Abstract
Background
This 5-year follow-up of the CCgenos cross-sectional study aimed to observe real-life outcomes in a cohort of 997 Han Chinese patients with chronic HCV infection and to explore the impacts of HCV genotype, patient characteristics and treatment status.
Methods
Clinical information and centralized HCV RNA measures were collected every 6/3 months for untreated/treated patients. Overall disease progression was defined as ≥1 of: de novo development of cirrhosis, Child-Turcotte-Pugh score increased by ≥2 points (if cirrhosis at baseline), progression to decompensated cirrhosis, hepatocellular carcinoma (HCC), liver transplant or death. Cox regression assessed risk factors for the time from estimated infection to cirrhosis or HCC. Logistic regression assessed risk factors for incidence rates of cirrhosis and overall disease progression.
Results
281 of 514 patients enrolled across China completed 5 years of follow-up. Overall disease progression occurred in 36/364 (9.9%) treated patients and 35/148 (23.6%) untreated patients (odds ratio = 0.35; 95% CI 0.21, 0.59; P<0.0001). Overall disease progression occurred in 6/231 (2.6%) patients achieving sustained virological response at 24 weeks (SVR24) versus 11/82 (13.4%) who did not (P=0.0002). Cirrhosis development was significantly associated with abnormal aspartate aminotransferase (AST), age ≥40 years, body mass index ≥28 kg/m2, HCV GT1, platelet count <100x109/l, and AST to platelet ratio index (APRI) ≥2 (multivariate Cox regression, P<0.05). HCC was significantly associated with HCV GT1 and platelet count <100x109/l (multivariate Cox regression, P<0.05).
Conclusions
Achieving SVR24 significantly reduced the probability of overall disease progression but no significant difference was seen for both cirrhosis and HCC during 5 years of follow-up.
Introduction
Chronic HCV (cHCV) infection presents a major global public health burden [1,2]: the current estimate of global prevalence is 71 million [1,2]. The development of highly effective, well-tolerated, direct-acting antivirals (DAAs) has changed the treatment landscape for HCV, but global availability of these agents has been limited [1]. More than half of the world's HCV-infected population is in Asia [3]. In China, the prevalence of HCV is estimated at 1% (10 million people) [4].
Newly reported cases of HCV in China increased from 20,000 cases in 2003 to over 200,000 in 2012 [5]. This trend is likely to continue with new diagnoses driven by disease progression [5].
HCV is genetically diverse and each genotype is associated with a different pattern of disease progression and different responses to antiviral treatment [6,7]. In China, more than half (58%) of cHCV infection is with GT1, most of which (98%) is subtype 1b [8]. The remaining infections are mostly with GTs 2, 3 and 6, with substantial regional and demographic variation [7,8]. Compared with other HCV genotypes, GT1 is associated with lower response rates to interferon (IFN)-based therapy, although GT1b may be more likely to respond than GT1a [9]; GT1b is associated with faster progression to cirrhosis and hepatocellular carcinoma (HCC) [10,11]; GT3 has a poorer response to some DAA regimens [12]; and GT6 increases the risk of HCC [6].
Achieving sustained virological response (SVR) after IFN-based treatments has been shown to result in reduced risk of HCC [13–18]. The impact of SVR following treatment with DAAs is less clear. Some studies observed an unchanged or even increased probability of recurrence of previous HCC in patients treated with DAAs [19–21], however, a growing number of reports suggest that successful DAA treatment results in a long-term reduction in the overall risk of de novo and recurrent HCC [22–26].
With the approval of several DAAs since 2017, the standard of care in China is likely to shift away from IFN and ribavirin towards the all-oral therapies [4]. To fully understand their potential impact we first need to understand the nature of disease progression under current disease management practices in China.
The cross-sectional CCgenos study (AI452-009/ NCT01293279) and the CCgenos 5-year follow-up study (AI425-018/NCT01594554) previously described patient characteristics, management, treatment patterns and associated preliminary outcomes observed during 4.3 years of follow-up [27]. Herein, we describe the progression of liver disease over time and evaluate the effect of risk-factors and treatment status on disease progression.
Methods
Study design and patients
The CCgenos cross-sectional study (AI452-009/ NCT01293279) included Han Chinese adults (≥18 years old) with recently confirmed cHCV infection, who were naive to HCV or HBV antiviral IFN-based treatment [8]. The original cross-sectional study included host and viral genotypic analyses and an analysis of patients’ HCV exposure history [8]. Patients who participated in the cross-sectional study were invited to enter this 5-year follow-up (AI452-018ST/NCT01594554) in 2012 [27]. Information on anti-HCV treatments received between completion of the cross-sectional study (June 2011) and enrolment in the follow-up (April 2012) was collected retrospectively for all patients. The primary objective of the 5-year follow-up study was to observe and describe long-term patient management, treatment patterns and outcomes. Secondary objectives were to describe liver disease progression over time and to evaluate the effect of risk-factors and treatment status on disease progression.
The protocols of the CCgenos study were approved by a central review board, the institutional review boards of each participating centre and by the China National Human Genetic Resource Management Office (2010). The study was conducted in accordance with the International Society for Pharmacoepidemiology guidelines for Good Epidemiology Practices and applicable regulatory requirements. All patients provided written informed consent before participating. All authors had access to the study data and reviewed and approved the final manuscript.
Observational assessments and outcome measures
The collection of clinical information and centralized HCV RNA testing was performed every 6 ±2 months for untreated patients and every 3 ±1 months for treated patients. The scheduled study visits were intended to match real-life practice and guideline recommendations in China. Study visits were scheduled from enrolment. Because treatment could have been initiated before or after enrolment into the follow-up study, the visit schedule did not always correspond to a specified treatment duration. Data for SVR at 12 weeks (SVR12) and at 24 weeks (SVR24) were based on the HCV RNA result collected at the closest time point occurring 12 or 24 weeks, respectively, after the last dose of treatment. HCV RNA measurement was carried out at a central laboratory (the Peking University People's Hospital) using the Abbott RealTime HCV assay (Abbott Laboratories, Des Plaines, IL, USA; lower limit of detection 12 IU/ml). Biochemistry, haematology and blood coagulation functions were performed at local laboratories. The use of concomitant medication for the treatment of hepatitis and other indications were also documented.
Overall disease progression was defined as one or more of the following: no cirrhosis at baseline but developed cirrhosis during follow-up, cirrhosis at baseline and Child-Turcotte-Pugh score was increased by 2 points or more, progressed to decompensated cirrhosis from compensated cirrhosis, HCC, liver transplant or death. Evidence of hepatic cirrhosis (compensated or decompensated) was based on biopsy results wherever possible. In the absence of biopsy results, diagnosis of decompensated cirrhosis was based on the following criteria: ascites, hepatic encephalopathy, upper gastrointestinal bleeding or Child-Turcotte-Pugh score >7. If none of these criteria were met, evidence of cirrhosis could be based on any two of: imaging showing features of cirrhosis (nodular liver or splenomegaly), platelet count <100x10 9 /l in the absence of other explanations, liver stiffness measurement >13 kPa and gastro-oesophageal varices in endoscopy.
Statistical analyses
Virological response analysis was based on the efficacy analysis set (EAS). Other analyses were based on the full analysis set, no imputation methods for missing data were used.
Descriptive statistics included frequency and percentage. Kaplan-Meier survival analyses were used to calculate the median time from baseline to cirrhosis or HCC; median time from estimated infection to post-baseline overall disease progression or HCC; 5-year post-baseline survival from estimated time of infection and from baseline. Comparisons of the time to cirrhosis or HCC between sub-groups based on treatment status, treatment response or HCV GT were carried out using the log-rank test. Univariate and multivariate Cox regression analyses were used to assess associations between the time from estimated infection to cirrhosis or HCC, and treatment status and risk factors such as alanine aminotransferase (ALT), aspartate aminotransferase (AST), age, body mass index (BMI), gamma-glutamyl transpeptidase (GGT), sex, alcohol use, diabetes history, viral genotype, hyperlipidaemia, platelet count and AST to platelet ratio index (APRI), and SVR24 status. The association of risk factors with the incidence rates of overall disease progression and cirrhosis were assessed using univariate logistic regression (Additional file 1).
Results
Patients
Of 997 patients invited to enter the 5-year follow-up, 514 patients from 25 university hospitals across China were enrolled. Two patients were removed to give a full analysis set (FAS) of n=512 (Additional file 2). There were minimal differences between the baseline characteristics for patients enrolled in this follow-up study and the initial CCgenos cross-sectional study [27].
Among the 512 patients of the FAS, 364 (71.1%) had received an anti-HCV treatment and 148 (28.9%) remained untreated. Out of the 364 patients who received anti-HCV medication, 328 (90.1%) patients completed the treatment regimen and 18 (4.9%) patients withdrew during the treatment. Of the 328 patients who completed treatment, 317 patients were followed-up for at least 24 weeks post-treatment. The EAS or intent-to-treat population included the patients who completed the treatment and had follow-up data for at least 24 weeks (n=317) and those who withdrew during the treatment (n=18; total of 335 patients). By the end of the study (29 March 2018), 45 untreated patients and 234 treated with anti-HCV medication completed the 5-year follow-up (Additional file 2).
Demographics and baseline disease states for treated and untreated patients are shown in Table 1. The characteristics of untreated and treated patients were broadly similar except that the untreated patients were of significantly greater mean age and had significantly lower mean ALT. The most common HCV genotype was GT1 (62.1%, 318/512) followed by GT2 (23.8%, 122/512), GT3 (8.0%, 41/512) and GT6 (6.1%, 31/512; Table 1). The GT1b subtype was detected in 97.8% (311/318) patients with GT1. Among the 364 treated patients, 340 (93.4%) received IFN-based treatment and 24 (6.6%) received DAAs.
Baseline demographics and disease characteristics

t-tests or Wilcoxon rank sum tests were used to compare continuous variables between groups. χ 2 or Fisher's exact tests were used for comparison of categorical variables.
Alanine aminotransferase (ALT) ≥ ULT defined for female as ALT ≥19 U/l or for male ALT ≥30 U/l.
Aspartate aminotransferase (AST) to platelet ratio index (APRI) = 100xAST/PLT/40. BMI, body mass index; CTP, Child-Turcotte-Pugh; GGT, gamma-glutamyl transpeptidase; IL28B, interleukin 28B; SNP, single nucleotide polymorphism; ULT, upper limits of normal.
In the EAS, 69.9% (234/335) patients achieved SVR24 (Additional file 3). Patients with cirrhosis either at baseline or who developed the disease during the study experienced a lower SVR24 rate compared with patients without cirrhosis, 42.6% (20/47) versus 74.3% (214/288), respectively.
Overall disease progression
In the FAS, a total of 36/364 (9.9%) treated patients and 35/148 (23.6%) untreated patients experienced overall-disease-progression during the study (odds ratio = 0.35; 95% CI 0.21, 0.59; P<0.0001). Furthermore, improved outcomes were observed for patients who achieved SVR24 after anti-HCV treatment, as overall disease progression was experienced by 2.6% (6/231) patients who achieved SVR24 compared with 13.4% (11/82) patients who experienced treatment failure (P=0.0002). The Kaplan–Meier analysis showed a higher probability of disease-progression free survival in patients who achieved SVR24 compared with those who were unresponsive to anti-HCV treatment (log rank P=0.0008; Figure 1).
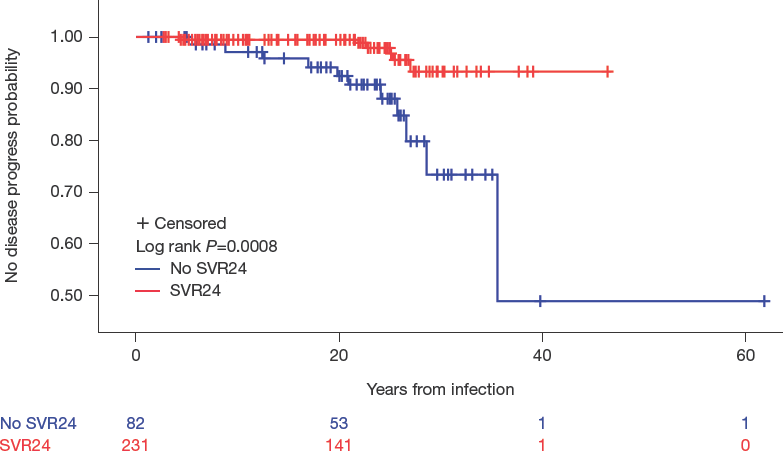
Time from estimated infection to post-baseline overall disease progression for treated patients who achieved SVR24 versus those who did not
Kaplan–Meier estimates showed a significant difference between the genotype of HCV and the time to disease progression (log rank P=0.025; Figure 2A). A higher proportion of patients with HCV GT1 experienced overall disease progression during the study (17.1%, 52/304), followed by 9.8% (4/41), 8.2% (10/122) and 3.2% (1/31) for patients infected with GT3, GT2 and GT6, respectively. In the GT3 subgroup, only patients with the GT3b subtype (15.4%, 4/26) experienced overall disease progression.
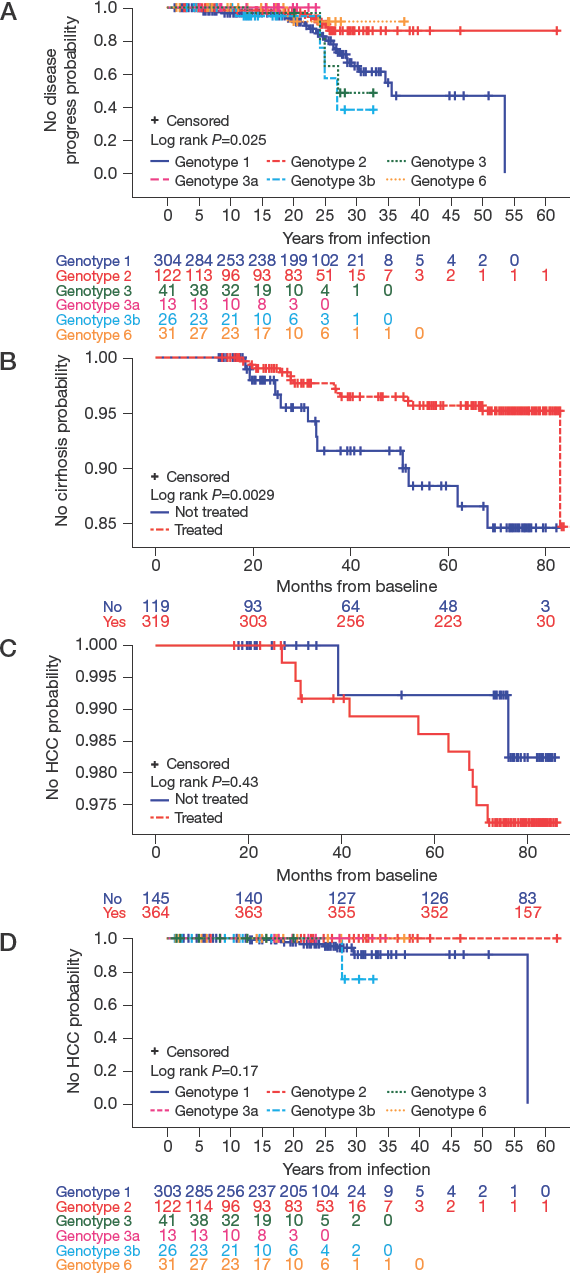
Kaplan–Meier curves
Logistic regression analysis found that the incidence rate of overall disease progression was significantly associated (P<0.05) with 18 of the assessed risk factors, with the strongest association (P<0.0001) seen for concentric obesity, high baseline ALT, AST/ALT ratio of greater than one, evidence of fibrosis (Fibroscan F4 versus F0–2), baseline APRI of at least 1.5, a baseline Fibrosis-4 score (FIB-4) at least 3.25 and baseline platelet count less than or equal to 90x10 9 /l (Figure 3). In addition, logistic regression analysis highlighted that the use of concomitant medication for hepatitis and other indications had a statistically significant association with overall disease progression (Additional file 4).

Forest plot for the logistic regression analysis of overall-disease-progression incidence (full analysis set)
Liver disease
Cirrhosis
In the FAS, 4.4% (14/319) treated and 9.2% (11/119) untreated patients developed cirrhosis during the study. The Kaplan–Meier estimate of the time to cirrhosis from baseline was 13.5–83.8 months for treated patients and 13.0–82.2 months for untreated patients. Treatment significantly reduced the probability of cirrhosis development when compared with no treatment (log rank P=0.0029; Figure 2B). The probability that a patient treated for HCV would develop cirrhosis within 5 years from baseline was 4.3% (probability of remaining free of cirrhosis within 5 years: 0.957, 95% CI: 0.933, 0.981), the corresponding probability for an untreated patient was 11.7% (probability of remaining free of cirrhosis within 5 years: 0.883, 95% CI: 0.811, 0.956).
Univariate Cox regression analyses highlighted that de novo cirrhosis was significantly associated with no treatment, abnormal ALT, abnormal AST, age of least 40 years old, BMI greater than or equal to 28 kg/m2, abnormal GGT, being female, being infected with HCV GT1, platelet count of less than 100x10 9 /l, and APRI ≥1.5 and ≥2.0 (P<0.05; Table 2). Moreover, multivariate Cox regression analyses uncovered an association between de novo cirrhosis and a BMI of at least 28 kg/m2, being over 40 years, abnormal AST, being infected with HCV GT1, platelet count of less than 100x10 9 /l and APRI ≥2.0 (Table 2). Multivariate analysis did not highlight a significant risk associated with SVR24 status and cirrhosis (Table 2). Logistic regression analysis of the incidence rate of cirrhosis found an association with 17 of the assessed risk factors (P<0.05), with the strongest (P<0.0001) seen for baseline APRI of at least 1.5, baseline FIB-4 of at least 3.25, and a platelet count greater than 90x10 9 /l (Additional file 5).
Summary of Cox model analyses of the impact of risk factors on time from estimated infection to cirrhosis and time to HCC
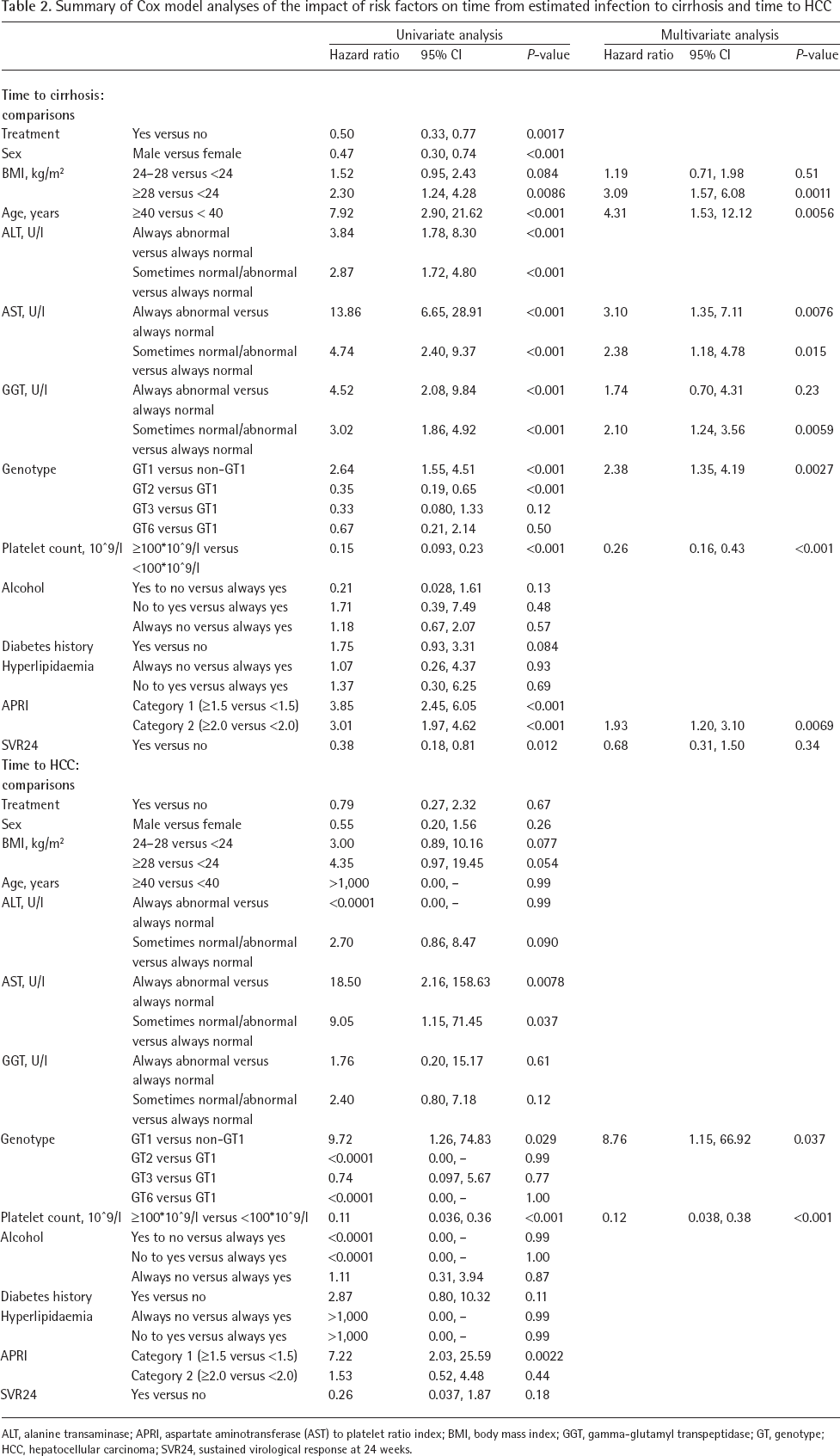
ALT, alanine transaminase; APRI, aspartate aminotransferase (AST) to platelet ratio index; BMI, body mass index; GGT, gamma-glutamyl transpeptidase; GT, genotype; HCC, hepatocellular carcinoma; SVR24, sustained virological response at 24 weeks.
HCC
In the FAS (none of whom had HCC at baseline), HCC developed post-baseline in 2.7% (10/364) treated patients and in 1.4% (2/145) untreated patients. The Kaplan-Meier estimate of the time to HCC from baseline was 16.9-86.6 months in treated patients and 17.9-86.4 months in untreated patients. There was no significant difference in the probability of HCC development within 5 years from baseline between treated and untreated patients (log rank P=0.43; Figure 2C). The probability for a treated patient of developing HCC was 1.4% (probability of remaining free of HCC within 5 years: 0.986, 95% CI 0.974, 0.998), and that for untreated patient was 0.8% (probability of remaining free of HCC within 5 years: 0.992, 95% CI 0.977, 1.007).
HCC developed post-baseline in 3.8% (1/26) patients with HCV GT3b and in 3.6% (11/303) patients with HCV GT1 but it did not occur in any patients with HCV GT2, GT6 or GT3a. Kaplan–Meier analysis of the time from the HCV infection to the development of HCC according to the HCV GT are shown in Figure 2D. Although there was no significant difference between the estimated year of HCV GT infection and probability of developing HCC (P=0.17), both univariate and multivariate Cox regression analysis highlighted that HCV GT1 increases the risk of HCC (univariate P=0.029; multivariate P=0.037; Table 2). Furthermore, HCC incidence was significantly associated with a platelet count less than 100x10 9 /l according to multivariate analysis (P<0.001; Table 2). Univariate analysis did not uncover a significant risk associated with SVR24 status and HCC (Table 2).
Discussion
Significantly fewer treated patients experienced overall disease progression over 5 years compared with untreated patients. The improved outcomes of the treated patients could be attributed to the younger age and higher mean ALT of this group at baseline compared with the untreated population. Among treated patients, as expected, patients who achieved an SVR were less likely to experience overall disease progression than those who did not. Previous studies have also demonstrated that SVR following treatment with IFN-based therapy significantly reduces the risk of disease complications in patients with HCV-related cirrhosis [28–31]. In these studies the risk of HCC was also reduced following SVR.
Our study did not demonstrate a significant reduction in cirrhosis or HCC risk for patients with SVR following treatment. A key reason why we did not uncover a link between the risk of cirrhosis or HCC with achieving SVR was due to the low number of disease progression events that occurred during the follow-up period. The small proportion of patients who developed cirrhosis or HCC during the study despite the modest sample size suggests that the 5-year follow-up time may not be long enough to determine if achieving SVR influences disease progression. Furthermore, this study observed the outcomes of patients who were on active anti-HCV treatment prior to enrolment or who commenced treatment during the study. Hence, an accurate comparison between the risk of cirrhosis or HCC and SVR24 status could only be conducted if participant baseline characteristics were recorded prior starting anti-HCV treatment and were matched in each group.
Most treated patients in the study received IFN-based therapy. It is uncertain that elimination of HCV infection with DAA therapy will be associated with the same long-term outcomes as elimination with IFN-based therapy; some studies have suggested increased rates of HCC occurrence and recurrence following DAA treatment [32,33]. However, evidence suggest that different outcomes following DAA versus IFN-based treatments, may be because of differences in the treated patient populations [34,35].
Consistent with previous observations [9–11,36], HCV genotype had an impact on the response to treatment and overall-disease-progression. SVR24 rates were higher among patients with HCV GT2 or HCV GT3 infection than among patients with HCV GT1b, which is typical of IFN-based treatments which are less effective against HCV GT1 compared with other subtypes (in this cohort treatments were 93.4% IFN-based and 6.6% DAA-based). Furthermore, both multivariate and univariate Cox regression analyses suggested a significant association between HCV GT1 and the likelihood of developing either cirrhosis or HCC. As we enter the era of DAA treatment, HCV GT1 will no longer be a treatment-refractory GT, whereas HCV GT3b, which was associated with the second highest overall-disease-progression rate in the current study, will be expected to be the most DAA-refractory GT [37].
Results from this follow-up study highlighted that fibrosis at baseline was associated with overall disease progression. Indeed, fibrosis has been described by others as associated with disease progression and HCC in both HCV- and HBV-infected individuals [38]. This association further implicates cirrhosis as a risk factor for more complicated liver disease and suggests that patients with markers of extensive fibrosis should be prioritized for treatment. An increased risk of cirrhosis progression with older age has also been reported elsewhere [36,39,40]. In a study on transfusion-related HCV infection, the time necessary to have a 50% probability of developing cirrhosis in patients aged 21–30, 31–40 and >40 years was 33, 23 and 16 years, respectively [40].
Evidence from our study has uncovered a statistically significant association between the use of concomitant medication and the increased odds of overall disease progression. Despite this strong statistical link, it is unclear whether the use of these drugs directly influences overall disease progression. In other words, the association of overall-disease-progression and the use of concomitant medication might be confounded by the comorbidities and complications experienced by this subset of patients. For instance, the use of hepatitis medications could suggest advanced liver disease in this population of patients, which in turn would increase the incidence of overall disease progression. Our study did not collect information on the types of complications experienced by patients who were using concomitant medication. Given the widespread prescription of additional medication in clinical practice and the high interaction potential of anti-HCV treatment regimens (DAAs), it is possible that the efficacy of HCV clearance and overall disease progression could be influenced by the use of such concomitant medication [41–44].
Limitations
While the methodology used to diagnose cirrhosis was consistent with Chinese clinical practice and guidelines, the lack of mandated biopsy confirmation of liver disease stage may have introduced some variation between sites. Furthermore, the five-year follow-up was not sufficient to clearly elucidate the impact of treatment (or other factors) on the development of HCC, longer-term follow-up is needed.
In conclusion, the impact of treatment on overall-disease-progression was dependent on the achievement of SVR, emphasizing the importance of effective anti-HCV treatment. Further, long-term studies are warranted to determine the impact of successful treatment with DAAs on HCV-related disease progression in China.
Footnotes
Acknowledgements
We would like to acknowledge Hong Li (currently at Global Health Economics Department, Amgen, Thousand Oaks, CA, USA, and previously at Global Health Economics and Outcomes Research Department, Bristol-Myers Squibb, Lawrenceville, NJ, USA at the time the study was conducted) for his contribution to the study design and CCgenos study investigators for their contributions to the study data acquisition, and the patients and their families for their support and dedication. Editorial assistance was provided by Esther Race (Oxford, United Kingdom) and Isuru Wijesoma (Meditech Media, Singapore), with funding from Bristol-Myers Squibb.
HR, RY, SZ, RL and L Wei were involved in the study concept and design; HR, QX, JS, ZG, HC, YS, JJ, JN, L Zhang, L Wang, L Zhao, JL, RY and L Wei were involved in the acquisition of the data; HR, QX, JS, ZG, HC, YS, JJ, JN, L Zhang, L Wang, L Zhao, JL, RY, SZ, RL and L Wei were involved in the analysis and interpretation of the data; HR, RL and L Wei were involved in the statistical analysis; HR, SZ, RL and L Wei were involved in the drafting of the manuscript; HR, QX, JS, ZG, HC, YS, JJ, JN, L Zhang, L Wang, L Zhao, JL, RY, SZ, RL and L Wei were involved in the critical revision of the manuscript. All authors agree to be accountable for all aspects of the work.
Clinical trials registration: NCT01594554 (BMS study ID: AI452-018ST).
This work was supported the China National Science and Technology Major Project for Infectious Diseases Control during the 12th and 13th Five-Year Plan Period under Grant 2017ZX10202202, 2012ZX10002003, 2012ZX10002005; Major Project of National Science and Technology Creation of Major New Drugs under Grant 2012ZX09303019, and Bristol-Myers Squibb.
HR has received speaking fees from Bristol-Myers Squibb, Gilead and AbbVie. QX, JS, ZG, HC, YS, JJ, JN, L Zhang, L Wang, L Zhao, JL and RY have nothing to disclose. SZ and RL are employees of BMS. L Wei has received grants from AbbVie, BMS and Roche to his institution; has personally provided consulting to AbbVie, Allegan, BMS, Gilead, JNJ, MSD, Roche, Pfizer; has been a speaker for Ascletis and Gilead.
Additional file 1: A list of factors included in the logistic regression analyses of the incidence of cirrhosis, and overall disease progression can be found at ![]()
Additional file 3: A table showing treatment efficacy (SVR24) by viral genotype subgroups can be found at ![]()
