Abstract
We report the first two paediatric cases of sofosbuvir treatment during high-intensity myeloablative conditioning and engraftment phases of haematopoietic stem cell transplantation. These reports highlight the safety of sofosbuvir during all phases of transplantation and the lack of interaction between sofosbuvir and alkylating or immunosuppressive agents.
Introduction
Active liver disease is a well-known major complication following haematopoietic stem cell transplantation (HSCT), with mortality rates ranging from 4 to 15% [1]. The risk is increased in patients with HCV infection: the rate of sinusoidal obstruction syndrome (SOS) is about 5x higher in HCV-positive than in HCV-negative patients [2].
Using direct-acting antivirals (DAAs) such as sofosbuvir is a recent practice for HCV-positive paediatric patients. DAAs offer a clear advantage in children who undergo HSCT and cannot be treated with interferon due to its adverse effects, such as myelosuppression and worsening of graft-versus-host disease (GVHD) [3,4].
The current approach is to treat HCV after HSCT to reduce the burden of the late complications, and the discovery of DAAs has greatly lowered the need to discontinue antiviral therapy, thus providing a higher rate of sustained virological response (SVR) [5]. However, using the treatment only after HSCT does not lower the risk of early complications, such as SOS or viral reactivation. Treating the infection during the conditioning therapy may lower the risk of these complications, but such an approach is not reported in the literature.
The current HCV treatment guidelines observe that ‘no recommendations can be made regarding overlap between the toxic effects of DAAs and the toxic effects of HSCT conditioning regimens or symptoms of GVHD because evidence is lacking’ [6].
We present two cases of paediatric patients with HCV infection who started treatment with DAAs before transplantation and continued the antiviral drugs during standard myeloablative conditioning for allogeneic HSCT. In the second case, the child underwent a double transplantation, first from a sibling donor and, after an early relapse, from a haploidentical donor (Figure 1).
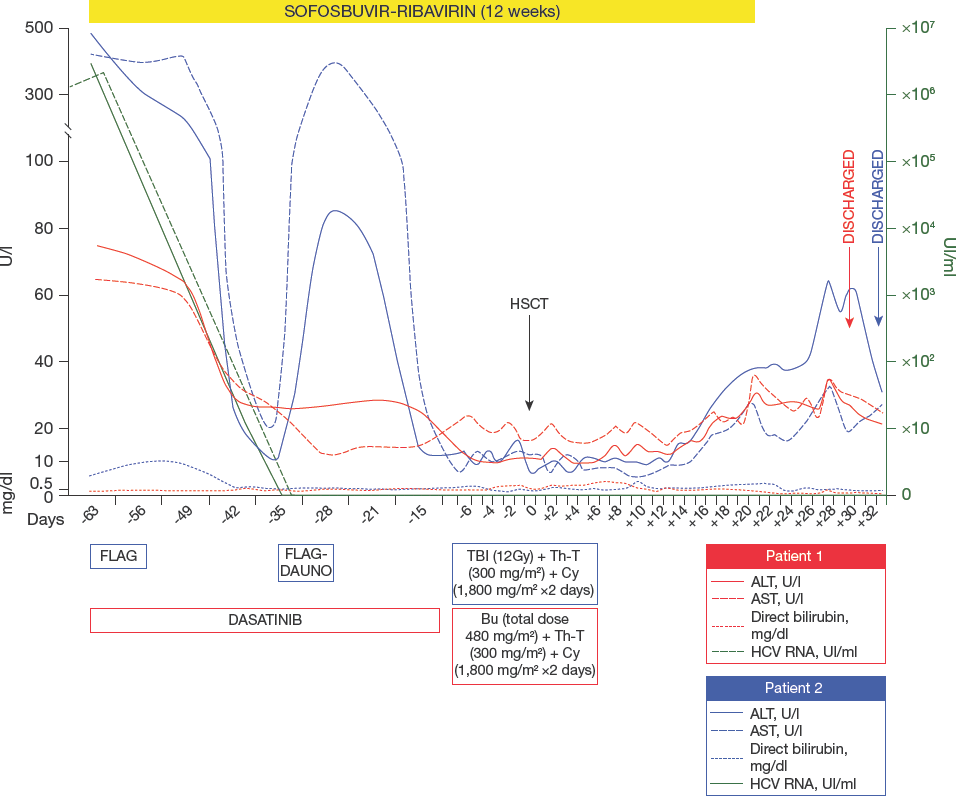
Two cases of integration of sofosbuvir-based antivirus therapy concurrently with allogeneic haematopoietic stem cell transplant-related therapy in the treatment course of HCV-positive paediatric leukaemic patients
The current post-transplant follow-up protocol in our center requires performing a liver biopsy in the case of persistent or progressive alteration, noted at 2-weekly follow-up assessments, of at least one clinical or laboratory marker of liver impairment or cholestasis, abnormal radiological findings, or one or more lesions suggestive of hepatic parenchymal infection [7,8]. Our standard practice of biopsy diagnostics has provided us the opportunity to document the histological features of hepatic parenchyma in all phases of treatment with DAAs.
Case Reports
The first patient, a 10-year-old girl, was admitted to our center with a diagnosis of chronic myeloid leukaemia (CML) positive for the Philadelphia chromosome with a high level of BCR-ABL transcript. She had experienced a good response to the targeted therapy with the ABL tyrosine kinase inhibitor (dasatinib) 100 mg/day. The girl was also infected by hepatitis C in chronic phase. At admission HCV RNA viral load was 2x10 6 UI/ml, genotype-2a/c. Liver biopsy confirmed the inflammatory infiltration of lymphomonocytes with some areas of lobular necrosis, compatible with a chronic HCV hepatitis with a hepatitis activity index (HAI) of 5/18 and Ishak stage 1 (minimal portal fibro-sis; Figure 2A).

Histological samples of liver biopsy before and after sofosbuvir treatment in two patients concurrently receiving allogenic haematopoietic stem cell transplantation
The girl was started on a therapeutic treatment of sofosbuvir (200 mg/day) and ribavirin (12 mg/kg/day in two administrations), in addition to dasatinib. 4 weeks later HCV RNA was undetectable in the blood and we could begin standard myeloablative busulfan-based conditioning. The therapeutic drug monitoring (TDM) of sofosbuvir metabolite GS331007 and ribavirin was performed at the end of the first week of DAA treatment and on the third day of busulfan conditioning. Moreover, the area under the plasma drug concentration–time curve (AUC) of busulfan was calculated after the first dose. All plasma drugs and metabolite concentrations were within the normal range. The post-transplant period was uneventful with rapid engraftment. Liver function tests (LFTs) remained unaffected from the first day of conditioning to the discharge from our unit.
No signs of GVHD were recorded with tacrolimus used as prophylaxis.
The girl continued the antiviral therapy for the scheduled 12 weeks. Subsequent re-evaluation of HCV RNA was negative, and a liver biopsy showed no signs of inflammation, fibrosis or cirrhosis (Figure 2B).
Two years following HSCT the girl is in remission from CML with HCV eradicated.
The second patient, a 5-year-old girl, came to our center to undergo HSCT for acute lymphoblastic leukaemia (ALL). She was infected by HCV, genotype-1b, acquired during previous chemotherapy. At admission HCV RNA viral load was 2.4x10 6 UI/ml. During the high-risk induction cycle she suffered a severe chemotherapy-induced hepatic injury, which was treated with defibrotide. The treatment successfully led to the remission of the clinical symptoms of SOS, but even afterward the level of aspartate transaminase (AST) remained 10x above normal range. For diagnostic evaluation we performed a liver biopsy (Figure 2C), which confirmed active HCV hepatitis with an HAI of 8/18 and Ishak stage 2 fibrosis.
A treatment for HCV hepatitis with sofosbuvir (200 mg/day) and ribavirin (14 mg/kg/day in two administrations) was started, and our tests after 4 weeks of treatment detected no HCV RNA in the blood and a normalization of LFTs. After a standard myeloablative total body irradiation-based conditioning, the girl underwent allogeneic HSCT. She continued the DAA therapy without interruption during myeloablative conditioning and the first month post-HSCT for a total of 24 weeks. Defibrotide was used for SOS prophylaxis. The post-transplant phase was uneventful with rapid engraftment, LFTs stably normal and HCV RNA negative. No signs of GVHD were recorded with tacrolimus used as prophylaxis.
On day +40 the girl had an early relapse of ALL. After a new cycle of rescue chemotherapy she underwent a second allogeneic HSCT from her haploidentical father after busulfan-based myeloablative conditioning. GVHD prophylaxis was done with post-transplant cyclophosphamide, tacrolimus and mycophenolate mofetil. Neither liver toxicity, nor HCV relapse nor other complications were observed. Liver biopsy performed 1 month after the second transplant showed no signs of inflammation (Figure 2D).
The child achieved HCV eradication but, unfortunately, died 1 year later due to leukaemic relapse.
Discussion
A knowledge gap still exists about the correct timing of antiviral therapy in HSCT paediatric recipients. Using chemotherapy in an HCV-positive patient could induce a faster development of hepatic fibrosis and cirrhosis in later years. Haematopoietic stem cell transplantation could offer a better prognosis in patients with malignant and myelodysplastic haematological disorders but could also result in a high incidence of liver complications if there is a concurrent active HCV infection. Follow-up studies show that the median time to cirrhosis in transplant recipients was 18 years as compared with 40 years in the control population of infected HCV patients.
We knew that starting the treatment of HCV infection after HSCT leads to a high rate of sustained viral response (SVR) that protects the patients from an earlier presentation of cirrhosis and liver fibrosis. Furthermore, treating the infection could also prevent relapse of HCV after the transplantation [9].
Using interferon-ribavirin treatment however is contraindicated after the HSCT in almost 60–70% of the patients because of GVHD or cytopenia and, in those treated, leads to a SVR in only 50% [4,10].
Unlike earlier treatments, sofosbuvir had no contraindications related to cytopenia and GVHD and could be used to treat HCV in HSCT recipients almost always with good tolerance. Kyvernitakis et al. [5] reported that in adult patients receiving an HSCT, using DAAs is a safe and effective option, achieving an SVR after 12 weeks from the treatment. However, there are some knowledge gaps about the correct timing of use of DAAs in HSCT recipients [6], and in their study all the patients were treated only after HSCT, exposing patients to a higher risk of early liver complication of HSCT such as SOS.
The role of HCV in developing SOS is debated in the current literature but evidence from the prospective study by Strasser et al. [11] suggests that HSCT patients with an active HCV disease with elevation of serum aspartate transaminase AST are at very increased risk (almost 10x higher). Due to the high mortality of this complication, almost 20–30% with defibrotide treatment, we think that this risk could not be underestimated. Thomas et al. [12] described a case of a 4-year-old girl who underwent HSCT for a relapsed LLA and who started sofosbuvir on day +88 due to an acute HCV hepatitis. It's interesting to note that the girl developed a SOS on day +20 that maybe could have been prevented by a pre-emptive treatment with sofosbuvir. In our second described case the patient had an SOS during the rescue chemotherapy to induce remission before HSCT and we observed an active HCV disease with persistent elevation of AST. To prevent a new flare-up of SOS during conditioning for HSCT we started HCV treatment and, after achieving HCV SVR after 4 weeks of sofosbuvir/ribavirin, he did not suffer from hepatic complications after two different myeloablative regimens for the two HSCTs. The drugs were tolerated very well. Blood levels of the drugs remained in range during conditioning as well. This finding may demonstrate that chemotherapy has no interaction with sofosbuvir and ribavirin clearance and metabolism. Suarez Benjumea et al. [13] describe using higher doses of tacrolimus during the sofosbuvir treatment due to an apparent significant tendency of sofosbuvir to reduce the levels of that immunosuppressive drug. This effect could be caused by the recovery of hepatic function rather than a direct interaction between tacrolimus and sofosbuvir [14]. In our cases we could adjust the effi-cacy of the immunosuppressive treatment using daily tacrolimus TDM without loss of efficacy.
The value of our cases is that they are the first reported paediatric patients treated with sofosbuvir started before undergoing HSCT. The patients showed an SVR at 4 and 12 weeks, a histological response on liver biopsy and had no precocious liver complication after HSCT. Weaknesses are the short follow-up for long-term complications in the two cases and using a combined therapy of sofosbuvir and ribavirin instead of sofosbuvir in combination with other DAAs or newer effective alternatives as stated by EASL 2018 guidelines [15]. We chose to not use ledipasvir because of the unknown interactions between the two DAAs and conditioning drugs, calculating that using only one DAA would make it simpler to measure possible side effects or interactions with other drug blood levels. Furthermore, accessibility of ledipasvir for children in Italy was limited at that time. SVR and the hepatic biopsy confirm the efficacy of the treatment with only ribavirin and sofosbuvir. This association may be currently the most efficacious therapy, keeping in mind potential drug–drug interactions.
Conclusion
The aim of the description of our cases is to show the feasibility of the sofosbuvir treatment during myeloablative and engraftment phases of HSCT. Efficacy of sofosbuvir was proven by the HCV clearance at week 4, by HCV RNA negativity at week 12 and week 24, by histological healing of the hepatic inflammation and by virological surveillance for at least 1 year. The drug was well tolerated without haematological toxicity, as demonstrated by a normal myeloid engraftment. We also demonstrated the lack of interaction between sofosbuvir and myeloablative drugs by performing AUC of busulfan and TDM of sofosbuvir. A larger cohort is needed to confirm the safety of sofosbuvir in paediatric HSCT recipients and to study the combination of sofosbuvir with other DAAs in this particular setting.
Footnotes
Acknowledgements
The funding agency had no role in the design or execution of this research.
The Sponsor did not receive any outside funding in support of this study.
Informed consent for publication was obtained.
There is no commercial relationship that might pose a conflict of interest in connection with the manuscript for all authors.
