Abstract
Background
Our objectives were to review the economic modelling methods and cost-effectiveness of second-generation direct-acting antiviral agents for the treatment of chronic HCV infection.
Methods
A systematic literature search was performed in February 2017 using Scopus and OVID to review relevant publications between 2011 to present. Two independent reviewers screened potential papers.
Results
The database search resulted in a total of 1,536 articles; after deduplication, title/abstract and full text screening, 67 studies were included for qualitative analysis. The vast majority of studies were conducted in high-income countries (n=59) and used Markov-based modelling techniques (n=60). Most of the analyses utilized long-term time horizons; 58 studies calculated lifetime costs and outcomes. The examined treatments were heterogenic among the studies; seven analyses did not directly evaluate treatments (just with screening or genotype testing). The examined treatments (n=60) were either dominant (23%), or cost-effective at base case (57%) or in given subgroups (18%). Only one (2%) study reported that the assessed treatment was not cost-effective with the given setting and price.
Conclusions
Despite their high initial therapeutic costs, second-generation direct-acting antiviral agents were found to be cost-effective to treat chronic HCV infection. Studies were predominantly conducted in higher income countries, although we have limited information on cost-effectiveness in low- and middle-income countries, where assessment of cost-effectiveness is even more essential due to more limited health-care resources and potentially higher public health burden due to unsafe medical interventions.
Introduction
According to the latest WHO estimates [1], more than 70 million people have been infected with chronic hepatitis C globally and approximately 400,000 people died in 2015 from HCV-related diseases, mostly from cirrhosis and hepatocellular carcinoma. Unsafe health-care procedures and intravenous drug use were the leading causes of the 1.75 million new infections in 2015. HCV infection is a worldwide public health problem, but its impact on regions are different. The Eastern Mediterranean and European regions are more affected with 2.3% and 1.5% prevalence, respectively, but there are significant variations both across and within countries.
Since 2011, HCV treatment has radically changed with the approval of a number of effective direct-acting antiviral agents (DAAs). In contrast to previous interferon-based HCV therapies, DAAs do not act directly on the immune response against the virus but directly target some of the main molecular components of HCV (for example, NS3 protease and NS5A protein) [2]. Because of the high risk of the virus developing resistance, DAAs are commonly used in combinations, which have been proven to cure >90% of patients with chronic HCV infection with 8–24-week treatments [3]. Oral administration, short duration of treatment and good safety profile of all regimens mean that most patients are eligible for therapy [4]. With these treatments, the WHO is aiming for an 80% decrease in new HCV infections and 65% mortality reduction by 2030 [5].
In our study, only second-generation DAAs were considered as active therapies because first-generation agents (boceprevir and telaprevir) were already discontinued in 2014 and 2015, respectively, due to the availability of better therapies. Second-generation DAAs (including sofosbuvir, velpatasvir, elbasvir, grazoprevir, daclatasvir, ombitasvir, paritaprevir, ritonavir, ledipasvir and simeprevir) with or without combination with pegylated interferon and ribavirin (PR) or ribavirin (R) alone were the focus of this study.
As new DAA therapies are effective, safe and the majority of patients are eligible for treatment, the major limitation remaining is economic. The high cost of therapies (for example, the cost of a 12-week regimen of sofosbuvir alone is 84,000 USD) is the main barrier [6]. Although the European Association for the Study of Liver (EASL) recommends that all patients without contraindications to therapy should be offered treatment [7], disease-based restrictions are still common in Europe, restricting DAAs to people with substantial liver disease (≥F2) and abstinence from drug or alcohol use [8]. By contrast, Australia offers unlimited number of treatment courses, with no restrictions on access according to disease stage, treatment history or drug use status [9].
Lower income countries have limited health-care resources, and usually have higher prevalence [1] of HCV; therefore, transparent decision criteria for innovative therapies like second-generation DAAs, including the assessment of cost-effectiveness, are essential. Transferability of good-quality technology assessments could be highly beneficial for preventing duplication of efforts and save human and financial resources for local technology assessment [10].
In health technology assessments, economic models have become essential tools for estimating the consequences (costs and outcomes) of policy decisions and evaluating whether particular health-care technologies should be provided within the context of an organized health-care system [11]. Decision-analytic models can project the long-term costs and health benefits of HCV treatment, and estimate the value of second-generation DAA therapies in terms of incremental cost-effectiveness ratios (ICERs) compared with other options. Several systematic reviews [12–16] on DAA economic models have been conducted, but all of them included first-generation DAAs and were restricted to genotype-1 [12] or focused on screening strategies [14]. Chhatwal and colleagues [13] examined the modelling approaches of DAAs without any restrictions, but they also included telaprevir and boceprevir studies and did not report the conclusions of the models about cost-effectiveness.
The objective of our study was to provide a systematic overview of the structural and methodological approaches used in economic modelling of second-generation DAA treatments and the results on cost-effectiveness of these agents for the treatment of chronic hepatitis C.
Methods
Sources and search strategy
We conducted a systematic literature search using Embase, Medline (via Scopus) and Evidence-Based Medicine Reviews (EBMR): Cochrane Database of Systematic Reviews, American College of Physicians (ACP Journal Club), Database of Abstracts of Reviews of Effectiveness (DARE), Cochrane Controlled Trials Register (CCTR), Cochrane Methodology Register (CMR), Health Technology Assessments (HTA) and NHS Economic Evaluation Database (NHSEED; via OVID). The search covered peer-reviewed original articles published from 1 January 2010 to 20 February 2017, to be able to identify all studies with DAA treatments. As the first DAAs for HCV treatment was approved in 2011, we did not want to miss studies that could have been published before the approval. The search strategy was built up as a combination of search strings related to ‘hepatitis C’ and ‘economic evaluation models’. The search terms are detailed in Additional files 1 and 2.
Study selection criteria
Two independent reviewers screened the titles and abstracts of selected papers to determine their eligibility for the study (exclusion and inclusion criteria), all disagreements were resolved by consensus. Potentially relevant full-text articles were retrieved and fully screened; non-relevant studies were excluded based on full-text exclusion criteria. The reference lists of included systematic reviews were also searched for eligible studies.
Eligibility
We included any articles reporting model-based economic evaluations on second-generation DAAs, regardless of comparator (old standard-of-care or another DAA, or therapy with the same agent started in different stage of disease). In the title abstract screening part, we included any kind of DAAs and articles that examined telaprevir or boceprevir as active treatment were excluded in full-text screening. We extracted data from all articles meeting the final inclusion criteria regardless of the quality of the report. New articles identified from the reference lists of included systematic reviews went through the same screening process as articles identified with the database searches.
The exclusion criteria in the title abstract screening were the following in hierarchical order: articles without English abstract, articles without new evidence (editorials, letters and non-systematic reviews), articles not related to hepatitis C, articles not examining DAAs, articles not reporting economic models. Articles not excluded reporting economic models and systematic reviews about the topic were included for full-text screening. During full-text screening, articles were excluded based on additional exclusion criteria: articles written in languages other than English and articles examining one of the two discontinued first-generation agents (telaprevir and boceprevir) were excluded.
Data collection
The following information was extracted from each included study: country setting, perspective of the analysis, characteristics of the modelled populations, HCV genotype, model type, cost-effectiveness analysis type, examined DAA regimens, time horizons, cycle length if applicable, health states definitions, discount rates used, base results and sensitivity analyses. Cost and outcome data were also collected including cost currency and year, cost types and source of the data and the usage of any kind of utility data and its source if applicable.
Results
Our literature search resulted in 1,536 unique references. After deduplication, 1,521 underwent title and abstract screening, and finally 67 studies were included in the final analysis (Figure 1). Table 1 summarizes the detailed findings of our review.
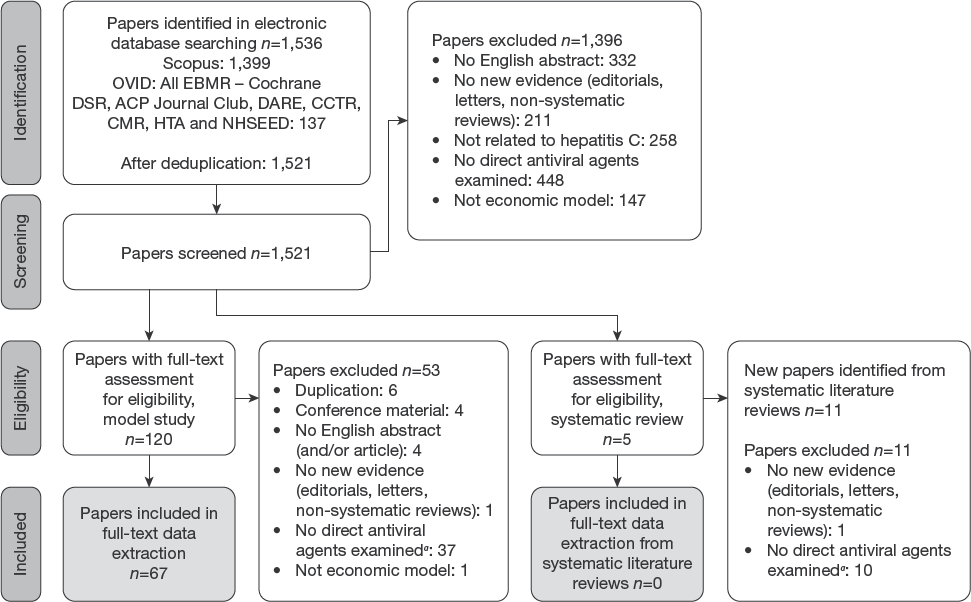
Flow diagram of the systematic literature search
Summary of the articles

Weekly cost of therapy could not be calculated based on the reported data.
Publication year; year of costing was not reported.
Reported as societal perspective, but only calculated with direct medical costs and did not take into account any true social cost. ASV, asunaprevir; CEA, Cost Effectiveness Analysis; CHC, chronic hepatitis C; CT, IEH, Continuous time individual event history model; CUA, Cost Utility Analysis; DAA, direct-acting antiviral; DCV, daclatasvir; DMM, Markov model evaluated deterministically; DSA, Deterministic Sensitivity Analysis; DSV, dasabuvir; DT, DES, discrete individual simulation- discrete event simulation in discrete time; DT, IEH, discrete-time individual event history model; DT, Decision Tree; EBV, elbasvir; GZV, grazoprevir; IFN, interferon; ISM, individual sampling model: Simulated patient-level Markov model; LDV, ledipasvir; LY, life year; NA, not applicable; NR, not reported; PR, pegylated interferon+ribavirin; PSA, Probabilistic Sensitivity Analysis; PWID, people who inject drugs; R, ribavirin; r, ritonavir; ScenA, Scenario Analysis; SMM, Simulated Markov model; SMV, simeprevir; SOF, sofosbuvir; SVR, sustained viral response; TE, treatment-experienced; TN, treatment-naive; VBA, Visual Basic for Applications; 2D, ombitasvir/paritaprevir/ritonavir; 3D, ombitasvir/paritaprevir/ritonavir + dasabuvir.
Study overview
Table 2 summarizes the study characteristics regarding the country and analysis perspective of the 67 studies included in the systematic review. Among the studies, 28 (42%) were conducted in Europe and 22 (33%) in the United States. Two studies [17,18] were conducted in Egypt, where the burden of disease is the highest, and only one study [19] was conducted in Central-Eastern Europe (in Croatia). One study was not country-specific, the analysis was conducted for ‘Asia’.
Summary of studies included in the systematic review

The majority of the studies (81%) used health-care perspective, additionally 10% of the studies reported societal perspective, but only calculated with direct medical costs and did not take into account any true social costs such as productivity loss or other indirect costs. Societal perspective or both health-care and societal perspective were used in 3 (4.5%) and 3 (4.5%), respectively.
Patient characteristics
Forty (59.7%) studies exclusively evaluated the cost-effectiveness of HCV treatment in treatment-naive patients, and 7 (10.5%) studies analysed screening; therefore these patients identified were also considered as treatment-naive. None of the studies analysed treatment-experienced patients only and 27 (40.3%) studies were conducted with both treatment-naive and treatment-experienced patients. The target population were people who inject drugs in three (4.5%) cases. Fifty-eight (86.6%) studies included genotype (G)1 patients, 21 (31.8%) studies included (not mutually exclusively) G2 patients, 24 (35.8%) studies included G3 patients, 25 (37.3%) studies included G4 patients, 7 (10.4%) studies included G5 patients and 6 (9%) studies included G6 patients.
Model types
We categorized the modelling approaches used by the studies on the basis of the taxonomy of model structures set by Brennan et al. [20]. Some of the modelling types were difficult to classify based on the articles, but all of them were placed in the most appropriate category based on the data extractor's judgement. The summary of the models is shown in Table 3. Fifty-nine (88%) studies reported cohort models, all but one [21] were Markov models with deterministic or stochastic analysis. Eight (12%) modelled the disease at patient level, two of them used simulated patient-level Markov models, and six used discrete or continuous time event simulation.
Model types in the identified studies

DT, decision tree.
Health states
We reviewed how the studies formulated health states to define stages of hepatitis C. We found that 41 (61%) models defined early HCV health states in terms of METAVIR fibrosis scores [22]. The others used either just chronic hepatitis C (10–15%), mild and moderate health states (6–9%) or just non-cirrhotic state (6–9%). Two studies did not report health states by name: one was a decision tree without any health states [21] and one did not consider early health states [23]. Fifty-five (82%) of the models considered liver transplant as a health state.
Treatments
Thirty-five (52%) studies analysed second-generation DAAs in combination with each other compared with second-generation DAA + PR treatments and first-generation DAA + PR treatments, only PR treatments or with no treatments. Ten (15%) analysed second-generation DAAs in combination with other second-generation DAAs or PR or R in earlier stages, or before transplant compared with later treatment initiation. Four (6%) analysed second-generation DAAs combined with R therapies (interferon-free) compared with second-generation DAA combination therapies containing interferon or no treatment. Nine (13%) of the studies used second-generation DAA + PR combination as active treatment compared with first-generation DAA + PR combinations of just PR. Two (3%) studies evaluated second-generation DAA + R combinations (interferon-free) compared with first-generation DAA + PR or no treatment. Seven (11%) studies evaluated different screening strategies or treatment scenarios involving treatments with second-generation DAAs alone or in combination.
Time horizon
The models which analysed treatments without screening used mainly lifetime horizon (55 studies), 1 [19], 2 [21,24], 1 [25] and 1 [26] calculated with 1 year, 5 years, 10 years (scenario analyses with 20 and 30 years) and 15 years, respectively. The studies also considered screening used lifetime horizon in 3 cases and 20 [27], 30 [28], 40 [17] and 100 [29] years in 1-1 cases, respectively.
Cycle length
Most of the models (35–52%) utilized 1-year cycles, the second most common was 1-month cycles used by 6 (9%), and 4 and 1 studies reported 3-month and 12-week long cycles (7.5%), respectively. Two (3%) models calculated with weekly cycles, six (9%) models used different cycle lengths for different stages, 3 (4.5%) used 3 months for the first 2 years and 1 year thereafter, 2 (3%) used 1 month cycles in the first 1.5 years, 3-month cycles for the next half year (2 cycles) and annual cycles thereafter, and one study reported weekly cycles during treatment and annually after. In 5 (7.5%) cases, cycles were not applicable due to modelling technique (decision tree and event simulations). Eight (12%) studies did not report any information about cycle length mostly because they used previously reported models.
Economic analysis type
The majority of the studies (n=60 [89.5%]) were cost– utility analyses, reporting only quality-adjusted life-years (QALYs) as outcomes; in 4 (6%) studies other outcomes in addition to QALY were used. Three calculated life year gain next to QALY gain and one calculated ICER as incremental cost/sustained viral response (SVR). Three (4.5%) studies did not use QALY as outcome, two of them calculated incremental cost/SVR and one cost/life year.
Sensitivity analyses
We found that 41 (61%) of the studies conducted both deterministic (DSA) and probabilistic sensitivity analyses (PSA) in-line with the recommendations of ISPOR-SMDM Modeling Good Research Practices Task Force [30] and an additional 2 (3%) included some scenario analyses in addition to DSA and PSA. Ten (15%) of the articles analysed uncertainty only with DSAs and 6 (9%) only with PSA with one addition with scenario analysis next to PSA. In 3 (4.5%) studies they only conducted different scenario analyses, one study did not specify the method of sensitivity analysis [18] and three did not report any kind of sensitivity analysis.
Software
Eighteen (27%) studies reported Microsoft Excel as the modelling platform and 17 (25.5%) used TreeAge software for model building. Three (4.5%) studies used one-one software, Java, Visual Basic (VBA) and Arena, respectively. Another 4 (6%) combined software, one used TreeAge and Microsoft Excel, one combined SAS with VBA and another one used R and Microsoft Excel. Twenty-five (37%) of the studies did not state clearly the software used.
Cost-effectiveness results
As 7 (10.5%) studies evaluated different screening or complex scenarios we only analysed the results of the remaining 60 studies that examined the value of DAAs. From these studies, the DAAs of interest were dominant in 8 (13%) cases and cost-effective in the given country setting in 30 (50%) cases. Studies examining DAAs given in earlier stages compared with the same given later found that earlier therapy is either dominant (6–10%) or cost-effective (4–7%). Eleven studies found that the examined DAAs are cost-effective with some conditions as in only some of the examined stages or genotypes. One study [23] stated that the examined DAA cannot be cost-effective with the given price.
Discussion
The availability of DAAs means a significant improvement compared with interferon-based therapies in HCV treatment, but the cost of DAAs is still high. Our approach was to systematically review HCV cost-effectiveness models analysing second-generation DAAs, focused on the methodological techniques and also the cost-effectiveness results. Previous systematic reviews limited their scope only to the modelling approaches of HCV evaluations [13], reviewed both, but were limited to one genotype [12] and/or included first-generation DAAs [15,16]. In our study, we systematically reviewed the results and also the methodological approaches of the included 67 studies that modelled the cost-effectiveness of treatment of HCV with second-generation DAAs.
We observed that the majority of the published models were developed for high-income countries and used Markovian techniques. Only a few models used a method where interaction between patients can be modelled, hence the majority of published models could not consider the benefits of HCV treatment in preventing or slowing disease progression as a result of lack of interaction. Consequently, the majority of studies could have underestimated the true value of HCV treatments.
The models used standard health states, mainly the classification based on the METAVIR score [22] to define the early stages of the disease, and all models used decompressed cirrhosis, hepatocellular carcinoma and death as advanced stages. Long-term benefit of second-generation DAAs could have manifested also in those patients who avoided liver transplants.
Most of the studies calculated costs and utilities and expressed the ICER in additional cost/additional QALY. With explicit or implicit willingness to pay thresholds in almost every country, decision about the cost-effectiveness of given therapies from different countries may be comparable. While the analysed therapies were heterogeneous, they were almost always cost effective (or even dominant), although the strength of evidence (and risk of bias) could vary across economic evaluations as we did not assess the quality of the studies. Only one article [23] came to the conclusion that at the published price the studied therapy could not be cost-effective.
The time horizon could play an important role in the cost-effectiveness of DAA therapies in HCV treatment due to the chronic nature of HCV infection and the huge initial costs of the relatively short treatments. Avoided major clinical events only occur several years after the treatment, thus a shorter time horizon could underestimate the true value of innovative treatments. In line with this, the analyses were mostly conducted on a lifetime horizon.
Our study certainly has limitations, the extraction was limited by the reported information in the included studies, in case the covered article used a model already published by others we did not review the original one. Our systematic literature review involved searching only studies that were published in English and indexed in the selected databases. Despite the broad database selection, the inclusion of unpublished material and studies from other resources was precluded. As we reviewed the cost effectiveness – not just the modelling methods – of the examined second-generation DAAs, we did not exclude multiple publications of the same model in different populations or countries. The quality assessment of the studies was not in our focus, so we did not evaluate the quality of reporting in the articles.
In conclusion, DAA therapies were considered as good value for money in almost all cases, and even could be cost saving in some settings. These results were calculated with similar modelling techniques and mostly in developed higher income country settings. Although we have limited information on cost-effectiveness in low- and middle-income countries, where assessment of cost-effectiveness is even more essential due to more limited health-care resources and potentially higher public health burden due to unsafe medical interventions. Therefore, future modelling efforts should consider cost effectiveness of HCV treatments in less affluent countries, where justifiable price of DAAs should be lower mainly due to lower willingness to pay for one unit of health gain. If we want to reach the global WHO targets [5] it is essential to scale-up second-generation DAA treatments in every country in a cost-effective way.
