Abstract
Background
Chronic hepatitis B (CHB) treatment adherence has been poorly studied worldwide. We evaluated long-term virological and adherence outcomes to antiviral treatment in CHB patients.
Methods
A prospective 183 Brazilian CHB patient cohort treated with monotherapy or combination adefovir dipivoxil, entecavir, lamivudine and/or tenofovir disoproxil fumarate was studied in a reference tertiary centre. Treatment adherence was evaluated by a validated questionnaire named ‘Assessment of Adherence to Antiviral Therapy Questionnaire’ (CEAT-HBV) within three yearly periods (2010/2011, 2013/2014 and 2014/2015).
Results
CEAT-HBV identified 43% (79/183) patients with non-adherence to antiviral treatment and among them, 67% (53/79) were viral load positive. The main causes associated with non-response to antiviral treatment were drug resistance variants followed by non-adherence, insufficient treatment duration and other causes. Single-dose pharmacokinetics demonstrated 35% (23/65) antiviral non-adherence. 2 years after the first assessment, the CEAT-HBV indicated that 71% (101/143) of subjects adhered to treatment (per-protocol population). However, 21% (40/183) of the patients could not be evaluated and were excluded. The main reasons for exclusion were death (20/183), 11 out 20 deaths due to hepatocellular carcinoma. HBV booklet was used for medical education. The third CEAT-HBV assessment (2014/2015) showed that 83% (112/135) patients were compliant with treatment adherence (per-protocol population). Long-term evaluation showed that adherence rate based on CEAT-HBV continue to increase after 4-years (P<0.001).
Conclusions
The results highlight the importance of CHB therapy adherence assessment monitoring. Long-term adherence outcomes were dynamic and it is possible to increase the migration rate to adherence/HBV-DNA-negative group.
Introduction
Chronic hepatitis B (CHB) affects over 257 million people worldwide. Long-term complications of infection include cirrhosis and hepatocellular carcinoma (HCC) deaths, which together cause over 887,000 deaths annually [1]. Long-term therapy with oral nucleoside analogues (NUCs) can effectively suppress HBV replication, improve hepatic function and may reduce the risk of disease progression [2–5] in individuals chronically infected. However, NUC discontinuation usually leads to loss of viral control and antiviral resistance variant development [6–8]. Conversely, adherence monitoring is a strategic action to avoid treatment failure [9–12].
Current antiviral therapy can suppress HBV in the blood but may not eradicate integrated HBV or covalently closed circular DNA (cccDNA) from hepatocytes. Thus, long-term (indefinite) treatment is needed in most individuals [13–15]. Therefore, most individuals with CHB require long-term treatment, which is associated with increasing risk of drug resistance, particularly when NUCs with low genetic barrier to resistance are used as monotherapy [16–18]. The virological breakthrough (VBT) is the first clinical manifestation of antiviral drug resistance and individuals with better adherence to NUC therapy have a trend towards lower rates of viral relapse [11].
The efficacy of novel approved NUCs was demonstrated in clinical trials [7,19–25], however, these trials generally evaluate 1-year or extended 2-year efficacy and safety end points, and the results may not be extrapolated to clinical practice due to challenges like persistent adherence and antiviral access, sometimes with high cost [26,27]. Therefore, post-marketing observational studies are needed to demonstrate the effectiveness of these agents long-term.
HBV therapy adherence has been shown to relate to clinical responses and treatment outcome [11]. A systematic review of HBV therapy adherence suggests that non-adherence in CHB could be less frequent than anticipated, but there is an increased risk of virological failure in poor adherent patients [28]. Communication and compliance appear central to treatment outcomes, and therefore strategies need to be implemented to improve ongoing adherence [29]. However, there are limited clinical data regarding the treatment adherence worldwide. The goal of our study was to evaluate long-term virological and adherence outcomes to antiviral treatment in adults chronically infected with HBV.
Methods
Study design
The target population of our cohort study included CHB individuals treated in a Brazilian public tertiary centre. The study was gender inclusive (both males and females were enrolled). The period of study was December 2010 to March 2015, and the sampling was consecutive (not probabilistic). Eligible individuals were enrolled during a scheduled clinic visit between December 2010 and August 2011, with follow-up until March 2015. At enrolment, subjects were asked to complete a confidential demographic and clinical questionnaire. The Brazilian government supported all costs of medical appointments, HBV therapy and laboratory exams. The health-care providers (four clinical pharmacy residents and two clinical pharmacy assistants) assessed the participants’ medication adherence during the clinic visit and medical staff (one hepatology faculty, one physician assistant and four medical residents) were informed about medication adherence. The adherence assessment protocol consisted of application of ‘Assessment of Adherence to Antiviral Therapy Questionnaire’ (CEAT-HBV) [9] over three medical appointments. The medical education and antiviral adherence communication were performed using an HBV booklet (HBV booklet is a printed material with information about the disease and medication obtained from the Drug Medical Label in Portuguese language and adapted for Brazilian patients’ understanding) in the second and third medical appointments. A blood sample for HBV viral load determination was collected at the same time. Medical records of each subject were reviewed to retrieve information regarding medical history, current and previous HBV treatments and VBT.
Subjects
Adult subjects with CHB (defined as a positive serum hepatitis B surface antigen [HBsAg] test for at least 6 months) attending the liver clinic at the University of São Paulo were eligible for enrolment if they were currently receiving any NUC therapy – that is, adefovir dipivoxil (ADV) 10 mg once daily, entecavir (ETV) 0.5 mg once daily, lamivudine (LMV) 150 mg once daily and tenofovir disoproxil fumarate (TDF) 300 mg once daily – for more than 3 months, to provide consent and had willingness/capacity to answer the questionnaire. Subjects were required to be at least 18 years of age. Ongoing NUC therapy in any combination was permitted. Individuals were excluded if they had coinfection with hepatitis C, hepatitis D or HIV. Following initial enrolment, individuals who stopped treatment because of demonstrated HBV clearance (defined as HBV undetectability and HBsAg seroconversion) were excluded from continued participation in the study.
Adherence assessment
Treatment adherence was evaluated using the validated questionnaire CEAT-HBV [9,30] over the three yearly periods – baseline December (2010)/August (2011), intermediate November (2013)/May (2014) and final follow-up July (2014)/March (2015).
The CEAT-HBV is a questionnaire with 20 questions that intends to assess the level of patient adherence to antiviral therapy. It was divided into two domains: one domain was called ‘degree of compliance with antiviral therapy’ with 5 questions [1–4,12] and the other domain was called ‘barriers to adherence’ with the other 15 questions [5–11,13–20]. The answers to the questions use the 5-point Likert scale (a higher score indicating greater adherence to the treatment) except questions 5, 19 and 20. On question 5, the score varies from 0 to 2: 0 indicates patients who did not remember the name and dosage of the antiviral administered; 1 point for those who knew only the name or dosage and 2 points for those who knew both the name and dosage of the antiviral. On questions 19 and 20, the score can be 0 or 1 (a negative answer on question 19 and an affirmative answer on question 20 scored 1). The full questionnaire ranges from 17 to 89 points. The receiver operating characteristic curve (ROC curve) for the CEAT-HBV demonstrated the capacity of the questionnaire in classifying adherent and non-adherent patients (P<0.001) and CEAT-HBV presented the sensitivity and specificity of the curve and set the cutoff at 80.50, which was associated with a sensitivity of 81.4% and a specificity of 67.3% [9,30].
Therefore, CEAT-HBV has a cutoff of <80 points on the global score questionnaire that indicates individuals who did not adhere to antiviral therapy and usually had a detectable HBV viral load. NUC therapy adherence was defined according to CEAT-HBV qualitative criteria: adherence or non-adherence [9,30].
HBV DNA Viral Load Assessment
Treatment adherence was also evaluated by analysis of virological response assessed by monitoring plasma HBV DNA at these three adherence assessments. Negative HBV DNA was considered an indirect method for adherence assessment due to the strong link between adherence rate and virological response [31]. Plasma HBV DNA was quantified using real-time polymerase chain reaction (RT-PCR) COBAS TaqMan HBV test (Roche Molecular Systems, Inc., Branchburg, NJ, USA). The HBV viral load detection limit was between 12 and 110 million IU/ml. When a subject was HBV-DNA-positive in the first adherence assessment, antiviral drug resistance variant and single-dose pharmacokinetics (direct adherence confirmation) were performed.
Classification of patients
Patients were classified in four groups: 1) adherence/viral load negative are patients who adhere to antiviral therapy according to CEAT-HBV questionnaire and have HBV DNA viral load negative. 2) Adherence/viral load positive are patients who adhere to antiviral therapy according to CEAT-HBV questionnaire and have HBV DNA viral load positive. 3) Non-adherence/viral load negative are patients who do not adhere to antiviral therapy according to CEAT-HBV questionnaire and have HBV DNA viral load negative. 4) Non-adherence/viral load positive are patients who do not adhere to antiviral therapy according to CEAT-HBV questionnaire and have HBV DNA viral load positive.
HBV Genotypes and Variants
HBV sequencing was performed in 54/65 specimens only at the first adherence assessment and at the Laboratory of Biochemical Pharmacology, Emory University, Atlanta, GA, USA. HBV genotypes and variants were analysed by direct sequencing. DNA was extracted from 200 μl of plasma using the Qiagen EZ1® Virus Mini Kit v2.0 (Qiagen, Hilden, Germany), according to manufacturer's instructions and eluted in 60 μl nuclease-free water (Qiagen). Extracted from plasma samples, the HBV DNA polymerase gene region was amplified by nested-PCR using specific primers. Briefly, the primers, forward primer: HBV0179FE (5′- CTA GGA CCC CTG CTC GTG TT −3’) and the reverse primer: HBV1286RE (5′- GCT AGG AGT TCC GCA GTA TGG −3’), were used in the first-round PCR. The inner primers: sense, HBVRes311FN (5′- GCT GCA GTC CCC AAC CTC CAA TCA CT −3’) and antisense, HBVRes1026RN (5′- GGA GCA GCA AAV CCC AAA AGW CC −3’) were used for nested PCR. The following PCR thermal-cycling programme was performed: PCR 40 cycles at 94°C for 45 s (first round) or 95°C for 30 s (second round), 55°C for 20 s (first round) or 58°C for 15 s (second round) and 72°C for 75 s. The intended fragments were amplified using 2 x PCR master mix contained 10 ml PCR reaction mixture (PerfeCTa SYBR Green FastMix, Low ROX [Quanta BioSciences, Gaithersburg, MD, USA]) with 10 ml of DNA extract (first round) and 3 ml of the first round PCR product (second round). After amplification of the polymerase gene, the amplicons (715 bp) were visualized by agarose gel electrophoresis and purified using QIAquick PCR Purification Kit (Qiagen), according to manufacturer's instructions. DNA were quantified by NanoDrop 2000 spectrophotometer (Thermo Fisher Scientific, Waltham, MA, USA). The purified PCR products were bidirectionally sequenced by GENEWIZ (South Plainfield, NJ, USA) and were subsequently analysed with the Deep-Chek®-HBVv1.4 software (ABL SA, Luxembourg City, Luxembourg), in order to perform genotyping, amino acid mutation detection using the HBV reference sequence X04615.1 and drug resistance assessment through up-to-date guidelines including the SeqHepB interpretation system and database.
Quantification of NUC by liquid chromatography tandem-mass spectrometry
This test was included only in conjunction with the first adherence assessment, and was performed at the Laboratory of Biochemical Pharmacology, Emory University, Atlanta, GA, USA. Briefly, 50 μl of plasma (LMV monotherapy samples) and 200 μl of plasma (other NUC samples) were extracted with 1 ml of methanol containing 10 ng/ml of emtricitabine (LMV monotherapy samples) or 10 ng/ml of emtricitabine (FTC) and 1 ng/ml of acyclovir (other NUC samples) as the internal standards. The supernatant of each extraction was collected, air-dried and reconstituted in 300 μl of 2 mM ammonium acetate with 0.1% formic acid, then subjected to LC-MS/MS analysis [32]. A Dionex Ultimate 3000 HPLC system (Thermo Scientific, Waltham, MA, USA) coupled with an AB SCIEX API 5000 triple quadruple mass spectrometer (AB SCIEX, Framingham, MA, USA), with an ESI interface operated in positive mode was used for the LC-MS/MS analysis. Analyst software version 1.5.2 was used to operate the mass spectrometer and to perform data analysis. Calibration curves were generated from standards of LMV, ADV, TFV and ETV by serial dilutions in blank plasma samples using the same extraction method described above. The calibration curves had r 2 value greater than 0.99.
Outcomes measures
The primary end points were long-term virological response (HBV DNA viral load undetectable) and adherence rates at baseline (2010–2011), intermediate and final follow-up. Secondary end point was to determine baseline predictors of CHB treatment failure. At enrolment, antiviral drug resistance variant and single-dose pharmacokinetics were evaluated for understanding of factors associated with treatment failure at baseline in individuals with HBV-DNA-positive samples.
Study approval
The study was performed in accordance with the principles of the Declaration of Helsinki and was approved by the Institutional Review Board of the Hospital das Clínicas of the University of Sao Paulo School of Medicine (protocol 0581/10). Written informed consent was obtained from all enrolled subjects.
Statistical analyses
All statistical analyses were performed using the Microsoft Excel 2016 (Microsoft Corporation, Redmond, WA, USA) and SPSS version 13.0 (IBM Corporation, Armonk, NY, USA). Continuous variables were expressed as median and IQR. Categorical variables were expressed as number and percentage. Categorical variables were analysed using the c 2 test, and Fisher's exact test when appropriate. The primary end point analysis was performed with data from the intention-to-treat (ITT) population (all individuals who were included in the first adherence assessment). To minimize bias and include all patients in the ITT analysis, imputation for group was necessary when a subject died, withdrew or had cleared HBV. For subjects completing the study as planned, the measurements taken at the third adherence assessment were used (per-protocol analysis). However, when a subject interrupted HBV therapy prematurely, the virological and adherence outcome data were transferred over to the ‘unfavourable outcome’ group (non-adherence and HBV-DNA-positive). When a subject died, or cleared HBV, the virological and adherence outcome was kept in the same group. When a subject was withdrawn due to loss of follow-up, the virological and adherence outcome was kept in the same group, providing the subject had transferred to another liver centre and was trackable. If no follow-up information was available for an enrolled subject, data for that subject was transferred to the ‘worst outcomes’ group. Individuals who withdrew consent were excluded from analysis. Markov model was used to do long-term dynamic analysis of virological and adherence outcomes. The P-value of <0.05 was considered statistically significant.
Results
Overall, 580 CHB individuals were screened and 183 subjects were enrolled in the study (Figure 1A). After the first adherence assessment, 40/183 subjects were excluded from the study due to subsequent deaths (20), lost to follow-up (16), cleared HBV (3) and stopped NUC treatment (1). The main cause of death was HCC (11), followed by unclassified shock (2), septic shock with abdominal focus (1), respiratory insufficiency (1), stroke (1), chronic renal failure (1), bacterial pneumonia (1), acute myocardial infarction (1) and suicide (1). Regarding subjects who were lost to follow-up, 12/16 were not found after three attempts to contact, 3/16 moved to another HBV reference centre in another city and 1/16 withdrew consent. After the second adherence assessment 8 out of 143 subjects were excluded; among them, 4 moved to another HBV reference centre in another city, 1 was lost to follow-up, 1 stopped NUC treatment, 1 died with unknown cause, and 1 withdrew consent. Therefore, 135 patients reached the third adherence assessment.

Study profile
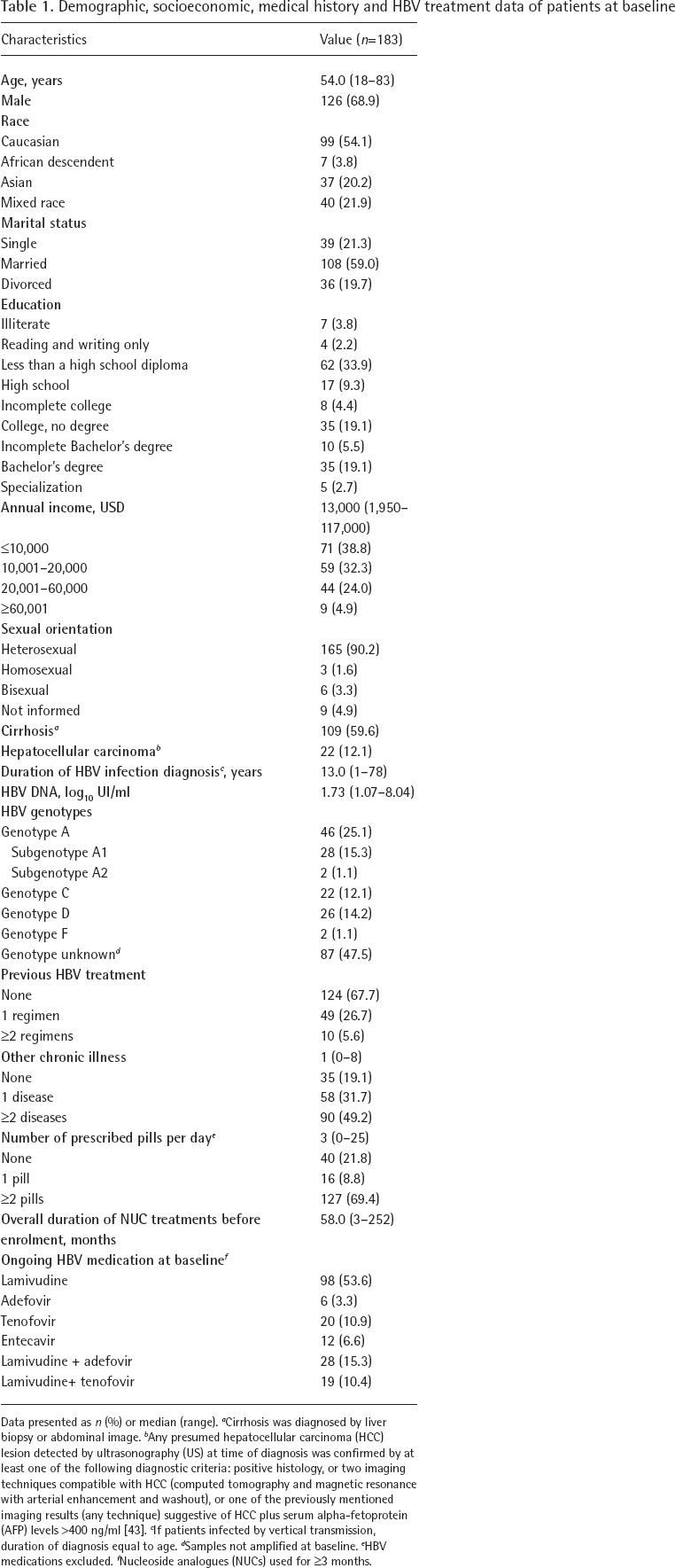
A total of 183 patients were enrolled and demographic and socioeconomic data were described in Table 1. The median age was 54 years and the population was predominantly male (68.9%). Approximately, half (54.1%) were Caucasian and 90% of the patients were heterosexual as sexual orientation. Only half of the patients (50.8%) had more than a high school degree and approximately 70% of the patients reported an annual income of less than 20,000 USD.
Demographic, socioeconomic, medical history and HBV treatment data of patients at baseline
Data presented as n (%) or median (range).
Cirrhosis was diagnosed by liver biopsy or abdominal image.
Any presumed hepatocellular carcinoma (HCC) lesion detected by ultrasonography (US) at time of diagnosis was confirmed by at least one of the following diagnostic criteria: positive histology, or two imaging techniques compatible with HCC (computed tomography and magnetic resonance with arterial enhancement and washout), or one of the previously mentioned imaging results (any technique) suggestive of HCC plus serum alpha-fetoprotein (AFP) levels >400 ng/ml [43].
If patients infected by vertical transmission, duration of diagnosis equal to age.
Samples not amplified at baseline.
HBV medications excluded.
Nucleoside analogues (NUCs) used for ≥3 months.
Approximately 60% of the 183 CHB patients had cirrhosis and 12% had HCC. The median duration of HBV infection was 13 years. Only 32.3% of the subjects had received prior HBV treatment. Three-quarters of the subjects were receiving NUC monotherapy and the others NUC combinations. The median duration of the current HBV treatment regime was 58 months, but 86.3% of subjects had been on the current HBV treatment for more than 1 year and half, of which 49.7% had been on the current treatment for more than 5 years. Comorbidities were common within the cohort including half (49.2%) of them with at least two chronic diseases and approximately 70% took more than two pills daily (Table 1).
Primary end point
The primary end points were long-term virological response (HBV DNA viral load undetectable) and adherence rates at baseline (2010–2011), intermediate and final follow up.
At baseline, intermediate and final follow-up evaluation the adherence rate determined by CEAT-HBV was 56.8%, 63.7% and 71.8%, respectively. The virological end point at baseline, intermediate and final follow-up evaluation was 61.7%, 60.8% and 69.6%, respectively (Table 2). CEAT-HBV identified 43% (79/183) subjects with non-adherence to antiviral treatment and among them, 67% (53/79) were viral load positive. However, 38% (70/183) had positive viral loads suggesting treatment non-response. Long-term evaluation showed that adherence rate based on CEAT-HBV in the second adherence assessment were 70.6%, on basis of an analysis of the per-protocol population; and 63.7%, based on an analysis of the intention-to-treat population with missing values imputed. These adherence rates continued to increase after 4 years, reaching 83% (P<0.001) and 71.8% (P<0.01), respectively. Adherence rate based on virological end point did not have statistically significant increase.
Adherence rate of long-term virological and adherence outcomes according to per-protocol and ITT populations
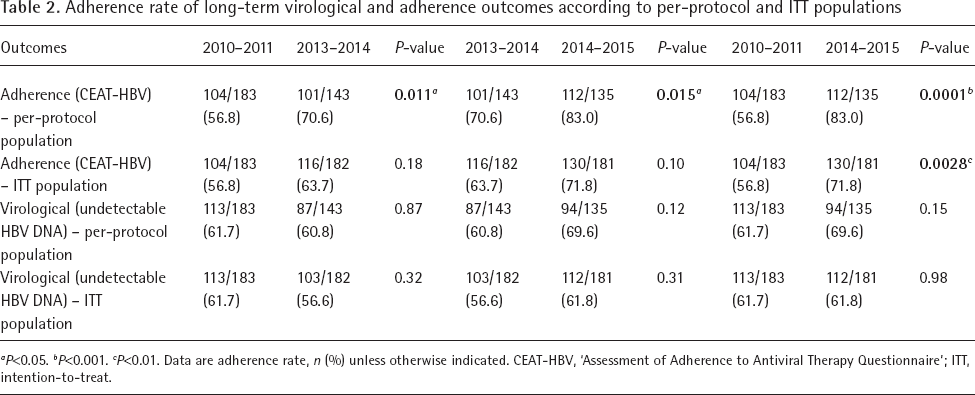
P<0.05.
P<0.001.
P<0.01.
Data are adherence rate, n (%) unless otherwise indicated. CEAT-HBV, ‘Assessment of Adherence to Antiviral Therapy Questionnaire'; ITT, intention-to-treat.
Secondary end points
The migration rate between the different groups according to viral load and adherence assessment can be seen in Figure 2 (per-protocol) and Figure 3 intention-to-treat (ITT). There was an increase of subjects’ flow to adherence and HBV-DNA-negative group, represented in light grey (Figures 2 and 3). In per-protocol population, the migration from dark grey to light grey groups was statistically significant increased (P<0.001) in the second and third evaluations, with better results after third evaluation due to migration from medium grey group to light grey group (P<0.001; Additional file 1). The ITT population showed similar results and statistically significant maintenance of patients from light grey and dark grey groups across the time (Additional file 2).
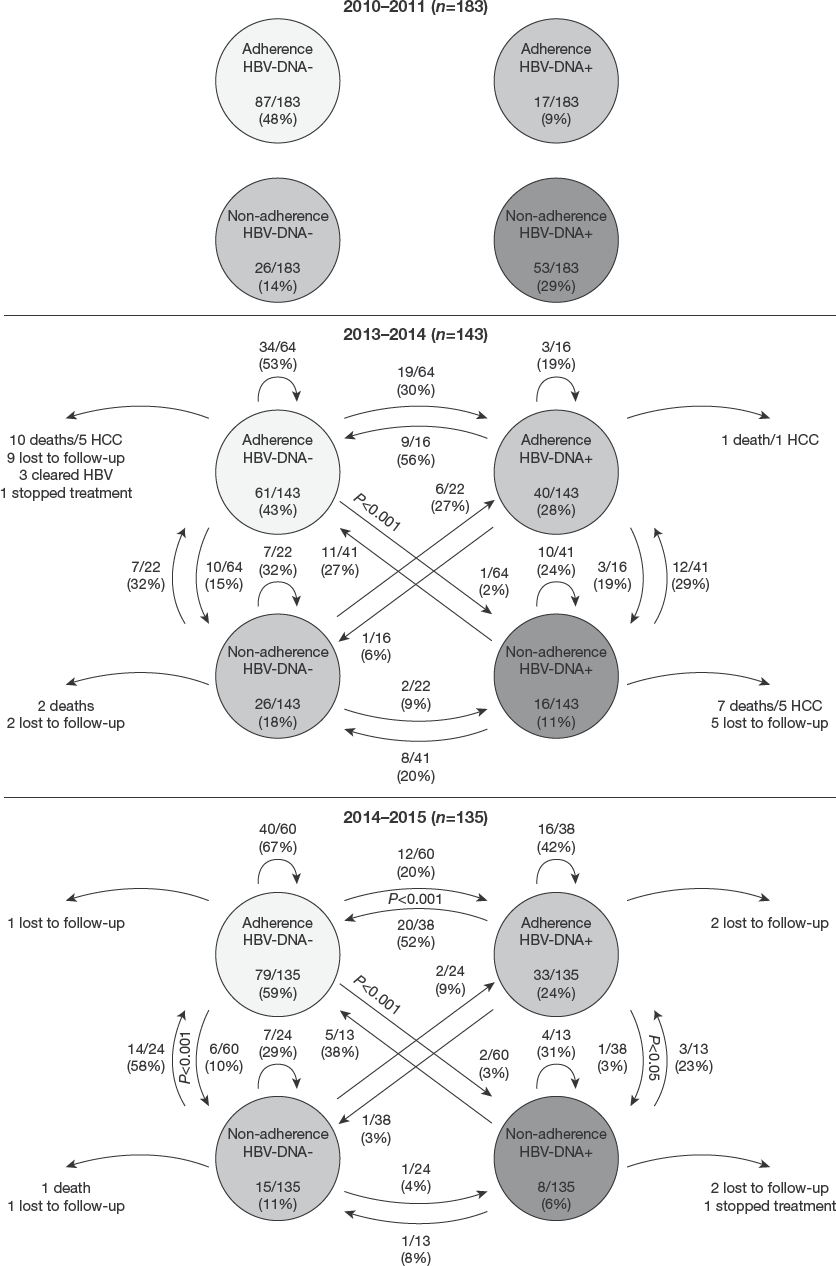
Dynamic analysis of viral load and adherence assessment outcomes in per-protocol population
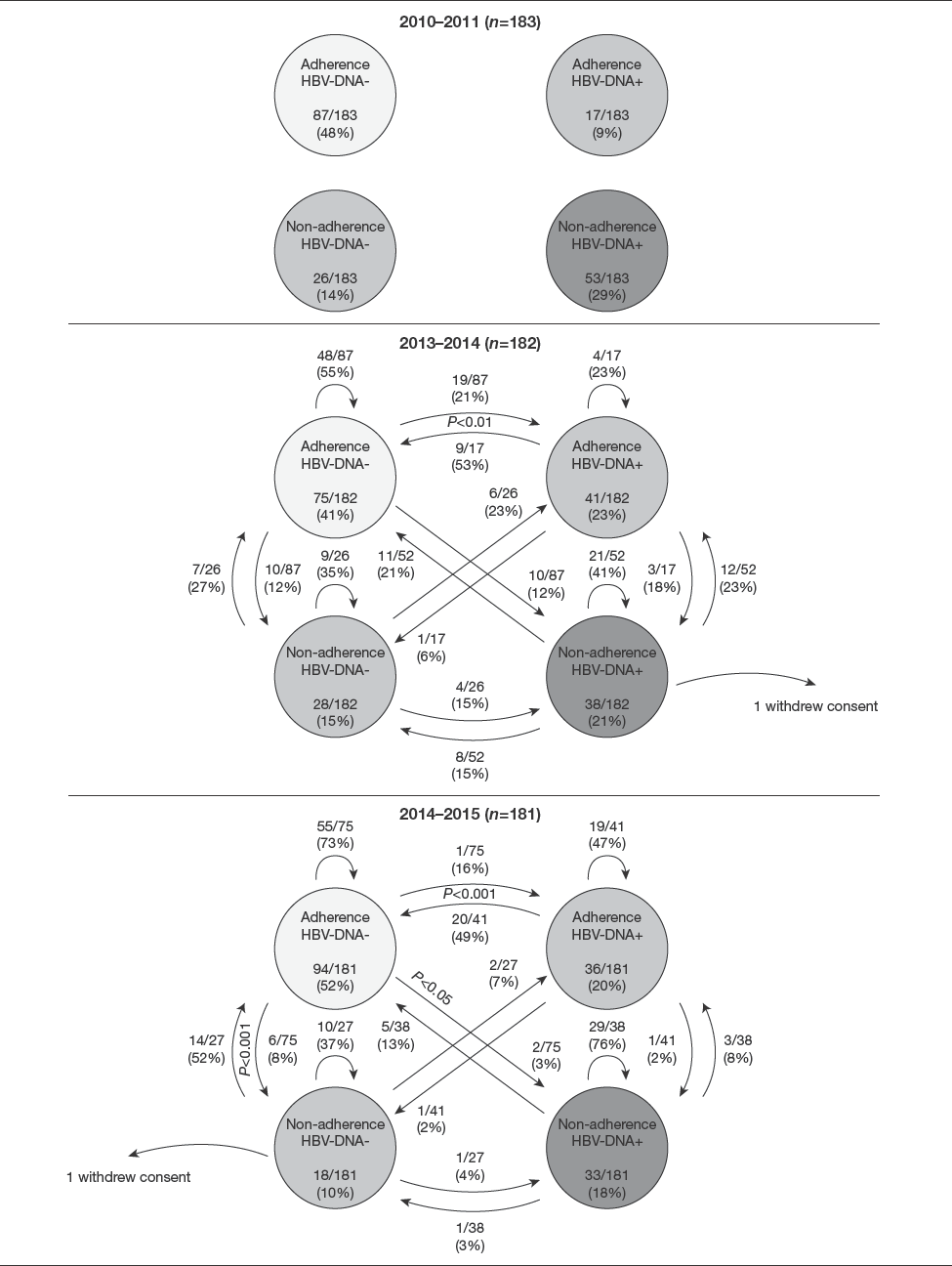
Dynamic analysis of viral load and adherence assessment outcomes in intention-to-treat population
A total of 143 patients had HBV therapy modified at the second adherence assessment as follows 37.8% (54) LMV+TDF, 30.7% (44) LMV, 11.2% (16) LMV+ADV, 10.5% (15) TDF, 5.6% (8) ETV, 3.5% (5) ADV and 0.7% (1) ADV+ETV. At the third adherence assessment, HBV treatment of 135 patients were 41.5% (56) LMV+TDF, 28.9% (39) LMV, 11.1% (15) TDF, 8.9% (12) LMV+ADV, 5.2% (7) ETV, 3.7% (5) ADV and 0.7% (1) ADV+ETV based on per-protocol population. The HBV therapy was performed according to current HBV Brazilian Guidelines at each time point. It is of note that the Brazilian protocol recommend a higher dose of lamivudine (150 mg once daily) in comparison with other countries that use 100 mg once daily.
The main reasons for exclusion were death (21/183), 10 out of 21 deaths due to HCC. The HBV genotype C had significant association with HCC development in this population (P=0.046). It is worth noting that 10 subjects deceased were adherent to antiviral treatment and had undetectable HBV DNA.
Of the 70 subjects with PCR HBV positive at baseline (Figure 1B), 5 subjects were excluded due to HBV plasma/serum unavailable. Only 54/65 specimens were amplified using nested PCR and the most frequent antiviral resistance variants were M204I/V (77.7%), L180M (59.2%), L80I (14.8%), V173L (7.41%) and Q215H (5.5%), as seen in Figure 4A. When we stratified the antiviral resistance variants according to antiviral therapy at baseline, HBV variants were present in 39.3%, 31.6% and 25.5% of subjects receiving LMV+ADV, LMV+TDF and LMV monotherapy, respectively, after a median of 58 months of HBV antiviral treatment. One subject from the TDF group had a variant of interest, but this individual had prior treatment with LMV and ETV monotherapy. Overall, 42/145 (29%) of subjects receiving LMV presented antiviral resistance variants at baseline (Figure 4B).
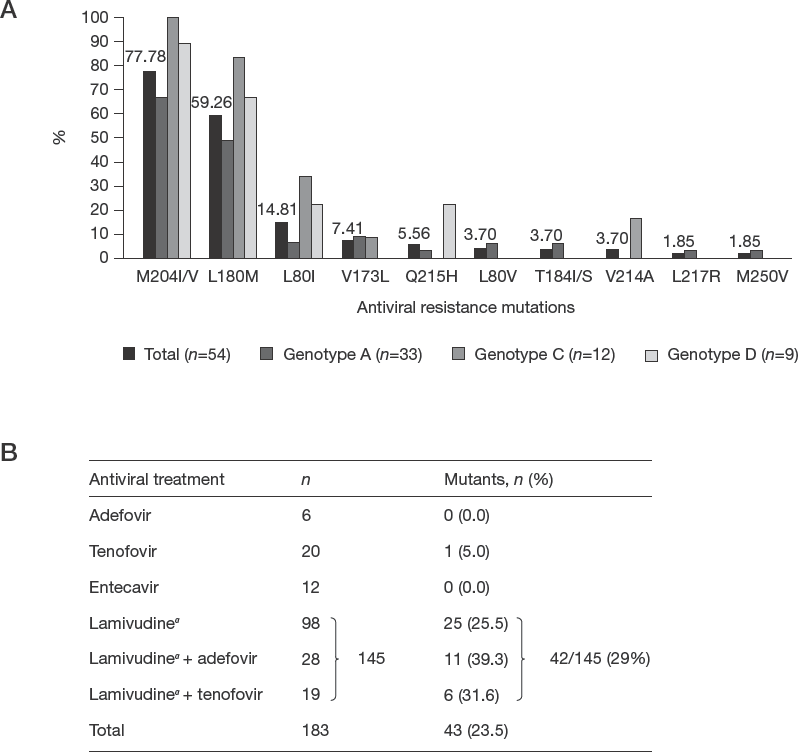
Drug resistance variants (mutations) analysis based on DeepChek–®HBV/HDV v1.4 software
The adherence rate based of single-dose pharmacokinetics (direct adherence method) at baseline was 64.6% (42/65 subjects).
Until now, few studies on chronic hepatitis B (CHB) treatment adherence have reported long-term adherence outcomes but in our 4-year cohort study with 183 patients, we found that the main factors associated with treatment failure (HBV-DNA-positive) at baseline were due to drug resistance variants (47.2%), followed by non-adherence (34.2%), insufficient HBV treatment duration (7.2%) and other causes (11.4%).
Discussion
Herein, we demonstrated that adherence to NUCs in CHB individuals being treated in an outpatient hepatology clinic was low at baseline. Other studies showed that the mean adherence reported ranged from 81 to 99%, with 66 to 92% of individuals being 100% adherent [28]. However, during this study the knowledge about drug resistance mutations and HBV therapy non-adherence promoted antiviral replacement and new interventions in order to improve antiviral adherence. The patient education based on antiviral adherence communication by an HBV booklet, at the second and third adherence assessment, was key to improve adherence rate and migration flow to adherence/HBV-DNA-negative group [29].
The virological outcome alone did not demonstrate statistical significance improvement across the time. The dynamic flow of individuals between groups, presence of drug resistance variants, non-adherence and insufficient treatment duration can explain this point.
Until 2009, the first-line treatment for CHB was only LMV 150 mg once daily monotherapy according to HBV Brazilian Guidelines. However, many individuals worldwide have developed antiviral resistance from the widespread use of less potent NUCs, such as LMV 100 mg or ADV, which have a low genetic barrier to resistance [13,14]. The resistance to LMV 100 mg once daily emerges in approximately 20% of individuals after 1 year and in 70% of individuals after 5 years of treatment [33], and long-term LMV therapy may select tyrosine-methionine-aspartate-aspartate (YMDD) variant [34]. Our study showed only 29% of resistance to LMV after an average of 58 months of treatment. This higher dose of LMV (150 mg) is poorly studied at this moment and future trials with larger samples should be encouraged.
Other countries including China and United States showed that antiviral therapy cost is one significant barrier to improved care for individuals with CHB and it was correlated to lack of treatment adherence [10,27,35]. This was not a limitation to our study because the Brazilian government provides free HBV therapy for all CHB individuals.
Based on the adherence rate at baseline, CEAT-HBV was 56.8%, the Morisky test was 53.5% and the virological outcome was 61.7% (P=0.14). We did not use the Morisky test because CEAT-HBV is a validated self-report for CHB individuals and it is the first specific instrument with greater sensitivity and specificity [9,31].
Several factors may explain the low rate of adherence to HBV NUCs observed in this study. First, the treatment stigma and psychological impact for HBV-infected individuals may be different as compared with HIV and persons with certain types of cancer. Another reason for the low adherence rate in this study may be related to the fact that most of the subjects had low education level, low annual income and other comorbidities [11]. Furthermore, over half of subjects had compensated cirrhosis and 12.1% had HCC at baseline. A third possible explanation for the low adherence rate is that the subjects were seen in a tertiary clinic that it is national reference for liver transplantation and HCC. To compound this scenario, many individuals had polypharmacy and experienced periods of antiviral withdrawal failure at the pharmacy and lost to follow-up at medical appointment [36,37].
A curious fact is related to deaths by HCC. At baseline, 12.1% of baseline had HCC not in end-stage of liver disease, and after 2 years, half of them had died. It is important to highlight that five HCC deaths occurred in adherence/HBV-DNA-negative group. Our subjects had median of 58 months of NUC treatment at enrolment and resistance genotyping was not routine clinical practice. This may lead to a delay in treatment failure perception. Although several studies [38–40] showed that long-term therapy with TDF and ETV was associated with reduced incidence of HCC, long-term real-life evidence may be affected by low adherence rate and antiviral resistance [41,42].
This study had several limitations. First, drug resistance mutations and quantification of NUC were only studied at the first adherence assessment. Second, in contrast to previous studies, the study population had free access to HBV drugs [11,27]. Third, enrolled subjects were all from a tertiary liver centre with more advanced disease. Fourth, there was heterogeneity of HBV treatment regimens. However, we have consistent long-term results that may be extrapolated to other study populations. Finally, drug resistance mutations, NUC quantification at other assessments, as well continuous follow-up data may confirm these results.
In conclusion, this study revealed that in a dynamic model, VBT was common in clinical practice. However, VBT was mainly related to antiviral drug resistance and non-adherence to treatment. In our Brazilian public clinical hospital, individuals with CHB receiving NUC therapy would likely benefit from being counselled on medication adherence at all medical appointments. Understanding of real VBT cause is prudent, since adherence can avoid unnecessary therapy replacement. The data highlight the importance of CHB therapy adherence assessment monitoring. Long-term adherence outcomes may be dynamic and it is possible to increase the migration rate to adherence/HBV-DNA-negative group. Thus, medical education on antiviral adherence communication may improve the effectiveness of HBV treatment in clinical practice.
Footnotes
Acknowledgements
We thank the Alves de Queiroz Family Fund for Research for support of studies, James J Kohler (Emory University, Atlanta, GA, USA) for manuscript review, Dimitri Gonzalez and Chalom Sayada (Advanced Biological Laboratories – ABL, Luxembourg City, Luxembourg) for the use of DeepChek®–HBVv1.4 software for genotyping and drug resistance analysis. All named authors meet the International Committee of Medical Journal Editors (ICMJE) criteria for authorship for this manuscript, take responsibility for the integrity of the work as a whole, and have given final approval for the version to be published. Finally, we greatly thank all patients for their consent and participation in the study and for the research use of their plasma specimens.
The findings and conclusions in this report are those of the authors and do not represent the official position of the Centers for Disease Control and Prevention. Use of trade names and commercial sources is for identification only and does not constitute endorsement by the U.S. Department of Health and Human Services, or the U.S. Centers for Disease Control and Prevention.
A part of this study was presented at the AASLD The Liver Meeting, San Francisco, on 16 November 2015. No commercial funding was involved in this study.
This research was supported by Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq number 201812/2014-2 to RMA), (CNPq number 308609/2018-2 and 551407/2007-7 to SKO), SPRINT-Emory FAPESP (2017/50042-2) and in part by NIH sponsored Center for AIDS Research Grant P30AI050409 and 1R01AI132833 (to RFS).
The principal investigators, RMA and SKO, were responsible for the conception and design of the study, the acquisition, analysis and interpretation of the data, and the drafting of the manuscript. LCB, ST, YJ, ASF, PCAH, LMG-R and YK helped with the design of the study, acquisition of the data, and critical revision of the manuscript regarding intellectual content. RFS, FJC and SKO guided the study and provided input on assays to be performed and helped write and review this paper.
The authors declare no competing interests.
