Abstract
Background
Tenofovir disoproxil fumarate (TDF) administration in the third trimester for pregnant women with high HBV DNA load has been accepted as a wise practice to prevent mother-to-infant transmission (MTIT). However, for those women who missed the optimal time window of antiviral prophylaxis, this treatment is lacking in the current clinical guidelines.
Methods
Forty-eight pregnant women who did not receive antiviral prophylaxis before 28 weeks of gestation were screened and were administrated with TDF plus hepatitis B immunoglobulin (HBIG; TDF+HBIG group) or TDF alone (TDF group). HBV DNA inhibition and the safety profile were compared between two groups.
Results
A decline of HBV DNA load was observed in both groups after a short period of treatment, and no infant had MTIT. However, compared with the TDF group, the speed of HBV DNA load decline was more rapid (P=0.002) and a much more striking HBV DNA load decline in the first 4 weeks of treatment was exhibited in the TDF+HBIG group (P=0.001). The percentages of mothers with HBV DNA <4 log10 IU/ml and 3 log10 IU/ml at delivery were both much higher in the TDF+HBIG group than the TDF group (P=0.034 and 0.024, respectively). TDF and HBIG were found to be well-tolerated with no safety concerns in the mothers and their infants.
Conclusions
TDF plus HBIG treatment resulted in a rapid HBV DNA load decline in high-risk women who missed the optimal time window of antiviral prophylaxis in pregnancy, which potentially protected infants from HBV infection.
Introduction
HBV infection has long been a serious public health burden with significant morbidity and mortality [1], with a high endemicity reported in most regions of Asia–Pacific and sub-Saharan Africa [2,3]. The transmission of HBV from infected mothers to their infants has been considered as a key contributor to this situation. The prevention of mother-to-infant HBV transmission is an efficient strategy to reduce the morbidity and mortality due to HBV infection. The combined immunoprophylaxis (hepatitis B immunoglobulin [HBIG] and hepatitis B vaccine) for neonates born to hepatitis B surface antigen (HBsAg)+ mothers have reduced the rate of HBV infection in infants to a great extent, however, there is still approximately 10% to 30% of infants born to mothers with high HBV DNA levels suffering from HBV infection [4,5]. The current clinical practice guidelines of European Association for the Study of the Liver (EASL) recommend all pregnant women with high HBV DNA levels should start tenofovir disoproxil fumarate (TDF) treatment at week 24–28 of gestation to prevent mother-to-infant transmission (MTIT) [6].
In daily clinical practice, it is not rare that high-risk pregnant women do not receive antiviral treatment before the 28th week of gestation due to ignoring the importance of antenatal reviewing and HBV screening. In Western China, where the economy is undeveloped and information circulation is limited, numbers of pregnant women miss the optimal time window of antiviral prophylaxis and it is challenging for doctors to administer prophylaxis in time for those high-risk patients. In a previous study, we distributed a questionnaire of knowledge about MTIT randomly to 300 HBsAg+ pregnant women in Shaanxi Province of Western China. Only 20% of mothers acknowledged the efficacy of antiviral treatment during pregnancy to prevent MTIT and 15% had in-depth knowledge about safety of tenofovir administration during pregnancy [7].
Maternal HBV DNA levels were significantly correlated with the risk of MTIT [8]; if antiviral prophylaxis administration during pregnancy did not result in maternal HBV DNA load decline to a safe threshold at delivery, the risk of MTIT is still not eliminated. In China, HBIG has been administrated during pregnancy to prevent MTIT for more than 20 years. It is thought that HBIG may act in the circulation by neutralizing circulating virions expressing HBsAg [9] and perhaps prevent cell-to-cell infection as well as reduce virion secretion upon endocytosis [10]. In addition, our previous study indicated that HBIG can be deposited in the human placenta and it may form a protective immunoglobulin ‘barrier’ between the maternal and fetal blood playing a complementary role to the placental barrier and thus preventing HBV components from permeating the placenta and thereby protecting the fetus from HBV infection [11]. HBIG administration during the antenatal period has been used to prevent HBV MTIT for over 20 years. However, in some studies, it was thought that the efficacy of HBIG to reduce the risk of MTIT was overrated in the mothers with high HBV DNA load [12–15]. However, there is no study to evaluate the efficacy and safety of antiviral treatment combined with HBIG administration during late pregnancy to prevent mother-to-infant HBV transmission, which may become an emergent prophylaxis for pregnant women missing the optimal time window of antiviral prophylaxis. Hence, we screened the high-risk women receiving TDF combined with HBIG starting after the 28th week of gestation and observed the kinetics of maternal HBV DNA load as well as the HBV transmission rate.
Methods
Subjects and treatment
From May 2013 to January 2017, chronic HBV-infected pregnant women undergoing routine consultation were recruited from the Department of Infectious Disease, First Affiliated Hospital of Xi'an Jiaotong University, Shaanxi, China, according to the following inclusion criteria: serum HBsAg-positive for more than 6 months, HBV DNA load more than 10 6 IU/ml; the exclusion criteria were coinfection with hepatitis A, C, D, E virus or HIV, or a history of antiviral therapy before 28 weeks of gestation.
The first-time consultation of these pregnant women was more than the 28th week of gestation. Considering the high risk of mother-to-infant HBV transmission, all of these women received TDF 300 mg daily (Viread; Gilead Sciences, Foster City, CA, USA) from 29-36 weeks of gestation, and some of them received injection of 200 IU hepatitis B immunoglobulin once every 2 weeks (Hualan Biological Engineering Inc., Henan, China) according to personal willingness. Infants born to these mothers had the following immunoprophylaxis schedule: HBIG 200 IU and the first dose of HB vaccine (10 mg; Shenzhen Kangtai Biological Products Co. Ltd, Guangdong, China) within 12 h postpartum, followed by additional doses of HB vaccine at 1 month and 6 months of age. Mother-to-infant HBV transmission was determined by HBsAg or HBV DNA testing from infant peripheral blood at 28 weeks of age, in accordance with well-accepted criteria [16–20]. No response to HB vaccine was defined as infantile anti-HBs titre <10 mIU/ml at 28 weeks of age [21].
HBV serum marker titre, alanine transaminase (ALT) level and HBV DNA load tests for these mothers were monitored every 4 weeks prior to delivery and HBV serum marker titre and HBV DNA load tests for their infants were examined at 28 weeks of age. The study protocol was approved by the ethics committee of the First Affiliated Hospital of Xi'an Jiaotong University.
Laboratory methods
Titres of HBV serum markers, including HBsAg, anti-HBs and hepatitis B e antigen (HBeAg), were quantified with ARCHITECT assays (Abbott Laboratories, Chicago, IL, USA), the positivities were defined as follows: HBsAg >0.05 IU/ml, anti-HBs >10.00 mIU/ml and HBeAg >1.00 s/co, respectively. The serum HBV DNA load was measured by real-time quantitative PCR (Zhongshan University Da An Gene Co. Ltd., Guangzhou, China) with the lower limit of 1.0x10 2 IU/ml. Liver function was performed with an automated bio-analyzer (Olympus AU5400, Mishama, Japan).
Outcome measures and end points
The previous studies indicated excellent efficacy of TDF treatment for pregnant women with high HBV DNA level to reduce HBV transmission rate and reported no infant had HBV infection due to MTIT [22–24]. Maternal HBV DNA load was the independent risk factor of MTIT [8]. In view of the low rate of MTIT due to TDF administration and the relationship between maternal HBV DNA load at delivery and the risk of MTIT [8], the primary outcome of this study was defined as the speed of HBV DNA load decline. In addition, the decline of maternal HBV DNA load from baseline to delivery, the decline of maternal HBV DNA load to the 4th week of treatment, the proportion of HBV DNA load less than limit of detection, 3 log10 IU/ml and 4 log10 IU/ml at delivery and the safety analysis was included in the secondary end points. Adverse events of mothers (including hypertension, gestational diabetes mellitus, fetal growth retardation and disorders in pregnancy) and infants (hypoevolutism and no response to HB vaccine) were recorded at each visit. Data of delivery (including premature delivery, premature rupture of membrane, postpartum haemorrhage, caesarean section) and newborns (including birth defects, Apgar scores, gender and birth weight) were collected from their medical records.
Statistical analysis
Data were analysed statistically with SPSS 13.0 (SPSS, Inc., Chicago, IL, USA). The χ 2 test or Fisher's exact analysis was used for comparison between two groups. The Shapiro–Wilk test and Levene statistic were used to evaluate the normality and homogeneity of the variance, respectively. According to the situation, the t-test or Mann–Whitney U test was used to evaluate differences between two groups. The significance level was set at P<0.05.
Results
Maternal baseline characteristics
A total of 48 pregnant women with HBV DNA load more than 10 6 IU/ml at baseline were included in the final analysis. The 48 women received TDF 300 mg daily after the 28th week of gestation (median: 32 weeks, range: 29–36 weeks). Among them, 22 pregnant women had HBIG injection every 2 weeks (the total doses of HBIG: 400–1,200 IU) before delivery and assigned to the TDF+HBIG group, 26 did not receive HBIG injection and were categorized as the TDF group. Table 1 summarizes the baseline demographics and clinical characteristics of the two groups’ mothers, and there was no statistic difference between two groups.
Comparison of maternal baseline characteristics between two groups

The values are expressed as the median (range) unless otherwise indicated. ALT, alanine transaminase; HBeAg, hepatitis B e antigen; HBIG, hepatitis B immunoglobulin; HBsAg, hepatitis B surface antigen; TDF, tenofovir disoproxil fumarate.
Rapid decrease in HBV DNA load during tenofovir combined with HBIG therapy
As shown in Figure 1A, the HBV DNA load declined significantly in women of both the TDF+HBIG group and the TDF group. Further analysis revealed a more significant HBV DNA load decline in the TDF+HBIG group to the 4th week of treatment (3.23 ±0.94 versus 2.53 ±1.19 log10 IU/ml, Z=-3.290, P=0.001; Figure 1B). To the time of delivery, seemingly more significant viral inhibition was also shown in the TDF+HBIG group, however, there was no statistical difference (5.13 ±1.65 versus 4.33 ±1.29 log10 IU/ml, Z=-1.655, P=0.098; Figure 1C). Whereas, the speed of HBV DNA load decline in the TDF+HBIG group was 0.53 ±0.16 (log10 IU/ml)/week, which was more rapid than the TDF group (0.53 ±0.16 versus 0.38 ±0.15 [log10 IU/ml]/week, Z=-3.166, P=0.002), and the proportions of pregnant women with HBV DNA <3 log10 IU/ml at delivery were higher in the TDF+HBIG group compared with the TDF group (50.00% versus 19.23%; c 2 =5.077, P=0.024), in addition, more women in the TDF+HBIG group showed the levels of HBV DNA <4 log10 IU/ml at delivery (72.73% versus 42.31%; c 2 =4.481, P=0.034).
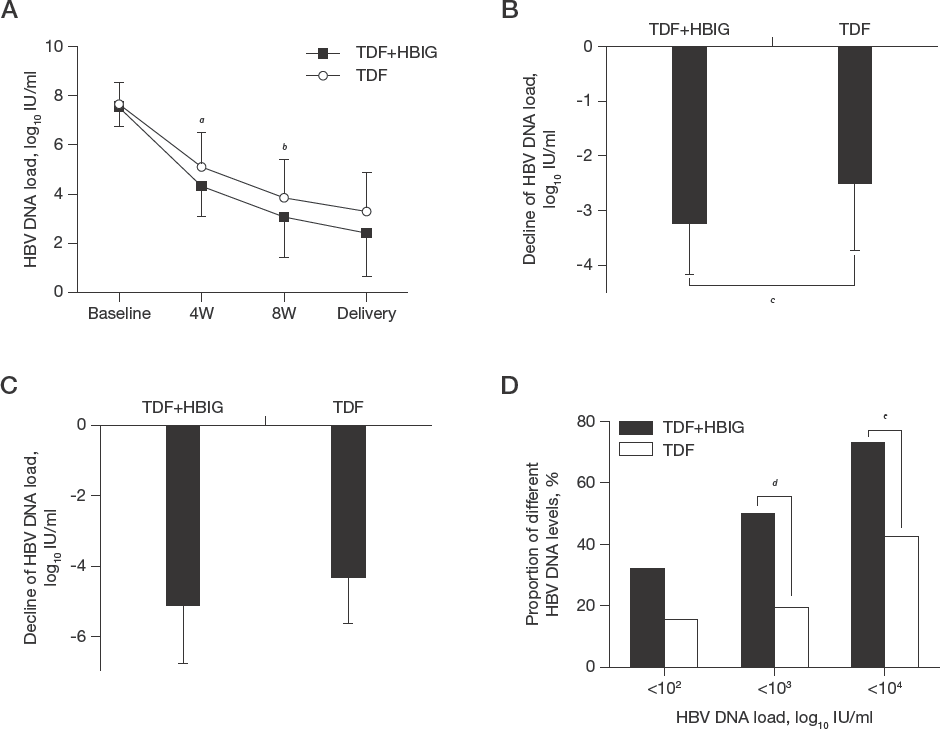
Comparison of HBV DNA load decline between the TDF+HBIG group and the TDF group
Among the 48 pregnant women, 20 subjects started TDF treatment after 32 weeks of gestation (9 in the TDF+HBIG group and 11 in the TDF group), the speed of HBV DNA load decline was more rapid in the TDF+HBIG group (1.02 ±0.25 versus 0.68 ±0.19 log10 IU/ml/week, t=3.743, P=0.003) and a more significant HBV DNA load decline in the TDF+HBIG group was observed, compared with the TDF group (−4.72 ±1.61 versus −3.10 ±1.45 log10 IU/ml, t=-2.336, P=0.031). The same outcomes were observed in 14 pregnant women who started TDF treatment from 36 weeks of gestation (6 in the TDF+HBIG group and 8 in the TDF group), the speed of HBV DNA load decline was more rapid in the TDF+HBIG group (1.14 ±0.11 versus 0.75 ±0.23 log10 IU/ml/week, t=3.823, P=0.002), and a more significant HBV DNA load decline was observed in the 6 TDF+HBIG group patients (−4.22 ±0.88 versus -2.88 ±1.08 log10 IU/ml, t=2.478, P=0.029).
Influence of TDF and HBIG on HBV markers and ALT levels
At baseline, serum HBsAg titre and HBeAg titre in women of the TDF+HBIG group averaged 4.12 ±0.55 log10 IU/ml and 2.46 ±1.24 log10 s/co, respectively, and remained at constant levels during therapy. In addition, similar dynamic patterns of HBsAg and HBeAg were observed in the TDF group (Figure 2A and 2B).

Dynamic change of HBV markers and ALT levels during the treatment
Eight women had elevated ALT levels (>40 U/l) at baseline (four in the TDF+HBIG group and four in the TDF group), seven of the eight women (87.50%) recovered to normal levels before delivery, and there was no difference in the proportion of ALT turning to normal levels between the TDF+HBIG group and the TDF group (75.00% versus 100%, χ 2 =1.143, P=1.000; Figure 2C and 2D).
Safety of TDF and HBIG in mothers and infants
In the current study, 48 mothers with chronic HBV infection had 48 infants. TDF and HBIG were generally well tolerated by the mothers and their infants. No maternal severe adverse effects were observed. None of the mothers experienced an adverse clinical outcome, and no infants had congenital deformities. In addition, there were no differences in maternal adverse events or data on children between the TDF+HBIG group and the TDF group (Table 2). After 28 weeks of follow-up, there was no mother-to-infant HBV transmission in the 48 infants, whereas, anti-HBs was undetectable in two infants (4.17%) after full immunization (one in the TDF+HBIG group and one in the TDF group).
Maternal adverse events and infant statistics
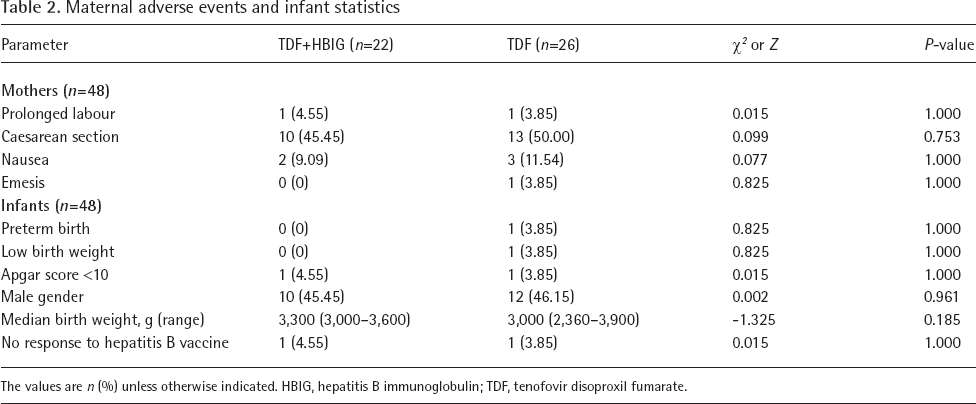
The values are n (%) unless otherwise indicated. HBIG, hepatitis B immunoglobulin; TDF, tenofovir disoproxil fumarate.
Discussion
In this study, high-risk pregnant women who received no antiviral treatment before the 28th week of gestation were screened. TDF combined with HBIG (TDF+HBIG group) or TDF alone (TDF group) was administered for these women and HBV DNA inhibition was compared between two groups. The data showed more potent viral inhibition in the TDF+HBIG group mothers, which potentially protected infants from HBV infection due to MTIT.
The combined immunoprophylaxis reduces the rate of mother-to-infant HBV transmission from 80–90% to 10–30% in mothers with high HBV DNA load [25,26]. When TDF was administrated during pregnancy to reduce the transmission rate in high-risk mothers, no infant with chronic HBV infection was reported in the previous studies [22,23,27,28]. However, the time of starting TDF treatment did not exceed 32 weeks of gestation in these studies. In this study, the median start of TDF administration was the 32nd week and the range was from the 29th to 36th week. The duration of TDF treatment was short and therefore rapid decline of maternal HBV DNA load before delivery was needed to block the MTIT.
Maternal HBV DNA load was the independent risk factor of HBV MTIT. Wen et al. [8] reported that the predictive rates of MTIT at maternal viral load levels of 5, 6, 7, 8, 9 log10 copies/ml were 0.9% (95% CI −0.9%∼2.7%), 2.6% (95% CI −1.1%∼6.2%), 6.6% (95% CI 0.5%∼12.6%), 14.6% (95% CI 5.6%∼23.6%) and 27.7% (95% CI 13.1%∼42.4%), respectively. The lower the maternal HBV DNA levels were, the lower the risk of HBV MTIT. In our study, compared to mothers with TDF treatment alone, the speed of HBV DNA load decline was more rapid in the TDF+HBIG group (P=0.002), and there was a significant HBV DNA load decline at the 4th week of treatment in mothers with TDF+HBIG treatment (P=0.001), which indicated potent viral inhibition of TDF combined with HBIG treatment. The percentages of mothers with HBV DNA <4 log10 IU/ml and 3 log10 IU/ml at delivery were both much higher in TDF+HBIG group than TDF group (P=0.034 and 0.024, respectively), HBV DNA <4 log10 IU/ml had been accepted as a relatively safe threshold for combined immunoprophylaxis to prevent MTIT [8,29,30]. In addition, there were 20 pregnant women starting TDF treatment after 32 weeks of gestation and 14 pregnant women starting prophylaxis at 36 weeks of gestation; limited antenatal prophylaxis time may result in much higher risk of MTIT. However, there was a more significant HBV DNA load decline in the TDF+HBIG group, no infant had HBV infection. For those pregnant women who had much more limited time of antenatal prophylaxis, it may be reasonable to administer TDF combined with HBIG to prevent MTIT.
The safety of TDF alone and HBIG alone used in pregnancy are high [12–15,22,23,27,28]. In the present study, TDF combined with HBIG was well tolerated by mothers and infants. No adverse clinical outcomes or birth defects were recorded and the rates of adverse events were not different between two groups. In addition, there was no difference in the proportion of ALT returning to normal level and ALT fare during pregnancy between two groups and the response to HB vaccine in the TDF+HBIG group infants was comparable to the TDF group of children. Thus, TDF combined with HBIG therapy demonstrated an excellent safety and tolerability profile.
In this study, our data indicated TDF combined with HBIG administration resulted in a more rapid and significant decline of HBV DNA load, but no decline of HBsAg and HBeAg titre, compared with TDF alone in late pregnancy, however, the mechanism of rapid decline of HBV DNA load due to TDF+HBIG administration was still unclear. There are some limitations of this study. It was not randomized and the number of mother–infant pairs was moderate. The difference of MTIT rate between two groups will be compared in our further study.
In conclusion, TDF combined with HBIG treatment exhibited rapid and potent viral inhibition in those high-risk mothers who missed the optimal time window of antiviral prophylaxis in pregnancy, which potentially protected infants from HBV infection due to MTIT.
Footnotes
Acknowledgment
This study was supported by the National Natural Science Foundation of China (No. 81702119, No. 81770594 and No. 81670537).
The authors declare no other funding or conflicts of interest relating to the publication of this manuscript.
