Abstract
Background
Influenza virus infections in lung transplant recipients (LTRs) have an increased risk of unfavourable outcomes. Early initiation of treatment is associated with improved outcomes. In clinical practice, empirical oseltamivir treatment is therefore commonly started prior to diagnostic microbiological confirmation. There is limited data on the patient characteristics, outcomes and safety of this practice. This study investigated outcomes and safety of this pre-emptive treatment strategy using oseltamivir.
Methods
Descriptive analysis of LTRs who received oseltamivir for ≥2 days for suspected influenza infection between July 2011 and June 2012. Analyses were based on data from electronic medical records and our standardized LTR database with prospective documentation of clinical information including medication, laboratory and radiological results, outcomes and adverse events.
Results
We included 133 patients with a total of 261 oseltamivir treatment episodes (87.4% as outpatients). Median duration of oseltamivir treatment was 4 days (range 2 to 67) and 98.5% had concomitant antibiotic pharmacotherapy. Indications for oseltamivir included acute respiratory infection (66.7%), non-distinctive inflammatory reaction (51.3%) and influenza-like illness (2.7%). Influenza virus infection was confirmed by PCR in only 7%. Rhinovirus was the most frequent pathogen detected (14.9%). We discovered a wide range of adverse events but none occurred in >5%, and most were mild and of questionable causal relationship to oseltamivir administration.
Conclusions
This non-controlled retrospective analysis suggests that the pre-emptive use of oseltamivir for respiratory tract infections pending microbiological results is safe in LTRs.
Introduction
The incidence of viral infections in lung transplant recipients (LTRs) is high [1]. Influenza is associated with significant morbidity and mortality in LTRs [1–5]. Seasonal influenza may be responsible for up to 10% of upper respiratory tract infections in LTRs and progression to lower respiratory tract infection is common, with viral pneumonia reported in 4% [1,6,7]. Complications of influenza may include bacterial pneumonia [6,8] and acute [2,5,6,8] or chronic allograft dysfunction most frequently presenting as obliterative bronchitis or bronchiolitis obliterans syndrome [3,4,6,8]. Hospitalization is more frequently required in transplant patients because immunosuppression predisposes these patients to complications of influenza infection [1,3,4,9,10]. Mortality secondary to influenza is observed in up to 21% of infected LTRs [4].
Antiviral prophylaxis may be protective against influenza infection in LTRs, and influenza vaccination is recommended for all immunocompromised individuals [2,11–13]. Studies supporting this practice are scarce [8,13], and impaired serological responses have been reported. Nevertheless, vaccination is generally considered protective or may attenuate the course of the infection [14–19]. Frequently pre-emptive treatment with oral oseltamivir is commenced prior to confirmation of influenza infection by polymerase chain reaction (PCR) from nasopharyngeal swab (NPS) samples, as early treatment appears to be more effective in reducing complications of influenza infection [2,8,13,14]. Oseltamivir is generally well tolerated in immunocompetent patients [20,21]. However, according to the package insert of the manufacturer hepatitis and abnormal liver function tests have been described [22]. In combination with other hepatotoxic medications such as itraconazole LTRs may experience such adverse events (AEs). Oseltamivir also has a low potential for clinically relevant pharmacokinetic interactions with co-administered medications [10,23]. In particular, there are no known pharmacokinetic interactions with commonly used immunosuppressive drugs [10].
Several studies have evaluated the safety of oseltamivir in healthy subjects and in solid organ and bone marrow transplant recipients [10,20,24–28]. However, such studies are scarce for LTRs [5,8]. The main objective of this study is to evaluate the safety of oseltamivir use in suspected and proven influenza infection in LTRs. Therefore, we retrospectively evaluated safety and outcomes data of oseltamivir treatments during a 1-year period in the LTR cohort at the University Hospital of Zurich.
Methods
Study design
This retrospective study was conducted to investigate safety, that is, frequency of AEs associated with oseltamivir treatment of lung transplant recipients managed by the physicians of the Division of Pulmonology at the University Hospital of Zurich between July 2011 and June 2012. The patients were questioned and examined in this 1-year period by one of four respiratory care physicians at the Zurich Lung Transplantation Program. The study was approved by the local institutional review board (KEK-No. 2016-00728).
Patient data
Consecutive data of LTRs who received either once or twice daily 75 mg of oseltamivir (depending on renal function) for at least 2 days were analysed during the treatment and the following 3 days, since the half-life of the drug's active metabolite is reported to be between 6 and 10 h [29]. The first dose in outpatients was typically administered in the hospital's outpatient clinic to avoid delay of treatment initiation, and patients were instructed to continue their treatment until they received further instructions which were based on laboratory findings, in particular, viral and bacteriological results [30]. Patients were excluded from this analysis if they had a treatment episode of less than 2 days or another antiviral treatment against influenza infection. Patient and treatment data were extracted retrospectively from the ongoing patient database. Follow-up data included oseltamivir treatment indication, duration, dosage and treatment location (outpatient or hospitalized), start or continuation of additional antibiotic therapy, and the final diagnosis based on all available information.
Additionally, hospitalized patients treated with oseltamivir were reviewed during treatment and 14 days post-treatment to analyse AEs as documented in hospital records and the evolution of laboratory findings. Documented and analysed information included duration of hospitalization and of treatment with oseltamivir, and the reason for admission.
Symptoms suggesting respiratory infection due to influenza virus were compiled [4,8,31–35]. The indication for treatment was determined according to the following categories: flu-like illness, non-specific respiratory symptoms, non-distinctive inflammatory reaction and other symptoms. Flu-like illness was defined as fever ≥38°C associated with cough. Non-specific respiratory symptoms were categorized as present, when either cough, sore throat, shortness of breath or coryza were noted and the involved physician considered it to be signs of an infection [36]. An elevation of the C-reactive protein (CRP) blood level was considered a non-distinctive inflammatory reaction. The list of AEs systematically searched for in the records was established based on previous studies [10,26,37–40]. Vaccination records were evaluated. According to our institutional policy the minimal time interval between transplantation and vaccination is 3 months.
Laboratory assessments and signs
Oseltamivir treatment was continued until either influenza virus was not detected by PCR from NPS samples from the first sampling, or in case of laboratory confirmed influenza infection (LCI) when two consecutive negative results were obtained, which has been standard practice for LTR in our cohort since 2009. This practice takes into account the frequently observed prolonged viral shedding and possibly also longer infectivity of LTRs with LCI due to triple immunosuppression. However, extended detection of influenza by PCR may represent non-viable virus, thus making the extended course of oseltamivir unnecessary [13]. Routine sampling from NPS for suspected respiratory infection includes a multiplex PCR for a panel of viruses (adenovirus, bocavirus, coronavirus, enterovirus, influenza A and B virus, metapneumovirus, parainfluenza virus, parechovirus, rhinovirus, respiratory syncytial virus) and bacterial analysis for gram-stains and culture [13]. In this influenza season the result became available mostly 18–48 h after sampling. The rapid PCR test providing results within 2 h or less was only introduced 4 years later [41]. Following a positive result serial swabs are obtained approximately every 7–10 days depending on the acuteness of disease at clinical presentation and in subsequent examinations as a part of our institutional policy. CRP blood level, imaging results from chest radiography or chest computed tomography scans (if available), body temperature, blood pressure and lung function results are usually considered when the decision is made to prescribe oseltamivir and determine the follow-up interval, whereby our internal guidelines recommend initial follow-up examinations at weekly intervals [13,42]. Follow-up assessments routinely included liver transaminases, creatinine, CRP, differential white blood counts. Patients with elevated transaminases, in particular, aspartate aminotransaminase (ASAT) increases to double the upper threshold value of 50 U/l were analysed in detail including pre-treatment and post-treatment values up until 14 days after treatment discontinuation. To assess the likelihood of drug-related AEs the Naranjo adverse drug reaction probability scale was used in all these cases [43]. A Naranjo score of 5/10 points is considered as a probable adverse drug reaction (ADR), a score of ≥9/10 points as a definite ADR. For the assessment of laboratory findings and AE severity the Common Terminology Criteria for Adverse Events was used [44].
Statistical analyses and graphical presentation
Patient characteristics were summarized in tables and graphs as appropriate, and we performed univariate analyses. We used Stata 14 (StataCorp LP, College Station, TX, USA) used for statistical analyses, and the scientific 2D graphing and statistics software GraphPad Prism (Version 5, 2014; GraphPad Software, San Diego, CA, USA) for graphs (Additional file 1).
Results
Baseline demographics and microbiological findings
A total of 133 (56.8%) out of 234 LTRs received oseltamivir for at least 2 days in the 1-year study period for 263 suspected respiratory tract infection episodes. Two treatment episodes had to be excluded from analyses due to insufficient safety information. Patient characteristics are presented in Table 1.
Patient characteristics for individual patients and oseltamivir treatment episodes
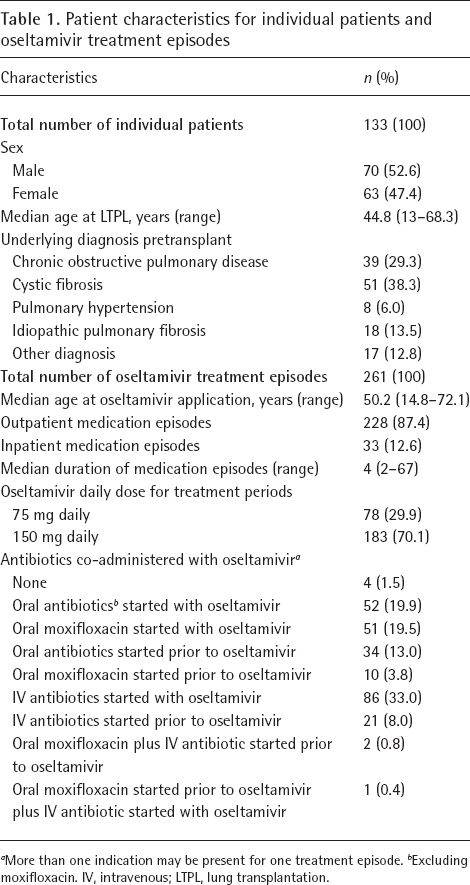
More than one indication may be present for one treatment episode.
Excluding moxifloxacin. IV, intravenous; LTPL, lung transplantation.
A total of 85 patients (63.9%) had received the seasonal influenza vaccine 2010/2011, the influenza season prior to the study period. Thirteen patients (9.8%) were not vaccinated for various reasons although vaccination had been recommended. In another 35 patients (26.3%) a vaccination was not feasible most frequently because they had recently received a lung transplantation and/or corticosteroid dose was too high (daily 20 mg prednisone or more) and therefore vaccination was not performed based on our internal guidelines. The seasonal influenza vaccine of 2011/2012 was given to 110 patients (82.7%). 10 patients (7.5%) did not receive the vaccine and 13 (9.8%) patients were not vaccinated due to a recent lung transplantation, which was performed within the last 3 months.
In 174 oseltamivir treatment episodes (66.7%) the indication to start treatment was signs of acute respiratory tract infection. The interquartile range of the duration of the treatment episodes was 5 days. The median duration between transplantation and the initiation of an oseltamivir treatment episode was 700 days, ranging from 39 to 18,685 days. The most frequent signs and symptoms prior to treatment start were elevated CRP in 105 (40.2%) episodes, sore throat or throat irritation in 85 (32.6%) episodes and rhinorrhoea in 81 (31%) episodes (Additional file 2).
In total, 21 had an LCI. Of these 21 LCI some presented with coinfections: 4 with influenza A virus and 3 with an influenza B virus had additionally a coinfection with another microbiological pathogen. Microbiological findings are presented in Table 2. One patient had a simultaneous infection with both influenza types. In 144 treatment episodes no microbiological pathogens were detected at all.
Safety
Overall, AEs occurred in 52 (19.9%) treatment episodes. The incidence of AEs is presented in Table 3. The most frequent AEs reported during treatment and within 3 days of discontinuation of oseltamivir were elevated liver transaminases in 13 (5.0%) episodes and oedema and headache, both in 6 (2.3%) episodes. Serious adverse events (SAEs) occurred in two (0.8%) episodes (acute coronary syndrome). These two patients were aged 61 and 70 years, and none had previous evidence of coronary artery disease (CAD) including coronary angiography in the pretransplant evaluation. No deaths occurred during oseltamivir treatment episodes and within 2 weeks of treatment discontinuation.
Table 4 shows the characteristics of the episodes with LCIs. The two most frequently observed findings before starting oseltamivir treatment were elevated CRP and lung function decline in nine (45.0%) and seven (35.0%) episodes, respectively. Only two radiological exams were performed in influenza-positive patients, and they showed pathological findings: in one patient a chest radiography showed a small, new consolidation in the right posterolateral lobe, which was considered by the radiologist to be post inflammatory or scarring, the main differential diagnosis was infection. A CT scan of another patient showed new bilateral infiltrations, partly tree-in-bud-pattern, and partly consolidations, which was interpreted as early signs of beginning bronchopneumonia.
Final microbiological findings from nasopharyngeal samples obtained prior to treatment initiation a
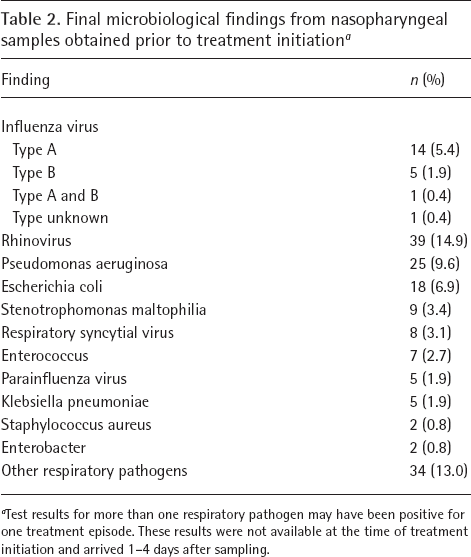
Test results for more than one respiratory pathogen may have been positive for one treatment episode. These results were not available at the time of treatment initiation and arrived 1–4 days after sampling.
Overall, in nine treatment episodes of LCI AEs were experienced. No severe AEs or deaths occurred within the treatment episodes or the 14 days after treatment in patients with LCI.
Sixteen patients (94.1%) with confirmed influenza infection had received the seasonal influenza vaccine of 2010/2011 at least 6 months prior to the infection. In one (5.9%) case the patient had refused vaccination. The seasonal influenza vaccination of 2011/2012 was given to 16 (84.2%) patients with confirmed influenza infection at least 2 months before the influenza infection occurred. In two (10.5%) cases the patients got infected in the months of June and July, before the seasonal vaccine had been available. None of these patients had travelled outside Switzerland, however, one patient spent a week on the border of Italy. In one (5.3%) case the patient received the lung transplantation 3 months before the infection and had not received the vaccination in the previous or current season.
Safety in hospitalized patients
Adverse events and signs occurring during and within 3 days after completion of oseltamivir medication episodes in outpatients and hospitalized patients a

More than one adverse event may be present for one treatment episode.
Abdominal pain, pruritus, insomnia and rash were also searched for in the database but not detected. ALAT, alanine transaminase; ASAT, aspartate aminotransferase.
Symptoms, signs, findings and adverse events in patients with documented influenza A or B virus infection
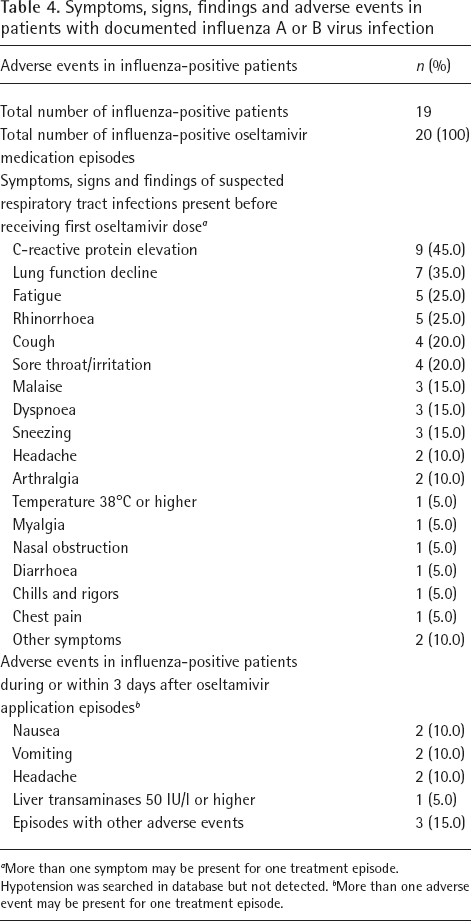
More than one symptom may be present for one treatment episode. Hypotension was searched in database but not detected.
More than one adverse event may be present for one treatment episode.
Adverse events and signs of hospitalized patients during or within 14 days after oseltamivir application episodes

More than one adverse event may be present for one treatment episode.
In 19 of 33 hospitalizations (57.6%) the reason for hospitalization was a decrease in lung function. The most frequent symptom in hospitalized patients during or within 14 days after a treatment episode with oseltamivir was pain in 29 episodes (87.9%). Cough was observed in 16 episodes (48.5%; Table 5). The most common AE in laboratory findings was a decreased haemoglobin level in 28 episodes (84.8%). Haemoglobin was already reduced below the reference value in 18 patients (54.5%) at the beginning of the treatment. During the evaluated period the AE intensity changed by one grade in 14 patients (42.4%) and by 3 grades in 1 patient (3.0%) according to the Common Terminology Criteria for Adverse Events [44]. Using the same criteria, seven patients (21.2%) developed a leukocytosis grade 3 whereby all had normal values at the beginning at diagnosis. In four patients (12.1%) a leukocytosis was found initially, but the grade did not increase during the evaluated periods. In eight patients (24.2%) normal leukocyte numbers were documented initially, and then leukopenia grade 2 developed. Creatinine increased in five (15.2%) patients from an elevated grade to a higher grade. In 26 episodes (78.8%) the creatinine level was already elevated at the beginning of the treatment. In three patients (9.1%) a normal thrombocyte number decreased to thrombopenia grade 1, in one patient (3.0%) to thrombopenia grade 2 and in another one (3.0%) to a thrombopenia grade 3. Four patients (12.1%) developed a new thrombocytosis. There was an increase from a normal initial CRP value to a pathological value after 2 weeks in three patients (9.1%), while in two patients (6.1%) an already elevated CRP level increased further.
Aspartate aminotransferase (ASAT) values in hospitalized patients were reviewed separately. In five episodes (15.2%) the ASAT increased in hospitalized patients above the level of twice the upper limit of reference of 50 U/l, that is, >100 U/l (Additional file 1). In all these patients elevated values decreased within 2 weeks of discontinuation of treatment to the reference values. The Naranjo score for these five cases was 3/10 points in four episodes and 2/10 points for one episode.
Discussion
In this retrospective study the pre-emptive use of oseltamivir for suspected and proven influenza infections showed no AEs in the majority of patients in LCI as well as in patients without a confirmed influenza infection. Although none of the individual AEs occurred in more than 5%, overall AEs were reported in 19.9% of treatment episodes. Severe AEs were experienced in 0.8%, none of which influenced mortality in the 6 months following the treatment episode. In most suspected respiratory tract infections Rhinovirus or Pseudomonas aeruginosa were detected. Since the majority of patients with LCI had received influenza vaccination of the respective season, it is likely that their influenza illness was ameliorated by the influenza vaccine, as evidenced by only a minority requiring hospitalization. In hospitalized patients there was evidence of hepatotoxicity in 5 of 35 LTRs with spontaneous and complete resolution after discontinuation of oseltamivir.
The rate of AEs in the current study was higher than observed in one earlier study, in which no specific AEs were documented and oseltamivir was well tolerated [8]. Other studies described similar outcomes regarding efficacy and safety for solid organ and haematopoietic stem cell transplant recipients [10,26,28,45]. Despite recent findings of oseltamivir affecting the innate, humoral and adaptive immune responses in mice, no secondary infections or transplant rejection have been described to date [46].
The most frequent AEs in our study were headache, vomiting, pain and nausea, which were also observed in other studies [10,20,26,47]. Interestingly, oedema was also found in our study and has not been described by others [10,20,26]. An acute coronary syndrome occurred twice (0.8%); in one patient at the age of 70 with previous subclavian vein thrombosis, there was previous intermittent tachycardic atrial fibrillation and overweight as relevant features in the patient's history. The other patient aged 61 had arterial hypertension, tachycardic atrial fibrillation and had a therapy with methotrexate due to rheumatoid arthritis. Both patients had been exposed to long-term immunosuppression and had a negative prior history for CAD. As CAD is not described in the other studies and with known risk factors in both patients, its causal relationship to oseltamivir remains uncertain. It is noteworthy that both patients took amiodarone due to atrial fibrillation, therefore an interaction with oseltamivir cannot be excluded, but appears unlikely based on known drug metabolism. The results of follow-up electrocardiograms are unknown. Due to the small number of observed patients with CAD no firm conclusion can be made in relation to oseltamivir. This possible association requires further research.
Additionally, we evaluated the episodes which occurred in hospitalized patients. The most common AEs of this subset were pain (mainly headache), cough and dyspnoea during and within 14 days following treatment with oseltamivir. In 19 of 33 cases the reason for hospitalization was a decrease in lung function which suggests that the described AEs may relate to other causes than oseltamivir treatment, such as the suspected or documented respiratory tract infection. Another aspect is the fact that the influenza virus infection itself has an effect on liver values [48]. Liver inflammation and elevated liver enzymes were also identified during post-approval use of oseltamivir. However, since these AEs were reported voluntarily from a population of uncertain size, it is according to the manufacturer not possible to reliably estimate their frequency or establish a causal relationship to oseltamivir exposure [22]. To evaluate if oseltamivir has an influence on transaminase levels in LTRs we looked at the course of ASAT levels in hospitalized patients and in 5 out of 20 patients ASAT levels rose to higher levels than twice the upper limit of normal. All these episodes of elevated transaminases were of short duration and showed a spontaneous regression over time. During these episodes, other medications were administered simultaneously including intravenous and oral antibiotics in all patients, as well as frequently prescribed itraconazole. This co-medication may have caused increased ASAT levels as well. The Naranjo score for these five cases were 3/10 points in four episodes and 2/10 points in one episode. According to the Naranjo evaluation an ADR with oseltamivir is therefore considered to be possible.
Immunocompromised patients, such as LTRs are more vulnerable to influenza infections. Previous studies have documented that influenza frequently presents with minimal or atypical symptoms in transplant recipients. In this study the principal symptoms in LTRs with suspected respiratory tract infection were sore throat and rhinorrhoea (Additional file 2). In LCIs fatigue and rhinorrhoea were mainly noted. In various other studies conducted with solid organ and haematopoietic transplant recipients cough and fever were the main symptoms [4,31–35], which can also be found in the general population affected by influenza virus infection [20,29]. Cough (27.2%) and fever (3.4%) were less frequently noted in our population.
The findings of the present study should be considered in the light of a few methodological limitations, which influence their general applicability. Given its retrospective design, we cannot provide a cause–effect relationship between oseltamivir intake and the described signs, symptoms and findings. Especially in hospitalized patients it is difficult to differentiate between AEs due to oseltamivir and clinical manifestations of the illness (regardless of the aetiology) requiring hospitalization, AEs of any other medical intervention (for example, antibiotics given) or any other illness complicating the hospitalization. Concomitant antibiotics in outpatients is also an additional limitation, making discerning safety of oseltamivir difficult. A retrospective chart review cannot robustly assess clinical signs and symptoms because the absence of documentation of a finding does not confirm its absence, as clinical documentation is not always thorough. The dataset of clinical, laboratory and lung functional data was prospectively collected and daily updated by physicians, nurses and data managers. However, neither the inpatient records nor the outpatient visit data collection method was designed to specifically capture oseltamivir side effects, other than those identified by laboratory tests. Therefore, a reporting bias by healthcare providers and a recall bias by patients cannot be excluded. Data were evaluated without comparison to placebo treatment, other drugs or no treatment at all. We did not test the effectiveness of oseltamivir, neither did we check for any emergence of resistance, which seems to occur occasionally [9,49]. Other than the fact that we did not have any influenza-related death in that year we cannot clearly delineate the benefit of this strategy with a pre-emptive use of oseltamivir in this susceptible population. Unlike in a randomized, controlled, prospective clinical trial, patients were included in the cohort on the basis of a provided and prescribed drug. The data reflect a real-life situation in a health system without major financial constraints.
Our study also has important strengths: a large number of LTRs were included with a high number of oseltamivir treatment episodes allowing reasonably precise estimate of drug safety, at least for frequent AEs. Data quality for all included patients is reliable since it was collected prospectively and all data for the hospitalized patients were documented by the treating doctors or as part of standard documentation of lab results. Furthermore, all patients were followed-up by our centre for all scheduled and unscheduled visits. Another strength is the evaluation of the ASAT levels, which were regularly determined during hospitalizations and all outpatient visits and allowed the analysis of potential hepatotoxicity related to oseltamivir intake. We are not aware of any other studies evaluating the course of ASAT levels in transplant recipients.
In summary, we found that oseltamivir is well-tolerated in the majority of LTRs with mainly minor AEs in approximately one-quarter of patients. Prospective studies and studies in larger cohorts are needed to ascertain the safety profile of oseltamivir in the population of LTRs.
Footnotes
Acknowledgements
We thank Nicolas Müller (Division of Infectious Diseases and Hospital Epidemiology, University Hospital Zürich, Zürich, Switzerland) for his valuable suggestions in the planning phase of this study.
The authors declare no competing interests.
