Abstract
Background
Frailty and falls occur commonly and prematurely in HIV-infected populations. Whether frailty in middle-age predicts future falls among HIV-infected women is unknown.
Methods
We evaluated associations of frailty with single and recurrent falls 10 years later among 729 HIV-infected and 326 uninfected women in the Women's Interagency HIV Study (WIHS) with frailty measured in 2005 and self-reported falls in 2014–2016. Frailty was defined as ≥3 of 5 Fried Frailty Index components: slow gait, reduced grip strength, exhaustion, unintentional weight loss and low physical activity. Stepwise logistic regression models determined odds of single (versus 0) or recurrent falls (≥2 versus 0) during the 2-year period; separate models evaluated frailty components.
Results
HIV-infected women were older (median 42 versus 39 years; P<0.0001) and more often frail (14% versus 9%; P=0.04) than uninfected women. Over 2 years, 40% of HIV-infected versus 39% of uninfected women reported a fall (single fall in 15% HIV+ versus 18% HIV-women; recurrent falls in 25% HIV+ versus 20% HIV-women [overall P=0.20]). In multivariate models, frailty independently predicted recurrent falls (adjusted odds ratio [aOR] 1.84, 95% CI: 1.13, 2.97; P=0.01), but not a single fall. Among frailty components, unintentional weight loss independently predicted single fall (aOR 2.31, 95% CI: 1.28, 4.17; P=0.005); unintentional weight loss (aOR 2.26, 95% CI: 1.32, 3.86; P=0.003) and exhaustion (aOR 1.66, 95% CI: 1.10, 2.50; P=0.02) independently predicted recurrent falls.
Conclusions
Early frailty measurement among middle-aged women with or at-risk for HIV may be a useful tool to assess future fall risk.
Introduction
As a result of widespread potent antiretroviral therapy use, growing numbers of people living with HIV (PLWH) are surviving into older age. In the US and Canada, life expectancy of PLWH on antiretroviral therapy (ART) approaches that of the general population [1]. PLWH experience an excess burden of comorbid conditions earlier in life that are typically associated with ageing, including cognitive impairment [2–4], osteoporosis [5] and geriatric syndromes such as frailty, falls and fractures [6,7].
Frailty, a geriatric syndrome characterized by diminished strength, endurance, and reduced physiological function, is associated with numerous adverse outcomes in the elderly HIV-uninfected persons, including falls, fracture, disability and death [8–10]. Frailty also predicts mortality in middle-aged PLWH [11–13]. We previously reported a high interval prevalence of falls in the WIHS, with at least one fall reported in the preceding 6 months in 19% of HIV-infected women and 18% of uninfected women [14], and one or more fall in 41% of HIV-infected and 42% of uninfected women over 24 months follow-up [15], despite a mean age of only 48 years. Among middle-aged, predominantly male HIV-infected adults, 24–30% reported at least one fall in the previous year [16,17], similar to rates in uninfected persons aged 65 years and older [18]. Moreover, after accounting for other fall risk factors, HIV-infected women may have greater risk of falls than HIV-infected men [16,17]. However, whether frailty in middle-age predicts future falls in HIV-infected women is unknown. We undertook this study to evaluate whether the Fried frailty phenotype predicts subsequent falls approximately 10 years later in HIV-infected and uninfected women in the WIHS, including the specific contribution of individual frailty components.
Methods
Study population
The WIHS is an ongoing, multicentre cohort study of the natural and treated history of HIV infection in women that initially enrolled women with and at risk for HIV infection at six consortia (Bronx/Manhattan NY, Brooklyn NY, Chicago IL, Washington DC, San Francisco CA and Los Angeles, CA) in 1994–1995, and subsequently in 2001–2002 and 2011–2012. WIHS methods and baseline cohort characteristics have been described previously [19]. At semi-annual visits, participants complete face-to-face interviews and physical examinations and provide biological specimens. Written informed consent was obtained from each participant using procedures approved by committees on human research at all of the collaborating institutions.
Study sample
Starting in 2014, 2,062 WIHS participants completed a questionnaire on self-reported history of falls. Of these, 729 HIV-infected and 326 uninfected women subsequently completed 4 falls questionnaires over 2 years (from 2014–2016), had previously completed at least 3 of 5 measures for determination of the Fried frailty phenotype in 2005, and are included in this analysis [8]. Women excluded from the current analyses (due to site disenrolment, loss to follow-up or death) were more likely to be frail (18% versus 12%; P=0.0001), of White race, and have HCV infection, renal impairment, neuropathy symptoms and cognitive complaints, as well as lower income and education levels than women included in the current sample, and were less likely to be obese. HIV-infected women excluded from the current analyses had similar rates of ART use and HIV viral load suppression but were more likely to have a prior AIDS defining illness and had lower CD4+ T-cell count and CD4+ nadir compared with those included in the current sample.
Frailty definition
Frailty was defined according to Fried frailty phenotype, which has been utilized in numerous population-based studies and cohorts including the WIHS [8,20,21], based on the presence of three or more of five characteristics: slow gait, reduced grip strength, exhaustion, unintentional weight loss of 10 pounds within 6 months and low physical activity. Mobility was measured using a 3–4 metre timed gait. Grip strength was measured using a hand-held dynamometer in the dominant hand with maximum force. Slow gait and reduced grip strength were defined as the lowest quintile of performance by site among HIV-uninfected women; exhaustion, weight loss and physical activity were based on self-report [20,21].
Falls ascertainment
Beginning in 2014, WIHS participants reported all falls within the prior 6 months at each semi-annual study visit. The current analyses include report of falls from 4 semi-annual visits over a 2-year period, from 2014 to 2016. Fall was defined as: ‘an unexpected event, including a slip or trip, in which you lost your balance and landed on the floor, ground or lower level, or hit an object like a table or chair’ and participants were instructed not to include falls resulting from a major medical event (for example, a stroke or seizure) or an overwhelming external hazard (for example, hit by a truck or pushed) [22]. Participants reporting any fall were then asked whether they had either ‘1’ or ‘2 or more’ falls in the prior 6 months, sought medical attention for any of these falls or any of these falls resulted in fracture.
Statistical analyses
Medians, interquartile ranges and proportions summarized study variables. c 2 tests compared proportions of categorical or categorized variables between HIV-infected and HIV-uninfected women and proportions with no fall, single fall (defined as exactly one fall during the 2-year period) and recurrent fall (defined as two or more falls during the 2-year period). Women who reported sustaining one fall in the prior 6 months at more than one of the four study visits, or who reported two or more falls at any of these study visits were considered to have recurrent falls. Bivariate analyses evaluated each predictor in association with single fall (versus no fall) and with recurrent falls (versus no fall), for HIV-infected and uninfected participants together and separately using univariate logistic regression.
Stepwise logistic regression models determined independent associations with fall risk after 10 years, over the 2-year follow-up for a single fall (1 versus 0) or recurrent falls (≥2 versus 0), forcing in baseline frailty and HIV status and allowing other variables in Table 1 (except frailty components) to enter and remain in the model if P<0.05. Primary outcomes were report of single fall compared with no fall, and report of recurrent falls over the 2-year period. HIV status and frailty status (the primary exposures of interest) were forced into models. Separate multivariable models restricted to HIV-infected women were constructed similarly and additionally evaluated the contribution of HIV treatment and disease-specific characteristics on fall risk. To evaluate the contribution of individual frailty components on fall risk after 10 years, we next constructed logistic regression models as described earlier that allowed frailty components to enter using stepwise regression, excluding overall frailty status [23–25].
Characteristics of 1,055 HIV+ and HIV- WIHS participants at index visit

Data are n (%) unless otherwise indicated. BMI, body mass index; CESD, Center for Epidemiology Studies Depression; CNS, central nervous system; eGFR, estimated glomerular filtration rate; N/A, not applicable.
Demographics (such as race/ethnicity and education level) were measured at WIHS enrolment; other covariates were time invariant and measured at the visit when frailty was assessed in 2005 (referred to as the index visit). Candidate covariates for multivariable models included demographics, behavioural factors, comorbid conditions, central nervous system (CNS) active medications and HIV-related factors, as follows: demographics included: age per 10 years, race/ethnicity (White, Hispanic/Other versus Black [reference]), annual household income ≤$12,000; having graduated from high school; and year of WIHS enrolment (2001-2002 versus 1994-1995). Substance use included tobacco use (current smoker, former smoker versus never smoker [reference]); cocaine, crack and/or heroin use (current, former versus never [reference]); marijuana use (current, former versus never [reference]); and recent (6 month) alcohol use: heavy (≥14 drinks/week), moderate (3-13 drinks/week) or light (<3 drinks/week) versus none [reference]). CNS active medication classes included anticonvulsants, antidepressants, antipsychotics, sedatives (including benzodiazepines, barbiturates and non-benzodiazepine sleep aids) and muscle relaxants. CNS active medications were analysed continuously as the number of classes being used at index visit. Comorbid conditions included: peripheral neuropathy (self-report of numbness, tingling or burning sensations in arms, legs, hands or feet lasting for more than 2 weeks); obesity (body mass index [BMI] >30 kg/m2); subjective cognitive complaints (self-report of major problems with memory or concentration that interfered with normal everyday activities and lasted for more than 2 weeks, or self-report of confusion, getting lost in a familiar place or inability to perform routine mental tasks); depressive symptoms (modified Center for Epidemiology Studies Depression [CES-D] score ≥15 [12,26], modified by excluding these two symptoms that overlap with the Fried frailty phenotype: ‘this past week I could not get going’ (overlaps with low physical activity) and ‘this past week everything was an effort’ (overlaps with exhaustion), diabetes mellitus as previously operationalized in WIHS [27]; renal dysfunction (estimated glomerular filtration rate [eGFR] <60 ml/min/1.73 m2 using the Modification of Diet in Renal Disease calculation) [28]; hypertension (self-report of hypertension with diastolic blood pressure ≥90 mmHg, systolic blood pressure ≥140 mmHg or current receipt of antihypertensive medication) [29]; and HCV infection (HCV antibody positive with detectable RNA).
Additional HIV disease specific covariates included: nadir CD4+ T-cell count (lowest CD4 measured prior to index) and current CD4+ T-cell count, current log10 HIV RNA level, current suppressed (<80 copies/ml) HIV RNA, prior AIDS-defining illness, current ART use.
All P-values reported are two sided, from χ 2 (for unadjusted proportions), Wilcoxon (for unadjusted continuous variables) and Wald tests (for logistic regression).
Results
Participant characteristics
The current analysis includes 729 HIV-infected and 326 HIV-uninfected women who completed the frailty measures in 2005 and subsequently completed the falls questionnaire at all four semi-annual study visits from 2014–2016 (Table 1). Median time between the frailty assessment and completion of first falls assessment was 9 years. At index visit in 2005, HIV-infected women were older and more likely to be HCV-infected, have renal dysfunction and report symptoms of peripheral neuropathy. HIV-infected women were less likely than uninfected women to be obese, or to report current smoking, current use of marijuana, heroin, crack/ cocaine or heavy alcohol use. Among HIV-infected women at the index visit, median CD4+ T-cell count was 462 cells/μl, 38% reported a prior AIDS defining illness, 67% reported taking ART and 50% had suppressed HIV RNA viral load.
Compared to uninfected women, HIV-infected women were more likely to be frail (14% HIV+ versus 9% HIV-; P=0.04), have weaker grip strength (28% HIV+ versus 19%; P=0.0025), and to report exhaustion (29% HIV+ versus 20%; P=0.002) and unintentional weight loss (14% HIV+ versus 7% HIV-; P=0.0015). The proportions of women reporting single and recurrent falls were similar for HIV-infected and uninfected women. A single fall was reported by 15% of HIV-infected women and 18% of uninfected women; recurrent falls were reported by 25% of HIV-infected women and 21% of uninfected women (overall P=0.20).
Factors associated with single fall and recurrent falls within a 2-year period
Overall, 61% of women had no fall (n=641), 16% had a single fall (n=167) and 23% had recurrent falls (n=247) during the 2-year period that falls were assessed. Median age at first falls assessment was 49 years for women without any falls, 50 years for women who had a single fall and 53 years for those who had recurrent falls (P<0.0001). Among women with frailty, 43% experienced recurrent falls, compared with 22% of non-frail women (P<0.0001; Table 2). All of the individual components of the frailty score were associated with greater proportion of falls in unadjusted analyses (Table 2).
Characteristics associated with single and recurrent falls a

Data are n (%) unless otherwise indicated.
Falls are reported over a 2-year period, approximately 10 years after measurement of frailty and other characteristics. BMI, body mass index; CNS, central nervous system; FFI, Fried Frailty Index.
Frailty as a predictor of single fall and recurrent falls 10 years later
In multivariable models, frailty was not independently predictive of having a single versus no fall over a 2-year period 10 years later, nor was HIV status. Additional factors at the index visit that were independently associated with greater likelihood of having a single fall 10 years later in multivariable models were current marijuana use (aOR 2.43; 95% CI: 1.38, 4.25; P=0.002), former marijuana use (aOR1.71; 95% CI: 1.06, 2.74; P=0.03), hypertension (aOR 1.72; 95% CI: 1.15, 2.57; P=0.009) and neuropathy symptoms (aOR 2.02; 95% CI: 1.17, 3.49; P=0.01; Table 3). In multivariable models of recurrent falls, women who were frail at the index visit had 1.84 times greater odds of sustaining recurrent falls 10 years later (aOR 1.84; 95% CI: 1.13, 2.97; P=0.01). Other factors at the index visit independently associated with increased odds of recurrent falls included older age (aOR 1.49 per 10 years; 95% CI: 1.23, 1.82; P<0.0001), neuropathy symptoms (aOR 2.30; 95% CI: 1.45, 3.66; P=0.0004), obesity (aOR 1.55; 95% CI: 1.10, 2.18; P=0.01), former marijuana use (aOR 1.61, 95% CI: 1.05, 2.47; P=0.03), current marijuana use (aOR 2.09. 95% CI: 1.23, 3.53; P=0.006) and greater number of CNS active medications (aOR 1.44; 95% CI: 1.17, 1.77; P=0.0006). HIV status was not associated with single or recurrent falls.
Overall frailty score and other factors independently associated with single and recurrent falls occurring approximately 10 years later a
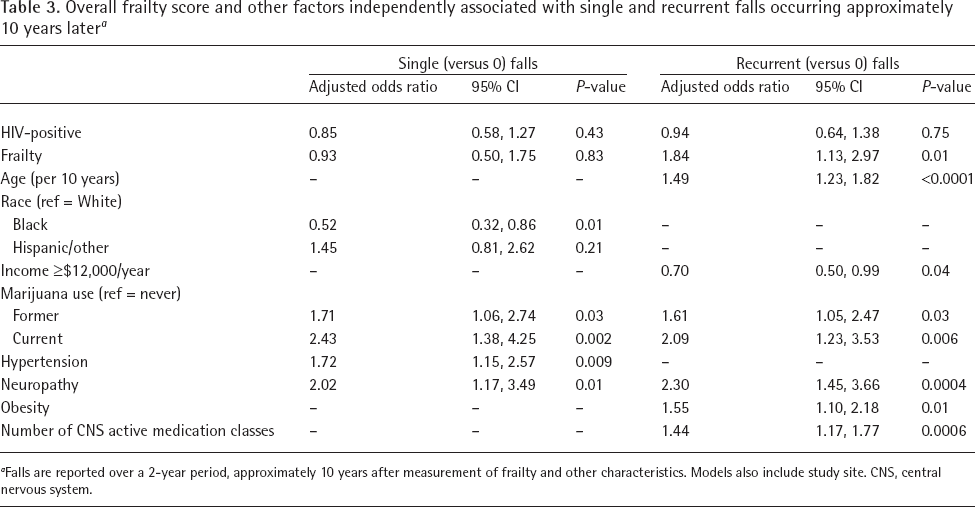
Falls are reported over a 2-year period, approximately 10 years after measurement of frailty and other characteristics. Models also include study site. CNS, central nervous system.
Individual frailty components as predictors of single and recurrent falls 10 years later
We separately evaluated the five components of the Fried frailty phenotype to determine the relative contribution of each on subsequent fall risk over a 2-year period, approximately 10 years later (Table 4). In multivariable analyses of single falls, at index visit, women who reported ≥10 pounds of unintentional weight loss had double the odds of sustaining a single fall approximately 10 years later (aOR 2.31; 95% CI: 1.28, 4.17; P=0.0054), compared to those without unintentional weight loss. Inclusion of unintentional weight loss in models of single fall rather than frailty score did not substantially change other results. In multivariable analyses of frailty components associated with recurrent falls, exhaustion (aOR 1.66; 95% CI: 1.10, 2.50; P=0.02) and weight loss (aOR 1.66; 95% CI: 1.13, 2.46; P=0.01) were associated with greater odds of sustaining recurrent falls approximately 10 years later (Table 4).
Frailty components independently associated with single and recurrent falls occurring approximately 10 years later a
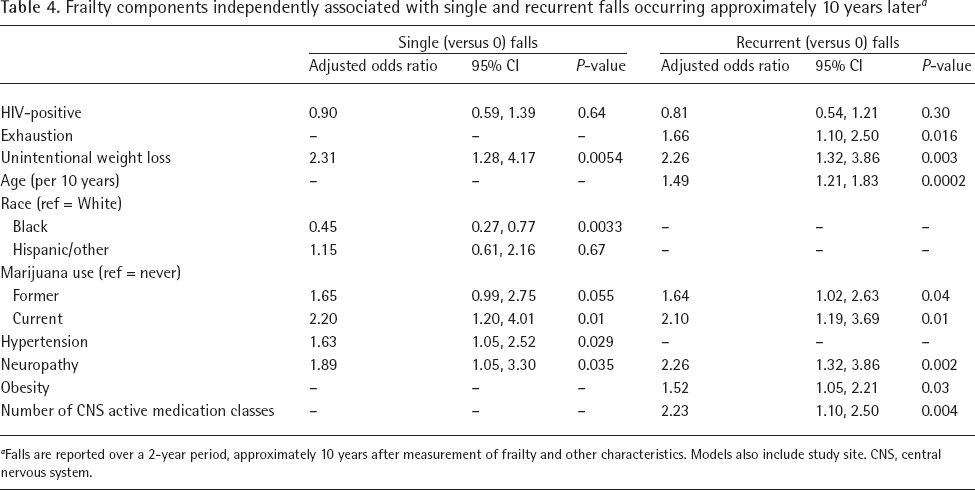
Falls are reported over a 2-year period, approximately 10 years after measurement of frailty and other characteristics. Models also include study site. CNS, central nervous system.
Frailty and individual frailty components as predictors of falls among HIV-infected women
In the subset of HIV-infected women, in multivariable models, compared to no falls, frailty was not significantly associated with 10-year risk of either single fall (aOR 1.16; 95% CI: 0.57, 2.35; P=0.68) or recurrent falls (aOR 1.32; 95% CI: 0.74, 2.33; P=0.34; Table 5). Other factors associated with single fall among HIV-infected women were similar to those in the full WIHS cohort. HIV disease severity and treatment measures were not associated with either single falls in models that included frailty or its components.
Overall frailty score and other factors independently associated with single and recurrent falls occurring approximately 10 years later among women with HIV only a

Falls are reported over a 2-year period, approximately 10 years after measurement of frailty and other characteristics. CNS, central nervous system.
Among the subset of HIV-infected women, as in the full WIHS cohort, age, neuropathy symptoms, obesity, greater number of CNS active medications and marijuana use remained associated with recurrent falls. In contrast, higher education level (aOR 0.58; 95% CI: 0.38, 0.88; P=0.01) and greater CD4+ T-cell count (aOR 0.92 per 100 cells; 95% CI: 0.85, 0.99; P=0.03) were associated with lower odds of recurrent falls among the HIV-infected subset, and income level was no longer statistically significant. In analyses of frailty components and fall risk among HIV-infected women only, unintentional weight loss was independently associated with 10-year risk of single fall (aOR 2.70; 95% CI: 1.40, 5.23; P=0.003), as well as recurrent falls (aOR 1.89; 95% CI: 1.02, 3.51; P=0.04; data not shown).
Frailty and individual frailty components as predictors of falls among HIV-uninfected women
Among HIV-uninfected women, frailty was independently associated with over triple the risk of sustaining recurrent falls over the next 10 years (aOR 3.63; 95% CI: 1.49, 8.85; P=0.005) but was not significantly associated with risk of single fall (Additional file 1). In analyses of frailty components and 10-year fall risk among HIV-uninfected women only, none of the frailty components were associated with single fall, however exhaustion (aOR 2.39; 95% CI: 1.1.4, 5.03; P=0.02) and unintentional weight loss (aOR 3.84; 95% CI: 1.24, 11.82; P=0.02) were associated with recurrent falls (data not shown).
Discussion
Ours is the first study to characterize the long-term relationship of falls with frailty and its components among HIV-infected women. Among predominantly middle-aged HIV-infected and at-risk uninfected women, frailty independently predicted having recurrent falls over a 2-year period approximately 10 years later, but did not predict odds of sustaining only a single fall during this time interval. Of the frailty components, self-reported exhaustion and unintentional weight loss independently predicted greater risk of recurrent falls over the 2-year period; self-reported weight loss also independently predicted single falls. Although falls were frequently reported in this cohort of HIV-infected and at-risk women, HIV serostatus and antiretroviral therapy did not independently predict either single or recurrent falls; however, we found that higher CD4+ T-cell count was associated with reduced risk of recurrent falls.
Frailty, as defined by the Fried phenotype using data from the Cardiovascular Health Study (CHS), has been associated with subsequent adverse outcomes in HIV-uninfected adults ≥65 years of age, including falls, hospitalization, disability and mortality [8]. Although women in the United States and other countries live longer than men [30], women are more likely than men to have activity restrictions in later life [31,32]. While the gap in life expectancy between men and women in the general population has narrowed in recent years, the gap in disability prevalence has increased in favour of men, and thus despite having longer lives, older women no longer live a greater number of active years than men [33]. Similarly, frailty is more frequent among women compared with men [8,34–37], associated with increasing age [8,35,38], and is more strongly associated with mortality among older women than in older men [34,39,40].
Our findings are important because this is the first study of frailty and its association with falls conducted in a racially and ethnically diverse group of women with HIV or at risk for HIV infection. Several large observational studies of older HIV-uninfected women have found associations between frailty and poor outcomes. In the Women's Health Initiative Observational Study, frailty predicted hip fractures, activities of daily living (ADL) disability, hospitalizations and death with 6 years average follow-up [38]. Frail women in the Women's Health and Aging Studies (WHAS) had higher risk of developing ADL disability, institutionalization and death but were not at higher risk of falls [41]. In the Study of Osteoporotic Fractures (SOF), frail older women were at increased short-term risk of recurrent falls, fractures and death across age and body weight categories [10]. Similarly, among older men in the Osteoporotic Fractures in Men (MrOS) study, frailty defined using either the Fried phenotype or a more parsimonious index, predicted short term recurrent falls in addition to incident disability, fracture and mortality [42]. In a meta-analysis of studies of community-dwelling elderly, frailty was associated with higher future fall incidence, including greater pooled risk estimates of any or incident first fall, as well as greater risk of recurrent or incident second falls [43]. Interestingly, in their meta-analysis, Kojima et al. [43] found that the magnitude of fall risk associated with frailty was higher in men than in women.
Our findings support the validity of the frailty phenotype as applicable to non-elderly populations, racially diverse populations including HIV-infected and uninfected minority women. Furthermore, our data are important as they highlight the long-lasting utility of the Fried frailty phenotype in identifying women at increased risk for recurrent falls approximately 10 years later, and suggest that early frailty assessment may be an important tool for identifying non-elderly HIV-infected and uninfected women at risk for falls, as they age. Among the components of the Fried frailty phenotype, self-report of unintentional weight loss, in particular, was a consistent predictor of both single and recurrent falls, suggesting that even among populations of women with prevalent overweight and obesity, with age, weight loss may predict adverse outcomes such as falls.
Frailty is common among HIV-infected women in WIHS, particularly when considering their median age of only 42 years, and has been associated with HIV disease severity including low CD4+ T-cell count and prior AIDS [20,21]. Even in middle age, frailty predicts mortality in PLWH, including among injection drug users in the ALIVE cohort [11], men initiating ART in the Multi-center AIDS Study Cohort (MACS) [13], and women on ART in WIHS [12]. Several cross-sectional studies have reported a high interval prevalence [14,16,17] and incidence [15] of falls among middle aged HIV-infected persons, however data is scarce on the relationship of frailty and falls in PLWH. Similar to our findings, Tassiopoulous et al. [25] reported that among older HIV-infected men and women, frailty was a strong independent predictor of recurrent but not single falls over a 12-month period, and that weak grip strength and slow 4-metre walk were associated with higher odds of recurrent falls.
Our study extends these findings to examine the long-term risk of falls associated with frailty, but differ from others in that neither gait speed, nor grip strength predicted recurrent falls in WIHS. It is unclear whether differences in findings between these studies result from the younger age of WIHS participants, the longer follow-up period, inclusion of HIV-uninfected controls, or sex differences in the effects of frailty components on fall risk. Taken together, these data highlight the importance of examining geriatric syndromes at younger ages in persons living with HIV and at-risk for HIV infection, which may either accelerate ageing via established pathways, or accentuate ageing as an additional risk factor for chronic disease conditions [44].
This study has several strengths. The WIHS is a racially and ethnically diverse cohort that includes HIV-uninfected women with similar demographic and behavioural characteristics to HIV-infected women, allowing comparison by HIV serostatus. Our analysis includes a large sample size with data covering approximately 10 years of follow-up. Participants are well characterized with regard to not only frailty, but also comorbid medical conditions that may affect fall risk. Thus, we are uniquely able to characterize the specific, long-term risk of falls associated with baseline frailty status in this non-elderly population of women, which persists 10 years after the initial frailty assessment. This approach is similar to other algorithms for risk stratification of outcomes of clinical interest, such as the 10-year estimate of cardiovascular risk calculated by the Framingham risk score [45], and the 10- year risk of major osteoporotic fracture estimated by the FRAX tool [46].
Our study also has several limitations. Frailty was measured at one time point only, however, frailty status may change over time. Falls were not ascertained until starting 9 years after the frailty assessment. Frailty is also associated with greater mortality in WIHS, and thus the frail women reporting falls approximately 10 years later reflect only those who survived this time period; this would likely bias our study to underestimate the effects of frailty on fall risk, as frail women may have fallen during the intervening years prior to death. Our study includes only women, and findings may not be generalizable to men.
We observed that frailty and its components observed in middle-aged women with and at-risk for HIV were predictive of increased risk of future falls during a 2-year period that started one decade after the measures were taken. Given greater vulnerability to geriatric syndromes such as frailty and falls experienced by women, future studies of ageing PLWH should examine whether the predictive ability of overall measures of frailty or its components differ by sex. As HIV-infected women continue to age, early frailty screening is an important clinical tool to identify those at greatest risk of falls, in order to develop targeted fall prevention strategies.
Footnotes
The authors declare no competing interests.
Acknowledgements
Data in this manuscript were collected by the Women's Interagency HIV Study (WIHS). The contents of this publication are solely the responsibility of the authors and do not represent the official views of the National Institutes of Health (NIH). Data in this manuscript were collected by five sites of the Women's Interagency HIV Study (WIHS). WIHS (Principal Investigators): Bronx WIHS (Kathryn Anastos), U01-AI-035004; Brooklyn WIHS (Howard Minkoff and Deborah Gustafson), U01-AI-031834; Chicago WIHS (Mardge Cohen and Audrey French), U01-AI-034993; Metropolitan Washington WIHS (Seble Kassaye), U01-AI-034994; Connie Wofsy Women's HIV Study, Northern California (Ruth Greenblatt, Bradley Aouizerat and Phyllis Tien), U01-AI-034989; WIHS Data Management and Analysis Center (Stephen Gange and Elizabeth Golub), U01-AI-042590. The WIHS is funded primarily by the National Institute of Allergy and Infectious Diseases (NIAID), with additional co-funding from the Eunice Kennedy Shriver National Institute of Child Health and Human Development (NICHD), the National Cancer Institute (NCI), the National Institute on Drug Abuse (NIDA), and the National Institute on Mental Health (NIMH). Contents of this publication are solely the responsibility of the authors and do not necessarily represent the official views of the National Institutes of Health. This research was also supported by National Institutes of Health K23AR06199301 (AS).
