Abstract
Background
Data is limited on the use of 8 weeks of therapy with ledipasvir/sofosbuvir (LDV/SOF) for special populations such as HCV–HIV-coinfected patients. The primary objective of this analysis was to compare sustained virological response at 12 weeks after end of therapy (SVR12) rates among HCV-monoinfected and HCV–HIV-coinfected patients in a real-world clinical setting. Additionally, we compared SVR12 rates among patients receiving 8 versus 12 weeks of therapy.
Methods
This was a single-centre, retrospective study of HCV-infected patients prescribed LDV/SOF at ambulatory clinics associated with the University of Maryland Medical Center (UMMC) from May 2015 to May 2016. Data were obtained from UMMC electronic medical records and outpatient pharmacy claims database. Comparisons between groups were made using χ2 or Fisher's exact test for categorical variables and Student's t-test or Wilcoxon rank-sum for continuous variables. All analyses were per-protocol; patients missing SVR12 data (25.2%) could not be evaluated for our stated objectives.
Results
A total of 274 patients were included. Median age was 58 years; 62.8% were male; 82.5% were Black. SVR12 data was available for 65 HCV–HIV-coinfected patients, of which 62 (95.4%) achieved SVR12. There was no difference in SVR12 rate between HCV–HIV-coinfected patients and HCV-monoinfected patients (86/90; 95.6%; P=0.959). Additionally, there was no difference in SVR12 attainment between HIV–HCV-coinfected patients who received 8 versus 12 weeks of therapy (P=0.101).
Conclusions
8 weeks of LDV/SOF was effective for treatment-naive, non-cirrhotic, HCV genotype-1 patients in this real-world setting, regardless of HIV status. Increased uptake of the 8-week regimen can decrease costs for patients and payers without compromising outcomes.
Introduction
Chronic HCV infection affects approximately 71.1 million people worldwide [1]. If left untreated, infection can lead to detrimental complications including decompensated cirrhosis and hepatocellular carcinoma [2]. Early intervention with appropriate treatment is necessary to decrease morbidity and mortality [3]. Within the past decade, the development of direct-acting antivirals (DAAs) has significantly changed the treatment landscape for HCV-infected patients. These drugs offer several benefits including a considerably improved side effect profile, once-daily oral administration and a dramatic improvement in sustained virological response (SVR) rates [4]. Since late 2014, the commercial availability of the oral fixed-dose combination ledipasvir (90 mg) plus sofosbuvir (400 mg; LDV/SOF) has created yet another niche in therapeutic advantage for HCV genotype-1 patients: the possibility of shortened 8-week treatment duration for select patients and a uniformity of treatment response across a wide range of patients [2].
The pivotal Phase III clinical trial, ION-3, demonstrated non-inferiority between 8-week and 12-week LDV/SOF treatment durations in terms of sustained virological response at 12 weeks after end of therapy (SVR12) [2]. Results of a subsequent post hoc analysis examining relapse rates between 8- and 12-week treatment groups led the Food and Drug Administration (FDA) to recommend use of 8-week courses only for treatment-naive, non-cirrhotic patients, with a pretreatment HCV RNA <6 million IU/ml [5]. The impetus to validate these clinical trial results in real-world settings is guided by several concerns. First, experience with other first-line DAA regimens, such as sofosbuvir plus simeprevir, has shown that clinical trial efficacy outcomes do not always translate equally into effectiveness in clinical practice, owing to the nuances of treating patients in non-controlled study environments [6–8]. Second, the statistical robustness of the aforementioned post hoc analysis have been challenged, suggesting that pretreatment HCV RNA viral load may not be an appropriate threshold measure by which to base the decision to treat for a shorter course [9,10]. Last, certain subgroups such as HCV–HIV-coinfected patients were excluded from ION-3 altogether [2]. While several real-world effectiveness studies have helped shed light on some of these concerns, questions still remain regarding how to best treat HCV–HIV-coinfected patients.
Current estimates suggest that 2.3 million HCV-infected individuals are coinfected with HIV worldwide [11]. Compared to HCV-monoinfected patients, HCV–HIV-coinfected patients experience higher all-cause mortality and higher rates of hepatic decompensation [12,13]. Lower response rates and high incidence of adverse effects associated with interferon-based regimens in coinfected patients led to traditionally decreased uptake of HCV treatment in this population [14,15]. However, the introduction of DAAs has removed these barriers and shortening treatment durations can increase adherence and access to care. Nonetheless, as of 2018, the updated American Association for the Study of Liver Diseases (AASLD) and Infectious Disease Society of America (IDSA) combined guidelines do not recommend an 8-week treatment duration with fixed-dose combination LDV/SOF for HCV–HIV-coinfected or Black patients due to a paucity of data [16]. While current real-world evidence on 8-week treatment outcomes for Black patients is conflicting, data for HIV–HCV-coinfected patients is limited [17–24]. In this retrospective observational cohort, we compared SVR12 rates among HCV-monoinfected and HCV– HIV-coinfected patients in a real-world clinical setting. Our secondary objective was to compare SVR12 rates among patients receiving 8 versus 12 weeks of therapy, both in the overall cohort and among the coinfected subgroup.
Methods
This was a single-centre, retrospective, observational cohort study of HCV-infected patients prescribed LDV/SOF at ambulatory hepatitis C clinics associated with the University of Maryland Medical Center from May 2015 to May 2016. All genotype-1, HCV-infected patients who received a prescription for LDV/SOF during this period were identified using our electronic medical record. Additionally, pharmacy claims data were reviewed for each LDV/SOF prescription dispensed during the study period. This included days’ supply of medication, third party payer and amount billed per prescription. Cost associated with treatment was extrapolated from this data.
Patients were only excluded from our analysis if they were missing the following data: either pretreatment HCV RNA viral load or both cirrhosis diagnosis and baseline fibrosis score. In accordance with the AASLD/ IDSA Hepatitis C Management Guidelines, patients were deemed ineligible for 8 weeks of therapy if they had cirrhosis, a baseline HCV RNA ≥6 million IU/ml, or if they were HCV treatment experienced. Cirrhosis was defined as a FibroSure® fibrosis score of ≥0.74 with an associated F4 Metavir stage, an ultrasound elastography (FibroScan®) result greater than or equal to 12 kPa, or as diagnosed via liver biopsy. End of treatment was defined as the last day covered by consecutive LDV/SOF prescriptions dispensed for each patient. SVR12 was defined as an undetectable serum HCV RNA at least 12 weeks after the end of treatment. Treatment failure was defined as a detectable serum HCV RNA at greater than or equal to 12 weeks after the end of treatment.
Demographic data collected included age, sex, body mass index (BMI) and race/ethnicity. Clinical variables collected included duration of LDV/SOF therapy (weeks), HCV RNA viral load at baseline (value closest to the treatment start date), HCV genotype, prior HCV treatment, fibrosis score, HIV coinfection, HIV RNA viral load at baseline, CD4+ T-cell count at baseline and cirrhosis diagnosis. The percentage of patients in each treatment duration group was calculated from the total days’ supply of all LDV/SOF prescriptions dispensed per patient. Patients considered to have completed 8 weeks of treatment received 56 days’ supply of LDV/SOF. This includes both patients that met guideline criteria for an 8-week duration and received 8 weeks, and those who met criteria for a 12-week duration, however, received only 8 weeks. Patients considered to have completed 12 weeks of treatment received 84 days’ supply of LDV/SOF.
Comparisons between groups were completed using χ 2 or Fisher's exact test for categorical variables and Student's t-test or Wilcoxon rank-sum for continuous variables. P<0.05 was considered statistically significant. SPSS Statistics V. 23 (IBM Corp., Armonk, NY, USA) was used for all statistical analyses. The University of Maryland Baltimore Institutional Review Board approved our research protocol.
Results
A total of 288 HCV genotype-1-infected patients received a prescription for LDV/SOF for either 8 or 12 weeks duration during the study period. Fourteen patients were excluded from the analysis due to non-genotype-1 infection (n=1), concomitant treatment with ribavirin (n=8), treatment for any duration other than 8 or 12 weeks (n=1), missing pretreatment viral load data (n=1), or discontinuation of therapy or loss to follow-up (n=3). One patient discontinued treatment early owing to severe headache as a medication side effect.
A total of 90 patients were included in the 8-week group. Patients were screened for 8-week treatment eligibility per LDV/SOF FDA package labelling criteria (Figure 1). Patients were deemed ineligible for 8 weeks of therapy if they had received prior treatment for HCV (n=34), had a baseline HCV RNA >6 million IU/ml (n=52) or had cirrhosis at baseline (n=111). Consequently, 114 (41.6%) met FDA criteria for shortened treatment duration with LDV/SOF, of whom 42 were women (36.8%), 99 were Black (86.8%) and 37 had HCV–HIV coinfection (32.4%). However, only 52 of these patients were prescribed 8 weeks of therapy. Therefore, the prescribing uptake rate of an 8-week course of LDV/SOF was 45.6% (52/114) during the study period. An additional 38 patients (13.9%) did not fulfil FDA labelling criteria for 8 weeks of therapy but received only two fills of medication (8 weeks). Of these, 9 (23.1%) were treatment experienced, 26 (66.7%) had cirrhosis at baseline and 15 (38.4%) had a pretreatment HCV RNA greater than 6 million IU/ml. Overall, 18 patients with HCV–HIV coinfection received 8 weeks of therapy.

Patients meeting eligibility criteria per LDV/SOF package insert
Entire Cohort
Median age was 58 years, 172 (62.8%) patients were male, 226 (82.5%) were Black and 90 (32.8%) were HCV–HIV-coinfected. Overall, 90 (32.8%) patients received 8 weeks of therapy with LDV/SOF and 184 (67.2%) received 12 weeks of therapy. The baseline characteristics of the entire cohort, stratified by treatment duration, are shown in Table 1. The only significant differences at baseline among the groups included cirrhosis diagnosis, mean fibrosis score and HIV coinfection, all of which were significantly higher in the 12-week treatment duration group.
Baseline characteristics of HCV genotype-1 patients receiving 8 versus 12 weeks LDV/SOF regimens

Continuous variables reported as mean ± standard deviation. Categorical variables reported as n (%).
Race unknown for one patient in each treatment duration group.
No subtype (n=1); mixed subtype 1a/1b (n=2); unknown (n=1). BMI, body mass index; LDV, ledipasvir; SOF, sofosbuvir.
Data on SVR12 attainment was available for 66 (73.3%) patients in the 8-week and 139 (75.5%) patients in the 12-week group, respectively (n=205; 74.8%), as shown in Table 2. There were no differences in baseline characteristics among patients with known and unknown SVR12 (Additional file 1). SVR12 was achieved in 95.6% of patients overall (196/205), 65 (98.4%) patients in the 8-week group versus 131 (94.2%) in the 12-week group (P=0.277). In univariate analyses, baseline characteristics including age, sex, race, HIV coinfection, prior HCV treatment and HCV subtype (genotype 1a versus 1b) did not impact SVR12 (Table 2). The SVR12 rate among Black patients was 95.3% (163/171) compared to 97.0% (32/33) among non-Black patients (P=0.991). Patients were significantly more likely to achieve SVR12 if they had a pretreatment HCV RNA less than 6 million IU/ml and did not have cirrhosis at baseline, regardless of treatment duration. All patients who met criteria for an 8-week LDV/SOF treatment course achieved SVR12. Of the 38 patients who did not meet eligibility criteria for 8 weeks but received 8 weeks, 26 had SVR12 data available; 25/26 (96.2%) achieved SVR12.
SVR12 rates by patient characteristics and treatment duration

Categorical variables reported as n (%).
Race unknown for one patient in the sustained virological response at 12 weeks after end of therapy (SVR12) group.
200 total patients with known HIV status at baseline.
Body mass index (BMI) unknown for three patients in the SVR12 group.
No subtype (n=1); mixed subtype 1a/1b (n=2); unknown (n=1).
HCV–HIV-coinfected (n=90)
The baseline characteristics of HCV–HIV-coinfected patients are shown in Table 3, stratified by treatment duration. The median age of this subgroup was 55 years, 32 (35.5%) were women and 79 (87.7%) were Black. The median pretreatment HCV RNA was 1.7x10 6 IU/ml. Of a total 90 HCV–HIV-coinfected patients included, 18 were treated for 8 weeks and 72 were treated for 12 weeks.
Baseline characteristics and SVR12 of HCV–HIV-coinfected patients receiving 8 versus 12 weeks LDV/SOF regimens
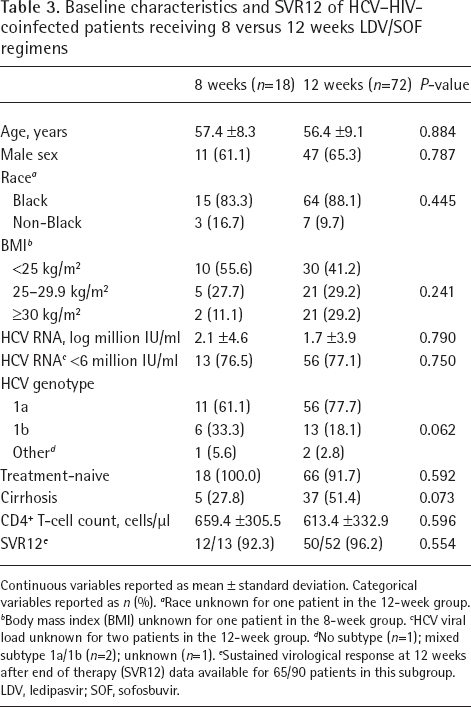
Continuous variables reported as mean ± standard deviation. Categorical variables reported as n (%).
Race unknown for one patient in the 12-week group.
Body mass index (BMI) unknown for one patient in the 8-week group.
HCV viral load unknown for two patients in the 12-week group.
No subtype (n=1); mixed subtype 1a/1b (n=2); unknown (n=1).
Sustained virological response at 12 weeks after end of therapy (SVR12) data available for 65/90 patients in this subgroup. LDV, ledipasvir; SOF, sofosbuvir.
All HIV-infected patients received antiretroviral treatment except for two patients who were long-term non-progressors. The median CD4+ T-cell count was 594 cells/μl (range 86–1,975). There was no difference in mean CD4+ T-cell count between patients receiving 8 and 12 weeks of therapy (Table 3). Eighty-two patients (91.1%) had an undetectable HIV viral load prior to receiving LDV/SOF treatment.
Data on SVR12 rates were available for 65 HCV– HIV-coinfected patients. Of these, 62 (95.4%) achieved SVR12, identical to the overall cohort. There was no difference in SVR12 rate between HCV–HIV-coinfected patients and known HCV-monoinfected patients (86/90; 95.6%; P=0.959). Additionally, there was no difference in SVR12 attainment between HCV–HIV-coinfected patients who received 8 versus 12 weeks of therapy (12/13 versus 53/52; P=0.554).
Discussion
In this real-world cohort of genotype-1 HCV-infected patients treated with LDV/SOF for either 8 or 12 weeks, we demonstrated an overall SVR12 rate of 95.6%, with no significant difference between treatment duration groups, or between HIV–HCV-coinfected and HCV-monoinfected patients. Our results add to the body of evidence demonstrating that clinical cure is equivalent for 8- and 12-week regimens in carefully selected patients, as recommended by FDA labelling criteria. Our results are comparable to those of a recent meta-analysis that included six real-world cohorts (two published studies; four abstracts) in which SVR12 rates were nearly identical for patients receiving 8 weeks and those of the overall cohort, with statistically similar relapse risk [17]. Despite this slowly accumulating data for shortened courses, there remains the question of whether certain subgroups can be appropriately treated with 8 weeks.
Due to limited data, the current AASLD/IDSA combined guidelines do not recommend shortening therapy to less than 12 weeks in HIV–HCV-coinfected or Black patients [16]. However, univariate analyses in our real-world cohort did not reveal these factors as negative predictors of SVR12. A similar cohort by Ingiliz et al. [18] included 35 patients, of whom 25 met eligibility criteria for 8 weeks of therapy. While these authors found similar SVR12 rates between monoinfected (99.4%) and coinfected (96.4%) patients, they were unable to stratify outcomes by treatment duration, as all patients received 8 weeks. The largest cohort to date examining SVR12 rates for coinfected patients receiving 8 versus 12 weeks was recently published by Buggisch et al. [19]. They too found that HIV–HCV coinfection did not influence SVR12 rates, regardless of treatment duration. However, a limitation of their study is that their cohort was nearly 98% Caucasian. Our study adds to the literature by including not only coinfected patients, but also a majority of Black patients. HCV infection in both of these groups has been traditionally considered more challenging to treat, excluding them from the AASLD/IDSA guideline recommendation for 8 weeks.
SVR12 rates in our population were similarly high across all subgroups, including Black race. There is a lack of adequate representation of racial minority groups in the LDV/SOF clinical trials, with Black patients representing only 19% (n=123) of all patients included in the ION-3 trial [2]. Furthermore, there is conflicting evidence on the efficacy of an 8-week LDV/ SOF duration for Black patients. In a large real-world cohort by Su et al. [20], Black race was identified as a predictor of treatment failure with LDV/SOF in patients receiving 8 weeks. On the other hand, a retrospective analysis of data pooled from the ION-1, 2 and 3 trials found no significant difference in SVR12 rates between Black and non-Black patients [21]. In addition, three separate real-world cohorts have reported similar cure rates regardless of race, with analyses limited to treatment-naive patients, without cirrhosis and baseline HCV RNA <6 million IU/ml [22–24]. It should be noted that the study by Su et al. [20] included over 6,000 Black patients, increasing the likelihood of exposing even small differences among subgroups. Furthermore, all racial subgroups achieved high SVR12 rates (>90%) and the difference was numerically small between Black and non-Black patients (93.1% versus 96.4%) [20]. Our study included 171 Black patients, of which 95.3% achieved SVR12. We call for consideration of shortened treatment courses for this subgroup.
The prescribing uptake rate of 8-week courses at our institution was 45.6%, which is comparable to other published real-world effectiveness studies [22,23]. Increasing the number of patients who are effectively treated with shortened courses can reduce costs and potentially allow more patients the opportunity to receive curative therapy. This is especially important in our resource-limited health-care system, in which not all patients have equal access to care. Based on data gathered from pharmacy claims, utilizing average wholesale price (AWP), the mean cost for an 8-week regimen of LDV/SOF in our cohort was $75,600, compared to $113,400 for a 12-week regimen. If all eligible patients had received 8 weeks of therapy, the gross cost-saving on drug acquisition alone would have been upwards of $2 million, over the course of 1 year. Larger real-world cohorts have demonstrated savings proportional to our study. For instance, Backus et al. [22] quoted a potential cost saving of $11 million in their cohort of more than 4,000 VA patients. From a payer standpoint, the overall cost equation includes the cost of drug acquisition and the cost of retreating patients who experience treatment failure or relapse. In our study, only one patient receiving 8 weeks of therapy experienced treatment failure; this patient had cirrhosis at baseline and did not meet criteria for a shortened course of therapy. Notably, 26 other patients in our cohort who had cirrhosis at baseline should have received 12 weeks of therapy but received only 8 weeks. This is attributed to process issues (insurance denials or lapses in prior authorization approvals) and lack of adherence, including no-shows for 4-week labs required by many insurance companies. This is largely representative of real-world practice experiences unseen in clinical trials. While treatment duration was at clinician discretion, all providers in our clinic adhere strictly to national guidelines.
Several new HCV drugs have been introduced in the past couple of years. Most notably, the fixed-dose combination of glecaprevir (300 mg)/pibrentasvir (120 mg; GLE/PIB) is the only drug that has been FDA approved for the treatment of HCV–HIV-coinfected patients for an 8-week duration. The EXPEDITION-2 study evaluated the safety and efficacy of GLE/PIB administered once daily for 8 weeks in 137 non-cirrhotic, HCV–HIV-coinfected adults [25]. The SVR12 rate among coinfected patients without cirrhosis was 100%. Moreover, the AWP of this drug is $15,840.00 per month; greatly reducing the cost burden associated with HCV treatment. However, as the HCV treatment landscape rapidly evolves, LDV/SOF remains one of the DAA agents with the most real-world data to validate its efficacy. Whether the efficacy of GLE/PIB seen in EXPEDITION-2 translates to real-world effectiveness remains unknown. Additionally, GLE/PIB is associated with a higher pill burden (three pills per day) than LDV/SOF, an important fact for the coinfected population that may be exposed to a high pill burden at baseline. Finally, GLE/PIB has drug– drug interactions with several protease inhibitors and non-nucleoside reverse transcriptase inhibitors used for the treatment of HIV, potentially eliminating it as an option for a substantial proportion of coinfected patients [26]. LDV/SOF, on the other hand, can be administered safely with nearly all antiretrovirals [26]. Therefore, an 8-week duration of LDV/SOF is still likely to be a viable treatment option for many patients, especially as market competition drives down the price of the drug.
Our study is not without limitations. Firstly, given that this was a contemporary cohort, we were unable to assess relapse rates in our population. Secondly, SVR12 data was missing for 25.2% of patients (n=69). However, this is unlikely to have affected results as the number of patients missing SVR12 data was nearly equivalent in the 8- and 12-week duration groups (26.7% versus 24.5%, respectively). Thirdly, the retrospective design of this study comes with inherent weaknesses, such as being unable to examine medication adherence or reasons for early discontinuation or failure of therapy. Lastly, due to gathering data from a single centre, our sample size is limited. Based on futility analysis, we would need more than 3,000 patients per group to show a significant difference between those receiving 8 and 12 weeks, based on observed SVR12 difference. This would not be feasible at our institution. Furthermore, this small numerical difference is arguably not clinically significant.
In conclusion, our study demonstrates that an 8-week treatment duration of LDV/SOF was effective for treatment-naive, non-cirrhotic, HCV genotype-1 patients in a real-world institutional cohort. Factors traditionally associated with treatment failure, such as HCV–HIV coinfection and Black race, did not adversely influence SVR12. In accordance with current national guidelines, coinfected patients in our cohort were significantly more likely to be prescribed 12 weeks of LDV/SOF. Our study shows that providers are more inclined to prescribe longer treatment courses for HCV–HIV-coinfected patients while 8-week durations may be effective. Future studies should focus on the cost–benefit ratio of a shortened course of therapy and include a greater percentage of patients from underrepresented subgroups.
Footnotes
The authors declare no competing interests.
Acknowledgements
This research received no funding or grants from any source.
This research was presented in poster format at IDWeek 2017 in San Diego, CA, USA on 5 October 2017.
