Abstract
Background:
The purpose of this study was to develop a new class of influenza A virus haemagglutinin (HA) blockers by tethering thiosialoside molecules to metal nanoparticles and producing glycoclusters that enhance the affinity of HA binding by N-acetylneuraminic acid.
Methods:
Oxygen of the glycoside bond of sialoside was replaced with sulfur to prevent hydrolytic digestion of the N-acetylneuraminic acid residue by viral neuraminidase. Two novel thiosialosides, α-2-S-[p-(N-levulinyl) aminophenyl]-5-N-acetylneuraminic acid (Neu5Ac-S-Lev) and α-2-S-[m-(N-levulinyl)aminobenzyl]-5-N-acetylneuraminic acid (Neu5Ac-S-CH2-Lev), were tethered onto the surface of metal nanoparticles via an aminooxy functionalized thiol linker in a glycoblotting reaction. Gold (Au) and silver (Ag) nanoparticles were coated simultaneously with 11-mercaptoundecyl phosphorylcholine to reduce non-specific adsorption of proteins. Phosphorylcholine self-assembled monolayercoated metals displaying clustered Neu5Ac (Neu5Ac-PCSAM-Au and Neu5Ac-PCSAM-Ag) were subjected to haemagglutination inhibition (HI) assays using the influenza A virus strain A/PR/8/1934 (H1N1).
Results:
Glyconanoparticles with thiosialosides had potent HI activities. In particular, Neu5Ac-PCSAM-Au with a diameter of 20 nm corresponding to 9.8 μM monosaccharide Neu5Ac was the most potent HA inhibitor. The versatility of this strategy was demonstrated by similar submicromolar HI activities of Neu5Ac-PCSAM-Ag with diameters of 50 nm and 150 nm.
Conclusions:
Glycosylated metal nanoparticles were designed and synthesized as potent influenza A virus HA blockers. This study may contribute to the acceleration of the discovery of a new class of nanoparticle anti-influenza drugs.
Introduction
Seasonal outbreaks and pandemics of influenza pose a significant threat to public health worldwide and have hastened the development of new anti-influenza drugs. Influenza viruses express the two enveloped carbohydrate-recognizing glycoproteins haemagglutinin (HA) and neuraminidase (NA). HA recognizes the terminal N-acetylneuraminic acid (Neu5Ac) residue associated with glycoconjugates of the upper respiratory tract and lungs [1,2]. Currently, the major anti-influenza drugs include oseltamivir and zanamivir, which are known NA inhibitors that affect the release of progeny virions from infected cells. However, resistance to oseltamivir [3,4] and zanamivir [5] has emerged, which reduces the efficacy of these influenza treatments.
The initial step in influenza infection involves viral HA-mediated adsorption of virus particles to Neu5Ac residues of glycoconjugates on host cell surfaces [6]. Therefore, HA is a potential target for the development of prophylactic anti-influenza virus drugs that prevent infection at the first stage. Such HA blockers have been investigated in detail [7–11]. As demonstrated for many lectins, the interaction between HA and Neu5Ac-terminated oligosaccharides is commonly weak [12] and improved binding affinity is necessary. Synthetic polymers [13,14], glycopeptides [15], and even simple scaffolds [16] that bear multiple Neu5Ac motifs have shown greatly enhanced binding to HA. However, development into drugs has been hampered by high molecular weights and/or complicated chemical structures and by digestion of natural O-glycoside bonds between Neu5Ac and adjacent galactose residues with viral NAs. In the present study, we increased the density of Neu5Ac residues by tethering them to metal nanoparticle surfaces [17] via artificial thioglycoside bonds. The resulting data facilitates systematic synthesis of a new class of glycoclusters with potent biological activities [18].
Materials and methods
General procedures for the synthesis of thiosialosides Detailed methods for chemical synthesis of the thiosialosides α-2-S-[p-(N-levulinyl)aminophenyl]-5-N-acetylneuraminic acid (Neu5Ac-S-Lev) (
Thiol monolayer assembly of the metal sialylglyconanoparticles Neu5Ac-PCSAM-Au and Neu5Ac-PCSAM-Ag
Gold (Au) nanoparticles with diameters of 5 nm and 20 nm were purchased from Sigma–Aldrich Company (St Louis, MO, USA). Silver (Ag) nanoparticles with diameters of 20 nm, 50 nm and 150 nm were purchased from Shinkou Chemical and Industrial Company (Osaka, Japan). The aminooxy-terminated thiol derivatives 11,11′-dithio bis[undec-11-yl 12-(aminooxyacetyl)amino hexa(ethyleneglycol)] (aoSH) and 11-mercaptoundecyl phosphorylcholine (PCSH) were synthesized according to a previously described method [17,19]. These were used to form mixed monolayers on the surfaces of Au and Ag nanoparticles [17]. Chemoselective oxime bonds between aminooxy functional groups of self-assembled monolayers and thiosialosides were prepared using the glycoblotting method established in our laboratory [19–21].
Haemagglutination inhibition assay
Prior to haemagglutination inhibition (HI) assays, substrates were incubated with 0.5% chicken red blood cells (CRBCs) in phosphate-buffered saline. HI tests were performed according to the microtitre method described by Sever [22]. Each test substrate was prepared as a 5-mM solution in phosphate-buffered saline. Serial twofold dilutions of the inhibitor were mixed with 4 HA units of A/PR/8/1934 (H1N1). CRBC suspensions (50 μl of the 0.5% solutions prepared above) were added after 30 min incubation at room temperature. HI titres were expressed as the reciprocal of the highest serum dilution, which completely inhibited haemagglutination of the virus. Comparisons of influenza A virus inhibitors were made on the basis of the minimum concentration of inhibitor required to inhibit agglutination of CRBC.
Results
Non-specific protein binding and self-aggregation of metal nanoparticles impairs the reliability and reproducibility of haemagglutination assays. To avoid this, PCSH and aoSH were used in combination as coating reagents that resist non-specific adsorption of protein [17,23–25] onto the surface of metal nanoparticles. As expected, attachment of thiosialosides 1 and 2 (Figure 1) to phosphorylcholine self-assembled monolayer (PCSAM)-coated metal nanoparticles proceeded smoothly without self-aggregation, and afforded novel glyconanoparticles, Neu5Ac-PCSAM-Au and Neu5Ac-PCSAM-Ag (Figure 2), of various ratios of PCSH to thiosialoside.
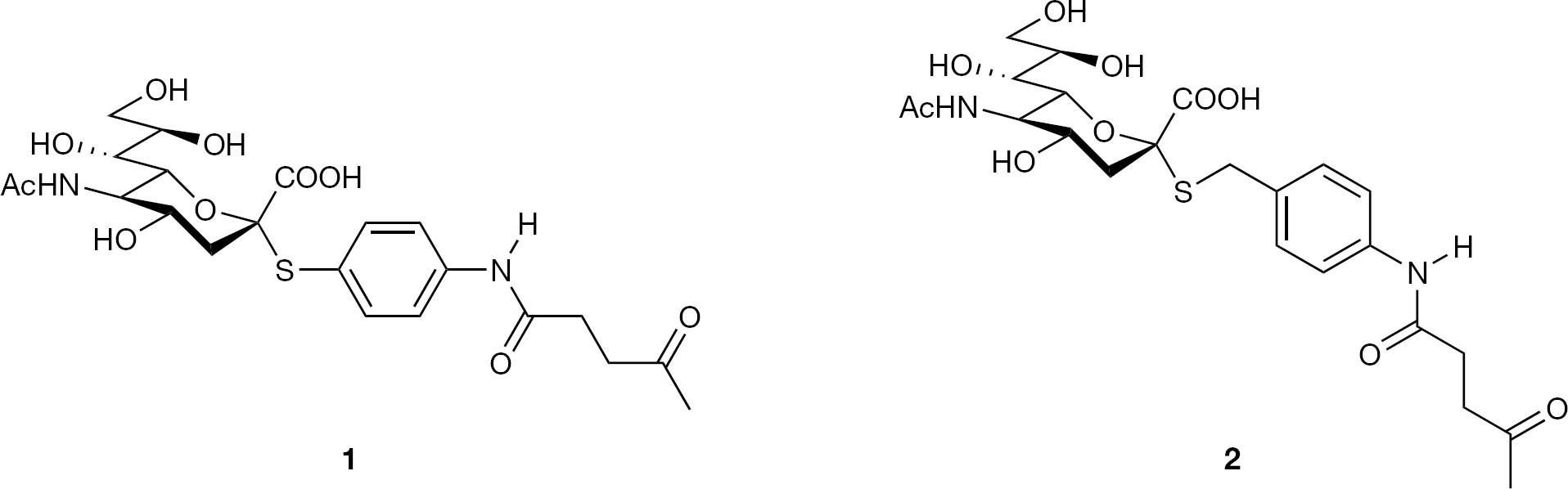
Chemical structures of thiosialosides
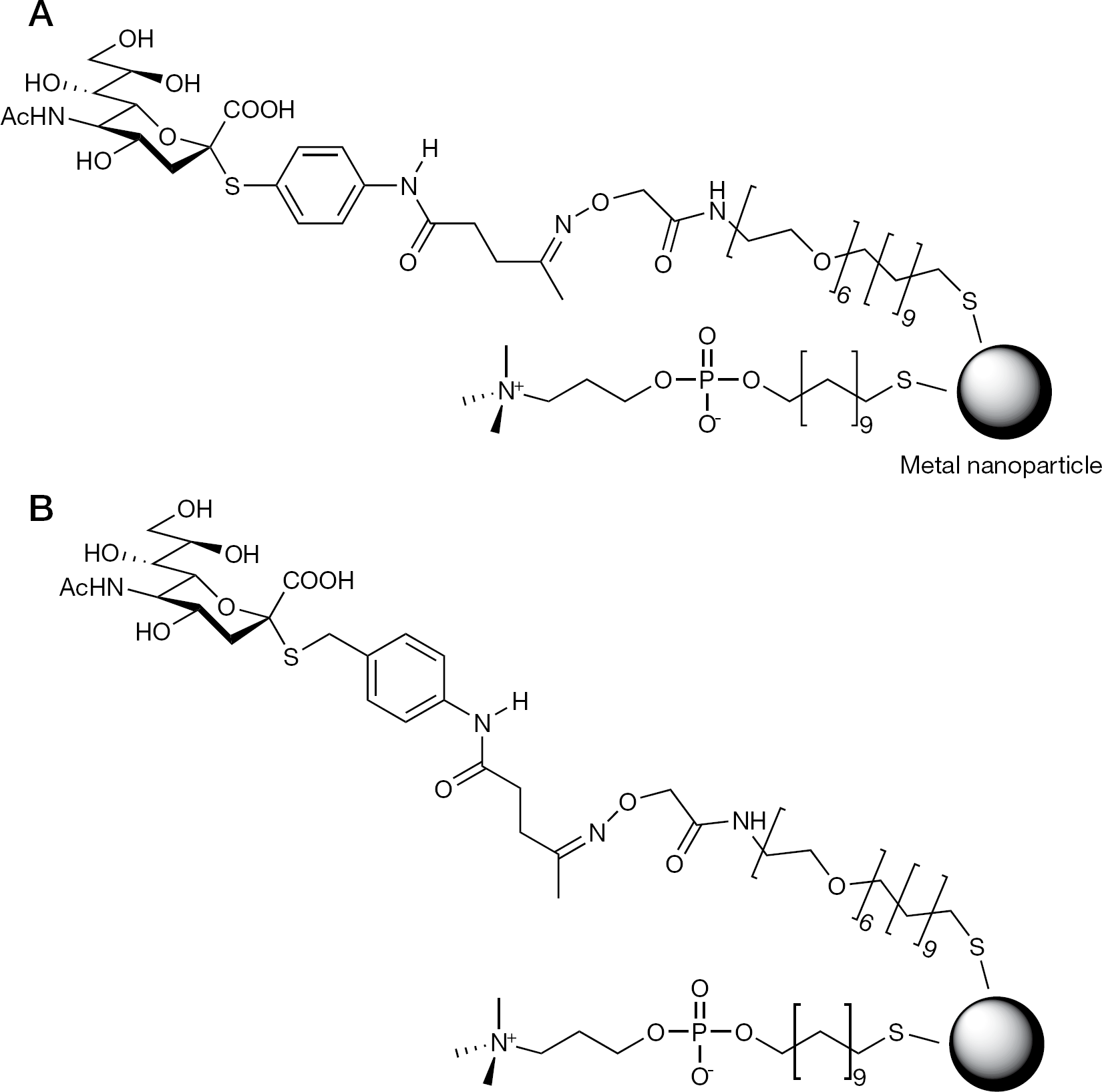
Glycosylated metal nanoparticles bearing phosphorylcholine self-assembled monolayers with thiosialosides
As shown in Table 1, Neu5Ac-PCSAM-Au (PCSH: Neu5Ac-S-Lev [1] =50:50) with a diameter of 20 nm corresponding to 9.8 μM monosaccharide Neu5Ac had the highest inhibitory activity. In addition, Neu5Ac-PCSAM-Au (PCSH: Neu5Ac-S-Lev [1] =30:70 and 20:80) corresponding to 13.7 and 31.2 μM monosaccharide Neu5Ac had similar efficacy, while HI activity was not evaluated with lower ratios of thiosialoside (PCSH: Neu5Ac-S-Lev [1]=90:10 and 80:20). Regardless of the PCSH to Neu5AcAu ratio, Neu5Ac-PCSAM-Au with a diameter of 5 nm did not inhibit HA effectively.
Haemagglutination inhibition activity of gold glyconanoparticles with α-2-S-[p-(N-levulinyl)aminophenyl]-5-N-acetylneuraminic acid (Neu5Ac-S-Lev) (1)
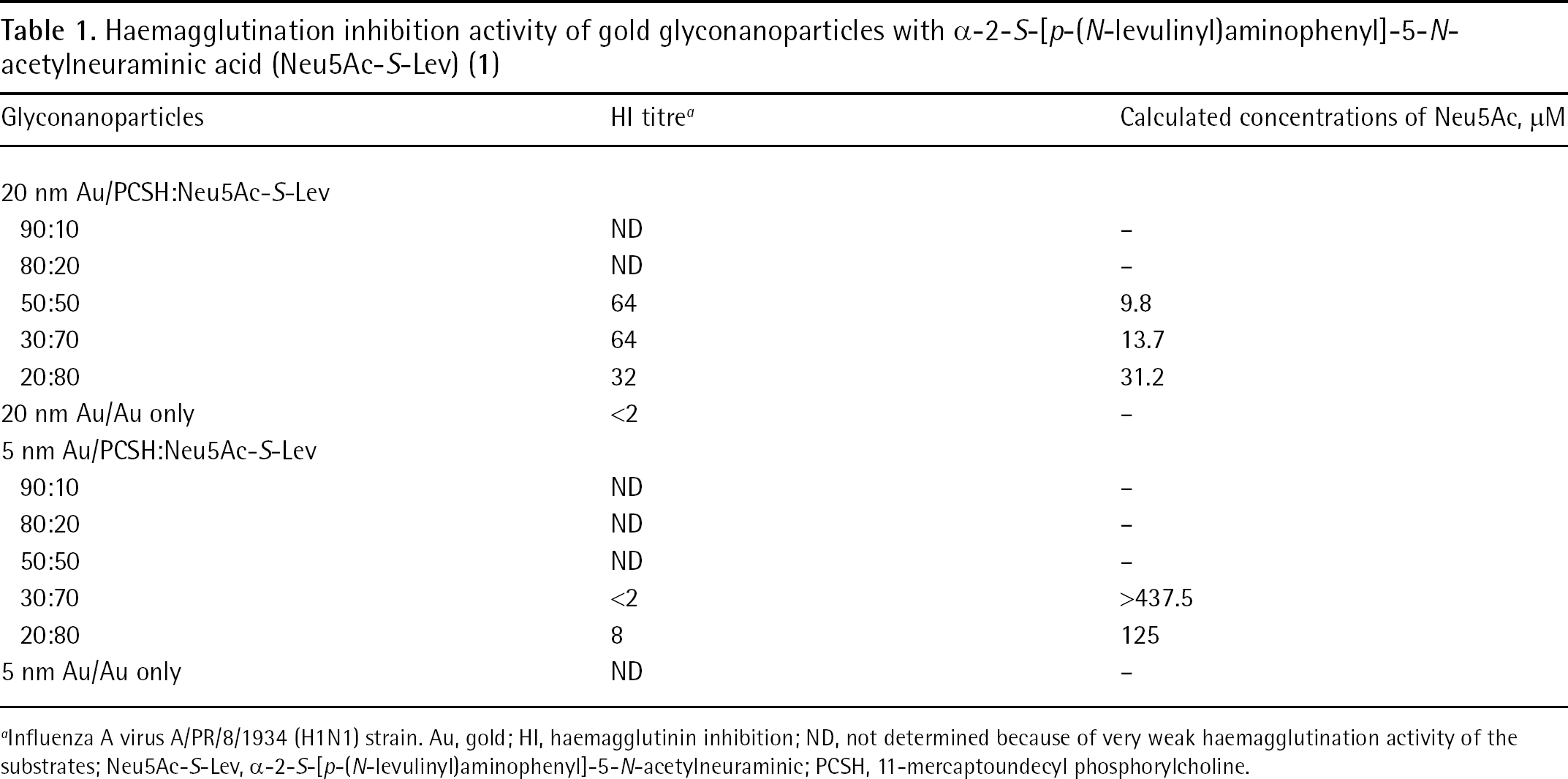
influenza A virus A/PR/8/1934 (H1N1) strain. Au, gold; HI, haemagglutinin inhibition; ND, not determined because of very weak haemagglutination activity of the substrates; Neu5Ac-S-Lev, α-2-S-[p-(N-levulinyl)aminophenyl]-5-N-acetylneuraminic; PCSH, 11-mercaptoundecyl phosphorylcholine.
In subsequent experiments, we investigated the use of commercially available Ag nanoparticles, because Ag is inexpensive and belongs to the same transition metals existing at the 11 group classification in the periodic table, which can be used as a scaffold for the self-assembled monolayer of common thiolates instead of Au. As expected, Ag-based glyconanoparticles (Neu5Ac-PCSAM-Ag) exhibited similar HI activities to that of Neu5Ac-PCSAM-Au (Table 2). Moreover, 50 nm and 150 nm diameter Neu5Ac-PCSAM-Ag particles had micromolar HI activities at higher ratios of Neu5Ac (PCSH: Neu5Ac-S-Lev [1] =50:50, 30:70 and 20:80), indicating that the density of Neu5Ac on the Ag nanoparticles strongly influences HI activity. Although 20 nm Neu5Ac-PCSAM-Au particles inhibited HA, 20 nm Neu5Ac-PCSAM-Ag particles did not. In addition, PCSH self-assembled monolayer coatings significantly enhanced the HI activity of Neu5Ac residues that were co-displayed on the Ag surface.
Haemagglutination inhibition activity of silver glyconanoparticles with α-2-S-[p-(N-levulinyl)aminophenyl]-5-N-acetylneuraminic acid (1)
Influenza A virus A/PR/8/1934 (H1N1) strain. Ag, silver; HI, haemagglutinin inhibition; ND, not determined because of very weak haemagglutination activity of the substrates; Neu5Ac-S-Lev, α-2-S-[p-(N-levulinyl)aminophenyl]-5-N-acetylneuraminic; PC, phosphorylcholine; PCSH, 11-mercaptoundecyl phosphorylcholine.
When the thiosialoside Neu5Ac-S-CH2-Lev (2), which mimics the skeleton of α-2,6 sialoside link-ages, was used instead of Neu5Ac-S-Lev (1) as an α-2,3 sialoside model, 50 nm Neu5Ac-PCSAM-Ag with PCSH: Neu5Ac-S-CH2-Lev (2) ratios of 90:10 and 85:15 corresponding to 15.6 μM and 23.4 μM monosaccharide Neu5Ac (Table 3) had HI activities. By contrast, Neu5Ac-PCSAM-Ag with higher ratios of Neu5Ac (PCSH: Neu5Ac-S-CH2-Lev [2] =50:50, 30:70 and 20:80) had reduced HI activity.
Hemagglutination inhibition activity of silver glyconanoparticles with α-2-S-[m-(N-levulinyl)aminobenzyl]-5-N-acetylneuraminic acid (2)
Influenza A virus A/PR/8/1934 (H1N1) strain. Ag, silver; HI, haemagglutinin inhibition; ND, not determined because of very weak haemagglutination activity of the substrates; Neu5Ac-S-CH2-Lev, α-2-S-[m-(N-levulinyl)aminobenzyl]-5-N-acetylneuraminic; PCSH, 11-mercaptoundecyl phosphorylcholine.
Discussion
Although multivalent and cooperative interactions of influenza virus particles with host cell surface receptors may occur naturally in the infection process, the affinity of Neu5Ac residues for influenza virus HA is low, as indicated by their millimolar dissociation constants [26]. Because low affinity binding of most monovalent sialosides [27] yields inefficient HI activities [28], a variety of compounds with multivalent sialosides were synthesized to enhance binding affinity and interaction with viral HAs [13,14,29]. It was also documented that the anti-influenza drug zanamivir tethered to 32 nm diameter microspheres as a bundled NA-inhibitor had higher affinity for both influenza A and B virus strains [30]. Thus, attachment of multiple Neu5Ac residues is a promising approach to enhancing the therapeutic efficacy of small monosaccharide ligands that target carbohydrate binding proteins. Metal nanoparticles are convenient scaffold materials for various biologically active molecules, with terminal thiol functional groups that can form specific metal-S bonds with Au-S and Ag-S [25,31]. To display NA resistant thiosialoside derivatives 1 or 2 on Au- or Ag-based nanoparticles, we used a glycoblotting method with aoSH according to previous reports [17,19]. Concomitant use of PCSH and aoSH greatly improved both solubility and stability of the present glycosylated metal nanoparticles as indicated by glycosylated quantum dots [17,21]. Thioglycosides are resistant to degradation by viral NAs; therefore, the thiosialosides were designed with a sulfur atom instead of an oxygen in the glycosidic bond [32,33].
The molecular composition and diameter required to achieve optimal HI activity differed between Au- and Ag-based nanoparticles. Neu5Ac-PCSAM-Au with a diameter of 20 nm was a better scaffold than the one with a diameter of 5 nm. The latter showed no HI activity regardless of the ratio of PCSH to Neu5AcAu, indicating that maximum multivalency of Neu5Ac displayed on the surface of nanoparticles may significantly influence HA binding affinity. However, Neu5Ac-PCSAM-Ag particles with a 20 nm diameter were inactive compared to 20 nm Neu5Ac-PCSAM-Au particles. These observations suggest that differences in the stability of S-metal bonds during the sugar attachment reaction under acidic conditions significantly affects the efficacy with which Neu5Ac derivatives are coupled and the density/distribution of Neu5Ac residues on the phosphorylcholine (PC)-coated metal surfaces. Considering that virus particles have a diameter of approximately 150 nm, Ag nanoparticles with a diameter of 50–150 nm may achieve a highly efficient packing structure or lattice with particles of similar curvature. Although these data suggest that Ag nanoparticles are an effective alternative scaffold material to expensive Au nanoparticles, further optimization is required for therapeutic application. Neu5Ac-PCSAM-Ag, which mimics the Neu5Ac-α2,6Gal linkage showed unexpected high HI activity with lower Neu5Ac-S-CH2-Lev (2) content. By contrast, high HI activity was maintained by nanoparticles coated with Neu5Ac-S-Lev (1) at higher ratios of Neu5Ac to PCSH. These data suggest that the methylene group between sulfur atoms and phenyl groups might provide the Neu5Ac residue with flexibility that improves contact with viral HAs. Because we reported previously that some synthetic glycopolymers with multiple Neu5Ac-α2,6Gal structures are recognized strongly by A/PR/8/1934 (H1N1) [34], the present compound (2), which mimics the Neu5Ac-α2,6Gal linkage, may be an alternative NA-resistant ligand of H1 HA.
The merits of glycosylated metal nanoparticles are evident because of the ease of synthesis, feasibility of molecular design, and the excellent solution properties of phosphorylcholine self-assembled monolayer coatings. We conclude that Neu5Ac-PCSAM-Ag bearing Neu5Ac-S-Lev (1) and Neu5Ac-S-CH2-Lev (2) are novel lead compounds for the development of anti-influenza A drugs that inhibit initial interactions between viral HA and glycoconjugates with Neu5Ac residues. Further in vitro and in vivo studies to evaluate inhibition of influenza A virus replication by the synthesized compounds and biochemical studies to elucidate improved binding constants are necessary in our future work.
Footnotes
Acknowledgements
The authors wish to thank S Oka at the Center for Instrumental Analysis of Hokkaido University for mass measurement. This work was partly funded by a Program of Founding Research Centers for Emerging and Reemerging Infectious Diseases grant and by grants for ‘Development of Systems and Technology for Advanced Measurement and Analysis (SENTAN)’ from the Japan Science and Technology Agency (JST).
The authors declare no competing interests.
