Abstract
Background
Direct-acting antivirals (DAAs) have revolutionized HCV treatment, but the impact of antiviral resistance at a population level is still not clear. The majority of patients who fail DAA therapy develop resistance-associated substitutions (RASs), which can impact re-treatment. There is potential for resistance prevalence to rise in the community with treatment scale up, due to transmission of resistant virus. Monitoring for increasing antiviral resistance requires a reliable baseline, yet there are few published data on the prevalence of HCV resistance in Australia. The aim of this study was to determine the prevalence of RASs among untreated Australians with HCV genotype-1a infection, to inform ongoing surveillance.
Methods
A cross-sectional study was performed at a single large university hospital pathology laboratory in Australia. Archived blood samples referred for HCV genotype testing were analysed. All patients were naive to DAAs. The prevalence of RASs in the HCV NS3 and NS5A regions was determined using Sanger based population sequencing.
Results
Of 379 samples tested, 34% contained DAA-resistant virus: 24% had resistance to NS3 protease inhibitors, 12% had NS5A inhibitor resistance and 4% of patients had resistance to both drug classes. Clinically relevant RASs conferring resistance against NS5A inhibitors ledipasvir, daclatasvir and elbasvir were detected in 5.8% of samples.
Conclusions
This is the largest study of HCV antiviral drug resistance in Australia, which differs from resistance prevalence in the USA. The results provide valuable data on the baseline prevalence of HCV resistance, which can be used in the future to monitor for increasing antiviral resistance.
Introduction
HCV infection is the primary cause of liver transplantation and hepatocellular carcinoma in Australia and now causes more deaths than HIV/AIDS [1], as in the USA [2]. Direct-acting antiviral (DAA) agents have revolutionized HCV treatment, offering sustained virological response (SVR), or cure rates of >95% with 12 weeks of oral therapy with minimal adverse effects. DAAs target specific HCV proteins, and are grouped into the NS3/4A protease inhibitors, NS5A inhibitors and NS5B polymerase inhibitors. Most HCV treatment regimens combine drugs from two or three of these classes. When the DAAs were listed on the Pharmaceutical Benefit Scheme (PBS) in March 2016, Australia became the first country to offer unrestricted treatment access to all people infected with HCV, including treatment in primary care settings. Since then there has been a rapid uptake in treatment, with over 58,000 patients being treated between March 2016 and March 2018, equating to approximately 26% of people with chronic HCV infection [3]. This rate of uptake is in line with the World Health Organization (WHO) goal of eliminating hepatitis C as a significant public health problem by 2030 [4], but increased prescribing in the community is crucial [5].
HCV is an RNA virus with a highly error-prone polymerase, so mutations appear frequently in the viral genome, resulting in a wide range of viruses (‘quasispecies’) within an infected person. Some mutations result in amino acid substitutions in viral proteins that confer antiviral drug resistance, termed resistance-associated substitutions (RASs). RASs can impact DAA treatment responses, particularly in difficult to treat patients, including those with cirrhosis and those who have previously failed older interferon-based therapy [6].
The first demonstration of clinically significant DAA resistance was in patients infected with HCV carrying the Q80K RAS, in whom adding the NS3 protease inhibitor simeprevir to pegylated interferon and ribavirin offered no treatment benefit [7]. However, with the availability of interferon-free treatment regimens, almost all of which contain an NS5A inhibitor, RASs in the NS5A region have emerged as the most clinically relevant. In HCV genotype-1a-infected individuals, the presence of NS5A RASs affects response to currently approved DAA combinations including ledipasvir/sofosbuvir, sofosbuvir plus daclatasvir and elbasvir/grazoprevir [6]. RASs have much less effect on treatment outcomes for genotype-1b infection, so this study focuses on genotype-1a.
There is little published data on the prevalence of HCV resistance in Australia, with data on NS5A resistance for genotype-1a from only 25 patients with acute HCV infection [8,9]. This study was performed to determine the prevalence of NS3 and NS5A RASs among DAA-naive Australian patients with HCV genotype-1a infection, prior to the availability of DAAs. The results provide a valuable baseline, informing future monitoring of antiviral resistance prevalence in the community.
Methods
A retrospective, single centre, cross-sectional, non-interventional study was performed, using samples from patients across a wide geographic region of New South Wales (NSW). The protocol was approved by our Local Human Research and Ethics Committee, which complies with the Declaration of Helsinki.
Sample size calculations were performed with the assumptions of precision = 5%, prevalence = 15%, population size = infinite and 95% CI specified limits (10%, 20%; these limits equal prevalence plus or minus precision). The estimated sample size needed to detect a resistance prevalence of 15% for genotype-1a (which is considered clinically relevant and is the level of detection achievable by Sanger sequencing) was determined to be 196.
Inclusion criteria were males or females >18 years of age with HCV genotype-1a infection or genotype-1 with undetermined subtype. The study was performed on remnant clinical plasma samples stored at −80°C at the HCV laboratory of a large urban Australian university hospital pathology laboratory. Samples had been sent for HCV genotype testing, so were presumed to be from people with chronic hepatitis C being considered for treatment. Samples from 2011–2012 were used, before the availability of DAAs in Australia, to ensure the patients were DAA-naive and could not have been infected by someone who had failed DAAs. Patients infected with genotypes other than genotype-1, indeterminate genotype or mixed genotypes were excluded. Samples were de-identified but demographic data including age, gender and patient's home postcode were recorded. In the case of Correctional Centers, the postcode of the Correctional Centre was used.
Plasma samples had viral RNA extracted using the EZ1 Virus Mini Kit v2.0 (Qiagen, Hilden, Germany). Extracted RNA was reverse transcribed into cDNA using SuperScript III reverse transcriptase (Life Technologies, Waltham, MA, USA). As shown in Figure 1, key regions of the HCV NS3 and NS5A genes were amplified using pan-genotypic primers and nested PCR, using published protocols [10,11]. The samples were separated on a 1.5% agarose gel, stained with GelRed and visualized under ultraviolet light, with minimal exposure. Bands corresponding to the predicted sizes of NS3 and NS5A were excised, purified (Wizard SV Gel and PCR Clean-Up System; Promega, Madison, WI, USA) and submitted for Sanger sequencing to the Australian Genome Research Facility (AGRF).

Summary of HCV genome, showing regions amplified for sequencing
Sanger sequence data were analysed for NS3 and NS5A HCV mutations using Geno2Pheno [hcv] [12]. The Geno2Pheno software identified these clades using NS3 sequence data. Choice of significant RASs for analysis were based on published data from the most current EASL guidelines [13], AASLD-IDSA guidelines [14] and expert reviews [6,15,16].
Results
A total of 461 samples met the selection criteria during the study period (2011–2012). This period was before the availability of DAAs in Australia, so was chosen to provide baseline data on antiviral resistance and to ensure the patients were DAA-naive. Twenty samples were not used because of insufficient plasma for testing. NS3 amplification was unsuccessful in 49 samples, and NS5A amplification was unsuccessful in 55 samples. In total, 379 genotype-1 samples had both the NS3 and NS5A regions successfully amplified and were included in the final analyses.
Characteristics of the study population
The average age of patients tested was 40.5 years (range 19–69), with 284/379 samples (74%) from males. Postcode information was available from 323 samples. Samples were all from NSW, both rural and urban in origin (Figures 1 and 2) and included 123 samples (32%) from NSW Correctional Centers.

Map of New South Wales, showing postcode origin of tested samples
The sample distribution across NSW is shown in Figure 2. The highest number of patients came from postcode 2828, which was the postcode that included the Wellington Correctional Centre. There were 182 samples from non-Sydney metropolitan areas, but none from north-eastern or southwest NSW.
197 samples (52%) were from patients living in greater metropolitan Sydney (Figure 3). The most common postcode was 2128, which corresponded to the postcode for the Silverwater Correctional complex. Samples came from across the city, although there was a predominance from Western Sydney.
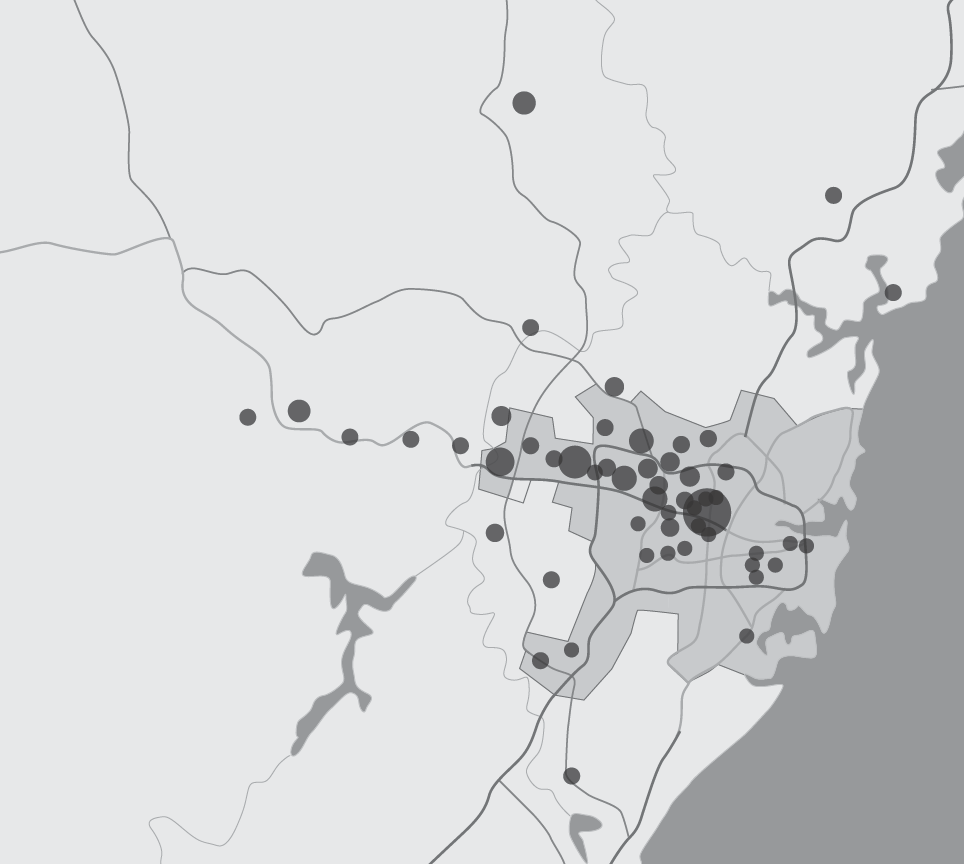
Map of Greater Sydney, showing sample origin according to postcode
Frequency of NS3 RASs
Within our cohort, 91 of 379 people (24%) had one or more NS3 RAS. The most common RAS was I170V, present in 25 of 379 people (6.6%). The most common RAS sites were V55 and I170. There were 79 of 379 people (20.8% of the cohort) with a single RAS. 13 of 379 (3.4%) had two NS3 RASs present and the most frequent combination of RASs was T54S and V55I, present in 8 people. No person had more than two RASs in NS3. 32 of 91 people with NS3 RASs were from Correctional Centers: the commonest RAS was Q80K, present in 11 persons (12.1% prevalence).
The spread of NS3A RASs is summarized in Figure 4, with individual RAS frequencies shown in Additional file 1.

Summary of individual NS3 RASs
Frequency of NS5A RASs
One or more NS5A RAS were found in 45 of 379 (11.9%) people (summarized in Figure 5, with a full list in Additional file 2), with the most common being M28V (4.8%). The most common RAS sites in NS5A were H58 and M28. 36 of 379 people (9.5%) had a single RAS, and 9 of 379 people (2.4%) had two or more NS5A RASs. There was no dominant RAS combination, with M28V+Q30H, M28V + H58R and Q30H + Y93H occurring twice each. Three persons had three NS5A RASs present at baseline. 13 of 45 people with NS5A RASs were from Correctional Centers, with M28V found in seven.

Summary of individual NS5A RASs
Combination NS3 and NS5A RASs
In our cohort, 15 (4%) had both NS3 and NS5A RASs; 4 were from Correctional Centers.
Prevalence of clinically relevant RASs
Definitions of what constitutes a RAS can vary across studies, with some studies reporting all RASs that may confer resistance to a drug class or all RASs that confer in vitro resistance [6]. For this study we used internationally recognized definitions of ‘clinically relevant RASs’ based on European and American guidelines [13,14]. Using this definition, 33% (125/379) of HCV-infected persons in our cohort were infected with drug-resistant virus. 24% (91/379) had substitutions conferring resistance to NS3 protease inhibitors. 12% (45/379) had NS5A inhibitor resistance, with 5.8% (22/379) having clinically relevant resistance mutations against the commonly prescribed drugs ledipasvir, daclatasvir and elbasvir. RASs conferring resistance against the pan-genotype NS5A inhibitor velpatasvir were seen in 6.3% (24/379), 7.9% (30/379) had RASs against ombitasvir, and 2.9% (11/379) had resistance to both NS3 and NS5A inhibitor drug classes. The prevalence of resistance to specific NS3 inhibitors is summarized in Figure 6, and to NS5A inhibitors in Figure 7.
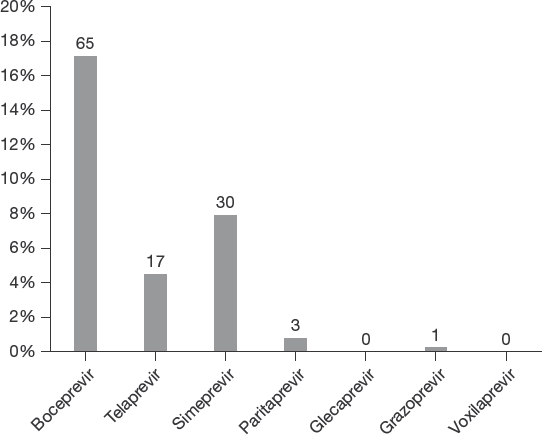
Prevalence of resistance to individual NS3 (protease) inhibitors

Prevalence of resistance to individual NS5A inhibitors
Analysis by patient demographics
Logistic regression analysis was performed to look for a possible effect of patient demographics on RAS prevalence, but no effect of age, gender, residence in a Correctional Centre, or urban versus rural setting was observed.
Correctional Centre analysis
Persons from Correctional Centers made up 30.9% (117 of 379) of the overall cohort. As shown in Additional file 3, compared with people in our cohort from the community, people from Correctional Centers were younger (mean age 35 versus 45; P<0.0001), and predominantly male (80%). The prevalence of NS3 RAS and NS5A RAS was similar in both populations. The commonest NS3 RAS differed between populations (I170V in the Correctional Centre population and V55A in the Community Population), although the commonest drug affected was the same, that is, boceprevir resistance. The prevalence of NS5A RASs was similar in both populations, occurring in 14.1% (37/262) in the Community Population, compared with 12.8% (15/117) in the Correctional Centre population.
The commonest NS5A RAS present in both populations was M28V, however, NS5A drug resistance patterns differed, with ombitasvir resistance most common in the Correctional Centre population, and velpatasvir resistance in the Community Population. There was no significant difference in the prevalence of RASs in both NS3 and NS5A regions of the virus: 3.4% in the Correctional Centre population, 4.2% in the Community Population.
Discussion
This study provides clinically relevant data on the baseline prevalence of antiviral drug resistance among Australians living with hepatitis C. In Correctional Centers (prisons), the prevalence of chronic hepatitis C infection among people who inject drugs is approximately 50% [17] and HCV transmission in the prison setting is well documented [18]. Treating HCV-infected people in prisons is a key strategy to achieve HCV elimination [5], so it is important to understand the prevalence of antiviral resistance in this group.
We analysed 379 HCV genotype-1a-infected people who were DAA-naive, making this the largest Australian study of HCV antiviral resistance. Importantly, the samples were collected prior to the introduction of interferon-free DAA therapy into Australia, so provide an important baseline to assess any future changes in prevalence of drug resistance in the community. In this study cohort, 24% of people were infected with HCV containing at least one resistance associated substitution in the NS3 region, 12% containing at least one NS5A RAS and 4% having both NS3 and NS5A RASs.
HCV has evolved into seven genotypes and 67 subtypes worldwide [19], which affect disease progression, complications and response to various treatment regimens [20]. Due to this genetic variation the prevalence of RASs, and hence innate drug resistance, also varies greatly around the world by geographic region [21]. Therefore, local prevalence data on antiviral resistance are key to individual patient management and to develop treatment guidelines and testing policies. In Australia, HCV genotype-1 is the most prevalent (55%), followed by genotype-3 (33%) [22]. To date there have been few data on the prevalence of HCV RASs in the Australian population, with a total of only 165 people with genotype-1a infection in all previous studies combined, only 25 of whom had their HCV sequenced for clinically relevant NS5A resistance [8,9,23]. Of note, most previous Australian studies used deep sequencing, whilst this study used Sanger sequencing as it is more readily available and sufficiently sensitive to detect clinically relevant RASs [6].
Early studies of antiviral resistance in hepatitis C focused on RASs in the NS3 region, as NS3 inhibitors were the first DAAs approved to treat hepatitis C. Most studies focused on the prevalence of the Q80K RAS, which affected treatment response to the second-generation NS3 inhibitor simeprevir [7]. Although simeprevir is no longer used in Australia, the worldwide geographic variation in Q80K prevalence highlights the importance of local data to inform treatment and testing guidelines. Globally Q80K is the most prevalent HCV RAS at 37.6% [21] but local prevalence varies widely, with 48.1% prevalence in the USA [24] and 19.8% in Europe [25]. In our study, we observed a Q80K prevalence of only 5% in Australia, which is consistent with 4% prevalence in a previous Australian study [8] and 7% prevalence reported in pooled data from clinical trials of simeprevir in Australia and New Zealand (n=11/155) [26]. Due to its high prevalence in the USA, Q80K testing was recommended in the US guidelines before prescribing simeprevir [14]. Baseline Q80K testing was not required in Australia, due to its low prevalence in Australian trials and the lack of suitable testing facilities. The low prevalence of Q80K we observed in Australian patients supports this recommendation.
Of note, most of the NS3 RASs we detected confer resistance against NS3 inhibitors no longer used in Australia (telaprevir, boceprevir and simeprevir), but we have included these data for completeness and because these drugs are still being used in other countries. In contrast, only three patients had RASs conferring resistance against paritaprevir (now used infrequently in Australia), one had a RAS against grazoprevir, and no patients had RASs against new pan-genotype NS3 inhibitors glecaprevir or voxilaprevir.
More recently, research on antiviral resistance has focused on the HCV NS5A region as all currently recommended DAA combinations contain an NS5A inhibitor and NS5A RASs have a greater impact on DAA treatment outcomes than NS3 RASs [6]. NS5A RASs typically exert a class affect, conferring clinical resistance against most NS5A inhibitors. Furthermore, NS5A RASs persist for years in patients who fail DAAs because these mutations have minimal effect on viral fitness [16]. Despite the emerging importance of NS5A RASs, they were not analysed in most early Australian studies of HCV resistance [23]. Indeed, the only previous Australian data on NS5A resistance for HCV genotype-1a is from 25 patients with recently acquired (not chronic) HCV infection, one of whom had an NS5A RAS [8,9]. Therefore, this sample of 379 people provides valuable data on the baseline prevalence of clinically relevant NS5A resistance among DAA-naive patients in Australia with chronic hepatitis C.
There was an 11.6% overall prevalence of NS5A RASs in this cohort. The prevalence of clinically relevant NS5A RASs, which confer resistance against the NS5A inhibitors ledipasvir, daclatasvir and elbasvir, was 5.8%. Ledipasvir/sofosbuvir was the most commonly prescribed DAA treatment combination in Australia up until July 2017 [3] and is still widely used in many countries. The impact of baseline ledipasvir RASs on treatment outcomes has been analysed in one large study, using pooled data from over 5,000 patients enrolled in Phase II/III clinical trials of ledipasvir/sofosbuvir [27,28]. Among patients with genotype-1a infection (n=3,507), the presence of ledipasvir-specific NS5A RASs did not significantly impact cure rates among treatment-naive, non-cirrhotic patients (92% versus 99%). However, ledipasvir-specific RASs reduced cure rates for non-cirrhotic patients who had previously failed interferon-based treatment (76% versus 97%) and for patients with cirrhosis (86% versus 99% for treatment-naive; 77% versus 96% for treatment-experienced) [27]. US guidelines recommend resistance testing where available for treatment-experienced patients with genotype-1a infection prior to prescribing ledipasvir/sofosbuvir, and if RASs are detected recommend adding ribavirin, or considering another treatment regimen [29]. Australian consensus guidelines do not recommend baseline resistance testing, due to limited access to HCV resistance testing and low clinical yield [30].
The treatment combination of elbasvir/grazoprevir is a regimen particularly impacted by NS5A resistance. In Phase II/III trials of elbasvir/grazoprevir, patients with HCV genotype-1a containing elbasvir RASs had a reduced cure rate of only 58%, compared with 98% for those without RASs [31]. Therefore, the US HCV treatment guidelines recommend NS5A resistance testing be performed for all patients before prescribing elbasvir/grazoprevir [14]. In contrast, baseline RAS testing for elbasvir is not recommended in the current Australian consensus guidelines [30], due to limited access to HCV resistance testing and a presumed low prevalence of elbasvir resistance in Australia. Our study confirmed a low 5.8% prevalence of elbasvir-specific RASs among untreated people in Australia, similar to the 5% prevalence observed in Australian patients in the international Phase II/III trials [31] and the 6.2% prevalence reported in a multi-centre Spanish study [32]. Elbasvir/grazoprevir accounted for 10–15% of DAA prescriptions in Australia in 2017–2018 [3], so the low prevalence of elbasvir-specific RASs is unlikely to have an impact on treatment outcomes at a population level. Nonetheless, if RAS testing is available we believe it should be considered in patients with genotype-1a infection prior to prescribing elbasvir/grazoprevir.
In our study the prevalence of RASs affecting the NS5A inhibitor ombitasvir was 7.9%. Ombitasvir is no longer prescribed commonly in Australia but may still be prescribed in other countries.
Of more concern, 6.3% of people had RASs conferring resistance against the new pan-genotype inhibitor velpatasvir, a pan-genotype regimen that is now the most commonly prescribed DAA combination in Australia [3]. This is higher than the 4.1% global prevalence observed among patients with genotype-1a enrolled in Phase III trials of velpatasvir/sofosbuvir [33]. In those trials, the commonest velpatasvir RAS was M28V, the same as in our study. In the Phase III trials of velpatasvir/sofosbuvir, NS5A RASs did not affect SVR rates among patients with genotype-1a infection, but in patients with genotype-3 the SVR rate was reduced from 98% to 93% when velpatasvir-specific RASs were present [33]. Therefore, although our data do not impact treatment guidelines for genotype-1a, more Australian data are needed to determine the prevalence of velpatasvir-specific RASs among patients with genotype-3 infection.
The HCV NS5B region was not analysed for RASs in this study, as the prevalence of clinically relevant NS5B resistance in untreated patients around the world is extremely low [6]. Resistance to non-nucleoside NS5B inhibitors like dasabuvir is not uncommon, although these drugs are no longer included in first-line DAA combinations in Australia and most other countries. The most commonly prescribed NS5B inhibitor in Australia is the nucleotide analogue sofosbuvir, which was present in approximately 90% of DAA combinations prescribed between March 2016 and March 2018 [3]. Sofosbuvir has a high barrier to resistance, and although the clinically relevant sofosbuvir RAS S282T has been observed in a few patients failing DAA therapy, it significantly reduces viral fitness, so does not persist beyond a few weeks and is rarely seen in untreated patients [6]. Indeed, in a global analysis of RAS prevalence, among almost 1,500 sequences analysed from Genebank, only one sample (genotype-1b) contained S282T [21]. Nonetheless, an increase in the prevalence of sofosbuvir RASs in the future would be of Australian and global importance, so monitoring for NS5B resistance should be part of ongoing testing, particularly in patients failing DAA therapy.
One limitation of this study is the lack of clinical data, as samples were obtained from remnant laboratory samples. However, as these samples had been referred to the laboratory for HCV genotype testing, they were most likely from patients with chronic hepatitis C who were being considered for antiviral therapy. Data are from patients in NSW only, so we cannot exclude that antiviral resistance may vary in other states of Australia. Another potential limitation of the study is that RAS prevalence was determined using population Sanger sequencing, which can only detect variants present in 15–20% of viral quasispecies. Although next-generation (‘deep’) sequencing allows for more sensitive detection of minor viral quasispecies, published data suggests that deep sequencing rarely adds additional useful clinical information, and the presence of mutations in at least 15–20% of quasispecies is recommended for reporting clinically relevant RASs [6].
In conclusion, this is the largest study to date looking at antiviral resistance among Australians living with hepatitis C. Its main finding is that clinically relevant RASs against NS5A inhibitors ledipasvir, daclatasvir and elbasvir are present in 5.8% of people infected with HCV genotype-1a. These data provide a valuable baseline level for HCV resistance, as blood samples were collected prior to the introduction of DAAs into Australia. This will inform future monitoring for increases in HCV antiviral resistance in the community, consistent with the approach taken for monitoring antiretroviral drug resistance for HIV infection in Australia [34].
Footnotes
Acknowledgements
Work on this manuscript was in part supported by Program and Project grants from the National Health and Medical Research Council (NHMRC) of Australia (358772, 1003767, 1047417 and 1053206) and by the Robert W Storr bequest to the Sydney Medical Foundation, University of Sydney. ATLO was supported by a NHMRC Postgraduate Scholarship.
ATLO, MWD, DED and JG designed the study. DED provided the samples. ATLO and ET conducted the laboratory work. ATLO, ET and MWD analysed the data. ATLO, ET, MWD, DED and JG wrote and edited the manuscript. All authors reviewed the draft and approved the final decision.
The authors declare no competing interests.
