Abstract
Herpes simplex virus-1 (HSV-1) is the most common cause of lethal sporadic encephalitis. Despite improved therapy with intraveneous acyclovir, HSV-1 encephalitis is associated with persistent severe neurological deficits. We report three cases of adult patients with HSV-1 encephalitis (HSE), discuss the current accepted guidelines for treatment as published by the Infectious Disease Society of America (IDSA) and review the literature pertaining to HSE. Our case presentations are consistent with the literature review noting a broad spectrum of clinical outcomes with HSE. We include the first published case of successful early transition to oral antiviral therapy. In the other two cases, repeat cerebrospinal fluid (CSF) analysis showed persistent lymphocytic pleocytosis necessitating prolonged viral suppression. Long-term neurological sequelae were noted in these two patients. The IDSA recommendation of 2–3 weeks of intraveneous acyclovir at 10 mg/kg every 8 h, depending on the clinical course, is sufficient for most cases of HSE. We recommend individualization of duration of treatment based on follow-up CSF analysis with quantification of HSV-1.
Introduction
Herpes simplex virus-1 (HSV-1) encephalitis (HSE) in adults is associated with severe morbidity despite improvements in mortality related to treatment with acyclovir. Overall mortality has decreased from 70% prior to the availability of effective antiviral therapy [1] to <20% currently [2]. Of survivors, >60% will have moderate-to-severe neurological deficits. Only in 2–3% of cases will a patient survive with fully normal neurological function [3]. Anterograde memory is most commonly impaired, but retrograde memory, executive function and language ability are also compromised. All of these outcomes are worsened when treatment is delayed [4].
Recommendations for therapy were published by the Infectious Diseases Society of America (IDSA) in 2008. The primary areas of concern in the guidelines relate to appropriate dosing and adequate duration of therapy. This is based on the review of multiple retrospective studies showing that inadequate dosing and duration of therapy are associated with worse outcomes in both the adult and paediatric populations. Another area of concern is with the management of patients who do not improve. Monitoring therapy by repeat cerebrospinal fluid (CSF) analysis with PCR for viral load is suggested for those patients who have not had an appropriate clinical response [5].
This article reviews the literature regarding current disease management. The potential for persistent infection and need for prolonged therapy is specifically addressed. We include three cases of HSE seen at Lincoln Hospital (Bronx, NY, USA) in a 15-month time period that illustrate the problems with management guidelines and suggest the benefit of repeat CSF analysis with PCR quantification.
Case 1
A 64-year-old woman with diabetes mellitus (A1c 16%) presented with generalized weakness, decreased appetite for 1 week and fever for 2 days. Physical examination on admission reported fever (102.0 F), normal speech, gait and motor responses. Computed tomography (CT) of the brain showed R posterior temporal hypodensity. On day 3 the patient developed left hemiparesis. Lumbar puncture showed lymphocytic pleocytosis with 213 cells (Table 1). Intravenous acyclovir at 500 mg every 8 h was started (patient weight 52 kg). CSF PCR confirmed the diagnosis of HSV-1 disease. Magnetic resonance imaging (MRI) of the brain showed R temporal lobe encephalitis (Figure 1). The patient significantly improved within 14 days and treatment was stopped. Over the next 10 days the L hemiparesis returned and a repeat MRI showed progressive change. Repeat CSF analysis showed persistent lymphocytic pleocytosis and increased red blood cells (Table 1). She was restarted on intravenous acyclovir and discharged with persistent L hemiparesis.
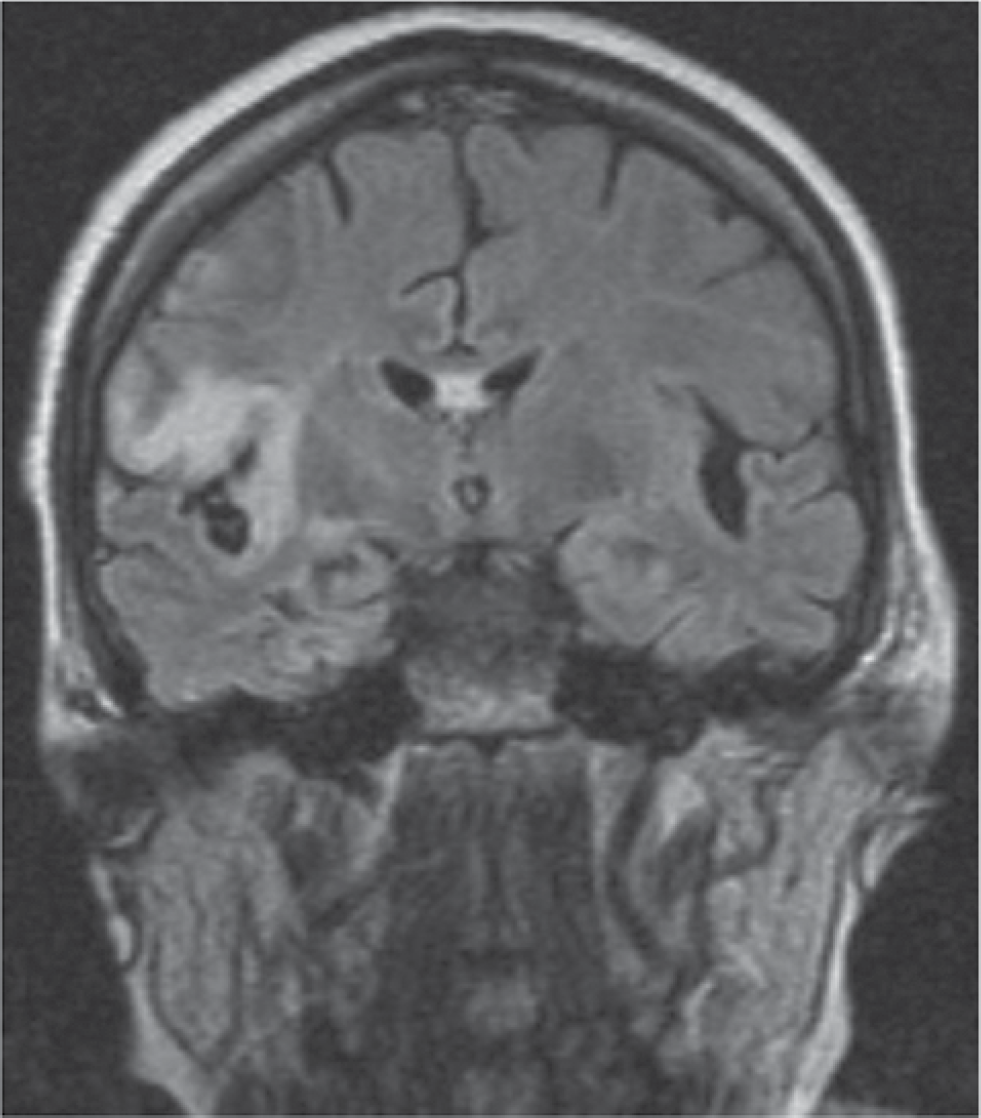
Case 1 on hospital day 4
Case 2
Case 2 was a 60-year-old man with AIDS (CD4+ T-cell count =116 cells/mm3) receiving HAART with undetectable HIV PCR. He presented with malaise, headache and fever for 3 days with altered mental status. On admission, the patient was found to have aphasia without focal weakness. CT scan of the patient's brain showed hypoattenuation in the left subinsular cortex. CSF analysis showed lymphocytic pleocytosis (Table 1). The patient was started on intravenous acyclovir, 500 mg every 8 h (admission weight 63 kg). PCR was positive for HSV-1. Brain MRI showed L temporal lobe encephalitis (Figure 2). Hospital course included persistent fevers and altered mental status. On day 12, repeat lumbar puncture showed persistent pleocytosis and PCR was positive for HSV-1 (Table 1). Intravenous acyclovir was increased to 600 mg every 8 h to complete 21 days. The patient improved clinically and was treated with valacyclovir orally (1 g three times daily) for 3 months as an out-patient. At 3 months the patient had significant short-term memory deficits without aphasia or motor impairment. The patient developed epilepsy 8 months after discharge. CSF PCR was negative for HSV-1 at 6 months after discharge.
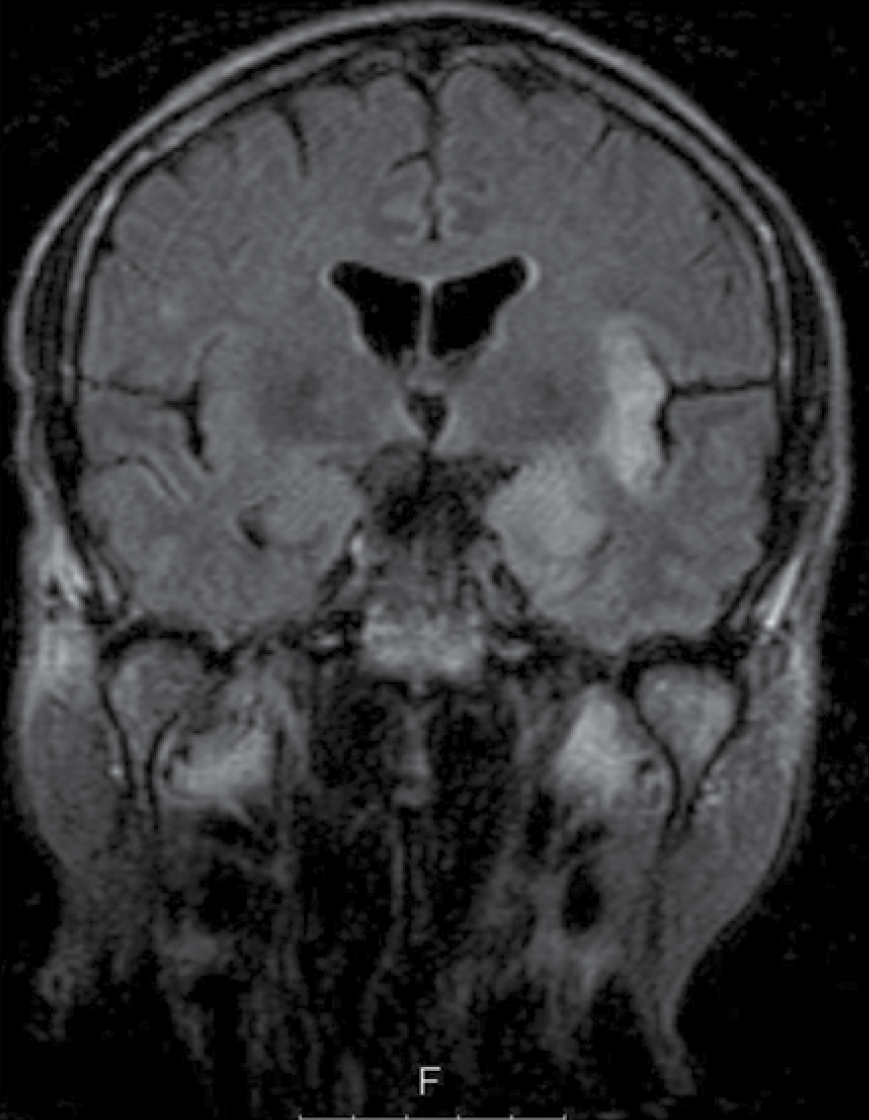
Case 2 on hospital day 3
CSF values for case patients
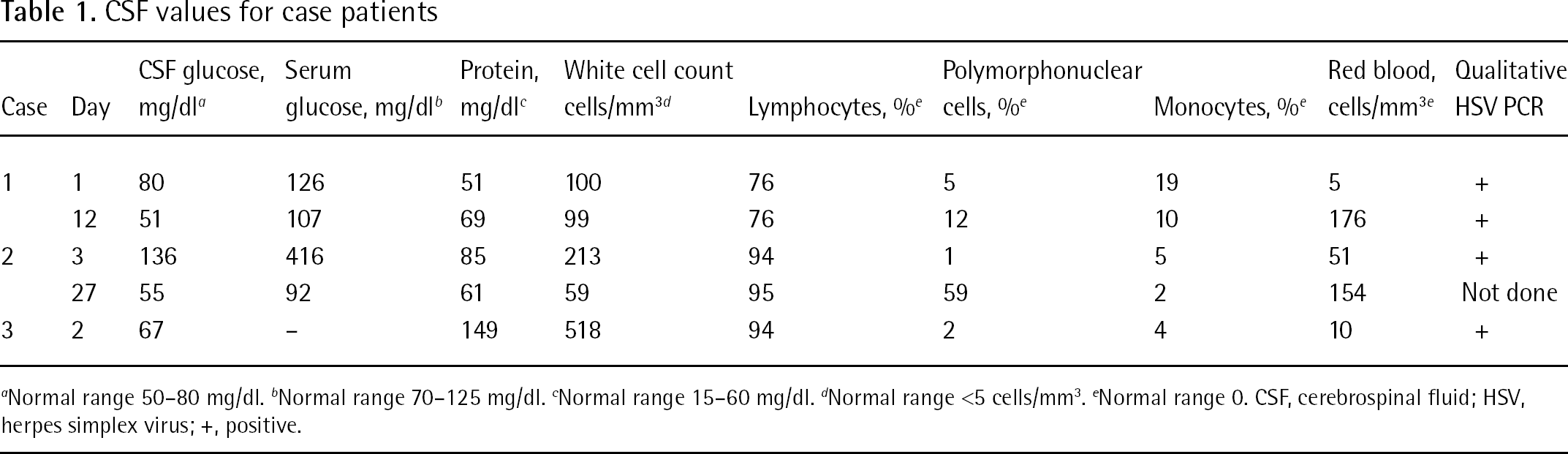
Normal range 50–80 mg/dl. b Normal range 70–125 mg/dl. c Normal range 15–60 mg/dl. d Normal range <5 cells/mm3. e Normal range 0. CSF, cerebrospinal fluid; HSV, herpes simplex virus; +, positive.
Case 3
Case 3 was a 46-year-old man with no significant past medical history who presented with neck pain, headache, vomiting and fever for 6 days. CT scan of the patient's brain showed temporal lobe oedema. CSF analysis revealed lymphocytic pleocytosis (Table 1). MRI was consistent with temporal lobe encephalitis (Figure 3). Intravenous acyclovir 800 mg every 8 h was started (patient weight 79 kg). CSF PCR was positive for HSV-1. The patient improved within 48 h and signed out of the hospital against medical advice on day 4. He was given valacyclovir 1,000 mg twice per day for an additional 2 weeks. He complained of mild headaches on day 15. On the next clinic visit 1 month after discharge, the patient was asymptomatic.
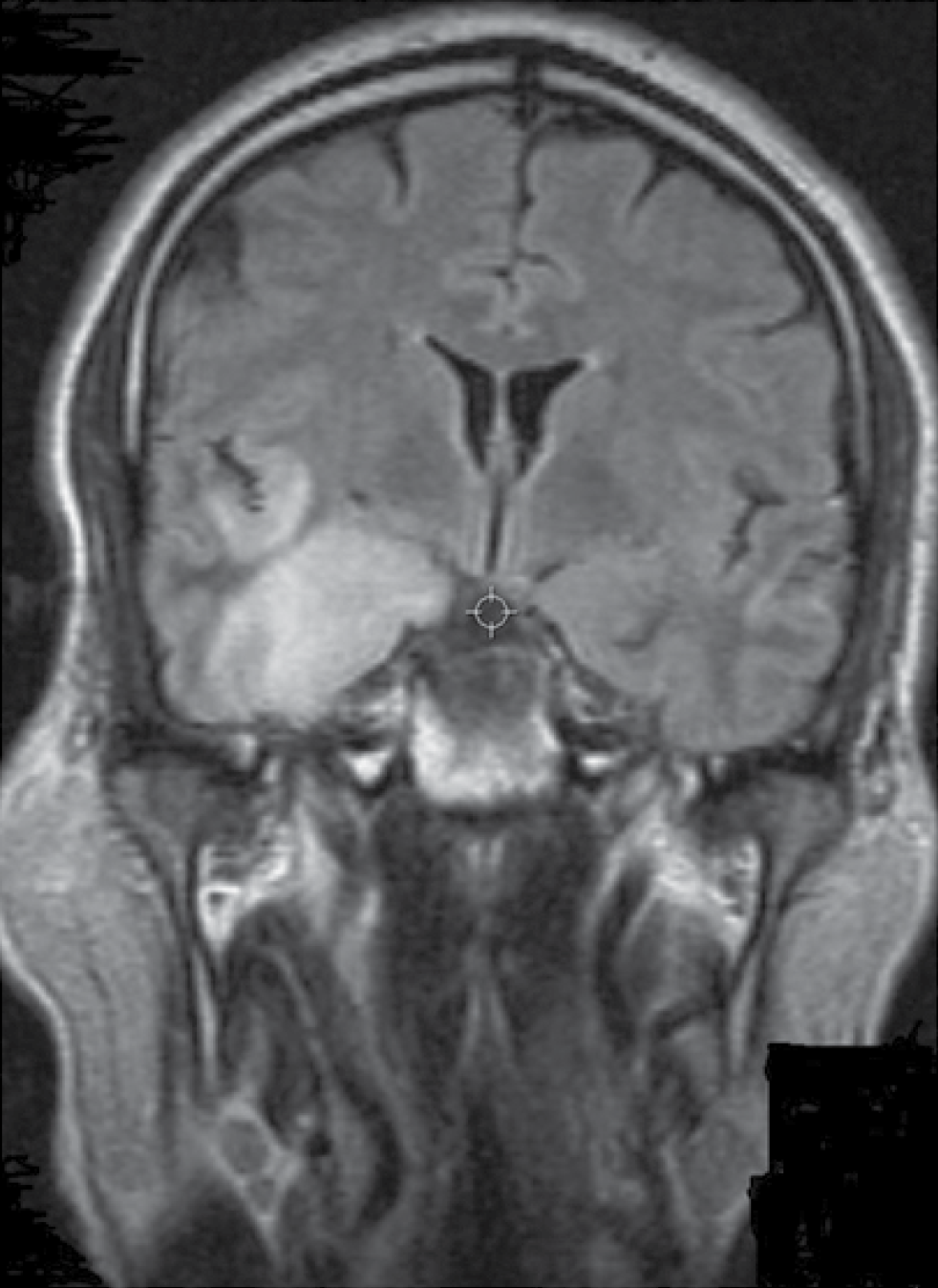
Case 3 on hospital day 3
Literature review
Clinical presentation
Fever and abnormal mental status are the primary signs and symptoms of HEV, occurring in >90% of patients [6–9]. Approximately 50–60% of patients will present with nausea, vomiting and meningismus. Seizures occurred in about 50% of patients and focal neurological deficits appeared in 30–50%. The rates of these findings are similar in other forms of viral encephalitis [9]. Clinically, the presenting symptoms do not allow suspected meningitis or encephalitis to be distinguished. CSF analysis is essential.
CSF examination will show a pleocytosis in 90–100% of cases with lymphocytic predominance. Protein elevation is found in >75% of patients. Glucose levels tend to be variable and less helpful for diagnostic purposes.
Temporal lobe abnormalities on brain MRI are found in >90% of cases. Non-contrast CT of the brain is positive in 46–66% of patients. The earliest abnormality is brain oedema, which may evolve to include haemorrhage and/or mid-line shift [3]. HSE has been reported with involvement of occipital and parietal lobes [10]. In cases 1 and 2, repeat CSF analysis showed >100 red blood cells consistent with micro-haemorrhage in the area of encephalitis (Table 1).
Treatment
Intravenous acyclovir 10 mg/kg every 8 h is the standard treatment for HSE [5]. The best outcomes occur when treatment is initiated before a prolonged duration of symptoms [6,11,12]. Intravenous acyclovir replaced vidarabine by decreasing mortality in HSE from approximately 50% to <20% [3]. It is a guanosine analogue that becomes phosphorylated first by viral thymidine kinase and then by cellular enzymes. The triphosphorylated product is a competitive substrate for viral DNA polymerase and after incorporation results in termination of viral replication. Herpes viruses have different susceptibilities to acyclovir with HSV-1 being the most susceptible virus [13].
In order to effectively inhibit viral replication, acyclovir must achieve an adequate concentration in the CSF. One study examining 77 clinical isolates of HSV-1 found that the mean 50% inhibitory concentration (IC50) to acyclovir was 0.38 μg/ml (sd 0.23, range 0.07–0.97) [14]. Intravenous acyclovir at 10 mg/kg every 8 h achieves a steady-state mean plasma concentration of 23 μg/ml (range 14–44). Concentrations achieved in the CSF are approximately 50% of plasma values, showing that the IC50 is far exceeded [15].
Oral acyclovir has poor bioavailability, achieving plasma concentrations that are only 15–30% of intravenous levels. The steady-state concentration in the CSF after oral acyclovir has been shown to be approximately 0.2 μg/ml [16]. This would not be adequate to treat many strains of HSV-1.
An alternative oral regimen may be valacyclovir. This is the valyl ester formulation of acyclovir and provides increased bioavailability (55%). It is hydrolysed to acyclovir by first-pass intestinal and hepatic metabolism [13]. Acyclovir concentrations in the plasma and CSF have been measured in studies using doses of 1,000 mg three times daily [17,18]. Plasma levels were measured as 5–6.5 μg/ml and the CSF levels at steady-state ranged from 0.5 to 1.5 μg/ml. These CSF levels would be above the IC50 for most but not all strains of HSV-1.
In neonates the dosage recommendations are to treat with 20 mg/kg dosage every 8 h. With the use of higher dosing, neonatal mortality and neurological outcomes have been significantly improved. Survival rates for neonates with disseminated disease were significantly better for the high-dose regimen than with standard dosing (odds ratio 3.3 with 95% CI 1.4–7.9). Patients treated with high-dose acyclovir were 6.6x as likely to be developmentally normal at 12 months as those treated with standard dosing [19]. In adults, the benefit of intravenous acyclovir doses above 10 mg/kg every 8 h has not been shown. A case series from France (Table 2) compared acyclovir dosing of 15 mg/kg every 8 h in 22 patients versus 10 mg/kg in 25 patients [20]. The higher dose was not associated with improved outcomes.
Adult case series of HSV-1 encephalitis
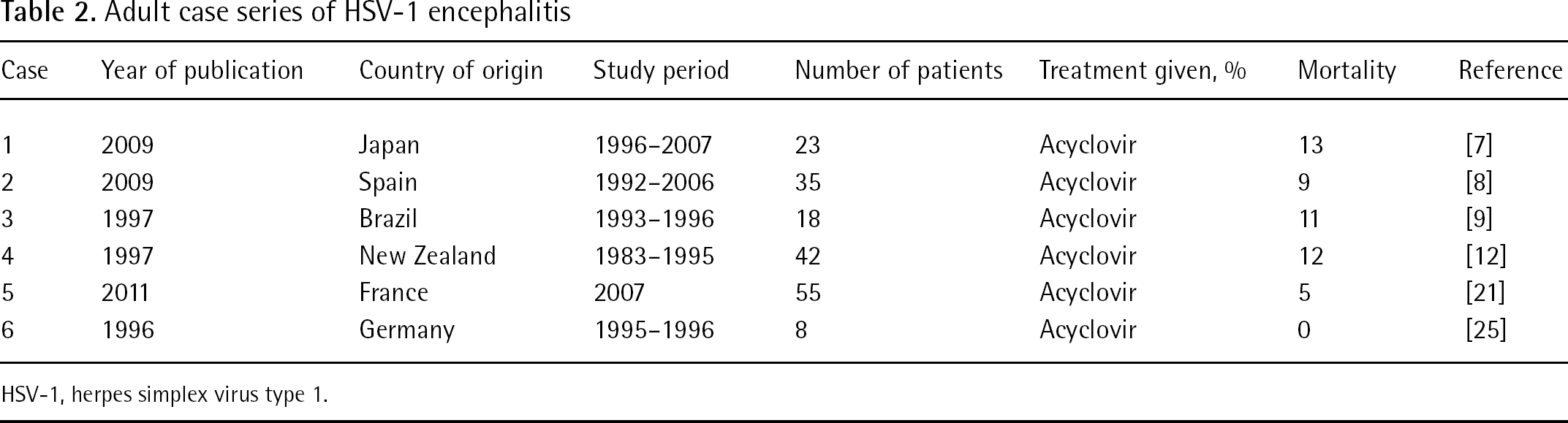
HSV-1, herpes simplex virus type 1.
The standard duration of therapy in adults as per IDSA is 2–3 weeks of intravenous acyclovir depending on the clinical response. In neonates, new recommendations based on a 10-year study published in the New England Journal of Medicine suggest 6 months of suppressive therapy with oral acyclovir after initial treatment of 21 days [21].
Duration of treatment in the adult population is not clearly defined in the literature. There are too few case reports of relapses in the adult population to support either increasing duration for acute treatment or prolonged suppressive therapy. The report of a fatal relapse in an adult with confirmed HSE published in the Journal of the American Medical Association in 1988 resulted in the change of initial treatment from 10 days of intravenous acyclovir (standard at that time) to the current 14–21 day recommendation [22]. Since 2003, there have been at least four case reports of adults with relapses of HSE after treatment durations between 14 and 21 days [23,24].
A 15-year review from Spain (Table 2) examined 35 patients with HSE who received a median duration of acyclovir of 14 days (IQR 11.75–18) [8]. Of the 35 patients, 32 had neurological sequelae at 6 months follow-up. A study of 42 adult and paediatric patients with HSE from New Zealand (Table 2) had initial treatment durations ranging from 7 to 28 days (median 14) [12]. Five patients died in the first month and 20 survivors (of the 29 evaluated) were left with neurological sequelae at 1 year. There were four relapses noted in this group ranging from 1 week to 4 months after completion of the initial treatment. In one case, after 26 days of treatment with acyclovir the CSF PCR was reported as still positive. The French study noted above with 42 patients treated for either 2 or 3 weeks, had 1 patient with positive PCR after 3 weeks of treatment. In a study of 23 patients with HSE from Japan (Table 2) examining risk factors requiring prolonged treatment, the criteria was ‘lack of improvement of neurological symptoms’ without CSF analysis [7]. Prolonged therapy did not improve outcomes.
A 2006 study from Sweden compared the CSF of four adult patients with clinical deterioration after initial therapy of HSE with four adult HSE survivors who had no clinical relapse [23]. The initial HSV viral loads were similar between groups. No HSV DNA was found on follow-up in either group. The relapsing group had significantly increased soluble CD8 suggesting a proinflammatory immunological response without evidence of cell destruction that would have indicated direct viral cytotoxicity. The relapsers were successfully treated with both corticosteroids and acyclovir. A similar phenomenon has also been reported in the paediatric population [3].
More information about the problem of persistent virus may be obtained by reviewing studies that use quantitative PCR. A 1996 study from Germany (Table 2) evaluated eight patients with HSE with repeat CSF analysis during and after treatment with intravenous acyclovir [25]. In four patients, the HSV-1 DNA disappeared from CSF before completion of therapy. Of the four patients who did not clear the virus during treatment, one received 14 days of acyclovir. In this patient, the viral load went from 522 copies/ml to 6 copies/ml on treatment day 11 and eventually to 0 copies/ml 4 days post-treatment. A second patient who was treated with acyclovir for 16 days went from 222 copies/ml to 5 copies/ml 5 days after treatment was stopped. No further therapy was given and there was no clinical relapse. A third patient who was treated for 21 days went from 264 copies/ml to 1 copy/ml on day 14 of treatment. The viral load for the fourth patient was initially 40 copies/ml and after treatment day 11 increased to 60 copies/ml. The patient completed 3 weeks of intravenous acyclovir without repeat CSF analysis.
Discussion
The current recommendations for dosing and duration of acyclovir for HSE appear adequate for most patients. The literature and cases 1 and 2 support the need for follow-up CSF analysis to assess continued central nervous system infection prior to discontinuation of antiviral therapy. Case 1 had responded but had an early relapse secondary to persistent central nervous system infection with poor outcome and residual hemiparesis. Case 2 responded clinically but had persistent HSV-1 in the CSF after 12 days. Intravenous acyclovir was therefore continued completing a 21-day course, followed by 15 days of oral valacyclovir 1 g three times daily. The patient was discharged on oral valacyclovir for 3 additional months of therapy. Despite prolonged treatment, serious sequelae included new onset seizures and short-term memory deficits. Whether delay in diagnosis or uncontrolled underlying medical conditions, (AIDS [CD4+ T-cell count 116 cells/mm3] or diabetes mellitus [Hgb A1c 16%]), contributed to or resulted in prolonged and invasive infection is unclear.
Case 3, in contrast to cases 1 and 2, showed that early diagnosis and treatment in the absence of medical confounds may result in a rapid recovery without serious sequelae. This represents the first case reported with the eradication of HSE with oral medication after only 3 days of intravenous acyclovir.
Since CSF PCR quantification of HSV-1 has become available, this should be requested initially and when repeat CSF analysis is indicated. Discontinuation of antiviral treatment prior to eradication of infection has been associated with significant neurological sequelae [25]. The viral load in the CSF may be a better indicator of the need for continued therapy more than the simple presence of the virus itself. Whether prolonged oral valacyclovir therapy improves long-term outcomes is currently under review [26]. The finding that some apparent relapses did not relate to persistent viral infection, possibly reflecting inflammatory pathophysiology, further supports the need for repeat PCR CSF analysis to assist in deciding on the optimal duration of treatment.
The IDSA recommendations are sufficient for most cases. We feel that individualization of treatment is necessary, with particular emphasis on repeat CSF analysis with quantification of the HSV-1 viral load.
Footnotes
The authors declare no competing interests.
