and 3 were younger and had a higher nadir CD4+ T-cell count; the females were more frequently observed in HCV genotype-2 than in the others (Additional file 2).
Achievement of SVR and variable associated
Of the 243 patients enrolled, 233 (95.9%) obtained an SVR12. The prevalence of SVR was very high in all demographic, clinical, viro-immunological subgroups of patients (Figure 1); only 10 subjects did not show an SVR. Table 2 shows the demographic, clinical and virological characteristics of these 10 non-SVR subjects (6 with a relapse and 4 with breakthrough). All but one were PWIDs, with a psychiatric comorbidity in four subjects and cirrhosis in five; all had a history of CD4+ T-cell count nadir of 300 cells/ml or less. In two subjects at failure an HCV genotype different from that identified at enrolment was identified (HCV genotype-3a instead of 1b in both), suggesting a re-infection also considering that both patients were PWID. Of the seven patients tested for NS3, NS5A and NS5B resistance-associated substitutions (RASs) by sequencing analysis, six showed at least one major RAS in one HCV region (all in NS5A, two in NS5B and one in NS3; Table 2).

Prevalence of SVR in different demographic, clinical, viro-immunological sub-groups of patients
Demographic, clinical and virological data of the 10 non-SVR patients
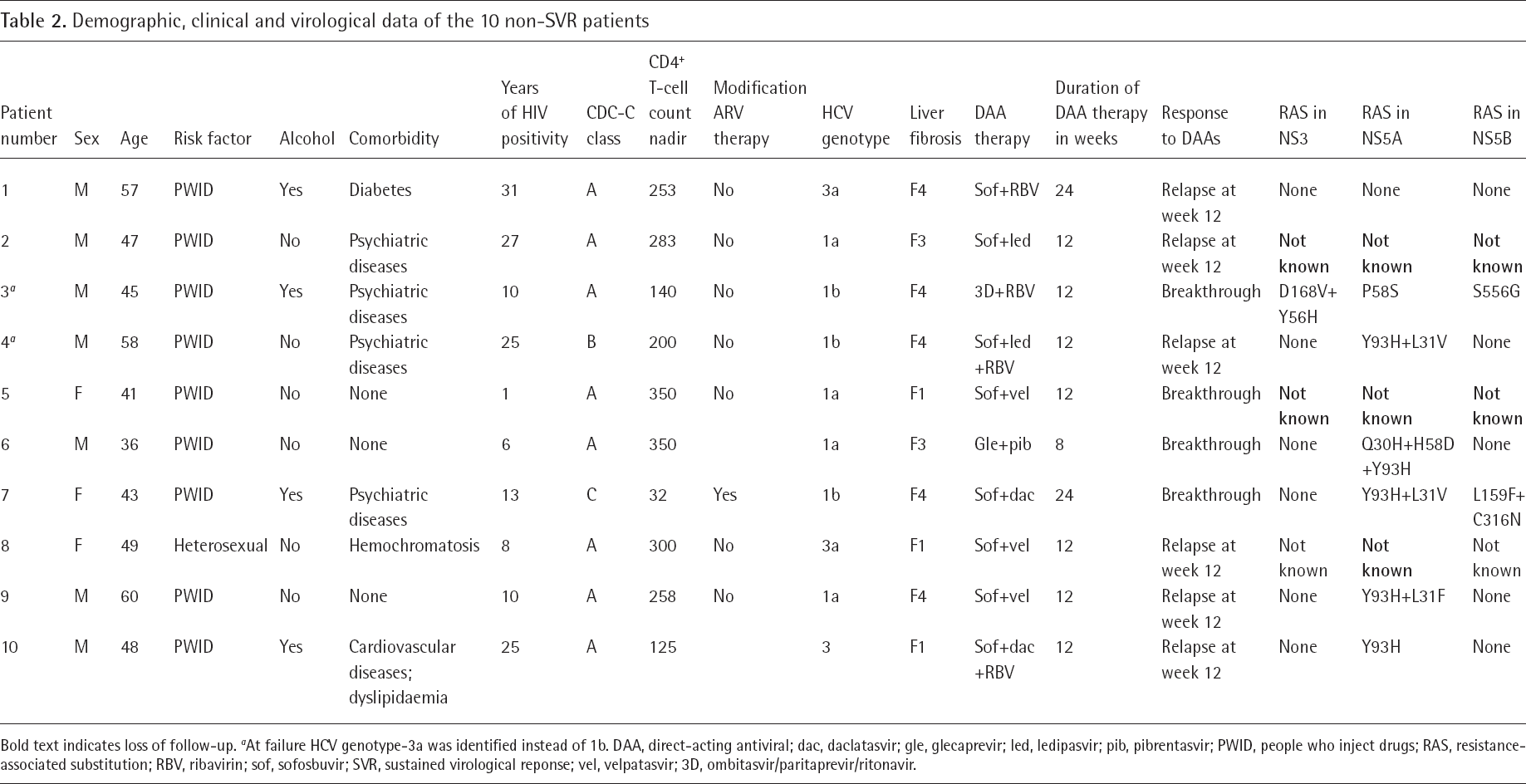
Bold text indicates loss of follow-up.
At failure HCV genotype-3a was identified instead of 1b. DAA, direct-acting antiviral; dac, daclatasvir; gle, glecaprevir; led, ledipasvir; pib, pibrentasvir; PWID, people who inject drugs; RAS, resistance-associated substitution; RBV, ribavirin; sof, sofosbuvir; SVR, sustained virological reponse; vel, velpatasvir; 3D, ombitasvir/paritaprevir/ritonavir.
Comparing the 233 patients who achieved SVR and the 10 who did not, no difference in demographic, biochemical or clinical characteristics was observed (Table 3). However, the 10 patients without SVR were younger (mean ±
Epidemiological and virological features of SVR and non-SVR HCV patients

Bold text indicates statistical significance. DAA, direct-acting antiviral; HCC, hepatocellular carcinoma; NA, not applicable; PWID, people who inject drugs; SVR, sustained virological response.
Logistic regression analysis for independent predictors of non-SVR
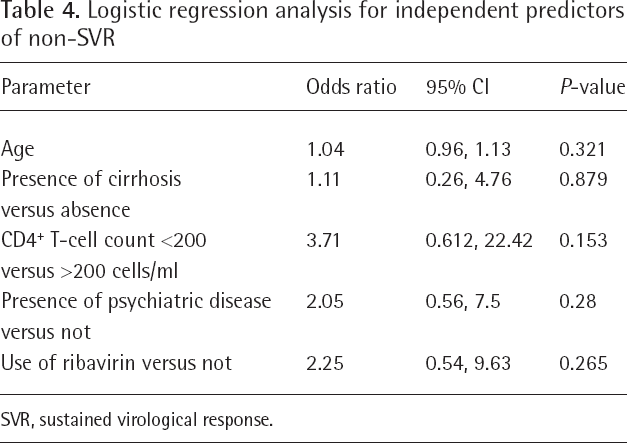
SVR, sustained virological response.
Biochemical and clinical follow-up during and after DAA regimen
During and after DAA regimen, no modification in the prevalence of HIV-RNA-negative subjects and of liver- and HIV-related events was observed (Table 5). Similarly, the mean CD4+ and CD8+ T-cell count, glycaemia, creatinine and triglyceridaemia were stable at all observational points. However, serum cholesterol and low-density lipoprotein (LDL) showed a statistical increase (from 159 ±41.3 mg/dl at baseline to 174 ±44.5 mg/dl at week 12 after stopping treatment, P<0.001, and from 92 ±34.6 mg/dl to 109.4 ±73.7 mg/dl, P=0.002, respectively).
Biochemical parameters and clinical events during and after DAA regimen
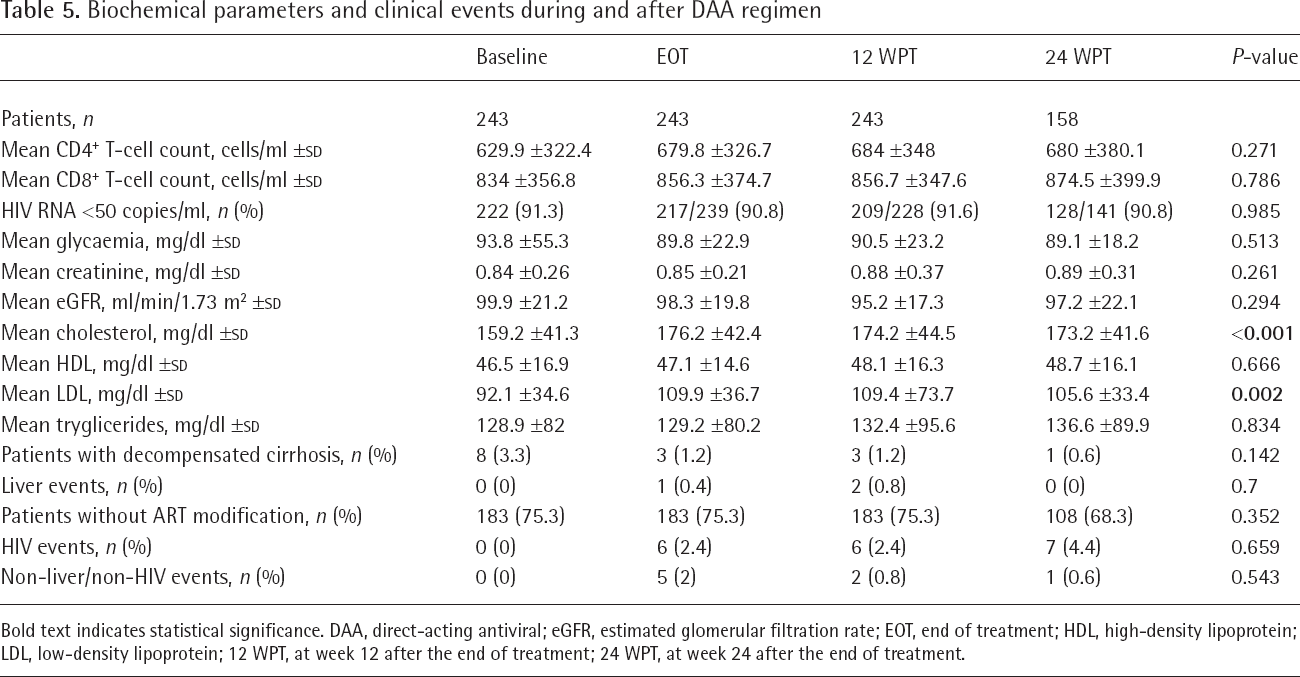
Bold text indicates statistical significance. DAA, direct-acting antiviral; eGFR, estimated glomerular filtration rate; EOT, end of treatment; HDL, high-density lipoprotein; LDL, low-density lipoprotein; 12 WPT, at week 12 after the end of treatment; 24 WPT, at week 24 after the end of treatment.
Discussion
The advent of DAA therapies has resulted in significant improvements in SVR rates also in subjects with HIV coinfection. In fact, the clinical trials showed SVR rates of more than 90% in HIV–HCV-coinfected patients treated with DAAs [25]. However, the data from real life on the efficacy of DAAs in coinfected patients and on the factors associated with SVR are scant. In our cohort 243 HIV–HCV-coinfected subjects were treated in 9 Infectious Disease Centers in southern Italy with the latest generations of DAA regimens; most of the patients showed control of HIV infection, but 37.4% had an advanced liver disease (cirrhosis or HCC). In our cohort the SVR rate was very high (more than 95%), with no demographic, biochemical, clinical or virological factors independently associated with non-SVR.
These results are in agreement with the data available in the literature. For example, in a multicentre real-life study on 536 HIV–HCV-coinfected patients who received DAA therapy in the US, the SVR rate (defined as HCV RNA <25 IU/l ≥10 weeks after stopping the DAA regimen) was 96.5% [37]. We underline that in our cohort the patients were mostly treated with the latest generations of DAA regimens: in fact, only 19 of the 243 patients enrolled were treated with sofosbuvir plus ribavirin or simeprevir, and 99 (45.3%) with a third generation agent (elbasvir + grazoprevir or pibrentasvir + glecaprevir or sofosbuvir + velpatasvir). Other cohort studies in the Veterans Administration system and Europe have demonstrated high rates of effectiveness with DAA therapy in HIV–HCV-coinfected patients similar to that of HCV-monoinfected patients [26,27,38–40]. Together, these data support the shift away from considering patients with HIV–HCV coinfection as a special ‘treatment-refractory’ population. Thus, the AASLD/IDSA Hepatitis C Guidelines recommend that coinfected persons should be treated and retreated in the same way as persons without HIV infection, keeping in mind only the drug–drug interactions between antiretroviral medications [33,34].
Several studies have suggested that vulnerable populations, such as PWID and/or patients with a psychiatric disorder, showed a lower rate of SVR, probably because of the lack of adherence or lack of last linkage to care of these populations [41–43]. In our cohort, the prevalence of PWID and of patients with a psychiatric disease was high (64.2% and 20.6%, respectively), but both variables were not associated with non-SVR. In fact, SVR was 94.2% in the 156 PWIDs and 90.9% in the 44 with a psychiatric disease. Experience in the management of vulnerable populations in the HIV centres participating in the study probably allowed obtaining high compliance and linkage to care in these difficult-to-manage populations and, thus, a high SVR rate.
HCV genotype-3 was another factor associated with non-response, especially in patients with cirrhosis. However, in our cohort of the 79 patients with HCV genotype-3, 95.9% presented SVR, demonstrating the high efficacy of the new DAA regimen also in these difficult-to-treat patients.
The virological analysis of the non-SVR patients showed that the prevalence of RASs were low in the NS3 and NS5B regions and high in the NS5A. This result is in line with the fact that the NS3 and NS5B inhibitors are DAAs with a high barrier to resistance [44,45], while those against NS5A have a low barrier to resistance. Furthermore, two patients showed a different genotype than that identified at baseline; also considering that they were PWID and had a psychiatric disorder, we may hypothesize a HCV reinfection during follow-up.
Finally, in the present study the DAA-related HCV clearance was associated with an increase in serum total- and LDL-cholesterol. These results are consistent with those in previous reports regarding monoinfected patients [46–49]. The changes in cholesterol and LDL levels were probably caused by increased delivery of lipids from the liver to circulating blood following viral eradication by DAA therapy. In fact, the lipid biogenesis is closely related to the RNA replication and assembly of HCV particles. Thus, the suppression of HCV replication by DAA treatment causes a reduction in the production of lipid droplets in HCV-infected liver cells, resulting in a rebound of circulating LDL and total cholesterol. These findings have important implications for hyperlipidaemia management of HIV patients, considering the role of cardiovascular diseases in this population and the association between serum LDL-cholesterol levels and the risk of coronary heart disease.
In conclusion, the DAA regimen was highly effective in a real-life setting of HIV–HCV-coinfected subjects, with a high SVR rate also in difficult-to-manage subjects, such as PWID and subjects with a psychiatric disorder. However, the HCV clearance was associated with an increase in serum LDL- and total-cholesterol, which should be taken into account in the management of HIV subjects.
