Abstract
Background
We aimed to project the 10-year future incidence of cardiovascular disease (CVD) and model several intervention scenarios based on a multi-site Asian HIV-positive cohort.
Methods
Analyses were based on patients recruited to the TREAT Asia HIV Observational Database (TAHOD), consisting of 21 sites in 12 countries. Patients on triple antiretroviral therapy (ART) were included if they were alive, without previous CVD, and had data on CVD risk factors. Annual new CVD events for 2019–2028 were estimated with the D:A:D equation, accounting for age- and sex-adjusted mortality. Modelled intervention scenarios were treatment of high total cholesterol, low high-density lipoprotein cholesterol (HDL) or high blood pressure, abacavir or lopinavir substitution, and smoking cessation.
Results
Of 3,703 included patients, 69% were male, median age was 46 (IQR 40–53) years and median time since ART initiation was 9.8 years (IQR 7.5–14.1). Cohort incidence rates of CVD were projected to increase from 730 per 100,000 person-years (pys) in 2019 to 1,432 per 100,000 pys in 2028. In the modelled intervention scenarios, most events can be avoided by smoking cessation, abacavir substitution, lopinavir substitution, decreasing total cholesterol, treating high blood pressure and increasing HDL.
Conclusions
Our projections suggest a doubling of CVD incidence rates in Asian HIV-positive adults in our cohort. An increase in CVD can be expected in any ageing population, however, according to our models, this can be close to averted by interventions. Thus, there is an urgent need for risk screening and integration of HIV and CVD programmes to reduce the future CVD burden.
Introduction
With the ageing of HIV-positive populations [1,2], comorbidities such as cardiovascular disease (CVD) are becoming more important in terms of clinical practice and public health policy. Studies have suggested that people living with HIV (PLHIV) have a higher risk of CVD events compared with the general population. For instance, studies from North America and the US found a 21% and 48% elevated risk of myocardial infarction, respectively, in those with HIV [3,4]. Furthermore, an Italian study found a twofold increased risk of CVD in PLHIV [5]. The increased risk may be in part attributable to HIV-specific factors, such as low CD4+ T-cell count, high HIV viral load and toxic effects of anti-retroviral therapy (ART), and to traditional risk factors such as smoking, hypertension, dyslipidaemia and diabetes [6].
Limited data exist on the burden of CVD in PLHIV in the Asian region and few studies have investigated actual numbers of CVD. In a small sample of hospital-admitted HIV-positive patients in India, cardiovascular abnormalities were observed in over half of the patients [7]. Furthermore, a large Taiwanese study showed that 1.2% of newly diagnosed HIV patients had a stroke during an average follow-up time of 4.6 years, which was significantly higher than what they found in HIV-negative controls [8].
Other studies suggesting a considerable burden of CVD comes from data on CVD traditional risk factors, which are highly prevalent in Asian PLHIV [9–11]. When information on risk factors is available, a person's future CVD risk can be estimated by risk prediction algorithms, such as the Framingham Risk Score (FRS), the Atherosclerotic Cardiovascular Disease (ASCVD) risk score, or – specifically developed for HIV-positive populations – the Data collection on Adverse events of Anti-HIV Drugs (D:A:D) risk equation. Using the FRS, 10% of a Thai HIV-positive study population had high risk of developing coronary heart disease in the next 10 years [12]. In Indian PLHIV, the D:A:D risk equation estimated a high 5-year CVD risk in 2.5% of the population [13] and the ASCVD risk score revealed a high lifetime CVD risk in 78% of those aged 40–59 years [14].
The emergence of cardiovascular comorbidity in Asian PLHIV may be challenging, particularly in regions with limited resources and lower health care accessibility [15]. This highlights a need to plan and implement treatment programmes that link HIV and CVD management. In the absence of large-scale data on actual numbers on CVD, risk prediction algorithms can be used to estimate the future incidence of CVD and in turn guide policy making and intervention planning. We aimed to project the incidence of new CVD events in PLHIV in the Asian region over a 10-year period and to model the impact of various intervention scenarios.
Methods
The study population consisted of patients enrolled in the TREAT Asia HIV Observational Database (TAHOD), a prospective observational cohort of the International Epidemiology Databases to Evaluate AIDS which has previously been described in detail [16,17]. Briefly, HIV-positive adults (≥18 years) were enrolled from 2003 onward at 21 clinical sites in 12 Asian countries. Data transfers occur biannually. Institutional Review Board approvals were obtained at all participating sites, the data management and analysis centre (Kirby Institute, UNSW Sydney, Australia), and the coordinating centre (TREAT Asia/amfAR, Bangkok, Thailand).
For this study, patients in the March 2018 data transfer were included if they were on triple ART for at least 3 months, were alive at the time of data transfer, had visited the clinic in the past 12 months, and had data available that were required for the model by which we estimated new CVD incidence rates. Patients were excluded if they were known to have experienced a CVD event previously.
CVD Event Incidence Model
The model was used to project incidence rates for new CVD events in 1-year increments from 2019-2028. We chose the D:A:D risk equation [18] to calculate CVD risk since it is the only available prediction tool that allows calculation of 1-year CVD risk, which permitted us to incorporate the effect of ageing and, based thereon, estimate the number of events per year. In accordance with the D:A:D CVD risk equation, CVD events were defined as myocardial infarction, stroke, invasive cardiovascular procedures or deaths due to CVD [18].
Our model follows Equation 1:
Where:
Where:
t=2018, 2019,…, 2027;
Thus, the estimates of future CVD events were based on this model which, along with individual CVD risk, considers mortality and CVD events in previous years.
Mortality risk
To project the number of patients at risk of CVD, we needed to estimate the number of people at risk per yearly interval (that is, the number of people still alive). Trends in mortality were assumed to continue in line with those observed in TAHOD up to 2018. We thus estimated sex and age-adjusted mortality risk based on data of all patients who initiated ART by performing Poisson regression. Overall mortality rates from 2019-2028 were then estimated for all patients included to model CVD projections. To do so, we time-updated age per yearly interval and calculated the annual risk of dying per patient with the retrieved Poisson regression coefficients.
Since CVD events can be fatal, we assumed that the annual number of CVD events partly overlaps with the annual number of deaths. Therefore, we corrected overall mortality for CVD-related mortality. According to estimations based on a large international sample of HIV populations, 11% of yearly overall mortality is CVD related [19]. Consistent with this evidence, we assumed that 89% of the estimated overall mortality in our sample was non-CVD-related.
Cardiovascular disease risk
To model CVD risk, we used the most recent available data for each CVD risk factor. Specifically, we used sex, age, diabetes (defined as one lab result of fasting plasma glucose >7 mmol/l), systolic blood pressure (in mmHg), high-density lipoprotein cholesterol (HDL) and total cholesterol level (in mmol/l), current use of abacavir (ABC), current use and duration of use (in years) of indinavir (IDV) and lopinavir (LPV), and current and ex smoking status. Data were also collected on CVD family history, if known. For descriptive purposes we reported the prevalence of high levels of systolic blood pressure (≥140 mmHg) and total cholesterol (≥5.2 mmol/l), and low levels of HDL (<1 mmol/l). Although other risk equations, such as the FRS [20] and the ASCVD [21] risk score, could not be applied in our CVD incidence model, we did use these scores to calculate 10-year CVD risk and compare it with 10-year CVD risk according to the D:A:D risk equation.
The variables of age and ART use were time updated for each yearly increment. In doing so, we assumed that patients remained on their current ART regimen throughout the 10 projected years. As such, if patients were currently using LPV or IDV, the total treatment exposure of these drugs was increased by 1 year for each yearly increment. Furthermore, it was assumed that unfavourable effects of ABC use would not diminish immediately; therefore, if a patient recently ceased using ABC (that is, <6 months prior), we coded that patient as using ABC in the first projected year, but not in the following years. Taken together, these assumptions allowed for increasing CVD risk over time.
Since family history of CVD was not recorded consistently in all clinics and the majority of these data was unavailable, we imputed missing data on this variable with a weight based on the prevalence of CVD family history in those patients for whom these data were reported.
Statistical methods
The estimated incidence rate of CVD events was projected per annum for 2019–2028. As with all projection models our model is based on assumptions and, as such, our projections are subject to a degree of uncertainty. Therefore, we conducted three sensitivity analyses:
In the main analysis patients were excluded if they did not have data available on CVD risk factors. Similar to what we did for family history of CVD in the main analysis, missing data on other CVD risk factors were imputed with a weight based on the prevalence or the median of the respective factors in those patients for whom these data were reported. Thus, we based our projections on a larger patient sample.
The D:A:D equation is based on a study cohort from high resource settings (that is, Australia, Belgium, Denmark, France, Italy, the Netherlands, Spain and the United Kingdom) and has not been validated in resource-limited settings. According to the Global Burden of Disease (GBD) study 2017 [22], CVD rates in the general population of TAHOD countries (Cambodia, China, India, Indonesia, Japan, Malaysia, Philippines, Singapore, South Korea, Taiwan, Thailand, Vietnam) are around 1.46-fold greater than in the D:A:D countries (Additional file 2) [22]. Assuming that the effect of HIV infection and ART on CVD risk is the same in D:A:D and TAHOD countries, we therefore adjusted the D:A:D risk equation estimated CVD risk by 69%.
Our projections are based on the most recent clinical assessment of CVD risk factors available for each patient and, apart from the time-updated variables age and duration on ART, they are assumed to remain stable over time. However, CVD risk factors may improve over time due to interventions that are implemented. In particular, availability of newer, less toxic types of antiretroviral drugs may be increasingly prescribed as they become more affordable for those in the Asian lower income regions. Furthermore, future CVD rates may be affected by interventions applied to the individual or public level, such as pharmacological treatment of high blood pressure and dyslipidaemia or smoking cessation interventions. To evaluate the impact of such interventions if they were to be successfully applied, we project CVD events while simulating various hypothetical scenarios. We accounted for scenarios of pharmacological interventions in which patients with high blood pressure, low HDL or high total cholesterol were treated to maintain more favourble levels of 130 mmHg, 5.2 mmol/l or 1.0 mmol/l, respectively. We accounted for scenarios of single drug substitutions in which patients on LPV or ABC were switched to another drug. As such, if patients were on LPV, total treatment exposure did not increase any further. We did not include IDV substitution due to the low number of IDV in our cohort. We accounted for a scenario of smoking cessation, that is, patients who were current smoker became ex-smoker. Smoking cessation does not have an immediate impact on risk. In the D:A:D study, the incidence rate ratio (IRR) compared with current smoking decreased to 0.68 at >3 years after smoking cessation, which was similar to the IRR of ex-smokers at baseline [23]. We thus incorporated a gradual reduction of smoking-associated CVD risk in the first 3 projected years until the CVD risk was equal to that of ex-smokers in the fourth projected year. We additionally modelled a scenario in which all interventions were combined.
Data management and statistical analyses were performed using SAS software version 9.4 (SAS Institute Inc., Cary, NC, USA) and Stata software version 14.1 (StataCorp, College Station, TX, USA).
Results
Study population
Of the 3,703 included patients who had sufficient data to estimate future CVD event rates, 69% were male, median age was 46 (IQR 40–53) years, and median time since ART initiation was 9.8 years (IQR 7.5–14.1). Furthermore, 22% had high systolic blood pressure, 19% had diabetes, 37% had high total cholesterol and 20% had low HDL. Median 10-year CVD risk score was higher when calculated by the FRS (7.1%, IQR 2.7–14.6) and lower when calculated by the ASCVD risk score (2.4%, IQR 0.7–6.7), compared to the D:A:D risk equation (4.6%, IQR 2.1–10.3).
Due to missing test data on one or more CVD risk factors, we had to exclude 2,515 (40%) patients. Specifically, data was missing on total cholesterol (11%), HDL (27%), blood pressure (16%), blood glucose (16%) and smoking status (13%). Compared with the included patients, excluded had similar sex distribution, were younger, had shorter time since ART initiation, had lower median CD4+ T-cell count, and had higher median HIV viral load. The prevalence of current LPV and ABC use were both lower in the excluded patients. Around 75% of excluded patients were from lower-income countries compared with 30% of included patients. Tables 1 and 2 provide a more detailed description of included and excluded patients.
Patient characteristics
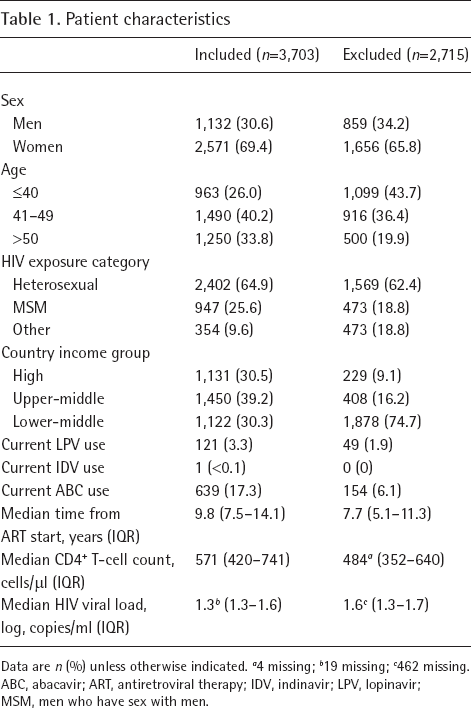
Data are n (%) unless otherwise indicated.
4 missing;
19 missing;
462 missing.
ABC, abacavir; ART, antiretroviral therapy; IDV, indinavir; LPV, lopinavir; MSM, men who have sex with men.
CVD risk profile of included patients (n=3,703)

Data are n (%) unless otherwise indicated. The most recent cardiovascular disease (CVD) risk factors are reported in this table. ASCVD, Atherosclerotic Cardiovascular Disease; D:A:D, Data collection on Adverse events of Anti-HIV Drugs; FRS, Framingham Risk Score; HDL, high-density lipoprotein cholesterol level.
Mortality in TAHOD
Of all 9,155 patients in TAHOD who initiated ART, 466 patients died over 73,659 person-years (pys). Mortality rates were higher in men than women (IRR 1.54, 95% CI 1.23, 1.92) and older patients (IRR 20.96, 95% CI 8.74, 50.28 per log years increase).
Projections of new CVD events
For 2019, our projections estimated approximately 12 deaths in our cohort, leaving 3,691 patients at risk of having a new CVD event. In 2028, after correcting for estimated mortality and CVD events in previous years, we expected that 3,178 patients will be at risk for a new CVD event. Our projections suggest that over the 10-year period 2019 to 2028, approximately 354 patients in our cohort (9.6%) will have experienced a new CVD event.
Overall, the annual rate of new CVD events was projected to double from 730 to 1,432 events per 100,000 pys from 2019 to 2028 (Figure 1). Thus, the cumulative CVD event rate is estimated to reach 10,420 per 100,000 persons in 2028. Sensitivity analysis 1 imputed missing data on CVD risk factors and, as such, additionally included all patients who were excluded from the main model due to missing data. The estimated incidence rate in this sensitivity analysis was lower than in the main model, with estimated incidence rates of 590 events per 100,000 pys in 2019 and 1,152 events per 100,000 in 2028 (Figure 1) which would result in a cumulative CVD event rate of 8,406 per 100,000 patients. Sensitivity analysis 2 adjusted predictions for the difference between D:A:D and TAHOD, resulting in decreased estimated incidence rates of 503 events per 100,000 pys in 2019 and 988 events per 100,000 in 2028 (Figure 1) which would mount up to a cumulative CVD event rate of 7,190 per 100,000 patients.

Estimated CVD event rate for 2019–2028
Simulation of interventions
In scenarios where we simulated pharmacological interventions, the number of new CVD events in 2028 reduced to 1,337, 1,377 or 1,409 events per 100,000 pys if patients were treated for high total cholesterol, high blood pressure or low HDL, respectively, compared with the 1,432 events per 100,000 pys that we projected in a scenario where no changes to the CVD risk factors would occur, apart from increasing age, over the 10-year time period (Figure 2A). Alternatively, in scenarios where we simulated antiretroviral drug substitutions, CVD events in 2028 were reduced to 1,226 or 1,334 events per 100,000 pys, when substituting ABC or LPV, respectively (Figure 2B). Lastly, in scenarios where we simulated smoking cessation CVD events were reduced to 1,193 per 100,000 pys (Figure 2C). Combining all interventions reduced the number of new CVD events to 837 per 100,000 pys in 2028 (Figure 2C).

Estimated CVD event rate for 2019–2028 after pharmacological, drug substitution, and smoking cessation and combined interventions
Discussion
Our projections suggest a rising and substantial CVD burden for the Asian HIV-positive population, with almost a doubling of new CVD events in the next decade. Estimates suggest that by 2028 about 10% of individuals in our cohort will have experienced a CVD event. Simulating various intervention scenarios showed that the number of CVD events can be greatly reduced by smoking cessation, antiretroviral drug substitutions, and, to a lesser extent, pharmacological interventions.
Our projections indicate that the CVD incidence in Asian PLHIV may rise from around 730 to 1,432 events per 100,000 pys from 2019 to 2028, should no changes occur in patients’ CVD risk profiles, apart from increasing age and exposure to ART regimens. No large-scale data is available on actual CVD incidence in Asian HIV-positive populations. However, the Global Burden of Disease (GBD) Study provides extensive data on CVD estimates in the general population [22]. According to these estimates, considerable variation exists across the Asian countries with combined incidence rates of ischaemic heart disease and stroke ranging from the lowest in Thailand (177 per 100,000 persons) to the highest in China (313 per 100,000 persons).
Our CVD incidence model was based on the D:A:D equation, which is unique in that it is developed for a HIV-positive population and it can provide a 1-year risk score, next to a 10-year risk score. Risk scores are known to vary between risk prediction tools, for instance, studies have reported the FRS to predict higher 10-year risk scores compared with the D:A:D [12,24]. When comparing 10-year risk scores across tools, we also found a higher median score using the FRS (7.1%) compared with the D:A:D (4.6%), whereas the ASCVD was somewhat lower (2.4%). As the D:A:D risk score was in the range of risk as identified by the other tools, the D:A:D risk score can be considered a ballpark figure of CVD risk in this study population.
It is well-established that ageing is one of the main drivers of CVD in populations [25] and the doubling of CVD incidence we projected can be considered largely a result of ageing over the next decade. In 2018, our study population already displayed high proportions of other CVD risk factors, including elevated blood pressure, unfavourable lipid levels, diabetes and smoking, which corroborates other findings on PLHIV in Asia [9–11]. Furthermore, relatively common was the use of ABC, which can be detrimental to cardiovascular health [26,27]. Strikingly, the intervention scenarios we modelled in our study showed that the majority of CVD events in the upcoming 10 years can be averted by implementing interventions that target such CVD risk factors, despite the inevitable effect of ageing.
According to our findings, the largest gain in CVD incidence reduction can be made by smoking cessation, followed by drug substitutions and pharmacological interventions such as dyslipidaemia treatment and anti-hypertensive medication. Smoking cessation and drug substitution may be favourable interventions since they can improve metabolic profiles as well [28–30]. However, caution should be taken with drug substitutions as substitute drugs could affect virological response or cause toxicity.
The extent to which events can be reduced with interventions mostly depends on the prevalence of risk factors in the population, which varies per setting and may change over time. This is illustrated by an earlier TAHOD study that investigated 5-year CVD risk [31]. In comparison with that study, the CVD risk profile in the TAHOD cohort has become somewhat more favourable. For instance, in the current study population we found a considerably lower prevalence of LPV use (3% versus 13%) and high total cholesterol (37% versus 46%).
It should be noted that past and future changes to the population may occur and at different rates depending on the country. For instance, newer drugs that are not detrimental to cardiovascular health may be increasingly prescribed, a trend which is likely to transpire earlier in the higher-income countries. Research also indicates increasing CVD awareness and control in high-income countries, but not in low- and middle-income countries [32]. In addition, pharmacological treatment for CVD risk factors is underutilized in low and middle-income countries [33,34], which may be due to the cost of treatment in these settings [35] and local standards of care. Thus, actual reduction in CVD incidence largely depends on the extent to which resources will be made available for the detection, prevention and treatment of CVD risk.
Several limitations should be noted when interpreting the results of this study. First, as with other risk prediction tools, there may remain inaccuracies when estimating individual future risk [36]. Nonetheless, they can be useful to guide clinical decision making and provide particularly valuable information when applied to the population level, as was done in this study. Second, we applied the D:A:D CVD risk equation, which is based on a mostly European cohort of HIV-positive individuals. Our population was indeed different to the D:A:D cohort in terms of sociodemographic characteristics, time on ART, and CVD risk profiles [18]. Furthermore, the study population was not necessarily representative of all PLHIV in Asia, especially since incomplete data ascertainment for CVD risk factors led to exclusion of many patients who were from lower-middle income countries. Two sensitivity analysis in which we used GBD country data to adjust projections for potential regional differences in CVD in the D:A:D countries and TAHOD countries and imputed missing data on CVD risk factors suggest our findings may overestimate actual CVD risk when generalizing to the wider region. It is possible that patients in our cohort were assessed on CVD risk factors if clinicians expected patients were at higher risk of CVD. Since the majority of included patients were from higher income countries, it is likely that the projections or more reliable for high-income regions in Asia.
Our projections suggest that CVD incidence rates in Asian HIV-positive adults will double in the next decade, which, according to our models, can be close to averted with combined interventions. This illustrates the urgent need for improved CVD risk screening among PLHIV to support timely intervention and prevent the future burden of CVD in this population. More research is warranted to investigate cost-effective measures to screen and control CVD in settings where resources may be limited. Additional research and other efforts are required to determine optimal approaches to the integration of HIV and CVD programmes and the development of joint HIV CVD management guidelines.
Footnotes
Acknowledgements
The TREAT Asia HIV Observational Database is an initiative of TREAT Asia, a programme of amfAR, The Foundation for AIDS Research, with support from the U.S. National Institutes of Health's National Institute of Allergy and Infectious Diseases, the Eunice Kennedy Shriver National Institute of Child Health and Human Development, the National Cancer Institute, the National Institute of Mental Health, and the National Institute on Drug Abuse, as part of the International Epidemiology Databases to Evaluate AIDS (IeDEA) [grant number: U01AI069907]. The Kirby Institute is funded by the Australian Government Department of Health and Ageing, and is affiliated with the Faculty of Medicine, UNSW Sydney. The PhD of RB has been supported through an Australian Government Research Training Program Scholarship. The content of this publication is solely the responsibility of the authors and does not necessarily represent the official views of any of the governments or institutions mentioned above.
Further acknowledgements can be found in Additional file 3.
The authors declare no competing interests.


