Abstract
Background
This study was aimed at evaluating the clinical significance of serum HBV RNA, hepatitis B core-related antigen (HBcrAg) and hepatitis B core antibody (anti-HBc) levels in chronic hepatitis B patients with undetectable HBV DNA during nucleoside/nucleotide analogue (NA) treatment.
Methods
Fifty-seven patients who received long-term NA treatment of median 5.83 (25%, 75% percentiles 4.67, 7.75) years were enrolled, and 285 serum samples at five time points for each patient were quantitatively analysed for the three serum markers together with serum HBV DNA and hepatitis B surface antigen (HBsAg) levels.
Results
The HBV RNA level significantly correlated with HBcrAg (r=0.629; P<0.001) but not HBsAg levels (P=0.1460). Nonetheless, the HBcrAg level significantly correlated with the HBsAg level (r=0.469; P<0.001). Hepatitis B e antigen (HBeAg)-positive samples showed higher HBV RNA, HBcrAg and HBsAg levels than HBeAg-negative samples did (all P<0.05). Nine patients with HBeAg loss manifested a significantly greater decline in HBV RNA and HBcrAg levels (median 1.84 [25%, 75% percentiles 1.02, 2.12] log10 copies/ml, 1.14 [0.62, 2.21] log10 U/ml, respectively) compared with those in seven patients without HBeAg loss (0.74 [0.10, 1.08] log10 copies/ml and 0.41 [0.21, 0.69] log10 U/ml, respectively). Overall, serum HBV RNA, HBcrAg, HBsAg and anti-HBc levels gradually decreased with time during NA treatment. At the end of observation, HBV RNA and HBcrAg reached an undetectable level in 26 and 6 (46% and 11%) patients, respectively. Conclusions: Monitoring of HBV RNA and HBcrAg levels is useful for NA-treated patients with undetectable HBV DNA. The attainment of HBV RNA undetectability usually occurs prior to HBcrAg undetectability.
Introduction
Chronic hepatitis B (CHB) affects approximately 292 million people worldwide [1]. HBV infection is a major pathogenic factor leading to CHB, cirrhosis, liver failure and hepatocellular carcinoma (HCC) [2]. Eradication of covalently closed circular DNA (cccDNA) has been suggested as the therapeutic goal in chronic HBV infection because cccDNA persistence in infected hepatocytes causes an off-therapy DNA rebound and hinders CHB cure [3]. A liver biopsy for quantitation of intrahepatic cccDNA is the most accurate measurement of the viral reservoir but is limited by its invasive nature. Noninvasive serological markers can serve as surrogate indicators of intrahepatic viral replicative activity. Quantitation of HBV DNA, hepatitis B e antigen (HBeAg) and hepatitis B surface antigen (HBsAg) as markers of viral replication is important but not sufficient for adequate management of patients with chronic HBV infection during nucleoside/nucleotide analogue (NA) therapy. Data on the quantitation of additional HBV markers such as serum HBV RNA, hepatitis B core-related antigen (HBcrAg) and hepatitis B core antibody (anti-HBc) accumulated in recent years [4–7].
We, and other investigators, recently demonstrated that serum HBV RNA is the encapsidated pregenomic RNA (pgRNA) that may exist inside virion-like particles [8,9]. The serum HBV RNA level may be an indicator of the intrahepatic transcriptional activity of cccDNA and is associated with liver histological changes in CHB patients receiving NA therapy [10]. Estimation of serum HBV DNA plus RNA levels much better reflects the activity of intrahepatic cccDNA in treatment-naive CHB patients [11]. In addition, the serum HBV RNA level has been found to be associated with the probability of a response not only to NA therapy but also to pegylated interferon treatment [8,12–15].
HBcrAg includes three species of related proteins sharing an identical 149-amino-acid sequence: hepatitis B core antigen (HBcAg; which is synthesized from pgRNA) as well as HBeAg and a 22 kDa truncated precore protein (p22Cr), which are synthesized from precore RNA [16]. HBcrAg has been proposed as a novel serum marker for disease monitoring and prognostication of CHB [9,16]. HBcrAg has been suggested to be a surrogate marker of the transcriptional activity of cccDNA [17]. HBcrAg correlates to various degrees with intrahepatic cccDNA, total HBV DNA and serum HBV DNA and to a lesser extent with HBsAg. A real-word cohort study has revealed that the HBcrAg level declines at a relatively higher rate as compared with the HBsAg level in the CHB patients receiving long-term entecavir (ETV) treatment [18].
Qualitative anti-HBc positivity is indicative only of a previous HBV infection. By contrast, quantitative anti-HBc analysis has been reported to be helpful for predicting an antiviral-treatment response in CHB patients and the risk of occult HBV infection reactivation in immunosuppressed patients [19,20]. In addition, the anti-HBc level has been reported to be useful for predicting significant liver inflammation in CHB patients with normal or near-normal alanine aminotransferase activity [21].
In clinical practice, classical serum HBV markers such as HBeAg and HBsAg are not fully satisfactory markers for monitoring HBV activity in the CHB patients with undetectable HBV DNA during long-term NA treatment. Longitudinal data are still limited for the above-mentioned emerging serum HBV markers regarding better management of these patients. In this study, therefore, we aimed to evaluate clinical implications of serum HBV RNA, HBcrAg and anti-HBc levels and to analyse the correlations among the serum HBV markers in the CHB patients who have achieved clearance (undetectability) of HBV DNA after long-term NA treatment.
Methods
Patients
Similar to what we previously described [22], consecutive CHB patients who underwent NA therapy and accepted drug-resistance testing in Beijing 302 Hospital from 2007 to 2017 were taken as study candidates. The enrolled patients were from the candidates and had to meet the following criteria: intact follow-up clinical information during long-term continuous NA therapy; available serially collected serum samples; persistent undetectable HBV DNA (<40 IU/ml) and no detectable HBV resistance mutation; and agreement to be taken into the Database of Beijing 302 Hospital and informed consent for the use of the samples for the study. Patients who were coinfected with other hepatitis viruses or HIV were excluded. Fifty-seven patients who received long-term NA treatment at Beijing 302 Hospital and had serial serum samples during the treatment were enrolled in the study.
Fifty-seven patients were finally enrolled in the study. Among them, 21 patients were treated with ETV monotherapy, five were treated with adefovir dipivoxil (ADV) monotherapy, 13 were treated with ADV+ETV, 11 were treated with ADV+ lamivudine, 5 were treated with successive ETV and ADV, and 2 were treated with successive ETV and tenofovir disoproxil fumarate. The study protocol was approved by the Ethics Committee of Beijing 302 Hospital. Serum samples were stored at −80°C until use. Serum samples at five proper time points were collected from each patient for the quantitation and analysis of HBV markers with other clinical information.
Assays for serological HBV markers and HBV DNA HBsAg, HBeAg, anti-HBe, anti-HBs and anti-HBc levels were determined on a Roche Cobas e601 electrochemistry luminescence immunity analyzer (Roche Diagnostics Ltd, Burgess Hill, UK), and the results were expressed either in IU/l for anti-HBs or according to a cutoff index for the remaining parameters, as described previously [23]. HBsAg was quantified using a Roche Cobas e601 electrochemistry luminescence immunity analyzer and an Elecsys for HBsAg quantitation (Roche Diagnostics) with a lower limit of detection (LOD): 0.05 IU/ml [24]. Plasma or serum HBV DNA was quantified by real-time fluorescent quantitative PCR (LOD was 40 IU/ml). All testing was implemented at the Department of the Central Clinical Laboratory of Beijing 302 Hospital.
Quantitation of serum HBV RNA
Briefly, HBV RNA was isolated with the EasyPure Viral RNA Kit (TransGen Biotech, Beijing, China) and treated with DNase I (Thermo Fisher Scientific, Waltham, MA, USA). For DNase I treatment, every reaction mixture comprised 1.1 ml of 10x DNase I buffer, 0.5 μl of RNase inhibitor (50 U/μl) and 8.4 μl of total nucleic acids. The reaction was carried out at 37°C for 30 min. Next, 1 ml of EDTA and 1 μl of the RT primer were added to each reaction mixture, and each mixture was incubated at 65°C for 10 min to inactivate DNase I. Finally, the microtubes were placed on ice for 2 min incubation. DNase-I-treated HBV RNA was reverse-transcribed by means of the RevertAid First Strand DNA Synthesis Kit (Thermo Fisher Scientific). Serum HBV RNA levels were measured using Applied Biosystems StepOne plus Real-Time PCR Systems (Applied Biosystems, Foster City, CA, USA). The serum HBV RNA level was determined as described previously [9,11]. The LOD of the assay was 300 copies/ml.
The HBcrAg assay
An HBcrAg assay kit was used with a fully automated analyzer system (Lumipulse System, Fujirebio Inc., Tokyo, Japan). This chemiluminescent enzyme immunoassay combines the antigenic reactivity of denatured HBeAg, HBcAg and a core-related protein p22cr, all of which are products of the HBV precore/core gene and share a 149-amino acid sequence [25]. The LOD of the assay was 1,000 U/ml with a linear range of 3–7 log10 U/ml.
Quantitation of anti-HBc
Serum levels of anti-HBc were quantified by a newly developed double-sandwich anti-HBc (total) immunoassay based on the Caris 200 Platform according to instructions from the manufacturer (Wantai, Beijing, China). The detection was calibrated by standards supplied by the World Health Organization. The LOD of the assay was 100 IU/ml with a linear range of 2–5 log10 IU/ml. In this analysis, <100 IU/ml value was still interpreted as 2 log10 IU/ml.
Statistics
All the results are summarized as medians. For univariate analysis, the Mann–Whitney U test was conducted for continuous variables and the c 2 test was carried out for categorical variables. Pearson's correlation analysis was applied to determine a correlation coefficient between two parameters. Data with a P-value or two-tailed P-value <0.05 were considered statistically significant. All statistical analyses were performed in IBM SPSS software, version 21 (IBM, Armonk, NY, USA). Graphical analyses and representation of data were performed in GraphPad Prism version 7.0 (GraphPad, San Diego, CA, USA).
Results
Patients
This study included 41 HBeAg-negative patients and 16 HBeAg-positive patients. Nine of the HBeAg-positive patients achieved HBeAg clearance (that is, loss; four of them achieved anti-HBe seroconversion) and one achieved HBsAg clearance during the observation. At the first time point of observation, 8 (14%) patients had a level of HBV RNA <LOD, 2 (4%) had a level of HBcrAg <LOD and 16 (28%) had a level of anti-HBc <LOD. The number of patients with <LOD levels of individual serum HBV markers gradually increased with time during NA treatment. By the end of the observation, the patient number with <LOD levels of HBV RNA, HBcrAg and anti-HBc increased to 26 (46%), 6 (11%) and 26 (46%), respectively (Additional file 1).
Correlation among serum levels of HBV RNA, HBcrAg, anti-HBc and HBsAg
We studied the correlation among HBV RNA, HBcrAg, anti-HBc and HBsAg. Data on samples below LOD were not included in the analysis. As presented in Figure 1, the coefficient of correlation between HBcrAg and HBV RNA was the highest (r=0.629; P<0.001), followed by the coefficient between HBsAg and HBcrAg (r=0.469; P<0.001). No correlation was detected between HBsAg and HBV RNA (P=0.146). Anti-HBc levels correlated weakly with HBsAg (r=0.323; P<0.001), HBcrAg (r=0.249; P<0.001) and HBV RNA levels (r=0.1945; P=0.041) among the analysed samples.

Analysis of correlation between the serum HBV markers
Serum HBV marker levels according to HBeAg status In general, HBeAg-positive status represents more active HBV replication as compared with HBeAg-negative status. Next, we tested whether serum HBV RNA, HBcrAg, HBsAg and anti-HBc levels were related to HBeAg status during NA treatment. As shown in Figure 2, the levels of HBV RNA, HBcrAg and HBsAg in HBeAg-positive samples were significantly higher than those in HBeAg-negative samples (median 3.92 [25%,75% percentiles 3.41,4.35] versus 2.79 [2.48,3.57] log10 copies/ml for HBV RNA, 5.51 [5.09,5.92] versus 4.13 [3.69,4.70] log10 U/ml for HBcrAg and 3.28 [2.97,3.73] log10 IU/ml versus 2.60 [2.23,3.07] log10 IU/ml for HBsAg; all P<0.05). In contrast, the anti-HBc level in HBeAg-positive samples was significantly lower than that in HBeAg-negative samples (2.00 [2.00,2.37] versus 2.28 [2.00,2.69] log10 IU/ml, respectively; P<0.05).
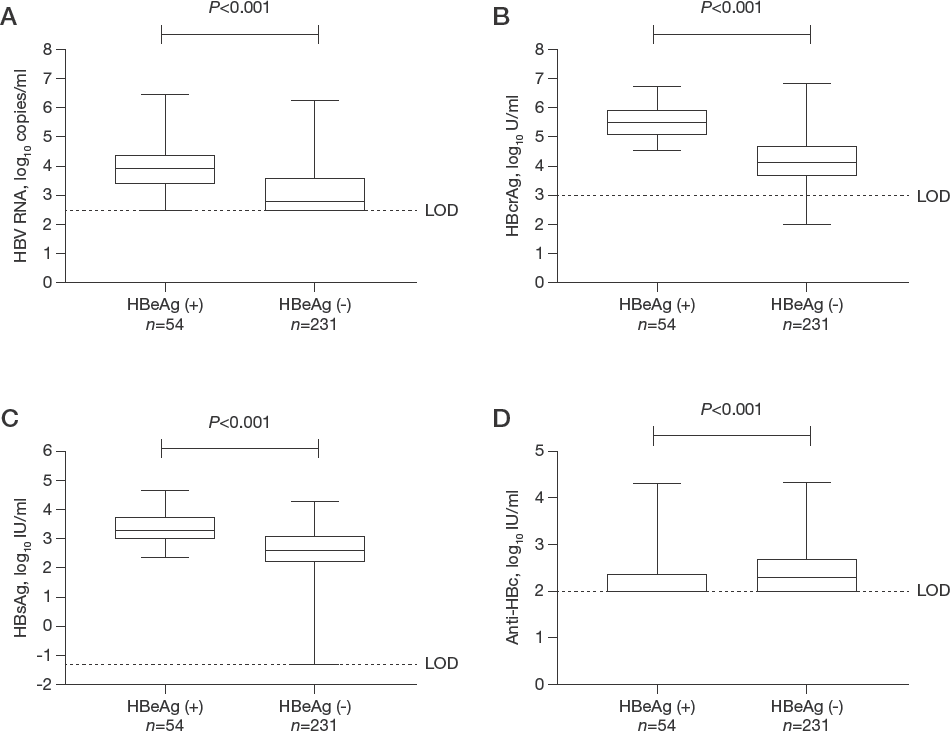
Comparative analysis of the serum HBV marker levels according to HBeAg status
Analysis of factors of HBeAg loss
HBeAg seroconversion marks an important spontaneous change and treatment end point in HBeAg-positive patients and is a prerequisite for HBsAg loss or functional cure. In this retrospective analysis, we identified the relevance of HBeAg loss using serum quantitative levels of HBV RNA, HBcrAg, HBsAg and anti-HBc in HBeAg-positive patients treated with NA. Among the 16 HBeAg-positive patients, 9 and 7 reached and did not reach HBeAg loss, respectively, during the observation of long-term NA treatment. The median age of the patients with HBeAg loss was significantly greater than that of the patients without HBeAg loss (median 49 [25%,75% percentiles 38, 55] years versus 42 [31, 43] years, respectively; P<0.05). The serum HBsAg level of the patients with HBeAg loss was significantly lower than that of the patients without HBeAg loss (2.95 [2.74,3.43] log10 IU/ml versus 3.40 [3.27,4.19] log10 IU/ml, respectively; P<0.05). The degree of decline of HBV RNA and HBcrAg levels in the patients with HBeAg loss was significantly greater than that of the patients without HBeAg loss (1.84 [1.02,2.12] log10 copies/ ml and 1.14 [0.62,2.21] log10 U/ml versus 0.74 [0.10,1.08] log10 copies/ml and 0.41 [0.21,0.69] log10 U/ml, respectively; both P<0.05; Table 1).
Comparison of clinical features between the patients with and without the achievement of HBeAg loss

Statistical description of continuous data, median (25% percentile, 75% percentile), statistical description of frequency data, n. Anti-HBc, hepatitis B core antibody; anti-HBs, hepatitis B surface antibody; COI, cutoff index; HBcrAg, hepatitis B core-related antigen; HBeAg, hepatitis B e antigen; HBsAg, hepatitis B surface antigen; NS, not significant; D, indicates the difference between start and end points of the observation.
Trends of serum HBV RNA, HBcrAg, HBsAg and anti-HBc levels during NA treatment
The trends of the serum HBV markers in CHB patients during long-term NA treatment are not yet fully clear. Our results show that all the serum HBV markers gradually declined during treatment, as illustrated in Figure 3 and Additional file 1. Negative correlations of the four serum HBV markers with the duration of NA treatment were observed either in all patients or in individual HBeAg-positive and HBeAg-negative patients, except anti-HBc in HBeAg-positive patients. Although marker anti-HBc was lower than the LOD of 100 IU/ml during antiviral therapy in 26 (46%) patients, it remained detectable. Simultaneously, HBcrAg was more persistent than HBV RNA in the serum of patients treated with antivirals as seen in Additional file 1. Dynamic changes of HBV RNA, HBcrAg and HBsAg were observed in all the patients (Additional files 2–7). Overall, HBeAg-positive patients had higher levels of serum HBV RNA, HBcrAg and HBsAg than did HBeAg-negative patients, but the decline of the marker levels was small in both groups of patients. In contrast, the patients with HBeAg loss and HBsAg loss manifested a greater decrease in the serum HBV markers (Additional files 6 and 7).
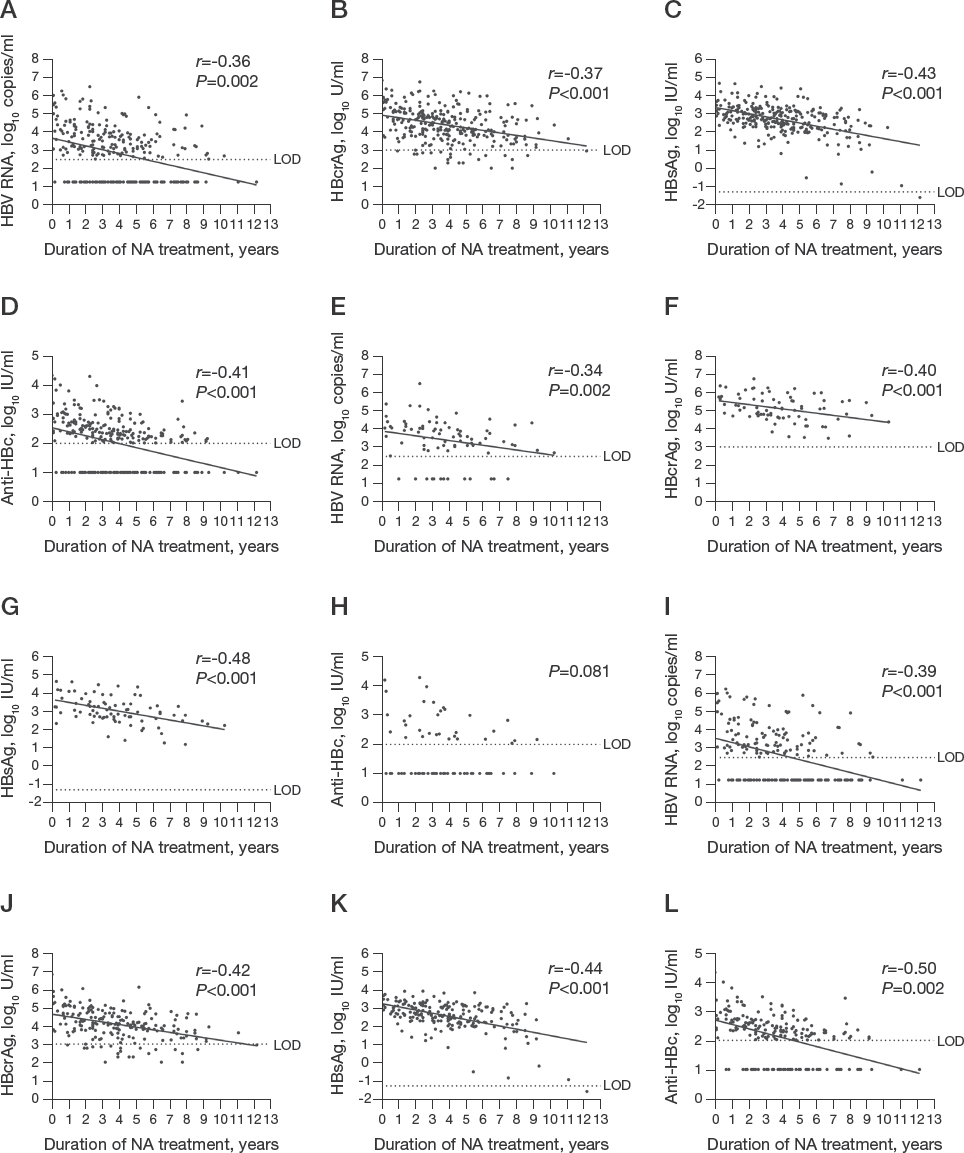
Correlations of serum HBV RNA, HBcrAg, HBsAg and anti-HBc levels with NA treatment duration
Discussion
This study revealed the profile of multiple serum HBV markers during long-term NA treatment in clinical practice. We found that the coefficient of correlation between HBcrAg and HBV RNA was the highest (r=0.629; P<0.001). HBsAg and HBcrAg levels correlated too (r=0.4693; P<0.001). In contrast, no correlation between HBsAg and HBV RNA was observed (P=0.146). Both HBV RNA and HBcrAg are derived from cccDNA, and this situation may explain the relatively strong correlation between serum HBV RNA and HBcrAg levels. For comparison, HBV RNA is enveloped in the nucleocapsid to become a template for the synthesis of the negative stand of HBV DNA. This process could be interrupted by NA treatment, and the amount of pgRNA-containing particles has been shown to increase in vitro and in transgenic mice, whereas no significant changes are detectable in patients [26]. HBcrAg is thought to constitute viral proteins derived solely from the transcriptionally active cccDNA mini-chromosome because HBcAg production and HBeAg production require the presence of the entire HBV genome. By contrast, HBsAg is known to be derived both from cccDNA transcription and from the sub-genomic fragments of integrated HBV DNA [27,28]. Our study indicates that the serum anti-HBc level only weakly correlates with HBV RNA, HBcrAg and HBsAg levels. A possible explanation is that the anti-HBc antibody is elicited by robust antigen HBcAg, leading to its relatively slow decline without a parallel decline of the other viral markers.
Although HBeAg status and HBV RNA, HBcrAg and HBsAg are all serum markers reflecting HBV activity, they may have distinct clinical implications. Our results suggested that serum HBV RNA, HBcrAg and HBsAg levels were generally higher in HBeAg-positive patients than in HBeAg-negative patients, confirming that the three serum HBV markers have a positive relation with HBeAg in patients undergoing long-term NA treatment. In contrast, the serum anti-HBc level was higher in the HBeAg-negative patients than in HBeAg-positive patients, and this phenomenon is consistent with the results of another study [6]. The mechanism may have to do with the finding that in comparison with HBeAg-positive CHB, HBeAg-negative CHB is more likely to undergo disease activity fluctuation, which will restimulate anti-HBc production.
HBsAg and HBcrAg have been suggested to be predictors of HBeAg seroconversion in HBeAg-positive patients treated with NA [29]. Our data corroborated significant differences between the patients who achieved HBeAg loss and those who remained HBeAg-positive. Factors associated with HBeAg loss in the 16 HBeAg-positive patients included age, the HBsAg level at the first time point of observation and the decline size of HBV RNA and HBcrAg levels. Patients at older age, with a lower HBsAg level and with a greater decline of HBV RNA and HBcrAg levels had a significantly greater chance to achieve HBeAg loss. The latter is an important clinical and virological ‘landmark’ during chronic HBV infection, meaning partial immunological control of HBV activity. Thus, our results indicate that HBV RNA and HBcrAg monitoring is useful for reflecting the attainment of a stable virological response in HBeAg-positive patients with undetectable HBV DNA during NA therapy.
So far, a number of studies have investigated clinical implications of HBV RNA or HBcrAg in NA-treated CHB patients [8,9,13–15,18,29–31]. Nonetheless, only a few studies simultaneously analysed the dynamic changes in the serum HBV markers (HBV DNA, HBV RNA, HBcrAg, HBsAg and anti-HBc) among CHB patients undergoing long-term NA treatment. Our results confirmed that although effectively NA-treated patients experienced a rapid decline of serum HBV DNA to an undetectable level, they manifested a relatively slow decline of HBV RNA and HBcrAg levels and a slow decrease of HBsAg levels. In the patients with persistent HBeAg positivity, the levels of the three serum HBV markers declined slowly and stayed at a relatively higher level. For comparison, in HBeAg-negative patients, the levels of the three serum HBV markers also declined slowly but stayed at a relatively lower level. In the patients who achieved HBeAg loss and HBsAg loss, their HBV RNA and HBcrAg levels underwent a relatively fast decline (Additional files 2–7). These findings reinforce the conclusion of other studies that the individual serum HBV RNA or HBcrAg level is useful for monitoring antiviral effects [13,29]. We observed that after HBV DNA became undetectable, HBV RNA usually turned undetectable earlier than HBcrAg did, and HBsAg loss occurred in one patient who successively attained HBV RNA and HBcrAg undetectability. On the basis of our results combined with data from other publications [5,7,32], we propose a schematic diagram for clinical relevance of serum HBV DNA, HBV RNA, HBcrAg and HBsAg (Figure 4). A single strand of pregenomic RNA is transcribed from cccDNA and then packaged into the core during viral nucleocapsid assembly; this RNA subsequently is reverse-transcribed into the first negative strand of HBV DNA. NA therapy effectively suppresses HBV replication by inhibiting HBV polymerase [33]. Thus, HBV DNA is the earliest marker to become undetectable in patients effectively treated with NA.

A schematic diagram for clinical relevance of serum HBV DNA, HBV RNA, HBcrAg and HBsAg during NA treatment
The role of HBeAg is associated with establishment of immune tolerance to HBV infection in infants. There is a report of HBV RNA detection in HBeAg-negative patients not treated with NA [34]. Our study revealed that among the patients effectively treated with NA, HBV RNA remained detectable in 59% (136/231) of HBeAg-negative samples. By contrast, the prevalence of detectability of HBV RNA among HBeAg-positive samples was found to be 93% (50/54), significantly higher than that among HBeAg-negative samples. HBV RNA remained detectable in the majority of HBeAg-negative samples, suggesting that HBV RNA could still serve as a serum marker for monitoring the patients with undetectable HBV DNA and negative HBeAg during NA treatment.
We found that 26 (46%) patients reached an undetectable HBV RNA level and 6 (11%) patients reached undetectable serum HBcrAg levels at the end of the study, indicating that HBV RNA undetectability was usually an earlier event as compared with HBcrAg undetectability. As mentioned in the Introduction, HBcAg is a major component of HBcrAg. HBV RNA is encapsidated to form a core particle, inside which HBV replication takes place, and then complete virions containing HBV DNA (Dane particles) are assembled accompanied by an overwhelming number (up to 100,000-fold greater relative to Dane particles) of subviral particles (10 14 /ml) that contain only the HBV surface protein. As a byproduct, empty (viral-genome-free) virion-like particles (10 11 /ml) that contain the surface proteins and HBcAg are typically found in 100-fold higher numbers in comparison with Dane particles in the blood of infected individuals. Among the three forms of virions or virion-like particles, the number of HBV RNA virion-like particles is usually much lower than that of the other two forms of particles (100–1,000-fold lower than the number of Dane particles, and the difference from empty particles is even greater) [35–37]. During NA treatment, the production of HBV DNA replication-based Dane particles was effectively inhibited. By contrast, the numbers of HBV RNA virion-like particles, empty virion-like particles and subviral particles were less affected. This result may be explained by the finding that among patients treated with an NA antiviral agent(s) in the long run, the decline to undetectability of the four serum HBV markers usually proceeds in the order HBV DNA, HBV RNA, HBcrAg and HBsAg. Accordingly, the achievement of undetectability of each additional serum HBV marker reflects approaching a functional cure.
Our study is based on real-life clinical practice. Unlike in a rigorously designed clinical trial, the patients enrolled in this study were following a variety of NA schedules and treatment duration varied extensively. Given that a lot of the patients that visited Beijing 302 Hospital originate from different regions throughout China and had received different NA therapies before they came to seek medical care here, their samples at baseline and early stage of treatment were unavailable. In addition, it was difficult for us to collect serial samples at fixed time points for many interesting patients. This weakness actually exists in many clinical practice-based retrospective studies. On the other hand, nevertheless, the time points and the duration for the detection of serum HBV RNA, HBcrAg, HBsAg and anti-HBc for each of individual patients were simultaneous and this provided comparability for evaluating which serum marker with closer clinical relevance.
In conclusion, the study showed that monitoring of HBV RNA and HBcrAg levels was useful for NA-treated patients with undetectable HBV DNA and the attainment of HBV RNA undetectability usually occurred prior to HBcrAg undetectability. It deepens our knowledge and understanding of clinical relevance of HBV RNA, HBcrAg, HBsAg and anti-HBc in the patients who maintained undetectable serum HBV DNA levels during long-term NA treatment.
Footnotes
The authors declare that they have no competing interests.
Acknowledgements
We are grateful to Mingjie Yao and Guiwen Guan from Department of Microbiology & Infectious Disease Center, School of Basic Medical Sciences, Peking University Health Science Center, and Le Li, Yayun Lin and Dan Luo from Institute of Infectious Diseases, Beijing 302 Hospital, for their technical assistance for this study. This work was supported by Application Research of Capital Clinical Characteristics and Promotion of Achievements (Z151100004015011) and the National 13th Five-Year Special Grand Project for Infectious Diseases (2017ZX10302201-001). The authors thank all the people who helped the study.
