Abstract
Background
Polypharmacy (use of ≥ five medications) increases the risk of drug-drug interactions and can lead to negative health outcomes. This study aimed to review the medications of people living with HIV (PLWH) and HIV-negative controls in the POPPY study and evaluate the frequency of polypharmacy and potential drug-drug interactions (PDDIs).
Methods
PDDIs between non-antiretroviral (ARV) drugs were analysed using the Lexicomp® database, and PDDIs between non-ARV and ARV drugs using the Liverpool drug interaction database. Between-group differences were assessed using χ2, Mann-Whitney U and Kruskal-Wallis tests.
Results
This analysis included 698 PLWH ≥50 years, 374 PLWH <50 years and 304 HIV-negative controls ≥50 years. The prevalence of polypharmacy was 65.8% in older PLWH, 48.1% in younger PLWH and 13.2% in the HIV-negative group. When ARVs were excluded, 29.8% of older PLWH and 14.2% of younger PLWH had polypharmacy. The prevalence of ≥1 PDDI involving non-ARV drugs was 36.1%, 20.3% and 16.4%, respectively, in older PLWH, younger PLWH and HIV-negative controls. In PLWH the prevalence of ≥1 PDDI involving ARV and non-ARV drugs was 57.3% in older PLWH and 32.4% in younger PLWH.
Conclusions
Polypharmacy and PDDIs involving non-ARV/ARV drugs and non-ARV/non-ARV drugs were common among older PLWH, highlighting the need for increased awareness and additional research on all types of PDDI.
Introduction
With the advancement of antiretroviral (ARV) therapy, people living with HIV (PLWH) are living longer [1]. The clinical focus has increasingly shifted from the management of HIV-related complications to managing comorbidities [2], polypharmacy and drug–drug interactions that may arise from medication use. There are many descriptions of the term polypharmacy, but it has commonly been defined in the literature as the regular use of five or more medications [3,4]. Polypharmacy among PLWH specifically has been identified as a clinical concern [5,6]. A correlation between the number of non-ARV drugs and adverse health outcomes has been demonstrated in both older PLWH and uninfected older individuals [7]. Although polypharmacy is often unavoidable in older patients, including PLWH, the potential negative consequences of multi-medication intake must be carefully considered. These include decreased adherence to treatments [8], increased risk of hospitalization, increased risk of adverse drug reactions or additive drug toxicities [9–12], increased costs [13], pill burden [14] and increased risk of geriatric syndrome, which is a collection of conditions traditionally associated with older age, including falls, delirium, incontinence and decreased nutrition status [15,16]. A major concern about polypharmacy is the risk of clinically significant potential drug–drug interactions (PDDIs) [17,18].
PDDIs are a well-recognized concern in PLWH [19–21] as ARVs can be inhibitors (for example, ritonavir, cobicistat), inducers (for example, efavirenz, nevirapine) or substrates (for example, rilpivirine, maraviroc and the newer agents bictegravir and doravirine) of the cytochrome P450 (CYP450) system isoenzymes [22,23]. Drugs may alter the function of enzymes (for example, UDP glucuronosyltransferases [UGTs]), transporters (for example, P-glycoprotein [P-gp], organic anion/cation transporters) etc. Interactions involving ARVs may also occur with drugs that alter gastric pH (for example, proton pump inhibitors), and drugs that act as chelating agents (for example, calcium and magnesium containing compounds) may affect ARV absorption [23,24]. The consequences of such PDDIs may be serious toxicities or a reduction in ARV drug concentrations that leads to virological failure and development of resistance to ARV therapy [25].
Although traditionally the focus has been on ARV/non-ARV PDDI, PDDI involving non-ARVs in PLWH are also of emerging concern, in view of the high levels of non-ARV polypharmacy encountered in this population. Our aim was to analyse the prevalence of PDDIs between both ARV/non-ARVs and non-ARVs/non-ARVs in three groups of people: PLWH ≥50 years of age, younger PLWH and people without HIV ≥50 years.
Methods
The Pharmacokinetic and Clinical Observations in People Over 50 (POPPY) study is a multicentre, prospective, observational study, with data collection taking place over a 3-year period from 2013–2016 [26]. Data from 1,376 participants were collected: PLWH ≥50 years (n=698), PLWH 20–49 years (n=374) and HIV-negative individuals ≥50 years (n=304). The study comprises a cohort that represents the overall clinic population, that is, did not over-represent those with more complex medical needs who either attended clinic more frequently or those who were more regular participants in research studies. Where possible, HIV-negative participants were selected from similar underlying populations. Participants were recruited across eight clinical centres in the UK and Ireland. Ethical, regulatory and informed consent, if indicated, was obtained from each study site.
Polypharmacy assessment
Information on medication usage was obtained at study entry by trained study staff; additional information on ARV usage among PLWH was obtained through linkage with the UK Collaborative HIV Cohort (UK CHIC) study [27]. Analyses were performed on data obtained at the baseline visit. For the purpose of this analysis the following medications were included: ARVs, non-ARV prescribed drugs, over the counter medications, dietary and vitamin supplements and homeopathic medications (recreational drugs were not included in the analysis). For both ARV and non-ARV combinations, individual components were counted and analysed separately, for example, Co-codamol® was inputted as paracetamol and codeine. Certain combinations, whose individual components could not possibly be determined, for example, ‘Multivitamin’ were counted as one medicine. Medications were recorded and classified based on the World Health Organization (WHO) Anatomical Therapeutic Chemical Classification (ATC) system. Certain medicines were reclassified, for example, aspirin was only included in the blood (anti-platelet) category of medications. Polypharmacy was defined as five or more medications in line with other published studies [3,28,29].
Potential drug–drug interactions
For PLWH, the University of Liverpool HIV drug interactions database was used to determine interactions between ARV and non-ARV medications. The interaction checker classifies specific ARV and non-ARV combinations as being contraindicated (colour code red), potentially clinically significant that are likely to require additional monitoring, alteration of drug dosage or timing of administration (colour code amber) or having no interaction (colour code green) [30]. The option to check interactions between ARVs was not used, as it was assumed these interactions were intentional, for example, darunavir and ritonavir, where ritonavir is used a pharmacokinetic enhancer.
For both HIV-positive and negative participants, the Lexicomp® database was used to check interactions between non-ARV/non-ARV medications. Lexicomp® classifies interactions as class A (no known interaction), B (no action needed), C (monitor therapy), D (consider therapy modification) and X (avoid combination) [31]. Examples in class X include coadministration of opioids with other opioids or co-administration of domperidone with quinolones. In this study the interactions categorized in class D were sub-divided into those requiring therapy modification and those that could be managed by separating administration times, for example, levothyroxine and calcium. All interactions were included, except interactions between two drugs that were duplicated due to different mechanisms of interaction, for example, two drugs which may both lower seizure threshold and prolong QTc interval. These combinations were only included once with the highest severity assigned to the interaction.
Statistical analysis
Proportions, frequencies, means and ranges were used for the descriptive analyses. Patient characteristics between groups were compared using Pearson's c 2 tests for categorical variables and non-parametric Kruskal–Wallis tests for continuous variables. Multivariable logistic regression models were used to assess the association of PDDI between non-ARV and non-ARV drugs and PDDI between ARV and non-ARV drugs with HIV group after adjusting for gender and race and current combination antiretroviral therapy (cART). Statistical analysis was performed using Microsoft Excel® and Stata 15 (Stata Statistical Software: Release 15; StataCorp., College Station, TX, USA).
Results
The characteristics of the study population are illustrated in Table 1. Reflecting the characteristics of the older HIV-positive population in the UK and Ireland, the older group of PLWH were predominantly male, men who have sex with men (MSM) and of White ethnicity; the younger group of PLWH and older HIV-negative controls were broadly similar in terms of these demographic characteristics.
Characteristics of the study population
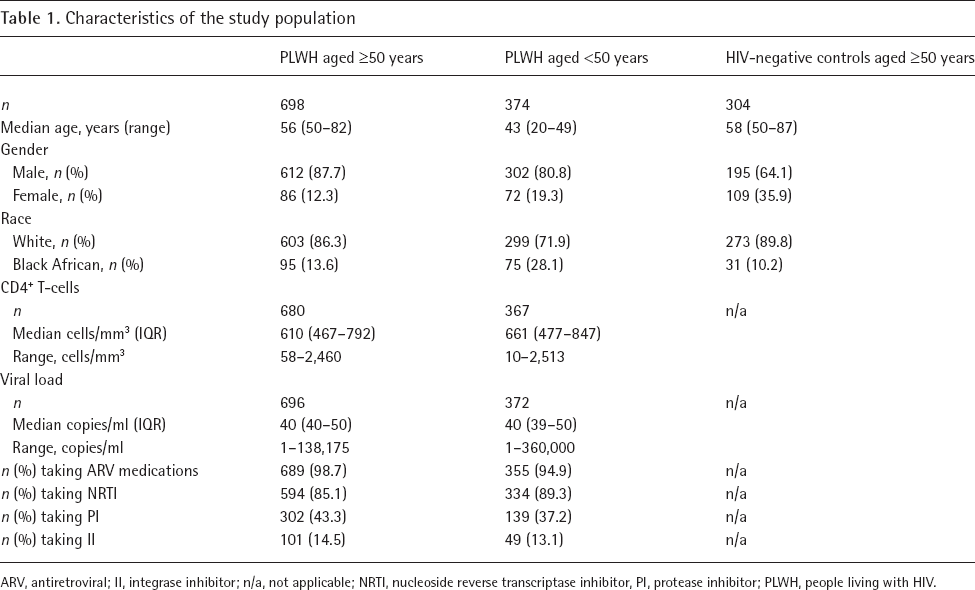
ARV, antiretroviral; II, integrase inhibitor; n/a, not applicable; NRTI, nucleoside reverse transcriptase inhibitor, PI, protease inhibitor; PLWH, people living with HIV.
Medication characteristics
Conventional medications
In PLWH ≥50 years ATC class C, the cardiovascular system, was the most common therapeutic class accounting for 19.7% (n=479) of the 2,426 co-medications in this group. ATC class N, drugs used to treat the nervous system, accounted for 16.8% (n=408) of all co-medications. ATC Class J, anti-infectives for systemic use, accounted for 10.8% (n=261) and ATC Class A, drugs for the alimentary tract and metabolism made up 8.9% (n=215) of all co-medications.
In PLWH <50 years, ATC class N was the most common therapeutic class, accounting for 20.1% (n=146) of the 726 co-medications. ATC class J made up 10.2% (n=74) of all co-medications, and ATC Class R, respiratory medicines, accounted for 9.4% (n=68).
In the HIV-negative group ≥50 years ATC class C was the most common therapeutic class, accounting for 17.9% (n=107) of the 598 co-medications. ATC Class N made up 8.5% (n=51) of all co-medications and ATC Class A accounted for 7.9% (n=47).
Non-conventional medications
A large proportion of all three groups were taking non-conventional medications (for example, vitamins, minerals, herbal and homeopathic remedies, dietary supplements). Among PLWH ≥50 years 228 (32.7%) were taking at least one non-conventional medication, compared to 91 (29.9%) of those in the HIV-negative group ≥50 years and 127 (34%) of PLWH <50 years.
Polypharmacy
The numbers of people with polypharmacy in each of the three groups is illustrated in Table 2.
Numbers of medications and interactions in the three groups

ARV, antiretroviral; n/a, not applicable; PDDI, potential drug–drug interaction; PLWH, people living with HIV.
In PLWH ≥50 years the group were taking a median of six medications including ARVs (IQR 4–9), PLWH <50 years were taking a median of four medications including ARVs (IQR 3–6) and the HIV-negative group ≥50 years were taking a median of one medication (IQR 0–3; P=0.001).
PLWH ≥50 years had more polypharmacy than the other two groups: 459/698 (65.8%) older PLWH had polypharmacy compared with 180/374 (48.1%) younger PLWH and 40/304 (13.2%) HIV-negative people ≥50 years (P=0.001).
When ARVs were excluded from the analysis in the HIV-positive groups, 208/698 (29.8%) of older PLWH had polypharmacy. This is considerably more than the HIV-negative group ≥50 years, of whom 13.2% had polypharmacy. In younger PLWH 53/374 (14.2%) had polypharmacy when ARVs were excluded.
Potential drug–drug interactions
Table 2 reports the frequency of PDDI in the studied population. A significantly higher proportion of older PLWH had at least one PDDI involving both an ARV/non-ARV than younger PLWH, and a non-ARV/non-ARV than younger PLWH and the HIV negative group ≥50 years.
Potential drug–drug interactions between non-antiretrovirals and antiretrovirals
Of the 913 total PDDIs between non-ARV and ARV medications identified in older PLWH, 25 were classed as contraindicated interactions and 888 were classed as potential interactions (Table 3). Clopidogrel (n=11) proton pump inhibitors (n=5) and inhaled corticosteroids (n=4) were the most frequently observed contraindicated co-medications in this group. Among younger PLWH, 201 total PDDI were measured with four being classed as contraindicated interactions and 197 classed as potential interactions. Three contraindicated interactions in this group involved inhaled corticosteroids and one involved clopidogrel. The ARV medications implicated in contraindicated interactions were all in the protease inhibitor (PI) class. White ethnicity and belonging to the PLWH aged ≥50 years group increased the odds of PDDI between ARV and non-ARV drugs compared with PLWH aged <50 years (odds ratio [OR]=2.00; 95% CI 1.26, 3.18; P=0.003 and OR=2.65, 95% CI 2.02, 3.47; P<0.001, respectively, after adjusting for gender and current ARV regimen). Moreover, the POPPY participants on a non-nucleoside reverse transcriptase inhibitor (NNRTI) without a PI, any integrase inhibitor (II) without a PI/NNRTI and participants on other ARV regimens compared with those on PI-containing regimens were 47%, 54% and 90% less likely to have a PDDI between ARV and non-ARV drugs (OR=0.53, 95% CI 0.41, 0.69, P<0.001; OR=0.46, 95% CI 0.28, 0.73, P=0.001 and OR=0.10, 95% CI 0.04, 0.26, P<0.001, respectively; Table 4).
Non-ARV drugs involved in interactions with ARVs

ARV, antiretroviral; PLWH, people living with HIV.
Association of PDDI between non-ARV and non-ARV drugs and PDDI between ARV and non-ARV drugs with HIV group, gender, race and ARV therapy

P-values obtained from multivariable logistic regression models. ARV, antiretroviral; II, integrase inhibitor; NNRTI, non-nucleoside reverse transcriptase inhibitor; OR, odds ratio; PDDI, potential drug–drug interaction; PI, protease inhibitor; PLWH, people living with HIV.
Potential drug-drug interactions between non-antiretrovirals and non-antiretrovirals
The odds of PDDI between non-ARV and non-ARV drugs was more than 3x higher in PLWH aged ≥50 years compared with HIV-negative controls after adjusting for gender and race (OR 3.08, 95% CI 2.16, 4.42; P<0.001; Table 4).
As seen in Table 5 older PLWH had more interactions that were classed as ‘Do not co-administer’ than the younger PLWH or older HIV-negative controls (2.6% [n=31] of all non-ARV drug interactions versus 0.7% [n=1] and 0.4% [n=1], respectively).
Non-ARV drug interactions

ARV, antiretroviral; PDDI, potential drug–drug interaction; PLWH, people living with HIV.
Discussion
In this analysis of polypharmacy from the POPPY study, we showed that among PLWH ≥50 years, 74.5% were receiving at least one co-medication (that is, medication other than ARVs), compared with 62% of PLWH <50 years. Furthermore, we showed that polypharmacy was more common among older PLWH than HIV-negative controls or younger PLWH, and that they were more at risk of a PDDI: involving non-ARV/non-ARV drugs than HIV-negative controls, and involving non-ARV/ARV drugs than younger PLWH. This shows the importance of considering co-medications when assessing appropriate ARV treatment choices for this group. While this has likely occurred because of the increasing age of PLWH and subsequently the increased prevalence of diseases of older age in this group, it must also be considered that older PLWH may attend more individual specialists to deal with comorbidities. While this practice may make it difficult to rationalize patients’ medication regimens, it should not preclude attempts to do so.
A large-scale Irish study of older people over 50 years found that 19% of participants had polypharmacy [32], the results from our own study show significantly higher rates of polypharmacy in PLWH. It is not surprising that all PLWH had significantly more polypharmacy rates compared with the HIV-negative group. Most of the studied PLWH were on ARV therapy, 98.7% of older PLWH and 94.9% of younger PLWH. Most ARV therapy comprises at least three medications, and these were counted separately for the purposes of this study. Consequently, many PLWH only need to have an additional two medications to meet the criteria for polypharmacy as we have defined it in this study. However, when ARVs were excluded 29.8% of older PLWH had polypharmacy. This finding suggests older PLWH are more likely to have a greater medication burden, independent of their ARV therapy. The reasons for this are multifactorial and could include the higher frequency of multimorbidity which seems to occur earlier in life in PLWH [33] and the involvement of various health-care providers that prescribe different medications. There also may be a reluctance on the part of prescribers to reduce medication that has been started by prescribers from other specialities.
There was a relatively small number of overall contraindicated interactions (2.7% [n=25] in older PLWH and 2% [n=4] in younger PLWH) between ARV and non-ARV medications. In older PLWH 3.6% (25/698) of people had a contraindicated ARV/non-ARV combination, and in younger PLWH 1.1% (4/374) of the group had a contraindicated combination. This is of a similar prevalence to that in the Swiss cohort study (2%) [19], and an Australian cohort study (3%) [34] which suggests good awareness of contraindicated combinations. Clopidogrel, proton pump inhibitors and corticosteroids were implicated in the majority of contraindicated interactions between non-ARV and ARV drugs, and it is clear that extra care should be taken to avoid these medications being co-prescribed along with PIs.
Furthermore, our study identified many PDDIs involving non-ARV/non-ARV medications. Although these interactions varied in terms of severity, it is important to underline that some of the interactions described may be associated with clinically significant events. Due to the higher rates of comorbidities, PLWH may be more vulnerable than the general population to the effects of complications associated with PDDI such as treatment failure or drug toxicity. The high prevalence of PDDI outside the use of ARVs in ageing PLWH suggests the need for better integrated, multidisciplinary care with regular review of co-medications.
Having identified the areas in which these interactions occur and the recommended actions that should be taken, pre-emptive measures can be put in place to minimize the risk of a person experiencing an adverse outcome as the result of a DDI. Many studies examining polypharmacy only examine conventional medications, and do not include herbal medications and supplements, which may result in underestimating poly-pharmacy. This study has shown that non-conventional medications make up a significant proportion of all co-medications, in all three populations in the study, and by including non-conventional medications a more accurate description of polypharmacy was obtained.
There were limitations associated with our study. One study limitation was the lack of data on dosing information on most medications. This means that some interactions may have been overestimated as the interaction may only apply where the medication is taken above a certain dose, for example, aspirin has a different interaction profile depending if it used as an anti-platelet or an analgesic. Furthermore, some medications may have been started and stopped at various time points over the course of the study, as the medication list was collected at study entry and not updated after this. The medication list obtained was a self-reported list. Prompts were given to participants to provide as accurate a list as possible, however, it is possible that medication use may have been under-reported, especially herbal and other non-conventional medications. Although many centres have excellent communication between health-care professionals, and between primary and secondary care, the lack of access to a complete prescription list that can be accessed by all health-care professionals remains a limitation. Lexicomp® was the only interaction checker used in this study for non-ARV interactions. It is a sensitive interaction checker, and many of the interactions flagged may not be clinically relevant in practice, especially those classified as no action needed. Pill burden was not determined as data on specific formulations was not specifically sought, and where combination product names were provided these medications were separated for the purposes of this study.
In conclusion, the findings from our study suggest that older PLWH are at higher risk for polypharmacy and PDDI than younger PLWH and older HIV-negative people. Importantly, even independently of ARV therapy, polypharmacy and PDDI are more common in older PLWH than older HIV-negative adults.
The findings above emphasize how imperative it is to focus on polypharmacy and PDDI in older PLWH who are taking multiple medications. There is a big focus on rationalizing medications in the older population [35] and PLWH should not be exempt from this process. Older PLWH may benefit from input from specialists in geriatrics and pharmacy to ensure that all medications are rationalized and deprescribed if appropriate. Polypharmacy and PDDI in the HIV population are likely to increase in importance as the age of this cohort continues to rise. Priorities for the future should include focus on non-ARV interactions in older people with HIV, and rationalizing medication use to ensure appropriate polypharmacy which would minimize the risk of PDDI.
Footnotes
Acknowledgements
We thank all participants in the study.
This work is supported by investigator-initiated grants from Bristol-Myers Squibb, Gilead Sciences, Janssen-Cilag, Merck Sharp and Dohme and ViiV Healthcare.
The research is supported by the National Institute for Health Research (NIHR) Biomedical Research Centre based at Imperial College Healthcare NHS Trust and Imperial College London and by a NIHR Senior Investigator Award to CS. The views expressed are those of the author(s) and not necessarily those of the NHS, the NIHR or the department of Health.
All the POPPY clinical sites in the UK are grateful for NIHR Clinical Research Network (CRN) support.
Further acknowledgements can be found in Additional file 1.
PM received funding for Advisory Boards, speaker panels, preparation of educational materials and/or research grants to his institution from Gilead Sciences, ViiV Healthcare, BMS, MSD, AbbVie and Janssen-Cilag. AW received honoraria or research grants from ViiV Healthcare, Gilead Sciences, BMS, Merck and Co. and Janssen. CS received funding from Gilead Sciences, ViiV Healthcare and Janssen-Cilag for the membership of Data Safety and Monitoring Boards, Advisory Boards, Speaker Panels and for the preparation of educational materials. MB has received travel and research grants from and has been speaker/advisor for Janssen, Roche, ViiV, Bristol-Myers Squibb, Merck Sharp & Dohme, Gilead, Mylan, Cipla, Teva. FAP has received research grants from Gilead Sciences and ViiV Healthcare, and has been a speaker or advisor for Gilead Sciences, ViiV Healthcare, MSD and Janssen. The remaining authors declare no competing interests.
