Abstract
Background
Two-drug regimens are increasingly used in clinical practice as switch strategies. We compared the efficacy and safety of two dolutegravir (DTG)-based dual therapies: DTG plus lamivudine (3TC group) versus DTG plus rilpivirine (RPV group).
Methods
In a multicentre cohort of virologically suppressed (HIV RNA <50 copies/ml) HIV+ patients switching to DTG+3TC or DTG+RPV we analysed the incidence of virological failures (VF) and treatment discontinuations (TD), as well as their predictors.
Results
We analysed 416 patients, 229 in the 3TC group and 187 in the RPV group. The 3TC group, during 344.4 person-years of follow-up (PYFU), had 10 VF without the emergence of resistance mutations, while 30 patients discontinued the regimen. In the RPV group, during 371.0 PYFU, there were 5 VF (1 developed non-nucleoside reverse transcriptase inhibitor mutations Y181C and E138Q) and 13 TD. The estimated probability of remaining free from VF at 48 weeks showed no significant difference between groups (log-rank 0.172). We found a higher risk of VF in patients with peak viral load >500,000 copies/ml in both treatment groups (log-rank P=0.004 in each group). The estimated probability of remaining in the study regimen at week 48 was 89.0% with DTG+3TC and 96.1% with DTG+RPV (log-rank 0.015). After adjusting for potential confounders, treatment group was not associated with TD. A significant decrease in total cholesterol was observed at week 48 in both groups while renal function remained unchanged.
Conclusions
DTG+RPV and DTG+3TC were compared in populations with different characteristics in clinical practice: both regimens showed good effectiveness and improved lipid profile.
Introduction
The association of dolutegravir (DTG) with either rilpivirine (RPV) or lamivudine (3TC), appears promising in maintaining virological suppression while reducing long-term toxicity in treated HIV-positive patients [1–3]. While randomized controlled trials comparing DTG plus RPV with standard triple therapy showed the non-inferiority of the two-drug regimen [4] and ongoing trials are exploring the efficacy of DTG plus 3TC as first-line and maintenance therapy, little is still known about the efficacy and tolerability of DTG-based two-drug regimens in clinical practice, since most studies conducted so far have limited sample size or short follow-up time [5–7]. In our multicentre cohort, we tried to investigate and compare the efficacy and safety of DTG plus 3TC versus DTG plus RPV.
Methods
A cohort of HIV-positive patients with viral suppression (HIV RNA <50 copies/ml) switching to 3TC+DTG (3TC group) or RPV+DTG (RPV group) from seven Italian centres was evaluated. The Kaplan–Meier method was used to estimate the time to virological failure (VF; defined as two consecutive HIV-1 RNA ≥50 copies/ml in a time frame of 3 months, or a single HIV-1 RNA ≥1,000 copies/ml), and time to treatment discontinuation (TD; defined as the interruption of any of the study drugs or the regimen intensification), and Cox regression to evaluate predictors of both VF and TD. Censor was established at last available visit, death or loss to follow-up; when considering the time to VF analysis, censor was also established at the time of TD. Changes in immunological and metabolic parameters at 48 weeks and their predictors were assessed by Student's t-test and linear regression analysis, respectively. The study was approved by each local Ethics Committee (protocol number of the promoter centre: 5284/15) and every patient signed an informed consent before data collection.
Results
A total of 416 patients were enrolled, 229 in the 3TC group and 187 in the RPV group. Patients’ characteristics are summarized in Table 1. Median age was 53.3 years (IQR 45.9–57.1), 298 (71.6%) patients were males, 86 (20.8%) had anti-HCV antibodies, 184 (44.3%) had at least one previous VF with a median time from HIV diagnosis of 16.4 years (IQR 9.2–16-3).
Patients’ characteristics at baseline
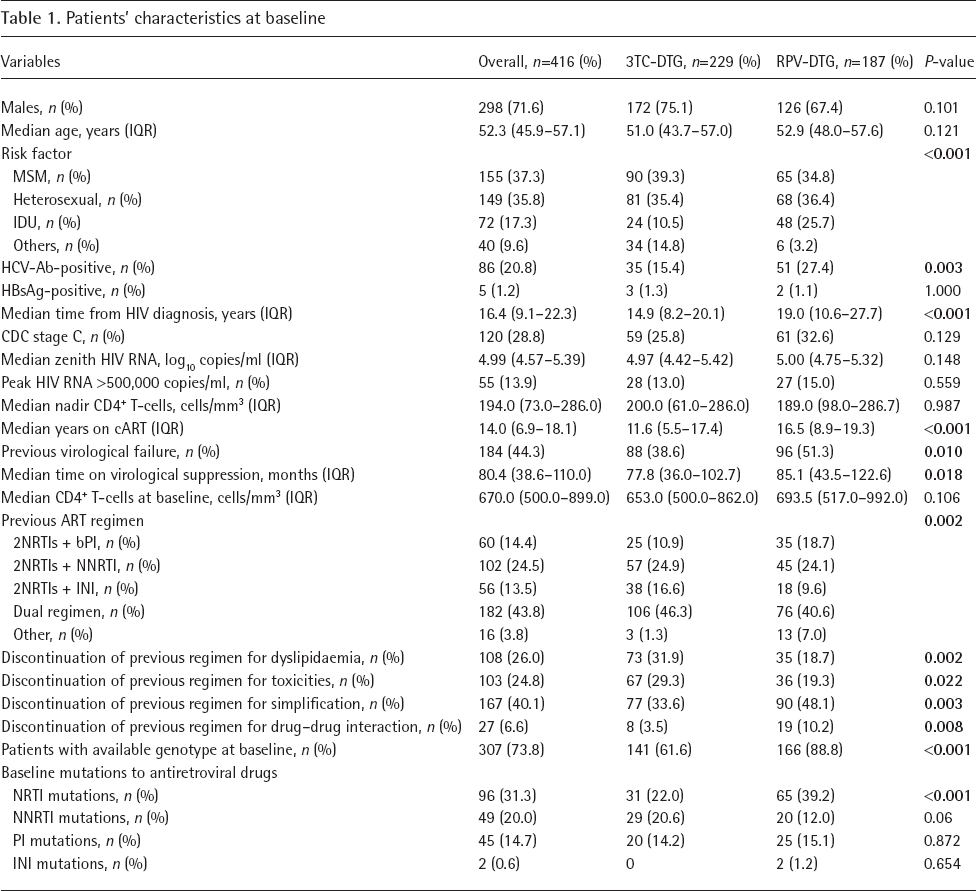
Bold represents significant post hoc comparison between the two groups. Ab, antibody; bPI, boosted protease inhibitor; DTG, dolutegravir; HBsAg, hepatitis B surface antigen; IDU, intravenous drug users; INI, integrase inhibitor; MSM, men who have sex with men; NNRTI, non-nucleoside reverse transcriptase inhibitor; NRTI, nucleoside reverse transcriptase inhibitor; PI, protease inhibitor; RPV, rilpivirine; 3TC, lamivudine.
After a median follow-up time of 22.3 months, 10 VFs were observed in the 3TC group during 344.4 person-years of follow up (PYFU), with an overall incidence of 2.9 VF per 100 PYFU; 3 VFs were due to two consecutive HIV-1 RNA ≥50 copies/ml, while 7 to a single HIV RNA ≥1,000 copies/ml. There was no evidence of new mutations in patients experiencing VF. In the RPV group, during 371.0 PYFU, we observed five VFs, an incidence of 1.3 per 100 PYFU; one VF due to two consecutive HIV-1 RNA ≥50 copies/ml and four to a single HIV RNA ≥1,000 copies/ml. One of those five patients, who self-reported severe lack of adherence to the study regimen, developed Y181C and E138Q resistance mutations after failure. None of the total 15 patients that experienced VF was hepatitis B surface antigen (HBsAg)-positive and all of them achieved virological control subsequently, 6 patients (3 per group) while maintaining the same regimen. Time to VF was not statistically different between groups with an estimated probability of maintaining virological suppression of 97.4% at week 48 and of 95.3% at week 96 in the 3TC group and of 98.2% and 96.9% at weeks 48 and 96, respectively, in the RPV group (log-rank 0.172). Time to VF (Table 2) was only predicted by peak HIV RNA (per 1 log unit higher, adjusted hazard ratio [aHR] 3.10, 95% CI 1.12, 8.55; P=0.029). An increased rate of VF was found in patients with a peak HIV-1 RNA ≥500,000 copies/ml (7.3 VF per 100 PYFU). In the 3TC group, the estimated probabilities of remaining free from VF were 98.1% at week 48 and 96.3% at week 96 in the group with peak HIV-1 RNA ≤500,000 copies/ml versus 95.2% at week 48 and 86.6% at week 96 in the group with higher peak viral load (log-rank P=0.004). Similarly, in the RPV group, the probabilities of remaining free from VF at weeks 48 and 96 were 99.2% and 98.4%, respectively, in the group with a lower peak viral load while in patients with peak HIV RNA above 500,000 copies/ml the probabilities were 91.8% at week 48 and 87.5% at week 96 (log-rank 0.004).
Factors associated with virological failure at univariate and multivariate Cox regression analysis

Bold represents significant predictors at multivariate analysis. Ab, antibody; aHR, adjusted hazard ratio; IDU, intravenous drug users; INI, integrase inhibitor; MSM, men who have sex with men; NNRTI, non-nucleoside reverse transcriptase inhibitor; NRTI, nucleoside reverse transcriptase inhibitor; PI, protease inhibitor.
A total of 30 TD occurred in the 3TC group, during 355.9 PYFU, with an estimated probability of remaining in the study regimen of 89.0% at week 48 and 85.8% at week 96. Reasons for discontinuation were represented by virological failure (6 of 229, 2.6%), hypersensitivity reaction (1 case, 0.4%), gastrointestinal toxicity (5 cases, 2.2%), neuropsychological events (8 cases, 3.5%), further simplification to single-tablet regimen (4 cases, 1.7%), other reasons (6 cases, 2.6%: 1 pregnancy, 1 drug–drug interaction, 4 unspecified reasons). In the RPV group, during 371.9 PYFU, we observed 13 discontinuations, due to VF (2 of 187, 1.1%), neuropsychological events (3 cases, 1.6%), gastrointestinal toxicity (2 cases, 1.1%), renal toxicity (1 case, 0.5%), further simplification (1 case, 0.5%), other causes (4 cases, 2.0%: 1 lipodystrophy, 1 drug– drug interaction, 1 malaise, 1 unspecified reason). In this group, the estimated probabilities of continuing study treatment were 96.1% and 94.2% at weeks 48 and 96, respectively. Survival analysis on time to TD showed a significant difference between the two treatment groups (log-rank P=0.015). In a multivariate analysis, HBsAg-positive serostatus (versus negative, aHR: 20.82, 95% CI 3.83, 113.10; P<0.001) independently predicted TD, after adjusting for significantly different parameters at baseline, treatment group and clinical centre. Of note, of the two HBsAg-positive patients who interrupted study regimen, one discontinued for further simplification and one for his own choice; also, no liver enzyme elevation occurred during the study period. Considering the possibility of a study bias due to the lack of association between hepatitis fares and TD, a second multivariate model for Cox regression was performed excluding HBsAg-positive patients, where no predictors of TD were found.
We observed a significant reduction in total cholesterol (TC) levels in both groups at week 48 (−8.35, 95% CI −13.86, −2.85, P=0.001 in the 3TC group; −15.24, 95% CI −23.04, −7.45, P<0.001 in the RPV group). A greater reduction in TC was independently predicted by higher baseline cholesterol values (per 1 μg/dl more, mean difference in change −0.53 μg/dl, 95% CI −0.67, −0.39; P<0.001) while an increase in TC was evidenced in patients switching from tenofovir disoproxil fumarate (versus other backbones, +53.5 μg/dl, 95% CI 8.14, 98.88; P=0.020) after adjusting for pre-switch regimen and nadir CD4+ T-cell count.
Regarding renal function, neither group showed significant changes in estimated glomerular filtration rate (eGFR) values at week 48 (−4.30 ml/min, 95% CI −17.80, 9.20; P=0.476 in the 3TC group and −3.49, 95% CI −7.53, 0.54; P=0.089 in the RPV group).
Discussion
To the best of our knowledge this is the first real-life study comparing two DTG-based two-drug regimens. In our observational cohort, we found a similar virological efficacy of the two strategies. Among patients experiencing VF, only one patient in the RPV group, with a history of non-adherence to ART, presented non-nucleoside reverse transcriptase inhibitor mutations after failure while we did not observe emergence of any integrase inhibitor resistance mutation. These data are in line with the results of the SWORD-1 and SWORD-2 clinical trials [4]. None of the patients in the 3TC+DTG group developed de novo mutations, confirming previous reports [8,9] about the safety of this strategy. Interestingly, patients with HIV-1 RNA peak ≥500,000 copies/ml showed a trend for an increased risk of V F. Since the decline in HIV DNA load during ART is negatively affected by higher zenith HIV RNA [10], and is predictive of the response to ART, those patients could have a significantly higher HIV DNA load, that could in turn justify a higher risk of viral rebound during treatment with a less-drug regimen. The lack of HIV DNA quantification in our study hampers definite conclusions; however, until more data are available, a prudent attitude when considering treatment switch to DTG-based two-drug regimens in patients with a long history of antiretroviral therapy and with very high zenith viral loads is warranted.
Reported rate of neuropsychological events was below 3.5% in both groups, which is lower than those observed from other studies with DTG [11–13].
Our secondary analysis on metabolic outcome at 48 weeks confirmed the improvement in the blood lipid profile in patients switching to 3TC+DTG [5,6] while also showing a reduction in TC in the RPV group, that was not previously evidenced [4,14].
Our study presents some limitations, such as the retrospective design and the possibility of study bias due to differences in patients’ baseline characteristics between groups. Moreover, data should be interpreted with caution, given that one-third of the patients switched from another two-drug regimen, suggesting that this was a more selected sample of patients.
In conclusion, our study shows that both analysed DTG-based two-drug regimens are effective and safe as maintenance strategies in clinical practice, with a cumulative low incidence of virological failures. Moreover, both study regimens show a favourable effect on fasting lipids profile with no influence on renal function.
Footnotes
GB received a travel grant from Bristol-Myers Squibb. A Capetti has received a personal grant from AB, Gilead and ViiV. S Rusconi has been an advisor for Gilead Sciences, AbbVie, BMS, Janssen-Cilag, ViiV Healthcare and MSD, has received speakers’ honoraria from Gilead Sciences, ViiV Healthcare, BMS, MSD, AbbVie and Janssen-Cilag, has received support for travelling to meetings from Gilead Sciences, BMS, Janssen-Cilag, MSD and ViiV Healthcare, has received research grants from Pfizer, Janssen, Gilead Sciences and ViiV Healthcare. GS received personal fees for lectures from Gilead, Janssen, MSD, ViiV and travel grants from Gilead, ViiV and MSD. Gd'E received consultancy fees from AbbVie, MSD, Janssen, ViiV, Gilead and BMS. MVC received a travel grant from Gilead Sciences. ADL received research grants via his institutions from ViiV Healthcare, Gilead Sciences (through a fellowship programme) and Merck Sharp and Dohme Italy while also receiving consultancy fees from ViiV Healthcare, Gilead Sciences, Merck Sharp and Dohme Italy, Janssen Pharmaceuticals, Bristol-Myers Squibb and AbbVie. SDG was a paid consultant or member of advisory boards for Gilead, ViiV Healthcare, Janssen-Cilag, Merck Sharp & Dohme and Bristol-Myers Squibb. All other authors declare no competing interests.
Acknowledgements
We would like to thank all the study participants, the investigators and their staff for contributing to the realization of this study.
This study was supported by ViiV Healthcare Europe, which provided an unrestricted grant to the Infectious Diseases Unit of the University of Siena. No additional external funding was received for this study. The funders had no role in study design, data collection and analysis, decision to publish or preparation of the manuscript.
