Abstract
Background
Neuropsychiatric AEs (NPAEs) leading to dolutegravir (DTG) discontinuation were seen more frequently in real-world use than in randomized clinical trials (RCTs). The recently approved fixed-dose combination bictegravir plus emtricitabine and tenofovir alafenamide (BIC/F/TAF) has shown comparable NPAE rates but some favourable patient-reported outcomes in RCTs compared with DTG. We were interested in its neuropsychiatric tolerability in clinical practice.
Methods
All patients starting BIC/F/TAF from June 2018 in a single centre (two subcentres) were followed retrospectively. Discontinuation rates due to any AEs and NPAEs were compared with those of patients initiating DTG-based regimens.
Results
As of May 2019, a total of 943 patients (852 males, 76 females, 15 transgender and gender diverse) initiated BIC/F/TAF outside RCTs. After a median follow-up of 6.2 months, 50 (5.3%) and 31 (3.3%) patients had discontinued BIC/F/TAF due to any AEs or to NPAEs, respectively. In multivariate analysis, a pre-existing depression and subcentre remained predictive for NPAEs, but not age, gender, ethnicity or prior DTG-related AEs. Compared with 1,043 patients treated with DTG-based regimens, the estimated NPAE-related discontinuation rate with BIC/F/TAF was comparable during the first 6 months (P=0.36). Cross-intolerance was low, and only 5/55 patients with prior DTG intolerability had to discontinue BIC/F/TAF due to NPAEs.
Conclusions
Short-term tolerability of BIC/F/TAF was comparable to DTG-containing regimens. As seen with DTG, discontinuation rates were higher than in RCTs. A pre-existing depression but also physician's awareness may have an impact on tolerability and continuation of BIC/F/TAF. In contrast, prior intolerability of DTG was of limited predictive value.
Introduction
Bictegravir co-formulated with emtricitabine and tenofovir alafenamide (BIC/F/TAF) is a new anti-retroviral fixed-dose combination (FDC). In Europe, BIC/F/TAF was approved in June 2018 for adults infected with HIV-1 without present or past evidence of viral resistance to the integrase strand transfer inhibitor (INSTI) class, emtricitabine or tenofovir. In Germany, BIC/F/TAF became very rapidly a preferred option for several reasons. High potency and good safety over 48–96 weeks were observed in five large randomized clinical trials (RCTs) conducted in both treatment-naive [1,2] and in treatment-experienced patients [3–5]. In contrast to other regimens, BIC/F/TAF does not require the addition of pharmacoenhancers or HLA-B*5701 testing and bears a relatively low potential for drug interactions. The in vitro barrier to resistance development appears to be high [6]. Moreover, during the second half of 2018, costs of BIC/F/TAF in Germany were markedly lower compared with dolutegravir (DTG) plus F/TAF (€366 difference per month) but also compared with other FDCs such as DTG/abacavir/lamivudine (DTG/ABC/3TC) or elvitegravir/cobicistat/F/TAF (EVG/c/F/TAF). Considering both medical and economic issues, many physicians have proactively modified ART regimens in their patients, resulting in a very rapid and broad prescription of BIC/F/TAF after marketing approval.
DTG, another second-generation INSTI, also has a high antiviral potency, high barrier to resistance and an excellent safety profile demonstrated in several RCTs [7]. Consequently, DTG rapidly gained an important place in the management of HIV since its approval in 2014. However, growing data from recent cohort studies have suggested that DTG is associated with a heterogeneous pattern of neuropsychiatric adverse events (NPAEs), leading to discontinuation in around 3.5% of subjects [8]. In our own experience, those NPAEs were observed more frequently in women, in older patients and in patients who initiated ABC at the same time [9]. In our analysis, discontinuation rates were also higher than with other INSTIs such as EVG or raltegravir.
In the three double-blinded RCTs comparing BIC/F/TAF and DTG-containing regimens, drug-related AEs during the first 48 weeks were less common with BIC/F/ TAF [4,10,11]. A secondary analysis of patient-reported outcomes in the two RCTs comparing BIC/F/TAF with DTG/ABC/3TC revealed a lower prevalence of bothersome symptoms and, to some extent, better patient-reported outcomes with BIC/F/TAF [12].
We aim to characterize the tolerability of BIC/F/TAF in clinical practice, compared with DTG-containing regimens.
Methods
We performed a retrospective analysis using the pseudonymized data of all HIV-infected patients under routine clinical care at the Infectious Diseases Center Hamburg (ICH) who initiated a DTG- or BIC-containing antiretroviral treatment regimen between January 2014 and May 2019. At the ICH, antiretroviral therapy is prescribed by a total of eight physicians at two different locations (hereinafter named as subcentres ICH-G and ICH-S). All physicians have more than 20 years of experience in HIV care and are also responsible for the patients’ general medical care. Patients were identified by screening the electronic patient database for all BIC and DTG prescriptions issued. BIC was prescribed as the FDC Biktarvy® (Gilead Sciences) including emtricitabine and TAF. DTG was prescribed as Tivicay® (ViiV Healthcare), the FDC Triumeq® (ViiV Healthcare) including ABC and 3TC and as the FDC Juluca® (ViiV Healthcare and Janssen Cilag) including rilpivirine.
Patients receiving both INSTIs within RCTs or who had initiated BIC or DTG elsewhere were excluded from the analysis, as were patients without a follow-up visit. Both treatment-naive and treatment-experienced patients were included. Patients who had received BIC and DTG at different times during the observation period contributed exposure time to each drug separately. Intermittent treatment interruptions were not accounted for if the patient was on BIC or DTG at the last follow-up.
Physicians at the ICH routinely document the main reasons for any ART discontinuation or modification. The start and stop date and all documented reasons or symptoms for discontinuation of each INSTI were extracted from the electronic database. All AEs leading to discontinuation were evaluated. The following symptoms were classified as NPAEs: insomnia, sleep disturbances, dizziness, anxiety/nervousness, headache, restlessness, depression, poor concentration, slow thinking and otherwise unexplained pain or paraesthesia. For further analysis, we considered only those NPAEs as such when at least some improvement was documented after withdrawal and/or treatment modification. In case of no documented improvement, these events were considered as AEs only.
In patients treated with BIC/F/TAF, other covariates potentially associated with drug discontinuation were evaluated. These included whether BIC/F/TAF was initiated first-line or in treatment-experienced patients, age (>60 years versus younger), gender (female versus male), ethnicity (Caucasian versus other), body mass index (BMI; ≥25 versus lower), CD4+ T-cell counts (≥500 cells/μl versus lower), HIV RNA (<50 copies/ml versus ≥50 copies/ml), prior DTG tolerability (yes versus no) and documented neuropsychiatric diseases (pre-existing depression versus no). Among treatment-experienced patients, we also documented the last regimen preceding BIC/F/TAF and the main reason for switch. Potential reasons were economic considerations, convenience (reduction of pill counts and food restrictions), prevention (of potential drug-drug interactions or cardiovascular events) and adverse events. Patients were also grouped according to prior exposure to DTG (no exposure, exposure with or without AE-related discontinuation).
We used Wilcoxon rank-sum (Kruskal–Wallis) and χ 2 test, respectively, for comparison of continuous and categorical data between groups. Kaplan-Meier analysis including log-rank and Wilcoxon (Gehan–Breslow) testing was used to compare exposure times on BIC and DTG with respect to the above covariates. Discontinuations due to NPAEs were categorized as event, and discontinuations for reasons other than AEs (that is, unrelated death, simplification, costs, prevention of interactions or of cardiovascular events) were censored. In the BIC group, a multivariate Cox regression analysis was used to identify risk factors for discontinuation due to any AE and due to NPAEs. All variables were included in the full model. Using stepwise backward selection, variables with P≥0.05 (except gender and age) were excluded to find predictors for the outcome of interest.
Results
Between June 2018 and May 2019, a total of 1,001 patients had received at least one prescription of BIC/F/ TAF. Among these, 37 had an insufficient follow-up and 21 had initiated BIC/F/TAF either within RCTs (n=12), in other centres (n=7) or as post exposure prophylaxis (n=2). The remaining 943 patients were further analysed, and outcome was compared with 1,043 patients who had initiated DTG-containing regimens between January 2014 and May 2019 at our centre and outside RCTs. The patient's characteristics initiated on BIC/F/ TAF and the proportion of patients with any AEs or with NPAEs leading to drug discontinuation are shown in Table 1. Most patients were male and median age was 50.2 years, with 151 (16.0%) and 79 (8.4%) patients being older than 60 and 65 years, respectively. The vast majority of patients were treatment-experienced (93.4%) and almost half (47.4%) had prior exposure to DTG. Among treatment-experienced patients, 93.8% had an HIV RNA below 50 copies/ml. A total of 35 and 12 patients had a viraemia above 50 or 200 HIV RNA copies/ml, respectively. A further 20 treatment-experienced patients were viraemic but had undergone prior treatment interruptions. The median CD4+ T-cells of all patients were 667/μl, with only 29 (3.1%) exhibiting a severe immune deficiency with a CD4+ T-cell count less than 200 cells/μl. Among 516 patients with available data on weight, 60 (11.6%) were obese (BMI >30) while only 10 (1.9%) were underweight (BMI <18.5). Neuropsychiatric diagnoses were highly prevalent, including depression in most cases (19.5%) but also mental and behavioural disorders due to opioids and/or alcohol (4.1%).
Numbers and characteristics of patients initiated on BIC/F/TAF and the proportion of patients with any AE or with NPAEs leading to drug discontinuation
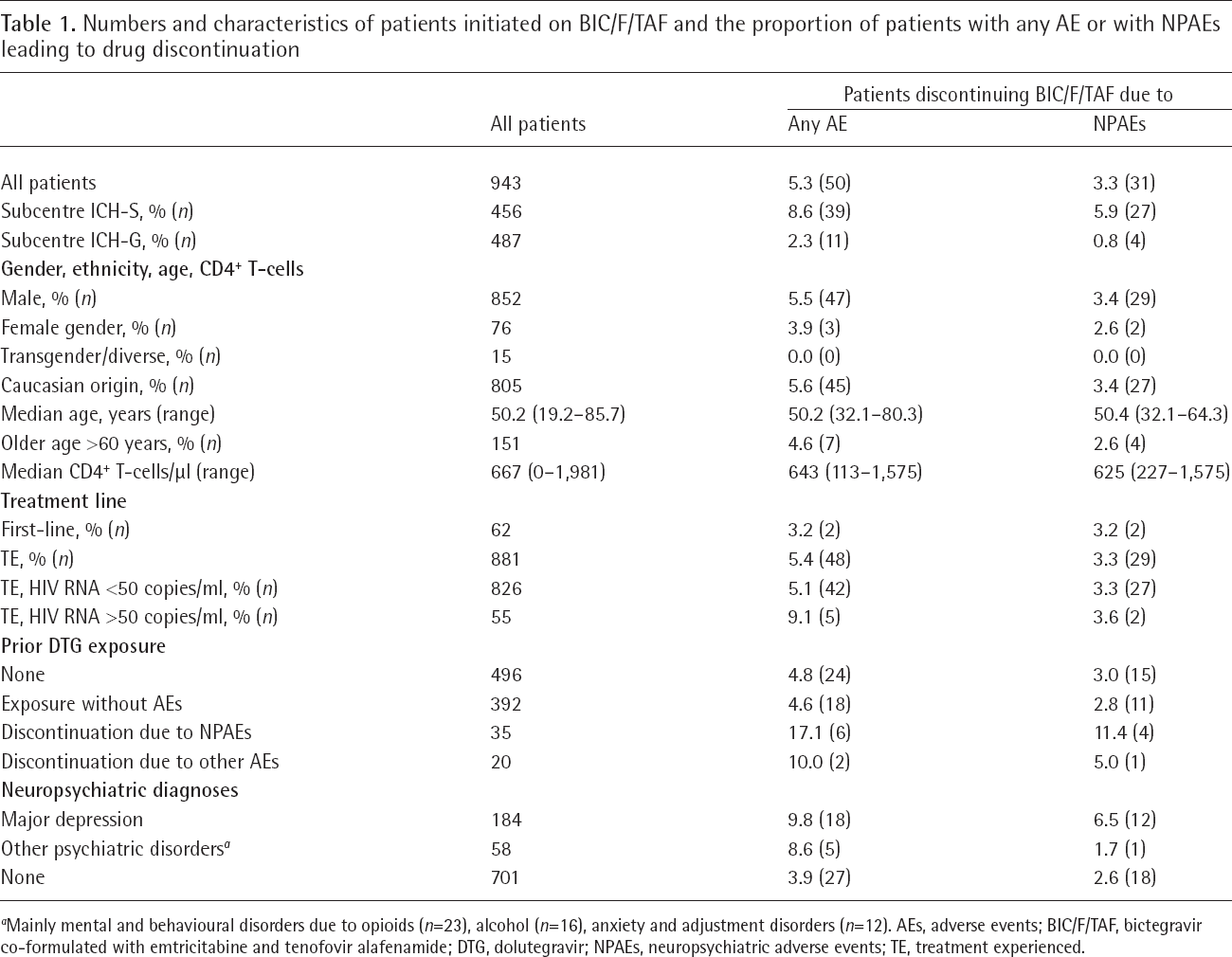
Mainly mental and behavioural disorders due to opioids (n=23), alcohol (n=16), anxiety and adjustment disorders (n=12). AEs, adverse events; BIC/F/TAF, bictegravir co-formulated with emtricitabine and tenofovir alafenamide; DTG, dolutegravir; NPAEs, neuropsychiatric adverse events; TE, treatment experienced.
The main reasons for switching to BIC/F/TAF and the preceding antiretroviral regimens are depicted in Figure 1. As shown there, the most frequent regimens were DTG plus F/TAF and EVG/c/F/TAF. The main reasons for treatment modification were economic, convenience and prevention of potential drug–drug interactions. After a median follow-up of 6.2 months, 50 had discontinued BIC/F/TAF due to any AEs, among them 31 patients due to NPAEs. Other AEs leading to discontinuation in more than one patient were gastrointestinal AEs (n=7) and weight gain (n=4).

The main reasons for switching to BIC/F/TAF and the preceding antiretroviral regimens
There were a total of six patients in which NPAEs did not show any changes after withdrawal of BIC/F/TAF, among them four patients with pre-existing neuropsy-chiatric conditions. One patient reported paraesthesia and dizziness not only immediately after taking BIC/F/ TAF but also with a darunavir-based regimen. In one patient, documentation of neuropsychiatric outcome after withdrawal was unclear. These six cases were not considered as to be NPAEs. In 5/6 patients, events were accompanied by gastrointestinal symptoms. In one patient, pruritus was reported.
Two patients died whilst on BIC/F/TAF, one from metastatic anal and one from pancreatic cancer. Both deaths were not considered to be drug related. These two patients were censored as well as three patients who stopped BIC/F/TAF for other reasons, among them one virological failure and two anticipated drug– drug interactions with rifabutin requiring treatment modification.
In the 31 patients who discontinued BIC/F/TAF due to NPAEs, the median time between start and discontinuation was 2.95 months. The most frequent symptoms included insomnia and sleep disturbances (73%) but the spectrum also included anxiety/nervousness (50%), depression (36%), dizziness (36%), poor concentration/slow thinking (28%), headache (19%) and musculoskeletal pain/paraesthesia (16%). No symptoms were life-threatening or led to hospitalization and most symptoms disappeared quickly after discontinuation of BIC. In 22/26 (85%) patients who were followed for a median of 3.8 months after BIC/F/TAF discontinuation, the subsequent antiretroviral regime was tolerated and effective for at least 3 months.
The estimated rates of discontinuation due to NPAEs (any AEs) at 6 months of exposure were 3.3% (5.3%) for BIC/F/TAF and 3.3% (4.7%) for DTG, respectively. In patients with a pre-existing depression, there was a trend towards more discontinuations due to NPAEs with BIC/F/ TAF (P=0.07). For the 1,043 patients exposed to DTG, the total estimated rate of NPAE-related discontinuation has now exceeded 10% at 5 years. Follow-up of patients with BIC/F/TAF was markedly shorter, with 50.6% and only 13.6% reaching 6 and 9 months, respectively. However, as shown in Figure 2, the risk for NPAE-driven discontinuations on BIC/F/TAF was comparable with those of DTG-based regimens (Wilcoxon test, P=0.36).

Time on BIC/F/TAF and on DTG-containing regimens, discontinuation due to neuropsychiatric adverse events (all other events censored)
There was no association between BIC/F/TAF discontinuation and gender, age, ethnicity, BMI, treatment line, prior regimen, main reason for switch or CD4+ T-cell count. However, patients without a pre-existing depression diagnosis had a lower rate of discontinuation due to NPAEs. This was also true for patients treated at the subcentre ICH-G, compared with patients treated at the ICH-S. In the latter subcentre, the risk of discontinuation was several times higher for both BIC and DTG (data not shown). In the absence of significant differences in patient profiles, we observed also considerable differences in discontinuation rates between individual physicians (data not shown). In univariate analyses, patients with prior intolerability to DTG had also a higher rate of NPAE-related discontinuation. In the multivariate analysis, however, only depression and subcentre remained predictive for discontinuation of BIC/F/TAF (Table 2) and a prior AE or NPAE related to DTG was no longer predictive.
RH of variables associated with BIC/F/TAF discontinuation due to any AE or due to NPAE and remaining in the final Cox model
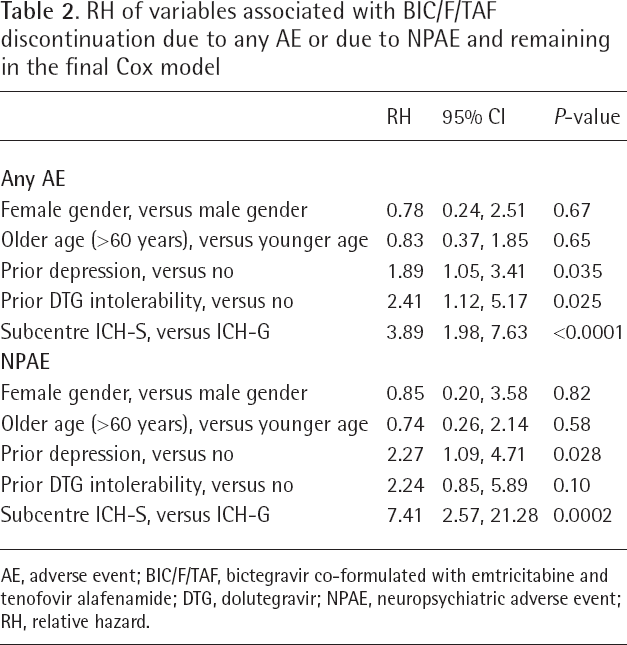
AE, adverse event; BIC/F/TAF, bictegravir co-formulated with emtricitabine and tenofovir alafenamide; DTG, dolutegravir; NPAE, neuropsychiatric adverse event; RH, relative hazard.
Cross-intolerance of BIC and DTG was low. Only 5/55 and 4/35 patients who had discontinued DTG due to any AE or due to an NPAE, respectively, had also stopped BIC/F/TAF due to an NPAE (two additional patients discontinued BIC/F/TAF due to weight gain). On the other hand, of 16 patients who had discontinued BIC/F/TAF due to NPAEs and who had been exposed to DTG-containing regimens, 11 had had no documented tolerability problems with DTG.
Discussion
In this large cohort of 943 HIV-1-infected patients who initiated BIC/F/TAF in routine clinical practice, the estimated rates of discontinuation due to any AE or NPAEs during the first 6 months of exposure were 5.3% and 3.3%, respectively. NPAEs were mild to moderate and quickly resolved after discontinuation. These real-life discontinuation rates were, however, markedly higher than seen in five Phase III RCTs encompassing 634 treatment-naive [1,2] and 806 treatment-experienced patients [3–5]. In the three double-blinded RCTs comparing BIC/F/TAF and DTG-containing regimens, drug related AEs had been less common in the BIC arm than with DTG [4,10,11]. In these trials, 11/916 (1.2%) patients stopped BIC/F/TAF due to AEs within the first 48 weeks. Only six events were considered to be neuropsychiatric. In addition, a secondary analysis of patient-reported outcomes in the two RCTs comparing BIC/F/TAF with DTG/ABC/3TC had revealed a lower prevalence of bothersome symptoms and, to some extent and at different time points, better patient reported outcomes and a somewhat better sleep quality with BIC/F/TAF [12].
We did not find significant differences between BIC/F/TAF and DTG-containing regimens in terms of NPAE-related discontinuations. In line with our previously published DTG experience [9] and with a number of other reported cohorts [8], our higher discontinuation rates may relate to the fact that patients enrolled in RCTs can differ substantially from broader treated populations. Moreover, limitations in access to health-care resources or predefined stopping criteria set up in RCTs may discourage premature discontinuations in cases of mild to moderate AEs. Barriers to treatment modifications in clinical routine may be much lower, especially in those patients who had switched from successful regimens to BIC/F/TAF for strategic reasons only.
In contrast to DTG [9], we did not find any impact of female gender or older age on discontinuation rates of BIC/F/TAF. Instead, a pre-existing diagnosis of depression was found to be independently associated with discontinuation due to NPAEs. In depressive patients, there was even a trend towards higher discontinuation rates with BIC/F/TAF than with DTG. However, high background rates of psychiatric conditions and the likely multifactorial pathogenesis make it challenging to assess the relationship between NPAEs and specific antiretroviral therapies such as INSTIs [13]. Depression has been associated with a threefold increase in nonadherence to medical treatment recommendations in general [14], and HIV research has consistently shown depression to be an impediment to ART adherence [15,16]. Many depressive symptoms overlap with the broad spectrum of NPAEs associated with INSTIs (for example, insomnia, headache, anxiety, cognitive problems). Although we focussed on NPAEs showing improvement after treatment modification, we could not rule out that some events were caused or triggered by the underlying depressive disorders and/or by the increased awareness of the treating physicians with regard to INSTI-related NPAEs. We had no information on prescriptions of antidepressant drugs. Moreover, sleep disorders are usually not coded in our database. However, the majority of our patients who discontinued BIC/F/TAF did so in the absence of any documented depressive disorder and most patients were seemingly well on the subsequent antiretroviral regimen.
The significant differences in NPAE rates between the two subcentres of the ICH merit further discussion. In the absence of significant differences of patient characteristics, we found a high variability in discontinuation rates between individual physicians. These findings were also true for DTG and indicate that differences in awareness, perception and assessment of NPAEs by health-care providers may impact substantially on discontinuation rates. Converging evidence suggests that the occurrence of unwanted adverse events during drug treatment is in part determined by nonpharmacological effects [17]. On the patient's side, it has been shown that beliefs and satisfaction with therapy modulate the impact of mild AEs on the discontinuation of antiretroviral regimens [18]. Nonpharmacological effects may explain our findings as well as the large differences in the ‘real-world’ reported to date for DTG. For example, in a large clinical cohort from London, 308/2,543 patients (12.1%) had discontinued DTG due to adverse drug reactions, among them 180 (7.1%) categorized as ‘sleep disturbance’, ‘psychiatric’ and ‘nervous system’ [19]. In contrast, in the Spanish PISCIS cohort prospectively following 1,650 patients on DTG from 12 HIV units in Catalonia, the corresponding rates for discontinuation were 4.3%, with only 1.9% considered to be NPAEs [20]. Of note, significant differences among centres were found in the PISCIS cohort, ranging from 0 to 6.9 DTG discontinuations/100 patient years.
Given the lack of objective, standardized criteria for NPAEs and the retrospective design of our study, selection and information biases may be important limitations. Further caution must also be applied when interpreting the results. Follow-up with BIC/F/TAF was relatively short with only 6.2 months, and as we had only data on AEs leading to discontinuation, the true rate of AEs related to BIC/F/TAF may be higher. Indeed, anecdotally several patients complained of transient NPAEs during the first weeks and others reported even persistent neuropsychiatric symptoms while continuing BIC/F/TAF. Regarding the comparison with DTG, the different observation periods with growing awareness of INSTI-related AEs and the growing number of FDC alternatives over time must also be considered.
Nevertheless, we believe that this study may be of value for clinical practice. First, the number of patients exposed to BIC/F/TAF within a few months in a single centre with a standardized documentation was large, exceeding those of RCTs in treatment-experienced patients. We have shown for the first time that prior intolerability for DTG was of limited predictive value. Very few of those who had discontinued DTG secondary to NPAEs went on to discontinue BIC/F/TAF and, equally, most patients developing NPAEs during BIC/F/ TAF had no reported tolerability problems with DTG, indicating a low cross-intolerance between the two agents. In addition, the large number of older patients treated with BIC/F/TAF adds further evidence to drug safety. In the RCTs on treatment-naive patients, for example, the number of patients older than 50 years who were exposed to BIC/F/TAF was below 100 [1,2]. Treatment-experienced patients enrolled in RCTs were somewhat older, but age rarely exceeded 60 years [3–5]. In all RCTs, no patient older than 74 years had been enrolled. In our cohort, none of the 79 patients older than 65 years withdrew from BIC/F/TAF due to NPAEs, among them 29 were older than 75 years. These findings indicate good tolerability of BIC/F/TAF in the elderly.
In conclusion, NPAEs with BIC/F/TAF seen in routine clinical care are heterogeneous, usually mild to moderate and rarely necessitate drug withdrawal, but could require drug discontinuation more frequently than expected from RCTs. Rates during the first 6 months were comparable with DTG-containing regimens, which have now exceeded 10% after 5 years of exposure in our centre. Prior intolerability for DTG was of limited predictive value, indicating low cross-intolerance between both INSTIs. Predisposing factors such as a pre-existing depression but also physician's awareness may have an impact on tolerability and continuance. Given the still unknown mechanisms underlying INSTI-related NPAEs, these side effects deserve further research.
Footnotes
CH has served as an advisor or speaker for AbbVie, Eusa Pharm, Gilead Sciences, Janssen Cilag, NSD, TAD and ViiV Healthcare. SF has served as an advisor for Gilead Sciences and ViiV Healthcare. HJS has served as an advisor or speaker for Gilead Sciences, Janssen Cilag, MSD and Teva. SH has received travel grants or has served as an advisor for Gilead Sciences, Janssen-Cilag and ViiV Healthcare. MS has served as an advisor or speaker for AbbVie, Gilead Sciences, Janssen Cilag and ViiV Healthcare. KS has served as an advisor or speaker for Gilead Sciences, AbbVie, MSD, Janssen Cilag, Hexal and ViiV Healthcare. TB has served as an advisor or speaker for AbbVie, Gilead Sciences, Janssen Cilag and ViiV Healthcare. AA declares no competing interests. The ICH has been awarded with grants for clinical research from AbbVie, Eusa Pharm, Gilead Sciences, Janssen Cilag, MSD and ViiV Healthcare.
