Abstract
Background
Methods
Conclusions
Introduction
Bovine herpes viruses type 5 (BoHV-5) is an enveloped double-stranded DNA virus, belonging to the genus Varicellovirus, subfamily Alphaherpesvirinae, family Herpesviridae [1,2]. These considerations lead us to suggest it can be the aetiologic agent of neurological disease, associated to fatal meningoencephalitis in cattle, which compromises their productivity and makes the slaughter for meat consumption impossible [3,4]. This viral infection has been reported in Australia [5], United States [6], Europe [7], Argentina [8] and Brazil [9,10], affecting mainly but not exclusively young animals. BoHV-5 has been identified from respiratory [11] and genital tracts [12], and the literature describes the presence of BoHV-5 in cryopreserved semen [12,13], after experimental infection in oocytes, embryos and also within spermatozoa [14,15].
Oxoquinolines are among the most common bio-active molecules that have dominated the market for more than four decades. Oxoquinoline derivatives are widely used as antibacterial agents since 1962 [16]. They have a broad and potent spectrum of activity and are also used as second-line drugs to treat tuberculosis [17]. Over the years, oxoquinolines have been reported to display ‘nonclassical’ biological activities [18], such as antitumour [19], anti-HIV-I [20] and anti-HSV-I [21].
As a part of our ongoing research, in this work we evaluated the cytotoxic effect and antiviral activity of a new series of 4-oxoquinoline derivatives (4a-k) and determined the mechanism of action of these compounds on BoHV-5RJ42/01 replication.
Methods
Compounds
The compounds tested in the present study (4a-k) were previously synthesized through the Gould-Jacobs methodology [22–25], using a three-step procedure that involved the condensation of anilines 1a-k with diethyl ethoxymethylenemalonate (DEEMM) in refluxing ethanol followed by thermal cyclization of the aniline acrylate intermediates 2a-k, as shown in the Figure 1. The nucleophilic substitution reaction between oxoquinolines 3a-k and the benzylamine in diphenyl ether, according to our previous works [19,20] afforded the respective 4-oxoquinolines-3-carboxamides 4a-k (Table 1) in 98–55% yields. The compounds 4a-c and 4f were previously described in the literature [20,26,27]. The structures of the new compounds (4d, 4e and 4g-k) were characterized by IR, NMR and mass spectroscopy. Experimental procedures and analytical data are available in the Additional file 1.
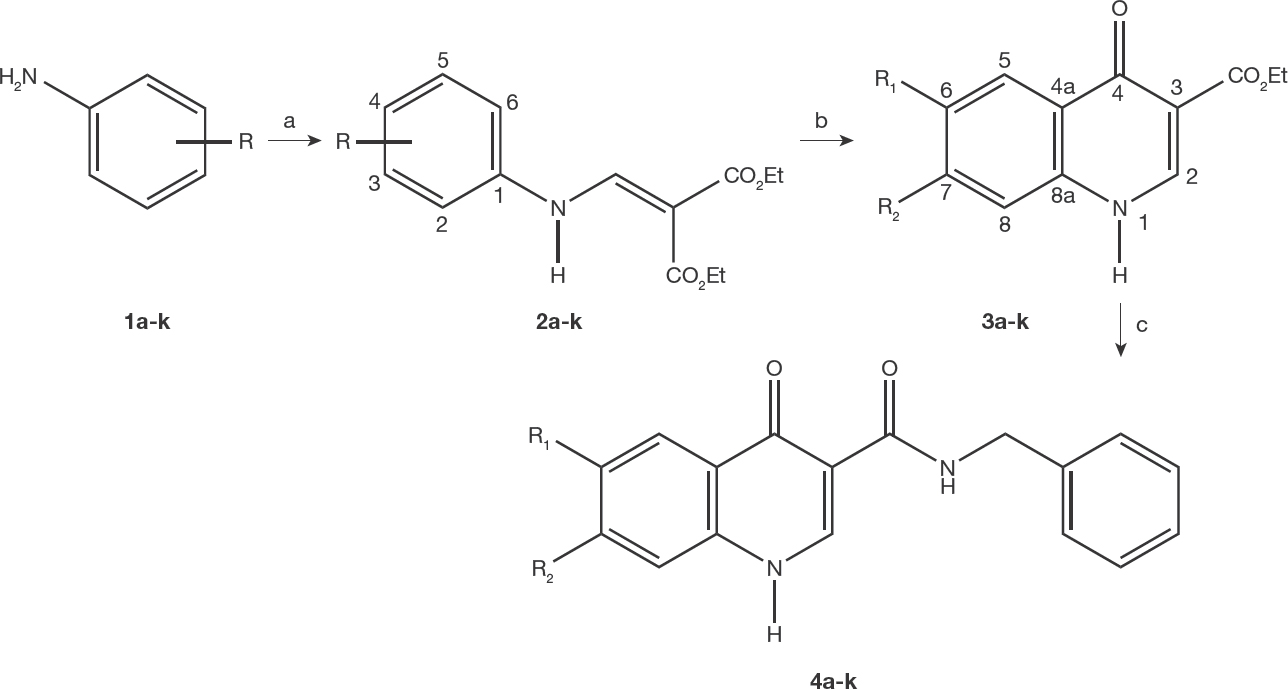
Synthesis of 4-oxoquinolines-3-carboxamides 4a-k
Yields of the reactions and melting points of 4-oxoquinolines-3-carboxamides 4a-k

Standard compound acyclovir (ACV) was purchased from Sigma-Aldrich Co. (St Louis, MO, USA). All compounds 4a-k previously synthesized were dissolved in dimethylsulfoxide (DMSO) purchased from Sigma Company, to obtain stock solutions at 50 mM and then diluted in culture medium without serum to obtain the test compound concentration before use. The final concentration of DMSO was <0.1%.
Cells
Madin–Darby bovine kidney (MDBK) cell line, originally obtained from ATCC (CCL-22) were grown and maintained in Eagle's Minimum Essential Medium (EMEM) containing 292 μg/ml of L-glutamine, 50 μg/ml of gentamicin and supplemented with 2–5% heated inactivated certified Fetal Calf Serum (FCS). Overlay medium for plaque assay consisted of EMEM 2x plus in agarose 2% (1:1) as previously described by Pinto et al. [28].
Virus
Bovine herpesvirus 5 (BoHV-5RJ42/01) was kindly provided by the Virology Section of Laboratório de Virologia Comparada, Fundação Oswaldo Cruz. Virus stocks were propagated in MDBK cells, and the virus titre was determined by plaque reduction assay (PA) [29]. Virus concentration was expressed as plaque-forming units (PFU) per ml.
Cytotoxicity of oxoquinolines 4a-k in MDBK cell line
Oxoquinoline 4a-k cytotoxic effect was assessed by monitoring the effect of compounds on MDBK cells by the conversion of 3-(4,5-dimethylthiazol-2yl)-2,5-diphenyltetrazolium bromide (MTT; Sigma-Aldrich) to formazan as previously described [30] with some modifications [31]. MDBK cells at a density of 3x10 3 were grown in 96-well microplates at 37°C under a 5% CO2 humidified atmosphere. After 24 h, MDBK cells were treated with different concentrations of compounds 4a-k and ACV (25, 50, 250, 500 and 1,000 μM) in triplicate. MDBK cells untreated were used as control. After 72 h of incubation the supernatant was removed and MTT (100 μl of 1 mg ml−1 in EMEM) was added to each well of the plates. Plates were re-incubated at 37°C with a 5% CO2 atmosphere for 4 h. The medium was discarded and DMSO (100 μl) added before a further incubation of 30 min at 37°C. Optical density was measured at 520 nm microplate reader and the result was expressed as the 50% cytotoxic concentration (CC50). The compound concentration required to reduce the optical density of MTT related to the cell control was calculated by a linear regression analysis.
Screening for antiviral activity of oxoquinolines 4a-k by plaque reduction assay
A primary antiviral screening was performed in MDBK cell monolayers grown in 24 well microplates inoculated with 200 PFU of BoHV-5RJ42/01 as previously described [29] with some modifications [28]. After 1-h of virus adsorption period, the inoculum was discarded, and the overlay medium containing different concentrations of oxoquinolines 4a-k or ACV (6.26, 12.5, 25 and 50 μM) was added. ACV was assayed at two additional concentrations (200 and 664 μM). After the cells were reincubated for 72 h at 37°C with a 5% CO2 atmosphere then cell monolayer was fixed and stained with 1% crystal violet in 10% formalin. The antiviral activity (EC50) was defined as the concentration required to reduce the plaque number by 50% of the untreated control and was calculated as follows:
percent of inhibition (%) = total number of control plaques - total number of tested plaques x 100/total number of control plaques.
Virus inactivation assay
The direct interaction and inactivation of BoHV-5RJ42/01 by the compounds tested were performed as previously described [28] with some modifications [31]. BoHV-5 RJ42/01 suspension containing 2x10 7 PFU ml- 1 was mixed with 25, 50 and 100 μM of the test oxoquinolines 4a-k or ACV and kept at 4°C and room temperature (24°C) for 3 h; a control of untreated infected cells was performed under the same conditions. Then the residual infectivity was determined by a limiting dilution assay as previously described [29].
Time-of-addition studies on viral replication
The effect of the oxoquinolines 4a-k on viral replication was evaluated at various time intervals up to 72 h, in accordance with procedures previously described [32] modified by Pinto et al. [28]. MDBK cells were grown in 24-well culture plates at a density of 2x10 4 cells per well. After 24 h MDBK monolayers were pretreated with oxoquinolines (4h = 25 μM; 4j = 25 μM; 4k = 25 μM) and four times the EC50 value of ACV = 664 μM for 1 h. MDBK cells without pretreatment were mock-infected or inoculated with BoHV-5RJ42/01 (200 PFU) and incubated for 1 h at 37°C with a 5% CO2 atmosphere for virus adsorption. The oxoquinoline derivatives were evaluated at different time intervals, as summarized in protocol (Figure 2).

Protocol for the evaluation of the time response assay used to investigate the mechanism of inhibition of the infection of BoHV-5 RJ42/01 at various time periods up to 72 h
Statistical analysis
All the data shown represent the mean ±
Results
Cytotoxicity and antiviral activity of compounds 4a-k on MDBK cells
Table 2 shows different cytotoxicity degrees of the oxoquinolines 4a-k on MDBK cells. The cytotoxicity was related to the nature of the substituents at C-6 or C-7 position (R1 or R2 radicals), respectively. The cytotoxicity results of compound 4a (CC50 = 985 ±3.7 μM) and of derivative 4i containing methyl substituent (R1; CC50 = 995 ±3.9 μM) were similar to that of ACV (CC50 = 989 ±2.0 μM).
Cytotoxicity of 4-oxoquinolines-3-carboxamide derivatives on MDBK cells, anti-BoHV-5RJ42/01 activity and selectivity indexes
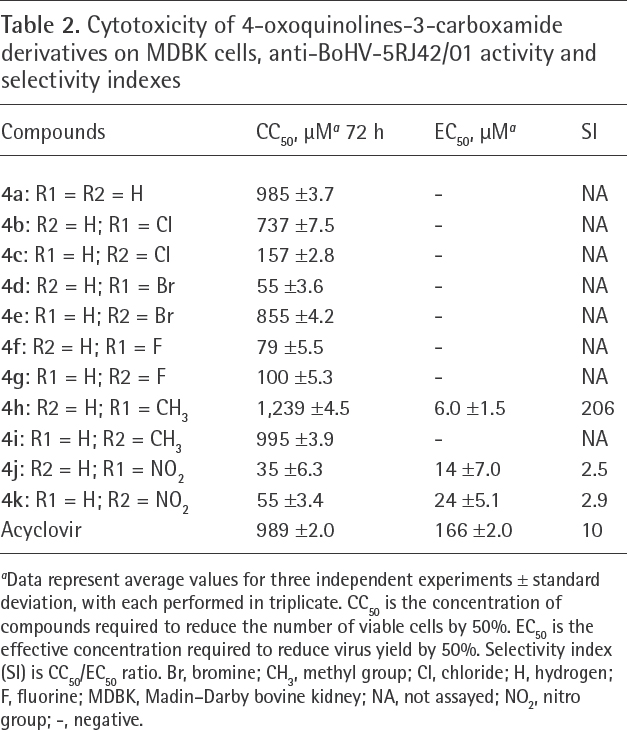
Data represent average values for three independent experiments ± standard deviation, with each performed in triplicate. CC50 is the concentration of compounds required to reduce the number of viable cells by 50%. EC50 is the effective concentration required to reduce virus yield by 50%. Selectivity index (SI) is CC50/EC50 ratio. Br, bromine; CH3, methyl group; Cl, chloride; H, hydrogen; F, fluorine; MDBK, Madin–Darby bovine kidney; NA, not assayed; NO2, nitro group; -, negative.
Halogenated oxoquinolines (4b-g) were shown to be cytotoxic, except 4e, and did not inhibit BoHV-5 production.
Nitro-oxoquinolines 4j (CC50 = 35 ±6.3 μM) or 4k (CC50 = 55 ±3.4 μM) showed high cytotoxic effects, but despite this fact, it is important to highlight that they inhibited BoHV-5 production (4j = EC50 = 14 ±7.0 μM; 4k = EC50 = 24 ±5.1 μM) at concentrations below their CC50 (SI = 2.5 and 2.9, respectively).
Table 2 also illustrates that the presence of a methyl group at C-6 or C-7 position resulted in less toxic oxoquinoline derivatives (4h and 4i), 4h being the less toxic one, with CC50 = 1,239 ±4.5 μM, EC50 = 6±1.5 μM and expressive SI value (SI = 206).
Inactivation of the BoHV-5 by 4-oxoquinoline-3-carboxamide and derivatives
The 4-oxoquinolines-3-carboxamides 4a-k showed slightly effects on the BoHV-5RJ42/01 infectivity assayed at 4°C and 37°C, for 4 h. The best results were obtained from compounds 4h at 37°C (25 μM = 10%, 50 μM = 16% and 100 μM = 20%); 4j (25 μM = 11%, 50 μM = 14% and 100 μM = 18%) and 4k (25 μM = 5%, 50 μM = 7% and 100 μM = 9%; Figure 3).

Free BoHV-5RJ42/01 inactivation by 4h, 4j, 4k derivatives and acyclovir
All compounds (4a-k) tested were not able to inhibit virus attachment and penetration on MBDK cells (data not shown).
Time-of-addition studies
Results of time-of-addition studies (Figure 4) showed BoHV-5RJ42/01 yield was inhibited by 4-oxoquinolines 4j and 4k more than 80% in all periods of kinetic studied. Figure 4 shows that although compound 4h was not able to inhibit viral expression during the first 2 h post-infection (pi) in MDBK cells, it was able to block BoHV-5 production in above 80% after 3 h pi.
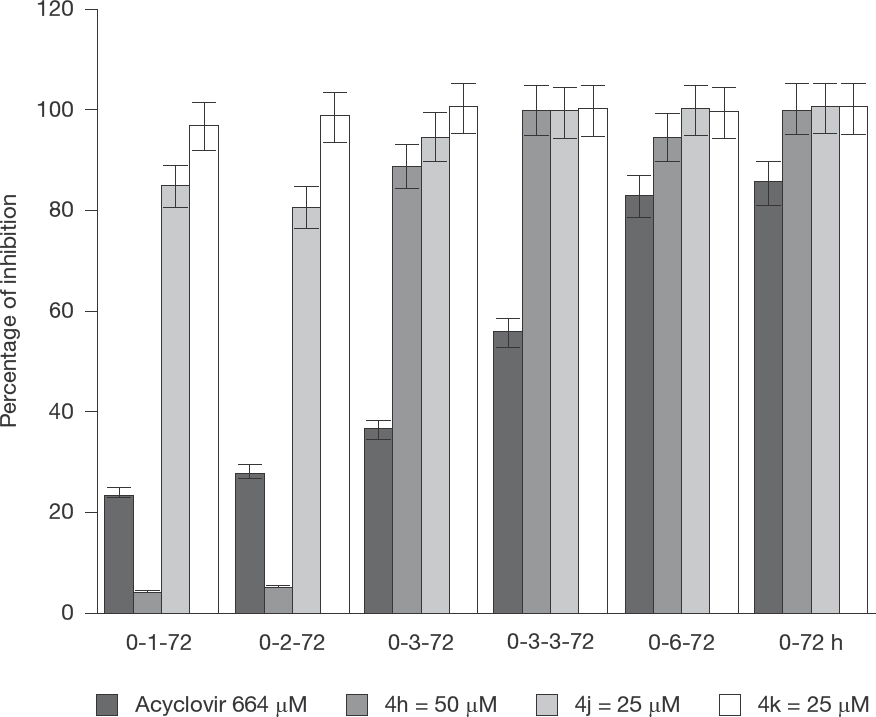
Results represent the reduction of BoHV-5 yield during viral multiplication kinetic analysis by treatment with 4-oxoquinolone-3-carboxamide derivatives 4h, 4j, 4k using ACV as positive control on MDBK cells infected with BoHV-5 after 1, 3 and 6 hpi
Discussion
The search for antiviral agents is still urgent in medical sciences. Antiviral compounds have been successfully applied in the chemotherapy for the treatment of several diseases caused by viral agents, such as the HIV, influenza A virus, HCV and human herpes simplex viruses (HSV). In veterinary medicine the vaccination is a widespread method in viral infection prevention. Slaughtering of livestock is also used to prevent and combat epizootic infectious diseases. By contrast, research aimed at the production of antiviral drugs for application in veterinary medicine therapy is scarce [28,33].
4-oxoquinoline derivatives represent an important family of compounds associated with a great versatility regarding biological activities. Substances containing the 4-oxoquinoline scaffold have been used as antibacterial agents for decades, but have also displayed potential as antimalarial, anti-toxoplasmosis, anti-leishmania, antifungal, anticancer and antiviral agents. Regarding this last bioactive profile, several examples can be found in the literature, such as: anti-HIV, anti-influenza A and B virus, anti-HCV, anti-porcine reproductive and respiratory syndrome virus, anti-human cytomegalovirus, anti-varicella zoster and anti-HSV derivatives [34–40].
In veterinary medicine, antiviral research has been already described for the treatment of herpesvirus. Nevertheless, the literature in this line of study is still scarce and limited to pet animals. Considering this, the antiviral research for application in livestock must be investigated [28,41]. In this work eleven oxoquinoline derivatives were evaluated for their cytotoxicity and antiviral activities against BoHV-5RJ42/01 on MDBK cells. The structure–activity relationships showed that the cytotoxicity of compounds was highly dependent on the nature of the substituents attached to C-6 (R1) or C-7 (R2) of the oxoquinoline core. The cytotoxicity results of compound 4a and of derivative 4i containing methyl substituent (R1) were similar to that of ACV but only derivative 4h showed clear inhibitory effects on BoHV-5 yield. All oxoquinoline derivatives 4a-k slightly interact and inactivate free BoHV-5RJ42/01 particles.
Results of time-addition studies showed reduction in the replication of BoHV-5 RJ42/01 around 90% during the first two hpi by compounds 4j and 4k (25 μM). In these periods herpesvirus a gene or immediate early herpesviruses genes expression begins. Compound 4h failed to block BoHV-5RJ42/01 replication during the first two hpi.
Expressive antiviral activities of oxoquinoline derivatives 4h, 4j and 4k were observed at 3 h pi (Figure 4). At this time begins the expression of early herpesvirus genes, important for the β proteins synthesis that are required for replication of viral genome. Herpesvirus genome is detectable at 3 hpi and reaches peak 5–7 hpi [42]. Temporal herpesvirus expression ends with expression of γ genes. All three compounds (4h, 4j and 4k) inhibited BoHV-5RJ42/01 around 100% after 6 h pi period corresponding to the synthesis of structural proteins, assembly and virion release [41]. In conclusion, antiviral activity and SI values (Table 2) suggests that oxoquinoline 4h is a promising and safe compound because it inhibited viral replication without promoting cytotoxic effect on MDBK cells. Kinect studies reveal that antiviral activity of 4h, 4j and 4k is related with intracellular events, affecting important steps of BoHV-5 replicative cycle since these compounds did not inhibit BoHV-5 infectivity.
The selectivity index of compounds 4h (SI=206) is higher than those of derivatives 4j, 4k and standard compound ACV. Therefore oxoquinoline 4h was consider safer and the most promising compound assayed in this study. However, further analysis is required to better understand the mechanism of action of these compounds against BoHV-5.
Footnotes
Acknowledgements
The authors thank the Brazilian agencies ‘Conselho Nacional de Desenvolvimento Científico e Tecnológico’ (CNPq), ‘Coordenação de Aperfeiçoamento de Pessoal de Nível Superior’ (CAPES) and ‘Fundação de Amparo à Pesquisa do Estado do Rio de Janeiro’ (FAPERJ), as well as FOPESQ-UFF and FIOCRUZ for financial support.
The authors declare that they have no competing interests.
