Abstract
Background
Recent studies suggest that serum lipids are associated with pegylated interferon-alpha (PEG-IFN-α) treatment response in chronic hepatitis C patients. However, the role of serum lipids in influencing the outcome of HBV treatment is not well understood. This study aims to investigate the association of serum lipids with the response to interferon-alpha treatment for chronic hepatitis B (CHB) patients.
Methods
We dynamically measured 11 clinical serum lipid parameters of 119 hepatitis B e antigen (HBeAg)-positive CHB patients, including 53 patients who achieved sustained response (SR) and 66 patients who achieved non-response (NR) induced by PEG-IFN-α treatment for 48 weeks.
Results
The dynamic analysis showed that the baseline serum total cholesterol (TCHO) level was higher in the NR group than that in the SR group (P=0.004). Moreover, the correlation analysis demonstrated a significant positive correlation between TCHO and hepatitis B surface antigen (HBsAg) at baseline (P=0.009). In addition, CHB patients with high baseline TCHO levels exhibited higher HBV DNA, HBsAg, HBeAg and hepatitis B e antibody (HBeAb) levels during early treatment periods (weeks 0, 4, 12 and 24) than those with the low TCHO levels. Furthermore, the logistic regression analysis identified that baseline serum TCHO was a risk factor for NR achievement (OR=4.94; P=0.047). Conclusions: Our results indicated that serum TCHO was associated with PEG-IFN-α therapeutic response in HBeAg-positive CHB patients which suggested that serum TCHO could be useful as an auxiliary clinical factor to predict poor efficacy of PEG-IFN-α therapy.
Introduction
More than 240 million people worldwide are chronically infected with HBV and HBV is a major cause of cirrhosis, which may lead to liver failure and liver cancer [1,2]. Thus far, interferons and nucleoside/ nucleotide analogues (NAs) are widely used to treat HBV [3]. Among them, pegylated interferon-alfa (PEG-IFN-α) possesses the highest off-treatment sustained response (SR) rate and relatively low risk of post-treatment relapse after a 48-week course of treatment in chronic hepatitis B (CHB) patients [4,5]. Nevertheless, the effect of PEG-IFN-α on HBV is still rather poor. For instance, the clearance of hepatitis B surface antigen (HBsAg) occurs in only 3% to 11% of CHB patients treated with PEG-IFN-α [6–8]. The loss of hepatitis B e antigen (HBeAg) occurs in only 30% of CHB patients treated with PEG-IFN-α [9,10]. Moreover, the underlying reason for this treatment non-response (NR) in HBV patients remains unknown.
Increasing evidence has supported that chronic HBV infection can influence serum lipid metabolism [11–13]. For example, HBV infection induces the expression of genes involved in cholesterol synthesis and promotes cholesterol provision [14]. The hepatitis B virus X protein can inhibit the secretion of apolipoprotein B (APO-B) and accelerate fatty acid synthase [15,16]. Conversely, serum lipids also influence the HBV infection [17–19]. However, serum lipid indexes that reflect the therapeutic efficacy of HBV treatments have not been so reliable. Currently, it has been confirmed that serum lipids are correlated with treatment response to PEG-IFN-α in chronic hepatitis C patients [20,21]. The low-density lipoprotein cholesterol (LDL-C) is a predictive factor of early and sustained responses to PEG-IFN-α plus ribavirin combination therapy in patients infected with HCV [22]. Higher baseline serum LDL and cholesterol levels may be significant prognostic indicators for the outcome of HCV treatment with a combination of IFN and ribavirin [23,24]. However, whether serum lipids play any role in influencing the outcome of HBV treatment has not yet been explored.
In this study, we aim to explore the levels of serum lipids in HBeAg-positive CHB patients receiving PEG-IFN-α treatment to investigate the association of serum lipids with PEG-IFN-α treatment response.
Methods
Patient characteristics
A total of 119 HBeAg-positive CHB patients who had received anti-HBV treatment by PEG-IFN-α for 48 weeks were enrolled in this study. CHB patients provided written informed consent and this study was approved by the Ethics Committee of the First Affiliated Hospital of Fujian Medical University. The inclusion criteria were as follows: the presence of serum HBsAg for 6 or more months; an age greater than 16 years old; a previous lack of antiviral treatment. PEG-IFN-α was commenced when the HBV DNA level was more than 20,000 IU/ml and when either the alanine aminotransferase (ALT) level was greater than 2x the upper limit of normal (40 U/l) or biopsy showed significant fibrosis/cirrhosis. The exclusion criteria were a coinfection with HCV or HIV, a history of organ transplantation, decompensated liver cirrhosis and a concurrent use of immunomodulatory drugs or corticosteroids. Two treatment strategies were performed among the 119 CHB patients. 65 patients received PEG-IFN-α monotherapy and the other 54 patients received PEG-IFN-α add-on NA therapy for 48 weeks. PEG-IFN-α was administered by intramuscular injection (PEG-IFN-α2a, 180 μg/week or PEG-IFN-α2b, 100 μg/week) for 48 weeks and NAs were given orally at the regular clinical dose (tenofovir disoproxil fumarate [TDF], 300 mg/day or adefovir [ADV], 10 mg/day or entecavir [ETV], 0.5 mg/day). According to curative effects at 24 weeks post-treatment (week 72), the 119 HBeAg-positive CHB patients were divided into two groups: SR group and NR group. A total of 53 patients achieved SR while the other 66 patients had NR after receiving 48 weeks of PEG-IFN-α monotherapy or PEG-IFN-α add-on NA treatment. SR to treatment was defined according to the EASL guidelines [25] as HBeAg <1 S/CO, HBV DNA <500 IU/ml and ALT <40 U/l when followed-up at 24 weeks post-treatment (week 72) in both of the treatment groups. Patients were considered to have NR to therapy if they did not meet all of the criteria above.
Measurement of clinical indicators
The clinical serum lipid indicators that were measured for this study included TCHO, triglyceride (TG), high-density lipoprotein cholesterol (HDL-C), HDL-C/C (the ratio of HDL-C to TCHO), LDL-C, very low-density lipoprotein cholesterol (VLDL-C), apolipoprotein A1 (APO-A1), APO-B, APO-A1/B, lipoprotein α (LP[α]) and non-esterified fatty acid (NEFA). The serum samples for clinical indicator measurement were obtained from fresh whole blood collected in tube without anticoagulants. All of the clinical indicators were quantified in samples taken at baseline and during the treatment period and follow-up periods. Sampling was performed at weeks 0, 4, 12, 24, 36, 48, 60 and 72 (week 0, namely baseline, was the time point at which treatment began). HBV DNA was detected by quantitative real-time PCR method (Sansure Biotech Inc., Hunan, China) and Roche Lightcycler 480 (Roche Corporation, Basel, Switzerland) with a lower limit of detection <500 IU/ml. HBsAg, HBeAg, anti-HBe and anti-HBc were quantified using an automated chemiluminescent microparticle immunology analyser (Abbott I2000, Abbott Laboratories, Chicago, IL, USA). Serum ALT and lipids were detected using an automatic biochemical analyser ADVIA 2400 (Siemens, Munich, Germany).
Statistical analyses
Data were performed using the SPSS version 20 software (SPSS, IBM, Chicago, IL, USA) and expressed as percentages and mean ± standard deviation (sd) or standard error of the mean (
Results
Clinical characteristics of HBeAg-positive CHB patients receiving PEG-IFN-α treatment
Among 119 HBeAg-positive CHB patients, 65 patients were treated with PEG-IFN-α monotherapy and 54 patients were treated with PEG-IFN-α add-on NAs. Baseline characteristics of patients are shown in Table 1. SR group had significantly lower mean HBsAg level (3.9 ±0.6 versus 4.1 ±0.5 log10 IU/ml; P=0.029), higher mean ALT level (270 ±203 versus 176 ±105 U/l; P=0.003), lower mean HBeAg level (2.7 ±0.6 versus 3.0 ±0.3 log10 S/CO; P<0.001) and lower mean anti-HBe level (29.3 ±22.5 versus 47.5 ±20.8 S/CO; P<0.001) than NR group. HBV DNA level was decreased in SR group compared with NR group although there were no statistical differences. No significant differences were observed in other baseline parameters between the two groups.
Clinical characteristics of the study cohort
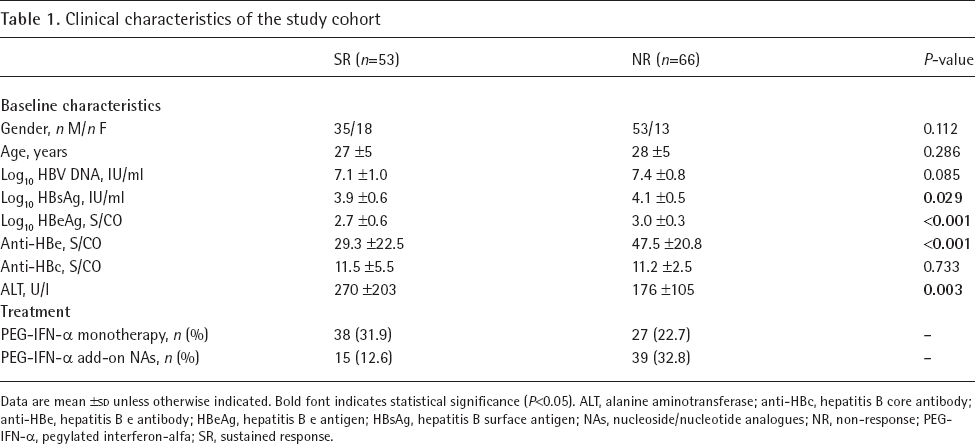
Data are mean ±
Correlation between serum lipid levels and therapeutic response
To identify whether there was a correlation between serum lipids and the anti-HBV therapeutic response, we compared the levels of all the clinical serum lipid indicators (TCHO, TG, HDL-C, HDL-C/C, LDL-C, VLDL-C, APO-A1, APO-B, APO-A1/B, LP[α] and NEFA) between SR and NR groups. We found that the baseline serum level of TCHO (4.62 ±0.85 versus 4.19 ±0.71 mmol/l; P=0.004), LDL-C (2.79 ±0.77 versus 2.45 ±0.63 mmol/l; P=0.011) and APO-B (0.88 ±0.22 versus 0.79 ±0.20 mmol/l; P=0.029) were higher in the NR group than those in the SR group (Figures 1A-C). While the baseline serum level of HDL-C/C was lower in NR group compared to SR group (0.31 ±0.09 versus 0.35 ±0.09, P=0.039; Figure 1D). No significant differences were observed in other clinical baseline serum lipid levels between two groups (data not shown). Furthermore, we analysed the dynamic variations of serum TCHO, LDL-C, HDL-C/C and APO-B levels throughout the course of treatment. We found that only baseline serum levels had significant differences between SR and NR groups (Figure 2). Besides, serum TCHO and LDL-C levels tended to decline throughout the course of treatment.

The baseline serum lipid levels correlated with the therapeutic response to PEG-IFN-α in HBeAg-positive CHB patients
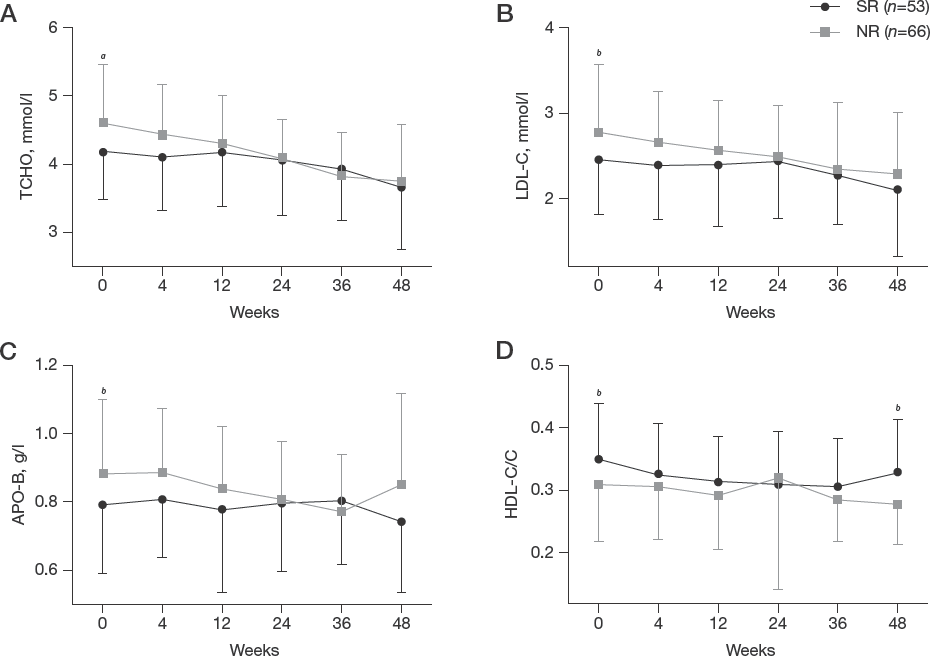
The dynamic variations of serum lipid levels throughout the course of treatment
Correlation between serum lipid levels and serum markers of HBV replication before treatment
Correlation analyses showed a significant positive correlation between TCHO and log10 HBsAg (r=0.247; P=0.009) as well as anti-HBe (r=0.255; P=0.008), and a significant negative correlation between TCHO and anti-HBc (r=-0.237; P=0.019; Table 2). LDL-C showed positive correlation with log10 HBsAg (r=0.207; P=0.031) and was negatively correlated with anti-HBc (r=-0.307; P=0.002; Table 2). HDL-C/C showed positive correlation with anti-HBc (r=0.245; P=0.016) and APO-B showed negative correlation with anti-HBc (r=-0.368; P=0.001; Table 2).
Correlation of serum lipids with HBV markers at baseline in HBeAg-positive CHB patients
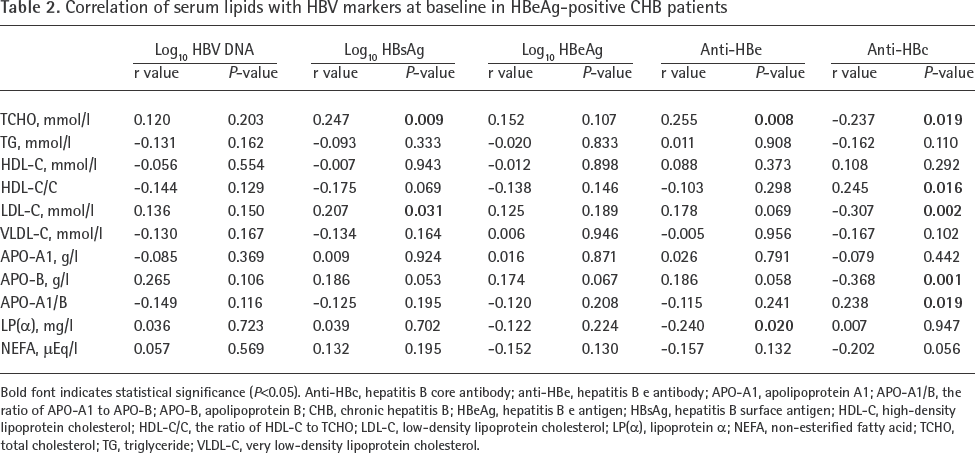
Bold font indicates statistical significance (P<0.05). Anti-HBc, hepatitis B core antibody; anti-HBe, hepatitis B e antibody; APO-A1, apolipoprotein A1; APO-A1/B, the ratio of APO-A1 to APO-B; APO-B, apolipoprotein B; CHB, chronic hepatitis B; HBeAg, hepatitis B e antigen; HBsAg, hepatitis B surface antigen; HDL-C, high-density lipoprotein cholesterol; HDL-C/C, the ratio of HDL-C to TCHO; LDL-C, low-density lipoprotein cholesterol; LP(α), lipoprotein a; NEFA, non-esterified fatty acid; TCHO, total cholesterol; TG, triglyceride; VLDL-C, very low-density lipoprotein cholesterol.
To investigate the influence of the baseline serum TCHO on chronic HBV infection in greater detail, according to the clinical reference range of serum TCHO (3.6∼5.69), the 119 HBeAg-positive CHB patients were divided into three groups: low TCHO group (baseline serum TCHO levels were <3.6 mmol/l); middle TCHO group (baseline serum TCHO levels were from 3.6 to 5.69 mmol/l); high TCHO group (baseline serum TCHO levels were >5.69 mmol/l). Then, the dynamic variations of six clinical serological and virological indices (HBsAg, HBeAg, anti-HBe, anti-HBc, HBV DNA and ALT) in different levels of serum TCHO throughout the course of treatment were analysed. We found that CHB patients with the high baseline TCHO levels exhibited higher HBV DNA, HBsAg, HBeAg and HBeAb levels during early treatment periods (weeks 0, 4, 12 and 24) than those with the low TCHO levels (Figures 3A–D). However, no correlation was found between decreases in two indices (anti-HBc and ALT) compared with the respective controls (Figures 3E–F), both during the course of treatment (0–48 weeks) and at 24 weeks post-treatment (week 72). The above analysis indicated that CHB patients harbouring high baseline serum TCHO levels exhibited a relatively poor therapeutic response to PEG-IFN-α.

The dynamic variations of clinical serological and virological indices in different levels of baseline serum TCHO throughout the course of PEG-IFN-α treatment
Baseline serum TCHO may serve as a risk factor for therapeutic response to PEG-IFN-α among HBeAg-positive CHB patients
Logistic regression analysis was used to identify risk factors of therapeutic response. Univariate analysis identified eight parameters that influenced the SR. The baseline serum HBsAg, HBeAg, anti-HBe, TCHO, LDL-C and APO-B were suggested as detrimental to SR achievement for CHB patients treated with PEG-IFN-α (OR=2.37, P=0.036; OR=6.50, P=0.001; OR=1.04, P<0.001; OR=2.02, P<0.005; OR=2.02, P=0.014; OR=7.24, P=0.033, respectively; Table 3). Conversely, we found two protective effects of baseline serum ALT and HDL-C/C to achieve SR (OR=0.996, P=0.004; OR=0.013, P=0.042, respectively; Table 3). Multivariate analysis that included the above variables identified three parameters that influenced the SR. The baseline serum HBeAg and TCHO were still suggested as detrimental to SR achievement (OR=19.15, P=0.022; OR=4.94, P=0.047, respectively) and ALT was still suggested as protective to SR achievement (OR=0.994, P=0.004; Table 3). These results suggested that baseline serum TCHO, as the only clinical serum lipid indicator that was correlated with a detrimental outcome in the multivariate analysis, can be used as an effective risk factor for identifying the non-response to PEG-IFN-α therapy before treatment for HBV infection.
Logistic regression analysis to identify risk factors of PEG-IFN-α treatment response in HBeAg-positive CHB patients
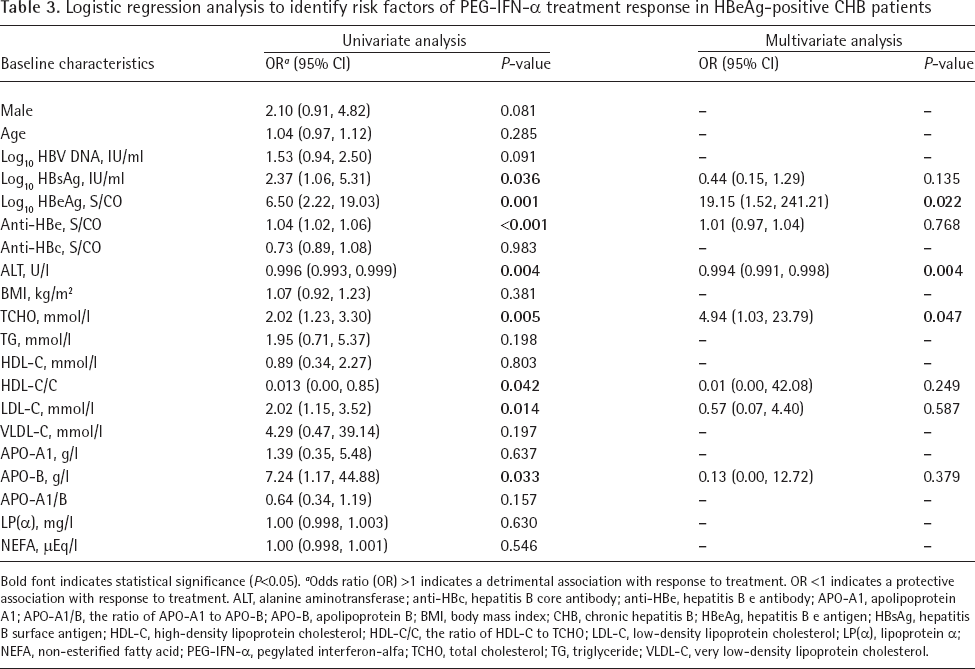
Bold font indicates statistical significance (P<0.05). a Odds ratio (OR) >1 indicates a detrimental association with response to treatment. OR <1 indicates a protective association with response to treatment. ALT, alanine aminotransferase; anti-HBc, hepatitis B core antibody; anti-HBe, hepatitis B e antibody; APO-A1, apolipoprotein A1; APO-A1/B, the ratio of APO-A1 to APO-B; APO-B, apolipoprotein B; BMI, body mass index; CHB, chronic hepatitis B; HBeAg, hepatitis B e antigen; HBsAg, hepatitis B surface antigen; HDL-C, high-density lipoprotein cholesterol; HDL-C/C, the ratio of HDL-C to TCHO; LDL-C, low-density lipoprotein cholesterol; LP(α), lipoprotein α; NEFA, non-esterified fatty acid; PEG-IFN-α, pegylated interferon-alfa; TCHO, total cholesterol; TG, triglyceride; VLDL-C, very low-density lipoprotein cholesterol.
Discussion
Currently, clinical studies suggested that several factors were associated with PEG-IFN-α therapeutic efficacy in CHB patients [26]. However, these factors were gained several weeks after treatment began. The ideal factors would be assessed before treatment to identify CHB patients that were not likely to respond to PEG-IFN-α therapy. We previously reported that polymorphisms of CYP27B1 were associated with PEG-IFN-α therapeutic efficacy in HBeAg-positive patients [27] and baseline serum HBV large surface protein (HBV-LP) level can be used for predicting virological response to PEG-IFN-α in CHB patients [28]. Nevertheless, these indicators have not been commonly detected in clinic.
Previous reports found that serum lipids played a key role in determining the outcome of HCV infection [29]. However, the sustained role of serum lipids in regulating HBV infection during PEG-IFN-α treatment is largely unknown. The present study demonstrated that a majority of NR CHB patients harbour high baseline serum TCHO levels compared to SR CHB patients. Besides, the dynamic analysis of serum TCHO levels throughout the course of treatment showed that only baseline TCHO levels had significant differences between SR and NR groups. We also examined the correlation between TCHO and HBV markers. The results discovered that baseline serum TCHO showed positive correlation with HBsAg, as well as anti-HBe. Moreover, CHB patients with the high baseline TCHO levels exhibited higher HBV DNA, HBsAg, HBeAg and HBeAb levels during early treatment periods (weeks 0, 4, 12 and 24) than those with the low TCHO levels. These results revealed that CHB patients harbouring high baseline serum TCHO levels exhibited a relatively poor therapeutic response to PEG-IFN-α. On the other hand, the univariate regression analysis suggested that low ALT concentrations, high HBsAg concentrations and high HBeAg concentrations at baseline associated with poor response to PEG-IFN-α, which are consistent with previous reports [30–32]. In addition, these data also recommended that high baseline HBV DNA and anti-HBe concentrations were concerned with NR to PEG-IFN-α. Interestingly, baseline serum TCHO was the only clinical serum lipid indicator that was statistically significantly correlated with a detrimental outcome to PEG-IFN-α therapy in the multivariate analysis.
The above analysis indicated that high serum TCHO levels were associated with failure to achieve durable HBeAg seroconversion induced by PEG-IFN-α therapy. To investigate whether serum TCHO levels were associated with spontaneous HBeAg seroconversion, we compared 81 patients with HBeAg-negative chronic HBV infection (inactive carriers) and 50 patients with HBeAg-positive chronic HBV infection (immune tolerant carriers). Clinical characteristics of patients were shown in Additional file 1. We found that the TCHO levels in the immune tolerant carriers group were significantly higher than those in the inactive carriers group (4.92 ±0.88 versus 4.56 ±0.73 mmol/l; P=0.011; Additional file 2). These data indicated that lower TCHO levels were associated with a state that is more likely to lead to spontaneous HBeAg seroconversion.
Considered together, our results suggest that the baseline TCHO can be used as a clinical risk factor that can be easily detected before clinical PEG-IFN-α treatment. However, the reason why serum cholesterol is concerned with therapeutic efficacy of PEG-IFN-α in CHB patients is unclear.
Previous studies have offered some evidence: bile acids increase HBV gene expression which counteracts the antiviral effect of IFN-α [33] and inhibits the induction of proteins involved in the antiviral activity of IFN, such as 2,5-oligoadenylate synthetase (OAS) [34]. It is noteworthy that bile acids derive from cholesterol, namely the cholesterol 7α-hydroxylase promotes bile acid synthesis by increasing the conversion of cholesterol to bile acids [35]. Therefore, we suppose that serum cholesterol affects the efficacy of IFN-α treatment through influencing the bile acid metabolism in CHB patients, which may need further validation.
In this study, a certain proportion of CHB patients were assigned to receive PEG-IFN-α add-on NAs. To investigate whether the treatment regimen affects the level of TCHO, we compared the serum TCHO levels from CHB patients treated with either PEG-IFN-α monotherapy or PEG-IFN-α/NA add-on at the indicated time points since PEG-IFN-α treatment. However, no significant differences were detected at any time point since PEG-IFN-α treatment (Additional file 3).
In conclusion, our findings identify a previously unrecognized role of baseline serum TCHO in HBeAg-positive CHB patients during PEG-IFN-α treatment. The high baseline serum TCHO levels can provide an explanation for the poor therapeutic effect of PEG-IFN-α treatment in some CHB patients. Moreover, these findings present an auxiliary clinical indicator for evaluating the efficacy of PEG-IFN-α therapy in CHB patients.
Footnotes
The authors declare no competing interests.
Acknowledgements
This work was supported by the National Natural Science Foundation of China (grant number 81572067); and the Joint Funds for the Innovation of Science and Technology of Fujian Province (grant number 2016Y9020).
