Abstract
Background
There is controversial data on possible occult HBV reactivation in HCV patients successfully treated with direct-acting antivirals (DAA). However, diagnosis of occult HBV infection (OBI) was not performed by gold standard procedures in any study.
Methods
By using several highly sensitive assays, we examined serially collected serum samples from 40 HBV-surface-negative DAA-treated HCV patients with OBI identified by testing liver biopsy specimens through nested-PCR technique. Serum samples were obtained at four time points from each patient (at baseline, at 4 weeks after starting, at the end and 12 weeks after stopping therapy) and tested for HBV DNA by nested-PCR and real-time PCR techniques.
Results
All tested serum samples were negative by both quantitative HBV surface antigen (HBsAg) and HBV core-related antigen assays. 26/40 patients were anti-HBs-positive and in all of them, the amount of this antibody was stable at the four time points evaluated. Serum HBV DNA was detected in 10 samples at baseline, in 6 samples 4 weeks after starting therapy, in 11 samples at the end of therapy and in 21 samples 12 weeks after stopping treatment (P=0.001). Aminotransferase values dropped within the normal levels at week 4 of therapy and persisted normal over time in all cases.
Conclusions
A slight increase in the amount of HBV DNA 3 months after stopping DAA therapy was the only parameter showing a possible reappearance of HBV activity in OBI patients cured for a concomitant HCV infection, but it was insufficient to lead toward a virological reactivation capable of inducing liver injury.
Introduction
Due to their worldwide diffusion and common mode of transmission, HBV and HCV coinfection is a fairly frequent occurrence, particularly among individuals at high risk of parenteral transmitted infection and in highly endemic areas [1–3]. A possible interplay between the viruses in the case of dual infection has been proposed, although contrasting data are available in this context [1–5]. In particular, several clinical reports and experimental data suggest a prevalent role of HCV in suppressing the HBV activity. On this basis, it has been hypothesized that the inhibitory effect of HCV on HBV replication and gene expression might lead to the development of an occult HBV infection (OBI), which is the phase of the viral life cycle characterized by long-lasting persistence of replication competent viral genomes with a strong suppression of their functional activities in the liver of HBV surface antigen (HBsAg)-negative individuals [6]. Indeed, a large body of evidence indicates that the prevalence of OBI is particularly elevated in HCV carriers [7–9]. The suppressive role of HCV on HBV has also been supported by a number of studies concerning HBsAg-positive patients treated with interferon-based therapy for a productive HCV coinfection, who showed the reactivation of the previous partially suppressed HBV when a favourable response to therapy and HCV RNA clearance were achieved [10–12]. Thus, the elimination of the HCV in these cases might produce the loss of the inhibitory effect exerted by this virus on HBV that may be reactivated.
Recently, clinical/virological HBV reactivation has been observed also in coinfected patients undergoing direct-acting antiviral (DAA) treatments [13–19]. This event has even been anecdotally reported in cases with OBI (particularly, in subjects with HIV coinfection) [20,21], in whom reactivation is characterized by HBsAg reappearance and serum HBV DNA detectability by diagnostic commercial assays with possible flare of hepatitis. However, a number of additional studies conducted on large cohorts of patients failed to confirm such a possibility [22–26]. All studies evaluating the risk of OBI reactivation in DAA-treated HCV patients have been performed examining HBsAg-negative individuals testing positive for antibody to HBV core antigen (anti-HBc) that is known to be just an indirect, surrogate marker of occult infection that can definitively be diagnosed only when viral DNA is detected [9].
In addition, these studies evaluated the HBV reactivation by routinely available diagnostic assays that are often insufficiently sensitive to reveal inconspicuous virological changes occurring over time. Indeed, we do believe that the possible occurrence of OBI reactivation in DAA-cured HCV patients has such relevant biological and clinical implications that it should be explored by more sensitive and specific approaches. The aim of this study was to investigate the possible OBI reactivation by multiple, sensitive methods applied in serially collected samples from HCV patients with documented persistence of intrahepatic HBV DNA who showed sustained virological response (SVR) to DAA therapies.
Methods
Patients
A total of 40 HCV-infected patients (35 males; median age 63 years, range 47–72) previously found to have OBI through the analysis of liver DNA extracts were consecutively and successfully treated with DAAs from April 2015 to September 2015 in the Division of Clinical and Molecular Hepatology of the University Hospital of Messina. All patients were HBsAg-negative and none of them were under anti-HBV agents. 36 individuals were positive for anti-HBc and 27 of them were also positive for antibody to HBsAg (anti-HBs), whereas 4 cases were negative for all HBV serum markers. In addition, none of the patients were coinfected with HIV. 27 patients had cirrhosis, and 13 had chronic hepatitis. 29 patients were infected with HCV genotype-1b, 5 with genotype-1a, 2 with genotype-2, 2 with genotype-3 and 2 with genotype-4. 18 patients were treated with sofosbuvir and simeprevir, 10 with sofosbuvir and lepidasvir, 9 with sofosbuvir and daclatasvir and 3 with Viekirax and Exviera. All patients had undergone needle liver biopsy for diagnostic purpose and – as usual in our Division – a fragment of each liver specimen had been stored in liquid nitrogen in the laboratory of the Division and subsequently tested for HBV DNA, applying methods previously reported and in accordance with the statements produced by an international panel of experts [9]. Briefly, liver DNA extracts were examined for the presence of HBV genomes by performing four different in-house nested-PCR amplifications to detect preS-S, preC–Core, Pol and X HBV genomic regions, respectively. The cases showing positivity in at least two different viral genomic regions were considered as OBI-positive [9]. Appropriate negative and positive controls were included in each PCR experiment and direct sequencing of all amplified HBV sequences were performed to confirm the specificity of the reactions.
All patients were followed-up with regular clinical and biochemical controls during and after the therapy cycles, and serum samples from each case were collected and stored at −80°C for subsequent analyses at four time points: the day of starting DAA therapy, 4 weeks after starting therapy, the day of stopping therapy, and 12 weeks after the end of therapy. At baseline, each patient showed elevated alanine aminotransferase (ALT) levels ranging from 0.5 to 5x the normal values, and all were HCV-RNA-positive (median HCV RNA levels, 1.4x10 6 IU/ml; range: 2.3x10 4 –6.6x10 6 ). However, all of them showed a drop of ALT values below the limits defined by Prati et al. [27] and undetectable serum HCV RNA since the 4th week of treatment, and they maintained this status at each time point of the follow-up. All cases were SVR as shown by undetectable serum HCV RNA 12 weeks after the end of treatments.
The study was approved by the Ethics Committee of the Messina University Hospital, and written informed consent was obtained from each patient.
Monitoring of HBV status
To reveal possible HBV reactivation, the four serum samples serially collected from each patient were analysed by applying different approaches.
Quantitative levels of HBV core-related antigen (HBcrAg) were determined using the LumipulseGHBcrAg assay (Fujirebio Italia, Pomezia [Rome], Italy), which measures simultaneously denatured HBeAg, HBcAg and the preC protein p22cr (aa 28 to aa 150). Samples were tested according to the manufacturer's instructions. The lowest limit of detection of this test is at 1.0 kU/ml.
Quantification of serum HBsAg was performed with the LumipulseG HBsAg-Quant assay according to the manufacturer's instructions (Fujirebio Italia), with the lowest limit of detection of HBsAg at 0.005 IU/ml.
Serum anti-HBs was quantitatively measured using the Liason XL Murex anti-HBs Plus according to the manufacturer's instructions (DiaSorin, Saluggia, VC, Italy), with the lowest limit of detection of serum anti-HBs at 3 mIU/ml.
For serum HBV DNA detection, viral DNA was extracted from 1 ml of each serum sample using the QIAamp UltraSens Virus kit (Qiagen, Milano, Italy) according to the manufacturer's instructions. All DNA extracts were analysed for the presence of HBV genomes by performing four different in-house nested PCR amplification assays in analogy to the above mentioned liver DNA testing, with limit of sensitivity of 10 genome equivalents/ml.
Serum HBV DNA levels were measured by in-house real-time (RT)PCR experiments performed in a Light-Cycler (Roche, Basel, Switzerland) with the LightCycler® FastStart DNA Master HybProbe kit (Roche Diagnostics, Mannheim, Germany), using a 20 μl reaction volume containing 10 μl of extracted DNA, 3 mM MgCl2+, 0.5 μM of forward and reverse primers and 0.25 μM of the labelled probe. Forward and reverse primers were 5′-CTCGTG-GTGGACTTCTCTC-3’ (nt 1548-1566) and 5′-CAGCAGGATGAAGAGGAA-3'(nt 401-418), respectively, whereas the hybridization probe was 5′-6FAM- AACCAGGACAAATTGGAGGACAGGA-TMR-3’ (nt 368-344). Amplification was performed as follows: 95°C for 10 min then 45 cycles at 95°C for 10 s, 58°C for 30 s and 72°C for 20 s. Serial dilutions of a plasmid containing a monomeric genotype D HBV insert (Alfa Wasserman, Milan, Italy) were used as quantification standards. A linear relationship from 1x10 to 1x10 7 copies/ml was obtained between cycle threshold values and numbers of HBV DNA copies used as standards.
Statistical analysis
Continuous numerical variables were expressed as median and range (minimum-maximum) and categorical variables as frequency and percentage. For the continuous variables that did not present a normal distribution, as verified by Kolmogorov Smirnov test, a non-parametric approach was used. For dichotomous or categorical variables the χ 2 test was used. In order to assess the existence of significant differences among the four time points for presence/absence of HBV genomes, the Cochran test was applied; subsequently, the pairwise comparison between two time points was performed using McNemar test. Wilcoxon test was applied in order to perform comparison between basal evaluation and the last time point, for HBV DNA serum levels.
Statistical analyses were performed using SPSS 17.0 for Windows package (SPSS Inc., Chicago, IL, USA). Two-sided P<0.05 was considered to be statistically significant.
Results
HBV DNA was found to be positive in the serum of 10/40 (25%) cases at baseline, in 6/40 (15%) at month 1 after starting therapy, in 11/40 (27.5%) at the time of stopping therapy and in 21/40 (52.5%) cases 3 months after the end of the therapeutic cycle (P=0.001). Therefore, the overall presence of circulating HBV sequences was significantly more frequent after than before and during DAA treatment in our patients, although in a certain number of cases HBV sequences were alternatively detectable over time. In particular, in 3/40 (7.5%) cases HBV DNA was detectable at baseline but it became undetectable in all the subsequent collected samples, in 10/40 (25%) it was detected only in the samples collected at the end of follow-up, in 1/40 (2.5%) it was found only in the sample collected at the end of treatment, in 13/40 (32.5%) cases it was alternatively detectable/undetectable in the serially collected samples, whereas it was stably negative or positive in all four samples in 12/40 (30%) and in 1/40 cases (2.5%), respectively (Table 1). We also performed RT-PCR quantification of HBV DNA levels in serum samples obtained at both basal time and 3 months after the end of DAA treatment in 14 out of the 40 patients from whom additional sera to perform this experiment were available. Quantification of HBV DNA serum levels confirmed the results obtained by nested-PCR. Indeed, only serum samples testing OBI-positive by nested-PCR had quantifiable amounts of HBV DNA (Table 2). Viral DNA could be quantified in the serum of 5/14 (35.7%) cases at baseline and in the serum of 11/14 (78.6%) cases 3 months after the end of therapy (P<0.05). Interestingly, median amounts of serum HBV DNA 3 months after the end of therapy (median: 188 copies/ml; range: 0-3,040 copies/ml; P=0.019) were significantly higher than median amounts of serum HBV DNA levels at basal time (median: 92 copies/ml; range: 0-880 copies/ml; Table 2).
Detection of HBV DNA by nested-PCR in longitudinally collected serum samples from 40 DAA-cured HCV patients

DAA, direct-acting antiviral.
Real-time PCR quantification of HBV DNA in serum samples from 14 DAA-cured HCV patients
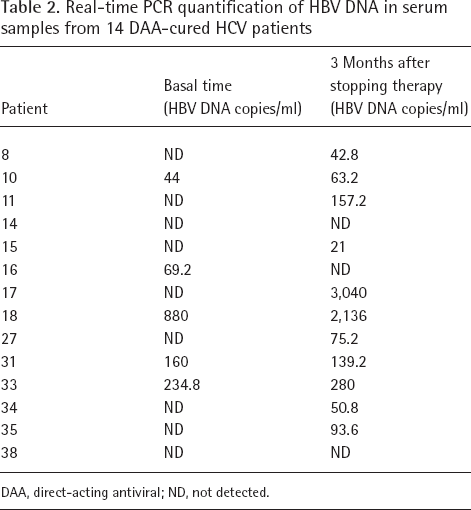
DAA, direct-acting antiviral; ND, not detected.
Quantitative measurement of HBsAg and HBcrAg showed that both these serum markers were negative at baseline and remained persistently negative (without any minimal fluctuation) in all the serially collected serum samples from each patient. In anti-HBs-positive patients, amounts of anti-HBs detected in the serum samples at baseline remained unchanged in all the other serum samples tested for all the patients.
Discussion
We evaluated HBV virological behaviour during DAA treatment in HCV patients with an occult HBV coinfection accurately diagnosed by liver DNA extract examination. We analysed serum samples serially collected before, during and after stopping the treatments by the use of different highly sensitive methods. HBV DNA testing by nested-PCR assays covering multiple, different HBV genomic regions was confirmed to be the best method to reveal OBI even when blood is examined, above all when the test is performed on 1 ml of serum and on multiple, serially collected serum samples. Under these circumstances, circulating HBV DNA may be alternatively detectable and undetectable over time, suggesting that (re)appearance of HBV viraemia is often just the expression of the natural OBI course, independently of the contemporary DAA therapy [28–30]. Indeed, overall analysis of the serially collected serum samples from the entire study population indicated that HBV DNA amounts were significantly higher 3 months after stopping therapy than at the other time points investigated, as also confirmed by RT-PCR quantification approach. Thus, a kind of stimulus of HBV replication may occur in OBI patients after HCV cure. This might be a consequence of the disappearance of a sort of direct inhibitory effect of HCV on HBV activity, or – more likely – of the fact that HCV infection activates an innate immune state that is favourable to a control of HBV replication [16,31]. Thus, anti-HCV effective DAA therapy might indirectly disrupt this immune state and the control on HBV activity might be impaired. In any case, however, we observed that only a very limited increase of HBV replication activity may occur in these cases, which is insufficient to induce a consistent reactivation of the viral replication and, consequently, to induce HBV-related liver damage, at least in immune-competent individuals. Furthermore, no evidence of HBV gene expression activation was found at any time point in our patients, as shown by the persistent absence of HBsAg and of HBcrAg serum markers, despite the fact that they were tested by highly sensitive techniques. In analogy, the persistence of quite high levels of circulating anti-HBs antibody (which is known to significantly decrease in several circumstances associated with potent immunosuppression, and this often precedes HBsAg re-seroconversion) [32,33] appears to exclude any relevant effect of the HCV cure on the specific anti-HBV immunological control.
Considering that HBV DNA values may rise 3 months after stopping therapy in some cases, one cannot rule out the hypothesis that HBV replication and gene expression may, subsequently, progressively reappear in some cases after HCV elimination. Noteworthy, however, is that all the 40 patients included in the study showed no evidence of clinical/biochemical reactivation during the follow-up with persistence of low ALT levels at 6 and 12 months after stopping therapy, thus weakening the above hypothesis.
OBI reactivation has been reported in many clinical conditions affecting the immune system, and in association with a quite large number of immune- and/or chemotherapy treatments [6,34]. DAA-induced HCV cure appears to depict a different scenario with very uncommon clinical consequences. Testing HBV status in HCV patients before starting DAAs is strongly recommended by the EASL guidelines, and there is a safety communication warning by the Food and Drug Administration concerning possible HBV reactivation in the setting of DAA therapy [35,36]. In this context, it is necessary to consider the quite large body of evidence indicating that OBI is a co-factor promoting progression of the liver disease toward cirrhosis and HCC development in HCV-infected individuals [37,38]. Such a negative influence on clinical outcome appears to be cancelled out if the patients are properly cured by anti-HCV treatment [39], thus rendering HCV patients with occult HBV coinfection a subset of patients that must quite urgently undergo DAA treatment. This aspect reinforces the need to evaluate the HBV serological profile in HCV patients.
In conclusion, this study shows that the HBV infection in its occult phase may only be marginally influenced by the elimination of a simultaneous HCV infection by DAA therapy, thus confirming that HCV does not play a major role in promoting the HBV suppression that leads to OBI development and indicating that HCV cure is an event insufficient by itself to induce virological and clinical reactivation of the HBV infection in its occult phase, at least in immunocompetent individuals.
Footnotes
Acknowledgements
This study was supported by Gilead Sciences, Fellowship Program 2015 and by BMS.
Conceptualization and design of the study: G Raimondo, TP. Investigation and acquisition of data: CM, GT, DL, G Raffa. Analysis and interpretation of data: CM, G Raffa, AA, IC, GS, G Raimondo, TP. Drafting the article and revising it critically for important intellectual content: G Raimondo, TP. Final approval of the version to be submitted: CM, IC, GT, DL, G Raffa, AA, GS, G Raimondo, TP.
The authors have no competing interests.
