Abstract
Background
Methods
Results
Conclusions
Introduction
Herpes simplex virus infection is caused by type-1 (HSV-1) and type-2 (HSV-2) viruses which are considered important human pathogens that belong to the Herpesviridae family, a large and diverse family of double-stranded DNA enveloped viruses. HSV-1 can establish latency in neuronal cells after primary infection causing progressive grey matter loss in the posterior cingulate gyrus and reduction in cognitive function besides increased liability to drug abuse [1,2]. Viral DNA and RNA activate the innate and adaptive immune systems inducing pro-inflammatory mediators that may trigger amyloid-β (Aβ) accumulation, phosphorylation of tau protein and neuronal injury [3]. Usually HSV infections start during childhood or youth by direct contact with secretions containing viral particles. HSV-1 infections can cause a variety of complications from mucocutaneous infections (herpes labialis and genitalis) to corneal blindness and life-threating diseases associated with encephalitis, neonatal herpes infection [4], and also increased risk for behaviour disorders [1,2].
The common mechanisms of antiviral action currently employed for clinical therapy are varied. However, most antivirals act on DNA polymerase inhibiting transcription or replication of the viral genome thereby interrupting viral protein synthesis or may affect the process of adsorption by impeding virus entry into the host cell and/or particle stripping by interfering with viral assembly, release or de-aggregation [5,6]. Approved HSV-1 antiviral therapy for immunocompetent host includes the nucleoside prodrug acyclovir (ACV) that requires prior activation by viral and cellular kinases for inhibiting the HSV DNA polymerase [7]. Orally bioavailable pre-prodrugs including the guanosine analogues valaciclovir, penciclovir, famciclovir, idoxuridine and trifluridine are specifically phosphorylated by the viral thymidine kinase and further by cellular kinases [7]. Nucleoside analogues (for example, amenamevir, pritelivir) target the viral helicase-primase enzyme complex comprising the HSV UL5, UL8 and UL52 gene products, thereby inhibiting viral DNA replication [4,7]. Other nucleoside prodrugs acting as direct DNA polymerase inhibitors such as phosphonoformate (foscarnet) and phosphonoacetate are very effective against HSV-1 and -2 viruses by binding in the pyrophosphate binding site of HSV Pol blocking polymerization of additional dNTPs [4,5]. However, both HSV-1 and -2 viruses can reactivate nearby the site of the initial infection. In particular, immunocompromised patients have frequent and long-lasting episodes of HSV reactivation with extensive lesions that eventually disseminate becoming life-threatening [7,8]. The efficacy of nucleoside analogue therapy is generally restricted, due to high probability of inducing a resistant virus by mutation in the UL23 gene that encodes GC hot spots repeats further resulting in a nonfunctional truncated TK enzyme, and/or also a mutation in the UL30 gene that encodes the viral target DNA pol enzyme [7,8]. HSV strains are characterized by exceptional capacity of latency associated with intermittent reactivation aggravated due to the persistence of drug-resistant mutant with increased pathogenicity and neurovirulence due to a latent infection in sensory neurons [9]. Therefore, development of innovative therapies based on new compounds with potent and effective antiviral activity but low toxicity are important due to emergence of resistant strains to current antiviral drugs [4,7,8,10]. A suitable antiviral compound with ample spectrum activity capable to prevent infectivity and drug resistance should specifically target viral fusion by altering lipid rearrangements on viral envelopes required for fusion [11,12].
Monoterpenes are hydrocarbons formed by junction of two isoprene units derived from the mevalonate pathway in plants, comprising volatile non-nutritive dietary components found in essential oils of citrus fruits and other plants, and used as fragrances and flavourings [13–15]. Monoterpenes diffuse across the membrane into the endomembrane system eventually binding to an intracellular target and thereby exerting remarkably diverse biological properties [16]. Perillyl alcohol (POH) presents chemopreventive, analgesic, wound-healing properties, anti-inflammatory, cytostatic, cytotoxic, antimutagenic, anti-angiogenic, proapoptotic, immunomodulatory and antioxidant activities [14,15,17,18]. POH also protects against experimental cerebral malaria and reduces cerebrovascular inflammation [19], inhibits virulence of Candida albicans by arresting cell cycle and altering mitochondria function of the opportunistic fungus [20]. POH also interferes with regulation of genes (bax, bad, bak) involved in activation/inhibition of apoptosis responsible for increased mitochondrial membrane permeability, cytochrome c release and activation of cell death pathways [18,21]. Due to its amphipathic characteristics POH acts as molecular glue in the aqueous phase forming clusters that continuously diffuse across the membrane binding to an intracellular structure [16,22]. In humans, POH is rapidly metabolized by cytochrome P450-type liver enzymes to perillyl aldehyde (perillaldehyde), perillic acid (PA), cis and trans-dihydroperillic acids with subsequent glucuronidation and excretion in the urine and to a lesser extent in bile [13]. PA is capable of blocking cell cycle and causes apoptosis in lung tumour cells [23], inhibits Ras protein isoprenylation [16,24,25] and IL-2 production [26]. Considering that HSV-1 is neuroinvasive and capable of causing lytic and latent infections [9,27,28] the present work aimed to analyse the cytotoxic and putative antiviral activity of POH and PA monoterpenes upon in vitro replication of HSV-1 KOS wild-type and the single-step syn- mutant of Glasgow 17 strain on Vero cells by plaque assay.
Methods
Cell Culture, virus and Compounds
Vero cells (ATCC) were cultured in Dulbecco's modified Eagle's medium (DMEM) supplemented with 5% fetal bovine serum (FBS; HyClone, Logan, UT, USA), 100 U/ml penicillin, 100 μg/ml streptomycin, and amphotericin B (25 mg/ml; Cultilab, São Paulo, Brazil) at 37°C in a humidified 5% CO2 atmosphere. HSV-1 strain KOS (ATCC) and 17+syn were used for experiments. The difference between wild-type KOS and the mutant syn 17 strain that presents at least 1,024 small nucleotide polymorphisms (SNPs) and 172 insertions/deletions contribute to its pathogenesis [28] especially on events related to the fusion [27]. VERO cells and virus stock cultures were prepared from supernatants of infected cells and stored at -80°C until use. POH and PA were bought from Sigma-Aldrich (Sigma Chem. Co., St. Louis, MO, USA), dissolved in sterile dimethylsulfoxide (DMSO) and stored at -20°C, and stock solutions (50 mM) were diluted in DMEM just prior to the experiment.
Cytotoxicity Assay
Vero cells cultivated in 96-multiwell plates were treated with various concentrations (50, 250, 500 and 1,000 μM) of POH or PA for 72 h, and 50 μl of 1 mg/ml solution of 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyl tetrazolium bromide (MTT; Sigma-Aldrich) was added to assess cell viability according to procedures described elsewhere [29]. The 50% cytotoxic concentration (CC50) was calculated by linear regression analysis of the dose–response curves.
Antiviral Evaluations and Plaque Reduction Assay
To determine viral titre 2x10 5 Vero cells were infected with various HSV-1 log10 dilutions for 1 h at 37°C in a 5% CO2 atmosphere. Thereafter, monolayers were covered with 2x DMEM medium with 5% FBS and 2% of methylcellulose (Fluka) for 72 h, particle forming unit per ml (PFU/ml) were counted and virus titre determined. Thereafter Vero cells (2x10 5 cells/well) maintained in 24-multiwell plates were infected with KOS strain at a high (1.0) multiplicity of infection (MOI) for 1 h, and treated for 24 h with different concentrations (50, 25, 12.5, 6.25, 3.12 and 1.56 μM) of POH or PA monoterpenes.
Monoterpene Effect on HSV-1 Viral Titration
Vero cells were infected with HSV-1 with a low MOI (0.1) for 1 h at 37°C, 5% CO2. After 1 h, viral inoculum was removed and 50 μM of POH or PA was added to cell monolayers for 24, 48 and 72 h post-infection. To determine the viral burden, 2x10 5 cell/well Vero cells maintained in 24-multiwell plates were infected with various log10 dilutions of collected samples, for 1 h at 37°C and 5% CO2 atmosphere. Monolayers were then covered with DMEM supplemented with 10% FBS and human IgG for 72 h. PFUs were counted and viral titration determined according to the number of PFU/ml.
Real-time Polymerase Chain Reaction
Total RNA and DNA were extracted with TRIzol® Reagent from infected and treated (50 μM) Vero cells with an MOI of 1 for 2, 4, 8 and 12 h. Then, to it was added 200 μl chloroform/isoamyl solution (V:V – 24:1), and after 10 min in ice samples were centrifuged at 12,000 x g for 15 min at 4°C and performed the following steps: briefly, for RNA extraction, the top aqueous phase were collected and transferred to a new tube with the same volume of isopropanol (Merck, Kenilworth, NJ, USA); samples were incubated at room temperature for 10 min and further centrifuged at 12,000 x g for 10 min at 4°C. Supernatant was then removed and RNA pellet washed with 70% ethanol, centrifuged at 7,500 x g for 5 min at 4°C, dried and resuspended in RNase-free water. RNA samples were treated with DNase and cDNA were obtained by reverse transcriptase reactions (Omniscript Kit, QIAGEN, São Paulo, Brazil); and for DNA extraction, the bottom organic phase was incubated for 3 min with 100% ethanol, centrifuged at 2,000 x g for 5 min at 4°C and washed with 0.1 M sodium citrate in 10% ethanol (pH 8.5) for 30 min. The preparation was further centrifuged at 2,000 x g for 5 min at 4°C, washed once more and 70% ethanol was added for 10–20 min at room temperature. Samples were then centrifuged at 2,000 x g for 5 min at 4°C to form DNA pellet, and after DNA pellet dried it was resuspended in 8 mM NaOH solution.
Western Blot
Monolayers of Vero cells were inoculated with HSV-1 KOS at an MOI of 1 and plates incubated for 1 h at 37°C. Infected cells were treated with 50 μM POH or PA for 3-24 h post-infection (hpi) and then incubated for 24 h. Cells were lysed with extraction buffer (10 mM Tris-HCl pH 7.5, 100 mM NaCl, 1% NP-40, 1% Triton X-100, 1% sodium deoxycholate, 0.1% SDS, 5 mM EDTA, 10 μg/ml aprotinin, 10 μg/ml leupeptin, 10 μg/ml pepstatin, 1 μg/ml sodium orthovanadate and 0.5 mM sodium fluoride). Each sample was analysed separately on a 12% SDS-PAGE gel and blotted onto nitrocellulose membranes. After blocking, membranes were incubated overnight with anti-HSV antibody (1:10,000; Abcam, OR, USA) conjugated to horseradish peroxidase. Protein bands were revealed by exposure to X-ray film using ECL substrate according to the manufacturer's protocol (Santa Cruz Biotechnology, CA, USA). Anti-actin antibody was used as control for total protein loading. Graphic was made according to results generated by the software ImageJ Fiji.
Monoterpene Effect on Viral Particle Release
To evaluate if the POH or PA compounds were disturbing virus release, Vero cells (2x10 5 cells/well in 24-multiwell plate) were infected for 1 h at an MOI of 1, followed by a 24 h treatment of the cells with the 50 μM POH or PA. After 24 h incubation, supernatant from each sample was collected to assess intracellular and/or membrane bound viruses, and then cells were washed with PBS 1xC and lysed with three cycles of freezing and thawing. The sample was then centrifuged and collected cell-bound viruses, vesicles or virus clumps attached to the cell membrane from lysed Vero cells. To determine viral titres of intra and extracellular viral particles, 2x10 5 Vero cells/well maintained in 24-multiwell plate were infected with 10-fold dilutions (1:10) of the collected samples for 1 h at 37°C and 5% CO2 atmosphere. Thereafter, monolayer was covered with DMEM, 10% FBS and human IgG for 72 h, and viral burden was determined according to PFU/ml.
Statistical Analysis
The programme GraphPad Prism 4.0 (GraphPad Software Inc., San Diego, CA, USA) was used and analysis of variance followed by multiple comparisons using the Kruskal-Wallis test. Differences were considered statistically significant when P<0.05.
Results
Evaluation of POH and PA Cytotoxicity and Antiviral Activity
Vero cells infected with two different virus strains KOS and 17syn+ were treated with various concentrations of POH and PA compounds (1,000, 500, 250 and 50 μM). We determined the effective concentration of the compounds that inhibited 50% (EC50) of particle forming units. PA showed a potential antiviral activity primarily against the 17syn+ HSV-1 strain (EC50 KOS: 2.84 μM; 17syn+: 1.08 μM; Table 1). POH was a promising compound with an antiviral activity with EC50 value near to ACV (EC50 – POH: 1.3 μM; ACV: 1.09 μM; Table 2).
Antiviral activity of monoterpenes

Vero cells were infected with KOS and 17syn+ virus strains and treated with various concentrations of perillyl alcohol (POH) and perillic acid (PA). Results represent mean ±
Antiviral activity and selectivity index of monoterpenes and acyclovir
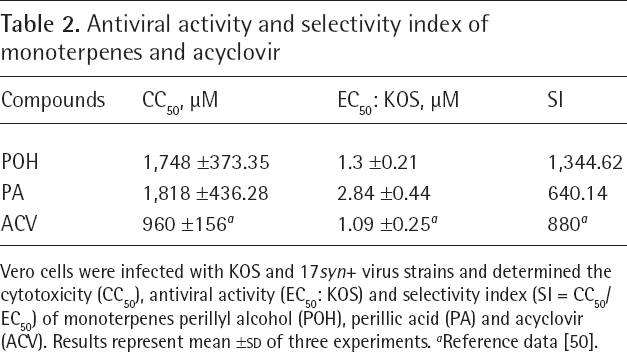
Vero cells were infected with KOS and 17syn+ virus strains and determined the cytotoxicity (CC50), antiviral activity (EC50: KOS) and selectivity index (SI = CC50/EC50) of monoterpenes perillyl alcohol (POH), perillic acid (PA) and acyclovir (ACV). Results represent mean ±
Reference data [50].
Based on CC50 and EC50 values it is possible to calculate the selectivity index value (SI), which represents the degree of safety for in vitro use of a given compound. Such pharmacological parameter was calculated from the ratio between CC50 and EC50 values. POH had higher SI values than ACV, indicating that POH is safer than ACV for use in in vitro systems (Table 2). PA was less cytotoxic (CC50 1,818 μM) than POH (CC50 1,748 μM) with a 2.1-fold increase of SI, but the difference was not significant (Table 2). Interestingly, POH and PA monoterpenes were less cytotoxic than the standard antiviral drug control, ACV with CC50 (μM) with 960 μM concentration (Table 2).
In addition, we performed a low MOI (0.1) plaque reduction assay [30] to confirm that the antiviral activity was due to the monoterpene activity, and not because formation of defective particles related to viral particle competition for the cellular machinery. We lowered the MOI to 0.1 and treated the cells after 1 h of infection with 50 μM POH or PA, and samples were collected after 24 h, 48 h or 72 h. The low MOI assay showed that (Figure 1) monoterpenes inhibited virus plaque formation within 24 h post-infection and continued its effect up to 72 h treatment. In addition, POH seemed more efficient during early time (24 to 48 h) whereas PA at 72 h post-infection. These results confirmed that the antiviral activity was due to POH and PA treatment. It is worth mentioned that POH is rapidly metabolized to PA inside the cells [16], which may explain low PFU numbers after treatment with PA in comparison with POH treatment. Interestingly, POH still induced stronger antiviral activity compared with control untreated infected Vero cells.
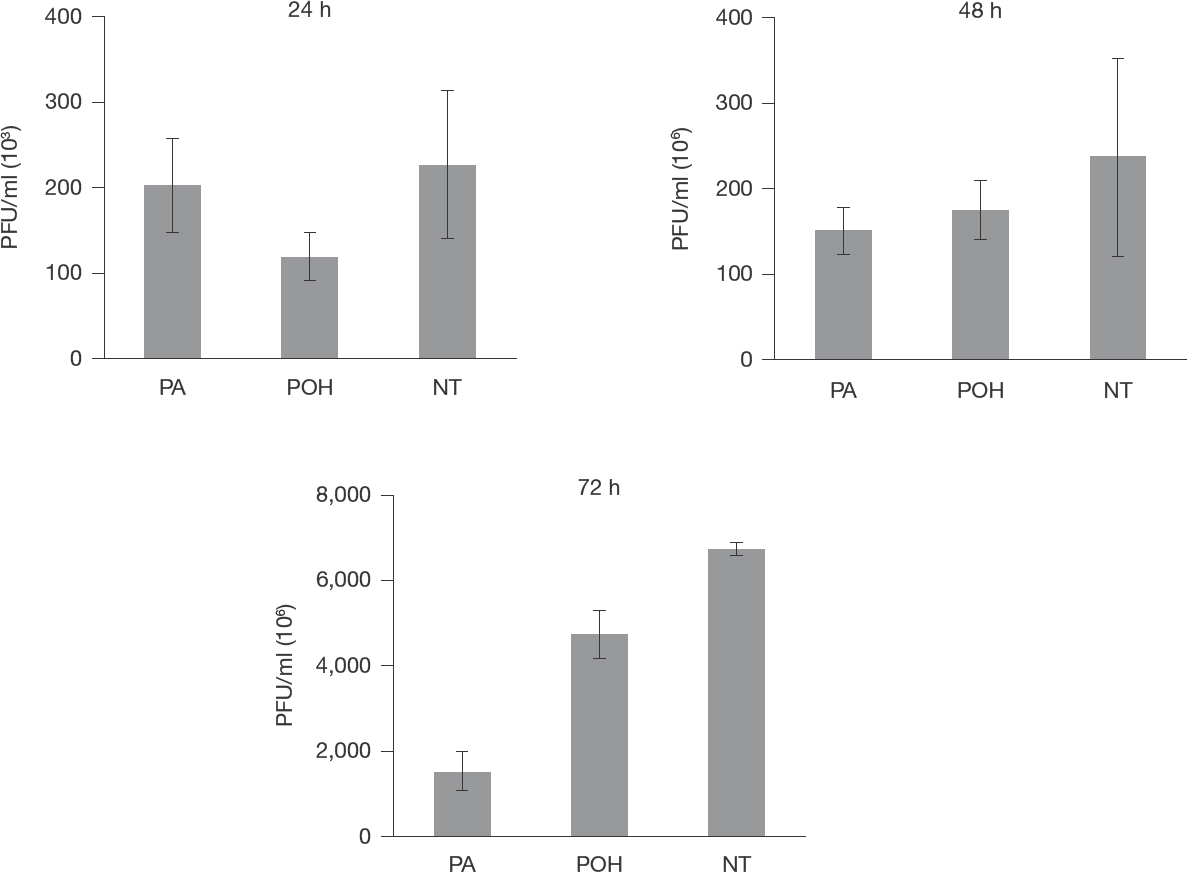
Antiviral activity assessed by plaque reduction assay
Real-Time PCR
Monoterpene Effect on HSV-1-Specific Gene Expression
Total RNA was collected from infected Vero cells treated or not with 50 μM POH or PA. cDNA was obtained with reverse transcriptase reaction as described in Methods. We analysed six relevant HSV-1-specific genes at different time points (2, 4, 8 and 12 h post-infection) to evaluate if POH and PA were interfering with mRNA production. The results showed that POH and PA did not affect HSV-1 gene expression (Figure 2).

Effect of monoterpenes on herpes simplex virus-1 specific gene expression
Monoterpene Effect on Viral DNA Synthesis
We further evaluated if 50 μM POH or PA were interfering with viral DNA synthesis. We extracted viral DNA from infected Vero cells treated or not with 50 μM POH or PA at different time points (2, 4, 6, 8 and 12 h post-infection). A significant reduction of viral DNA production after POH or PA treatment was not observed (Figure 3).
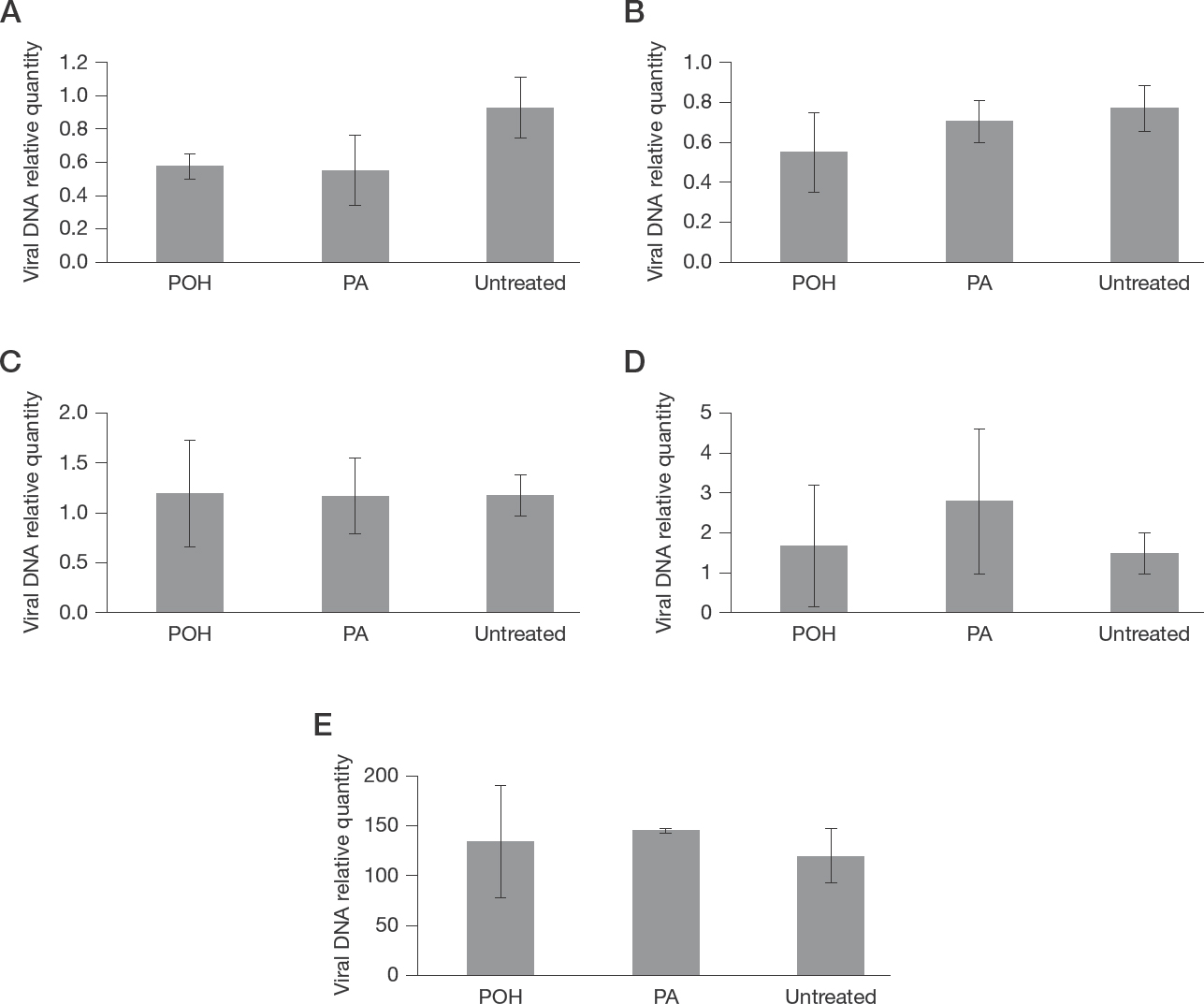
Monoterpene effect on viral DNA polymerase
Western Blot
We further investigated the viral protein profile of infected cells treated or not with the compounds at different time points (3, 6, 12 and 24 h post-infection). Both monoterpenes did not change total protein profile of infected cells (Figure 4).

Western blot analysis for total HSV-1 proteins
Monoterpene Effect on viral Particle Release
Next it was relevant to assess if the compounds were blocking viral particles release from Vero cells. We carried out a plaque reduction assay, and just before lysing the cells we collected the supernatant to titre the virus release and further washed the monolayer and Vero cells were lysed with three cycles of freezing and thawing to collect cell-bound viruses. After 24 h of treatment, 50 μM POH caused a higher content of cell-bound virus plaques than untreated control, but lower extracellular particle forming units when compared with the untreated samples. However, after 24 h treatment, 50 μM PA caused low intra and extracellular viral content in comparison with untreated control (Figure 5). As expected we observed that POH or PA were capable of reducing the amount of particle forming units on the extracellular media, thus confirming that both compounds reduce virus yield, as observed on antiviral activity assays. Moreover, samples treated with POH had a large number of PFUs on the intracellular media, an indication that POH affects HSV-1 viral release from infected cells.

HSV-1 plaque reduction assay to evaluate viral particle egress from infected cells treated or not (untreated) with POH and PA for 24 h (n=3)
Discussion
Monoterpenes are important class of compounds with pleiotropic biological properties [13,14,17,19,20,23]. Additional data showed that combined monoterpenes [31], limonene metabolites and diterpenes isolated from the Brazilian marine alga Dictyota and Plocamium genus presented antiviral activity to HSV-1 and HIV [32–35], and also against a highly pathogenic and neurovirulent RNA mosquito-borne West Nile flavivirus [31]. Herein we present evidence that POH and PA may also be considered as potential antiviral compounds.
Hence, we confirmed the antiviral activity of POH and PA on Vero cells and high selectivity index (CC50) values for POH (1,748 μM) and PA (1,818 μM) was a clear indication that both compounds were not toxic. A promising antiviral agent ought to present high CC50 value but low EC50 value. Interestingly, both compounds presented high relative in vitro antiviral effectiveness compared with ACV, a standard drug but with relative neurotoxicity and inability to prevent neurological sequelae among HSV-1 encephalitis [36]. POH antiviral activity was further tested in a low MOI (0.1) to verify long-term action on infected cells to confirm its antiviral activity rather than action upon the formation of defective particles due to viral particle competition. Such experiments showed that POH exerted a strong HSV-1 antiviral activity during the first 48 h infection, whereas PA was more efficient later (72 h). It was further appropriate to verify whether POH and PA exerted any relevant effect upon viral DNA and RNA expression of specific HSV-1 genes (ICP0, ICP34.5, UL20, ICP4, TK, gC), and also to evaluate whether POH and PA were directly inhibiting viral protein production. Both POH and PA did not alter the expression of viral RNA and DNA. We postulate that the antiviral activity of monoterpenes was not related to inhibition of the replication cycle, but probably by interfering with egress and maturation of HSV-1 viral particles. Interestingly UL47 (or VP13/VP14) is considered the main viral structural protein regulator for HSV-1 nuclear virus release [37]. Therefore, it is not ruled out that both POH and PA may be exerting an effect upon UL47 which further affects HSV-1 release from Vero cells [38,39]. We present evidence that POH and PA have low cytotoxicity but high antiviral activity against HSV-1 without any interference on viral DNA, RNA expression and protein profile therefore indicating an effect on late stages of HSV maturation and the release of infective viral particles.
Most antivirals target viral proteins of a specific viral type; however, virion lipids are not encoded in the viral genome and maintain their rearrangements during fusion. Therefore, antivirals that inhibit lipid rearrangements could establish a barrier to resistance and have broad-spectrum activity [11]. POH exert remarkable biological properties modifying structure and dynamics of the plasma membrane bilayer continuously diffusing across the membrane and eventually binding to an intracellular structure [16,22,40]. Moreover, POH also exerts multiple antioxidant functions by inducing several classes of genes implicated in detoxifying reactive oxygen species [16] including metabolic changes that occur during the onset of endoplasmic reticulum stress either by protecting human renal cells against ischaemia/reperfusion injury [41] or inducing cytotoxicity in human glioma cells through the ERS pathway via GRP78 protein [42]. Phase I and II trials with oral POH treatment caused undesirable adverse effects such as gastrointestinal toxicity, nausea, vomiting [43–45]. However, Phase I/II clinical trial with intranasal POH treatment regimen on a continuous basis showed efficient therapeutic activity without long-term severe adverse events even following daily administration for more than 4 years continuous treatment of patients with brain tumour [46,47]. The intranasal POH is a non-invasive strategy for delivering therapeutic agents to the brain because of the unique anatomic connection provided by the olfactory and trigeminal nerves. These nerves connect the nasal mucosa and the central nervous system rapidly targeting parenchymal brain tissue, spinal cord and/or cerebrospinal fluid (CSF) within minutes using an extracellular route through perineural channels bypassing the blood–brain barrier and minimizing systemic exposure [48,49]. Interestingly, the trigeminal nerve is the main nerve used by HSV-1 to establish latency. In latent HSV-1 infection, the viral genome is present but only one set of transcripts is formed, and no viral proteins are detectable. Yet, virus can reactivate from latency and acute infection ensues after replication and synthesis of viral proteins with whole virion being produced and released [3]. In summary, it is conceivable to envisage a promising therapeutic strategy by using intranasal delivery of POH as antiviral agent or bioactive POH hybrid molecules acting on virion lipid tails and genome thereby inhibiting maturation, fusion and HSV-1 infectivity responsible for intermittent reactivation and neurovirulence.
Footnotes
Acknowledgements
This study was supported by grants from CAPES (Coordenação de Aperfeiçoamento de Pessoal de Nível Superior), FAPERJ (Fundação de Amparo a Pesquisa do Rio de Janeiro) and CNPq (Conselho Nacional de Desenvolvimento Científico e Tecnologico). CPM had a fellowship from CAPES attached to PPBI Science and Biotechnology postgraduate programme. We are grateful to Samara Braga do Nascimento and Hania Cristina Rosado Silveira (both from Departamento de Biologia Celular e Molecular, Instituto de Biologia, Universidade Federal Fluminense, Niteroi, Brazil) for technical assistance.
The present manuscript has not been published previously except in Portuguese in the form of an academic thesis (CPM). The present version was approved by all authors.
The authors declare no competing interests.
