Abstract
Background
An increase in pretreatment drug resistance (PDR) to first-line antiretroviral therapy (ART) in low-income countries has been recently described. Herein we analyse the prevalence of PDR and risk of virological failure (VF) over time among migrants to Italy enrolled in ARCA.
Methods
HIV-1 sequences from ART-naive patients of non-Italian nationality were retrieved from ARCA database from 1998 to 2017. PDR was defined by at least one mutation from the reference 2009 WHO surveillance list.
Results
Protease/reverse transcriptase sequences from 1,155 patients, mainly migrants from sub-Saharan Africa (SSA; 42%), followed by Latin America (LA; 25%) and Western countries (WE; 21%), were included. PDR was detected in 8.6% of sequences (13.1% versus 5.8% for B and non-B strains, respectively; P<0.001), 2.1% of patients carried a PDR for protease inhibitors (PIs; 2.1% versus 2.3%; P=0.893), 3.9% for nucleoside/ nucleotide reverse transcriptase inhibitors (NRTIs; 6.8% versus 2.1%; P<0.001) and 4.3% for non-nucleoside/ nucleotide reverse transcriptase inhibitors (NNRTIs; 6.3% versus 3.1%; P=0.013).
Overall, prevalence of PDR over the years remained stable, while it decreased for PIs in LA (P=0.021) and for NRTIs (P=0.020) among migrants from WE.
Having more than one class of PDR (P=0.015 versus absence of PDR), higher viral load at diagnosis (P=0.008) and being migrants from SSA (P=0.001 versus WE) were predictive of VF, while a recent calendar year of diagnosis (P<0.001) was protective for VF.
Conclusions
PDR appeared to be stable over the years in migrants to Italy enrolled in ARCA; however, it still remains an important cause of VF together with viral load at diagnosis.
Introduction
HIV-1 pretreatment drug resistance (PDR) to anti-retroviral therapy (ART) is one of the major public health issues in the field of HIV-1 infection currently, as it can increase the risk of virological failure in treated patients compromising the efficacy of ART at the individual and population level [1]. PDR refers to the presence of drug resistance in people initiating ART for the first time or in HIV-infected individuals with prior ART exposure reinitiating first-line therapy; therefore, PDR is either transmitted, acquired or both [1]. Remarkably, PDR is considered one of the best indicators to guide the prescription of an effective first-line ART regimen [2].
Despite the reported decrease of PDR in both the United States and Europe in recent years [3–6], an alarming increase of this phenomenon has been reported in low- and middle-income countries, like sub-Saharan Africa [7,8], Latin America [9] and Asia [10]. In conjunction, an increase of non-B subtypes and circulating recombinant forms has occurred in previously clade B-homogeneous areas due to multiple reasons, among which waves of migration could have played a role [11,12]. Unfortunately, viral load monitoring and genotypic resistance testing (GRT) are not easily affordable in low-income countries [13] and cannot be regularly performed at patient entry in care and at virological failure, as recommended by current guidelines [14]. As a consequence, the wider use of ART in the context of a universal ‘test and treat’ strategy may provoke the increment of PDR, and of drug resistance globally [15,16]. In fact, the overall PDR surveillance data show that ART resistance has surpassed the worrying threshold of 10% in many low- and middle-income countries [2] and this could negatively impact the management of HIV-infected individuals in these settings. Moreover, the increase of PDR could also cause severe economic damage; in fact, some mathematical models have predicted that PDR will account for half a million new infections in sub-Saharan Africa, causing an additional ART cost of $6.5 billion between 2016 and 2030 [17], continuing this trend. Due to the important migration flows, a coordinated approach to supervise and prevent the spreading of PDR should be implemented both in high- and in low-income countries and should involve local people and migrants, as some recent data on this specific group seem to suggest [18]. Therefore, the aim of this study is to describe the prevalence and trend over the years of PDR among migrants to Italy and to estimate the impact of this phenomenon on the risk of virological failure in this population, retrieving data from a large Italian cohort of HIV-infected individuals (ARCA database).
Methods
Patients
ARCA (Antiviral Response Cohort Analysis) is a public observational database created for surveillance of HIV-1 drug resistance and for the implementation of models predictive of virological response to antiviral treatment. Currently ARCA collects data of more than 40,000 HIV-1-infected patients followed at >50 clinical centres in Italy, including both university centres and public hospitals, whose data are systematically collected and revised every 6 months. ARCA contains demographic, clinical and treatment data, HIV-1 RNA load, CD4+ T-cell counts and drug resistance information, providing a framework for comparative evaluation of recommended as well as less common treatment regimens. Since major Italian centres involved in HIV infection care are included in ARCA, this database can be considered highly representative of an Italian cohort of HIV-infected subjects currently on ART.
HIV-1 sequences from ART-naive patients of non-Italian nationality were retrieved from ARCA database from 1998 to 2017. In addition, information about country of origin, demographic, clinical and treatment data, HIV-1 RNA load, CD4+ T-cell count and drug resistance were also retrieved from ARCA for each patient.
Migrants were defined as those born outside Italy, based on their geographical origin which was derived from nationality and classified as follows: Western countries (WE: Europe, North America, Australia and New Zealand), North Africa and Middle East (NAME), sub-Saharan Africa (SSA), Latin America (LA) and Asia (ASIA).
HIV RNA Quantification
HIV-1 RNA quantification was performed at the individual centres by using last generation assays allowing the detection of ≤50 copies of HIV-1 RNA/ml.
Drug Resistance Genotyping
HIV-1 genotypes were performed at each participating centre on the basis of local procedures and checked for consistency upon sequence uploading into the ARCA database, which stores the sequences, automatically extracts the mutations with respect to HIV-1 consensus B using a built-in local alignment script based on ClustalW and returns a 5-level susceptibility score based on the built-in AntiRetroScan algorithm [19].
PDR was defined as the detection of at least one mutation included in the World Health Organization (WHO)-recommended surveillance drug resistance mutation (SDRM) list, updated in 2009 for nucleoside/nucleotide reverse transcriptase inhibitors (NRTIs), non-NRTIs (NNRTIs) and protease inhibitors (PIs) [20].
Statistical Analysis
χ2 or Fisher's exact tests were used, as appropriate, to detect potential differences in PDR between B and non-B subtypes, and according to migrant origin. Differences in PDR prevalence over the years were evaluated by χ 2 test for trend. Survival analysis (Kaplan–Meier curves and Cox regression model) was used to evaluate the probability and predictors of virological failure (the first of two consecutive viraemia >200 copies/ml after at least 180 days of therapy regardless of therapy change or interruption, intention to treat approach [ITT]) according to PDR, by using as confounders gender, age, HIV-1 risk factor, geographic area, HIV-1 subtype, HIV RNA and CD4+ T-cells count at GRT and year of GRT. Survival analysis was performed in patients starting a first-line regimen and with available viraemia follow-up. All analyses were performed by using the opensource statistical environment R (v. 3.5.1 or greater) and by using a statistical significance threshold of α=0.05.
Ethics
The database was approved by the Ethics Committees at each clinical centre and written informed consent was obtained from all patients before participation. The study was performed in accordance with the ethical guidelines of the Declaration of Helsinki (7th revision) and with the International Conference on Harmonization Good Clinical Practice guidelines.
Results
General Demographic and Immuno-Virological Features of Patients
Protease/reverse transcriptase sequences from 1,155 ART-naive patients enrolled in ARCA database from 1998 to 2017 were analysed. General demographic and immuno-virological characteristics of subjects from whom sequences were taken are shown in Table 1.
General characteristics of enrolled patients

Boldface values represent statistically significant estimates (P-value <0.050). GRT, genotypic drug resistance test; IDU, intravenous drugs users; MSM, males who have sex with males; NNRTI, non-nucleoside/nucleotide reverse transcriptase inhibitors; NRTI, nucleoside/nucleotide reverse transcriptase inhibitors; PDR, pretreatment drug resistance mutations; PI, protease inhibitors; Q1–Q3, first–third quartile; Sex, sexual intercourse.
Overall, 54% of patients were males, with a median (first quartile [Q1] – third quartile [Q3]) age of 34 years (28–40). For the most, subjects came from Africa (48%) and America (27%), while the remaining came from Asia, Australia, Oceania and Europe (25%), including 141 subjects from East European countries, 56.6% of the European patients. The main risk factor for HIV-1 infection was sexual transmission (70.5%), predominantly heterosexual (40.9%).
A HIV-1 non-B strain was detected in 713 patients (61.7%), with an increasing trend in recent years (from 59.5% before 2006 to 62.1% in 2017, P=0.060; data not shown).
Median HIV-1 viral load at GRT was 4.7 (4.0–5.3) log10 copies/ml (without difference between B and non-B strains; P=0.82), and median CD4+ T-cell count was 296.5 (128.2–474.8) cells/mm3 (336 versus 270 for B and non-B respectively; P<0.001).
Analysis of Drug Resistance Mutations
PDR was detected in 8.6% of sequences, with a significant difference between B and non-B strains (13.1% versus 5.8% respectively; P<0.001). As reported in Table 1, the prevalence of resistant mutations was 2.1% for PIs, 3.9% for NRTIs and 4.3% for NNRTIs.
In particular, the frequency of mutations for NRTIs was 6.8% in B compared with 2.1% in non-B variants, P<0.001 and for NNRTIs was 6.3% in B compared with 3.1% in non B sequences (P=0.01).
By performing a post hoc analysis, the frequency of PDR was not associated with a specific transmission route in this cohort (data not shown).
As shown in Figure 1, over the years, PDR in B strain (Figure 1A) at first GRT remained stable for NRTIs (P=0.180), NNRTIs (P=0.786), while a decreasing trend was observed for PIs (P=0.085). Among non-B strain (Figure 1B), PDR remained stable for NRTIs (P=0.293), NNRTIs (P=0.743) and PIs (P=0.505).
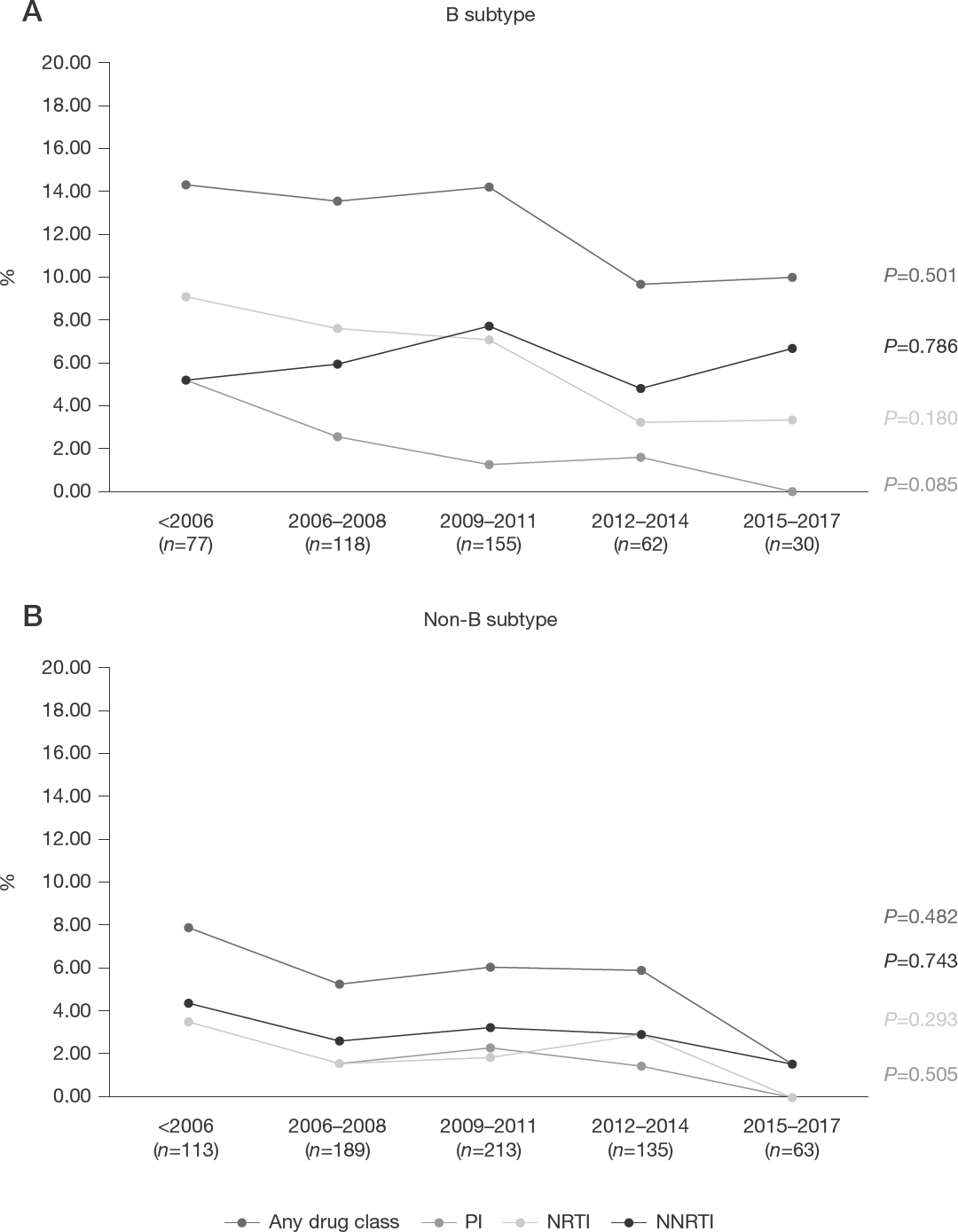
Trend of pretreatment drug resistance over the years among B and non-B subtypes
The pattern of drug resistance mutations was different between B and non-B strains (Figure 2). In 6/442 (1.4%) subtype B sequences, the major PIs resistance mutation detected was M46I, followed by L90M (3/442, 0.7%); on the contrary the M46I was one of the rarest mutations in non-B subtypes (0.1%), while the L90M was the most frequent (in 3/713, 0.4%). Likewise, among the NRTI mutations, M41L was the most common mutation (2.3%) in B strains, followed by M184V and T215D (1.6% and 1.6%, respectively). Among non-B subtypes, M184V was the most prevalent PDR (1.1%), followed by M41L (0.6%); finally, mutations at position 215 (one patient showed T215F, two T215S, one T215F) were rare. On the contrary, resistance pattern for NNRTIs was similar between B and non-B strains, showing the K103N mutation as the most frequent PDR (2.7% and 1.7% among B and non-B subtypes, respectively). Similarly, no particular association was found between K103N and gender (P=0.144) or transmission route (data not shown).

Drug resistance mutations for different drug classes among B and non-B strains
Trend of PDR over the Years According to Country of Origin
The overall prevalence of PDR varied according to the origin country of migrants (11.1% for LA and ASIA, 10.4% for WE, 8.8% NAME, 5.7% for SSA, respectively; P=0.042). No significant difference in PDR distribution was evidenced by stratifying the analysis for diverse drug classes.
The evaluation of PDR over the years for the different countries showed a decline in migrants from WE (any drug class; P=0.026), particularly significant for NRTIs (P for trend =0.010; Figure 3A). On the contrary, among migrants from LA, PDR remained stable, with a slight trend toward an increase, although a decreasing trend in PDR was noticed only for PIs (P=0.021; Figure 3B). Likewise, the trend of PDR over the years among migrants from SSA showed a stable course (Figure 3C). Finally, sample size was too small to allow the analysis of PDR trends over the years in the NAME and ASIA groups.

Trend of pretreatment drug resistance over the years according to country of origin
PDR and Risk of Virological Failure
A survival analysis was performed in 681 patients starting a first-line antiretroviral regimen and with available viraemia follow-up. By Kaplan–Meier curves estimate (Figure 4), patients having more than one class of PDR showed a significant higher probability of experienced virological failure compared with those having only one class of PDR and those having no PDR (55% versus 22% versus 26%, respectively; P=0.013) Moreover, by performing a Cox regression multivariate model, after evaluating the proportional hazard assumption (Table 2), having more than one class of PDR (adjusted hazard ratio [aHR], 3.65 [95% confidence interval [CI]: 1.29, 10.34]; P=0.015 versus absence of PDR), higher viral load at diagnosis (1.24 [1.06,1.46]; P=0.008) and being migrants from SSA (2.62 [1.51,4.54]; P=0.001 versus WE) were predictive of VF. On the contrary, a recent calendar year of GRT (0.9 [0.85,0.94]; P<0.001 per 1 year higher) was protective for V F.
Multivariable Cox regression model

Boldface values represent statistically significant estimates (P-value <0.050).
Adjusted for gender, age, HIV risk factor, country of origin, HIV subtype, number of pretreatment drug resistance (PDR) classes at first genotypic resistance test (GRT), viral load and CD4+ T-cell count at first GRT, calendar year of first GRT.
Reference group (dummy). aHR, adjusted hazard ratio (HR); cHR, crude HR.
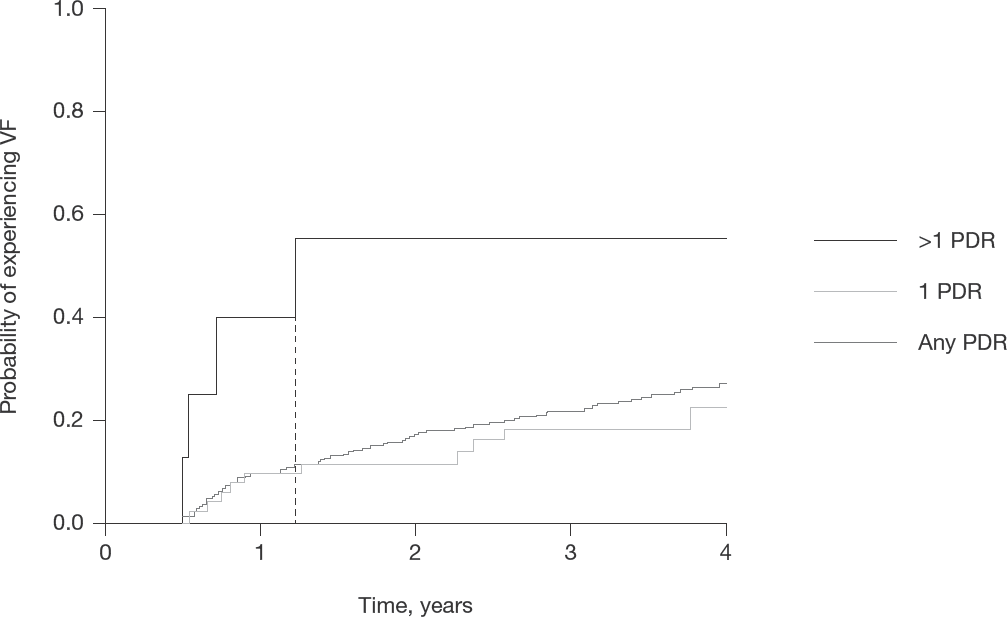
Kaplan–Meier survival analysis
Discussion
In this study, temporal trends in the prevalence of PDR among ART-naive HIV-infected migrant patients in Italy and entering care during the period 1997–2017 were reported, retrieving data from ARCA, a large multicentre cohort. Moreover, the impact of PDR on the risk of virological failure in this particular population was assessed.
In recent years waves of migration to Europe remained stable in terms of country of origin of migrants [21]; overall, in this study, sub-Saharan Africans represented almost 40% of all migrants diagnosed with HIV-1 infection, carrying mainly non-B viral subtypes. Conversely, the majority of subjects carrying B subtype enrolled in this work were Latin Americans, who accounted for almost 30% of migrants diagnosed with HIV-1 infection. This data are in line with recent studies [22,23] which showed that a considerable proportion of the infections occurred inside well-defined clusters, with a very limited mutual contribution.
Globally the prevalence of PDR among both B and non-B subtypes remained almost stable in our cohort over time. Our analysis showed that subjects with subtype B HIV-1 infection had a significantly higher prevalence of PDR at first GRT (13.1%) if compared with non-B subtypes (5.8%). Therefore, although not significantly increasing over the years, PDR in B subtypes is permanently above the worrisome threshold of 10% [2]; this result suggests that more effort should be made to revert, or at least to limit, the spreading of PDR.
Overall, resistance to PIs was rare among both B and non-B subtypes, and it was mainly attributable to the M46I/L mutation, followed by L90M. These mutations, which contribute to resistance to a broad spectrum of PIs (with the exception of darunavir), were expected because of their low viral fitness cost and consequently persistence, even in untreated patients [24,25].
On the contrary, both B and non-B subtypes demonstrated a similar NNRTI PDR pattern, pointing to K103N, which confers resistance to efavirenz and nevirapine but not to etravirine and rilpivirine, as the main resistance mutation.
Finally, as previously reported [10,26,27], the most frequent NRTI resistance mutations among B subtypes belonged to the thymidine analogue mutation (TAM) group (revertant mutants at codon 215, followed by M41L), which are long-term persistent mutations in ART-naive individuals [28]. Conversely, in non-B strains, the main resistance mutation was M184V, which is selected in the course of previous WHO-recommended first-line ART regimens when administered in the absence of virological monitoring [29,30]. Hence, the presence of M184V in GRT of patients naive to ART migrants from sub-Saharan Africa could indicate a possible circulation of this mutation and, as a consequence, the need of specific interventions in this setting to counteract this phenomenon.
It should be noticed that the prevalence of M184V in this study could be underestimated; indeed, because of its high fitness cost, viruses harbouring this mutation often tend to revert to wild type but persisting minority variants harbouring the M184V can be archived or replicate at low levels hampering future treatments.
Initiating ART with regimens with both greater potency and higher genetic barrier [31], and having a greater availability of drugs for second- (or third-) line regimens, along with viral load monitoring and genotyping at baseline and after treatment, would be pivotal enhancement in terms of long-term global success of ART.
Strategies to reduce the spread of HIV-1 drug resistance should be differentiated according to different countries since a higher PDR incidence could be present in low-income countries if compared with the high-income ones. In fact, in agreement with previous studies [3–6], the prevalence of PDR among migrants from western and industrialized countries has had a stable decline in the ARCA database reaching a percentage below 4% for any drug class among patients enrolled in the last 5 years. On the contrary, GRTs from migrants from Latin America demonstrated a slight but persistent increase in overall resistance to antiviral drugs (excluding PIs), reaching 18% of median prevalence to any drug class (12% and 6% to NNRTI and NRTI, respectively) in subjects enrolled in recent years.
Finally, the prevalence of PDR among sub-Saharan Africans seems to be stable over the years; however, the number of migrants from any country included in our study decreased in the last observation period, probably refecting a reduced number of performed drug resistance testing rather than a real reduction in new infections in this population. In fact, according to data from our National Health System [32], the absolute number of migrants newly diagnosed with HIV in our country remained stable in the last 10 years. One possible explanation could be that clinicians are now less prone to request baseline GRT due to the increased availability of antiretroviral drugs with high genetic barrier to resistance. However, in order to both maintain a high level of PDR surveillance and reduce the risk of virological failure, genotypic resistance testing should be performed in all HIV-infected subjects at diagnosis and at virological failure, especially in vulnerable populations, like migrants. In addition, global access to the care could be unfortunately lower for migrants compared with local people, increasing the already complex management of HIV infection in these groups. Therefore, an extended access to care for migrants in all industrialized countries is pivotal in order reduce the spread of PDR.
Our study confirmed that having more than one PDR at first GRT is an important predictor of virological failure [33]. Moreover, being a migrant from sub-Saharan Africa is associated with failure, thereby underlining the specific vulnerability of this population in comparison with subjects coming from industrialized countries [34,35]. It should be noticed that previous studies [36] reported an increased incidence of virological failure in sub-Saharan Africans mainly because of their lack of adherence to ART and not due to their viral subtype [37]. Probably, as already discussed, our results could refect the same phenomenon in our cohort; nevertheless, it should not be forgotten that migrants from low-income countries are not fully integrated in the health-care system; therefore the lack of adherence could derive also from difficulties in access to care [38].
The strengths of the study are the large sample size, the wide time span analysed and the epidemiologically relevant findings, as they are representative of patients entering ‘real life’ clinical care. The main limitations are the retrospective design of the study, and the lack of information about the calendar year of migration and acquisition of HIV-1 infection. These latter can greatly influence the epidemiology of different patterns of PDR; recent studies, in fact, seem to suggest that more than 60% of infections in migrants are acquired after migration in Europe [39].
Second, it is quite difficult to estimate which proportion of migrants with HIV infection are represented in ARCA, especially among illegal migrants. In fact, although in Italy equal access to ART and health-care facilities is assured by the universal health-care system for all HIV-infected patients, including undocumented individuals, access barriers to the health system still exist. However, it is reasonable to suppose that, since all consecutive subjects diagnosed at participating centres are included into the database, the majority of migrants who gained access to care at least once were equally included, independently from their legal status.
Moreover, in this study PDR to integrase strand transfer inhibitors was not evaluated; nonetheless, in line with other studies on this topic [40,41] the prevalence of transmitted mutations to this class is particularly low in HIV-1-infected subjects enrolled in ARCA database, with no evidence of increasing trend in recent years [6].
Similarly, recently emerged drug resistance mutations for second-generation NNRTIs were not evaluated in this study since they are not included into the 2009 WHO SDRM list. However by conducting a post hoc investigation of these mutations (according to recently updated USA-IAS list [42]) we identified only 5 patients harbouring the E138K, 60 cases of E138A (reaching an incidence of 5%, that is in line with previous data reported [43]), 4 cases of E138G and 1 case of V179L. Conversely, any case of E138QR, H221Y or F227C was detected. Therefore, only a limited circulation of these mutations associated with new NNRTIs was observed in our population.
Finally, it should be considered that ART compliance was unavailable among variables analysed as a predictor of virological failure. However, the limited virological failure rates in this cohort stand for a good adherence overall.
In conclusion, PDR appeared to be stable over the years in migrants to Italy, however, it remains an important cause of virological failure together with higher viral load at diagnosis and being a migrant from a low-income country. In addition, these data suggest that baseline genotyping should still be performed for all new HIV-1 diagnosed patients and in particular in vulnerable populations, also in the ‘rapid-ART’ era.
Footnotes
Acknowledgements
This work was presented as an ‘oral communication’ at 11° Italian Conference on AIDS and Antiviral Research (ICAR 2019), 5–7 June 2019.
This research did not receive any specific grant from funding agencies in the public, commercial or not-for-profit sectors.
The authors declare no competing interests.
