Abstract
For over 50 years, viral infection has been recognized as an important trigger of acute myocarditis, inflammatory dilated cardiomyopathy (DCM) and congestive heart failure. Nevertheless, viral heart disease remains challenging to diagnose and treat. Improved diagnostic methods for myocarditis have led to a better understanding of its pathophysiology. The recognition of virus-mediated damage, inflammation and autoimmune dysregulation in these patients highlights the importance of differentiating between virus-positive and virus-negative inflammatory DCM. These insights have led to the development of novel treatment strategies, including intravenous immunoglobulin and interferon therapy for virus-positive patients. This article will focus on the pathogenesis of viral myocarditis, especially parvovirus B19-induced, its progression to inflammatory DCM and future treatment strategies.
Introduction
Myocarditis by definition is an inflammatory disease of the cardiac muscle. It has a variety of causes of both infectious and non-infectious origins [1]. Most often, myocarditis results from a viral infection, but also other pathogens, cardiotoxic or hypersensitivity drug reactions, or autoimmune reactions such as giant cell myocarditis, sarcoidosis and Churg-Strauss [1–3] may induce myocarditis. At a histological level, it is characterized by either focal or diffuse inflammation of the myocardium. In the long-term, myocarditis may evolve towards an inflammatory dilated cardiomyopathy (DCM), ultimately leading to chronic heart failure [4–7].
The true incidence of myocarditis is difficult to assess due to the extreme diversity of its clinical manifestations, with symptoms ranging from mild dyspnea or chest pain to cardiogenic shock and sudden death [1,2]. Myocarditis may lead to chronic heart failure [8] and may be the cause of sudden death of young adults in 8–12% of cases [9,10].
Endomyocardial biopsies (EMB) are the gold standard for the diagnosis of virus presence and inflammation in the heart. Due to the lack of sensitivity and specificity of traditional staining methods – haematoxilin and eosin – to diagnose myocarditis, its criteria have been updated. They consist of EMB with immunohistochemical counting of inflammatory cells and viral PCR analyses in cardiac tissues [11]. This has led to a renewed interest in inflammatory cardiomyopathies and the recognition of overlap in virus-mediated damage, inflammation and autoimmune dysregulation in these patients. These insights allow the development of novel aetiology-directed treatment strategies, both for virus-positive and virus-negative inflammatory DCM. Herein, we will focus on viral myocarditis, especially parvovirus B19 (B19V)-induced, the possible progression to virus-induced inflammatory DCM and novel treatment strategies.
Viral myocarditis
For over 50 years, viral infection has been recognized as an important factor of congestive heart failure, nevertheless it remains a challenging disease to diagnose and treat [12,13]. Until the 1990s, the most frequently reported viruses in patients in the developed countries were adenoviruses and enteroviruses [14,15]. Recently, B19V and human herpes virus 6 (HHV6) are increasingly found in a significant percentage of patients diagnosed with both acute and chronic cardiomyopathy [16–18]. However, the pathogenetic significance of B19V genomes in those patients is still a matter of debate [19]. To better understand the variable clinical outcome of viral myocarditis, one has to first address its underlying pathophysiological process (Figure 1).
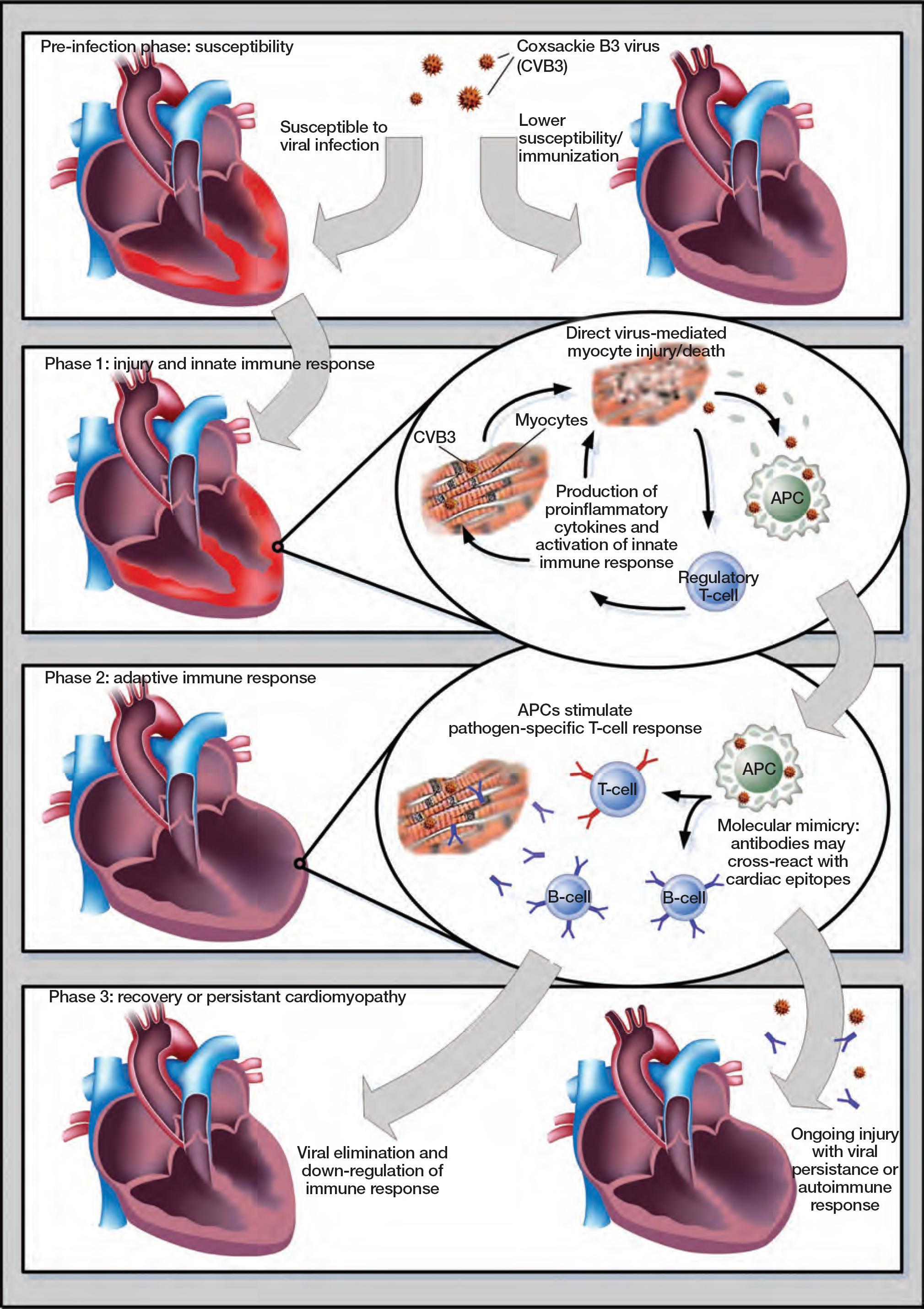
Pathophysiological phases of viral myocarditis
Pathophysiological phases
The pathophysiological progression of virus induced-myocarditis is mainly derived from experiments with Coxsackie virus B in murine models. To date, in vivo animal models for other cardiotropic viruses such as B19V or HHV6 are lacking. It is important to note that enterovirus is known to infect cardiomyocytes, whereas B19V targets endothelial cells [20,21]. The pathogenesis involves a pre-infection phase and three distinct phases that occur once the virus comes into contact with the cardiomyocytes or endothelial cells [20–22]. The pre-infection phase is the time when susceptibility is determined for the development of adverse inflammation in response to viral infection. Identifying the immuno-genetic background that makes people susceptible to myocarditis is essential. Preventive measures, such as immunization for virus-mediated heart disease, could be instituted in those who are susceptible to the development of exaggerated cardiac inflammation and the resulting injury [20].
During phase one, enterovirus experiments demonstrate active virus replication within the myocardium [20,22]. Direct virus-mediated lysis induces the destruction of the cardiomyocytes. Here, viral proteases induce apoptosis and enhance the proteolytic activity in cardiomyocytes [23–25]. Viral entry through receptor-mediated endocytosis triggers the innate immune response through proinflammatory cytokines [26,27], suppressors of cytokine signalling proteins [28,29] and Toll-like receptors (TLRs). These TLRs recognize damage-associated molecular patterns (DAMPs) and pathogen-associated molecular patterns (PAMPs) of microbial pathogens [30]. This innate immune response enhances cardiac cell injury, but also may result in efficient clearance of the virus, which correspond clinically to a non-symptomatic or subclinical myocarditis [20,22]. B19V infection of endothelial cells also induces cellular dysfunction [31,32], which might contribute to cardiac injury and failure after B19V infection.
The second phase is characterized by an immune shift towards an adaptive immune response with inflammatory cellular infiltration. Persistence of the viral genome without detectable virus replication potentially leads to immune dysregulation. The latter is characterized by the maintenance of an immune response with continuous injury of the myocardium, leading to progressive failure [20,22]. Immune dysregulation is, in part, induced by molecular mimicry [33], where mimicked epitopes are shared between the viral and cardiac antigens [34]; this may induce the autoimmunity.
The last phase is characterized by remodelling of the heart, possibly in the absence of replicating virus or viral genome within the myocardium. Circulating cross-reacting autoantibodies together with the virus-induced cardiomyocyte injury and subsequent release of cytokines, lead to the development of a DCM [35,36].
Treatment strategies
Conventional therapy
The goals of treatment in patients with inflammatory DCM are to improve survival, slow disease progression, minimize risk factors and alleviate symptoms. Standard supportive heart failure therapy should be initiated in all inflammatory DCM patients, including ACE-inhibitors angiotensin II receptor blockers, aldosterone antagonists and β-blockers [37,38]. Additionally, patient lifestyle modifications are important to control symptoms of heart failure. In certain cases, such as inflammatory DCM patients with cardiac conduction abnormalities and/or increased risk of sudden cardiac death, cardiac resynchronization therapy combined with an implantable cardioverter device significantly reduces both morbidity and mortality [39,40].
Immunization therapy in susceptible persons
One possible strategy for the prevention of viral myocarditis is immunization. The feasibility of such an approach was first demonstrated in mouse models of viral myocarditis [41,42]. However, there is currently no vaccine or therapeutic reagent for viral myocarditis in real clinical practice, mainly due to the inability to identify persons at risk of developing myocarditis in the presence of these common respiratory viruses [43]. It also remains quite unclear which specific serotypes of these cardiotropic viruses cause myocarditis, heart failure or sudden death. Therefore, future immunization programmes should aim to prevent myocarditis in susceptible persons and target specific viral serotypes. Research on this topic should also result in an accurate prediction of the risk-benefit ratio for immunization against specific virus-induced cardiac disease.
Antiviral therapy
Beside the conventional heart failure regimen and possible preventive immunization strategies, specific treatment strategies must target the presence of the underlying aetiology of inflammatory DCM. Therefore, distinction between virus-negative or virus-positive inflammatory DCM is crucial, since it will lead to different treatment strategies and outcome [44,45].
To date, few studies have investigated antiviral therapy in virus-positive inflammatory DCM patients. Early reports on relatively small patient cohorts of enterovirus-positive inflammatory DCM patients indicated a beneficial haemodynamic effect and partial elimination of viral infection with interferon-α treatment [46]. In addition, the potential antiviral effect of interferon-β using 18 injections of 106 U during 6 weeks was shown in an open-label pilot study of 22 patients with chronic heart failure and enterovirus or adenovirus persistence [47]. After these results, a prospective, placebo-controlled randomized multicentre study was initiated in 2002 and ended in 2007. The trial has not yet been published, but the first study results were presented at the American Heart Association meeting in 2008 [48], demonstrating that viral elimination or substantial decrease of the viral load resulted in an improvement of functional performance of the patient.
Because B19V is the most prevalent virus in heart disease, our group recently investigated the beneficial effect of high-dose (2 g/kg) intravenous immunoglobulin (IVIg) in patients with chronic unexplained heart failure related to a significant B19V myocardial persistence (>250 copies/μg DNA) in an open-labelled uncontrolled study of 17 patients [49]. The mode of action of IVIg is complex, owing to its broad range of activities including anti-infection, anti-inflammatory and immunomodulating properties [50,51]. The IVIg treatment in this pilot study resulted in a significant decrease in B19V load accompanied by an improvement of clinical symptoms and cardiac function, and decrease in end-systolic diameter. Additionally, in some patients a coinfection with HHV6 was noted, which was also eliminated after IVIg treatment, suggesting that IVIg might also have a beneficial effect on other cardiotropic viruses. Based on these favourable results of this pilot study, a randomized, double-blind, placebo-controlled study has been initiated in our centre in patients with chronic B19V-related idiopathic DCM (Clinical Trial number NCT00892112).
Immunosuppression and immunomodulation
Successful treatment of virus-negative inflammatory DCM with immunosuppression further supports the clinical benefit of patient stratification, with an increase in left ventricular ejection fraction and improvement in NYHA class [52,53]. By contrast, immunosuppressive therapy in patients with virus-positive DCM may deteriorate cardiac function, stressing the need for determining the specific aetiology of inflammatory DCM [54]. Additionally, immunoadsorption therapy with subsequent immunoglobulin substitution has proven to be an effective immunomodulatory regimen in inflammatory DCM patients, although the presence of myocardial virus genome was not addressed in these trials [55,56].
Conclusions
Myocarditis is a disease characterized by myocardial inflammation, with a highly variable natural history, but frequently evolving to chronic heart failure and inflammatory DCM. The advancement in molecular and immunohistological biopsy techniques allows us to better differentiate between virus-positive and virus-negative DCM. Antiviral therapy holds great promise for reducing clinical symptoms and systolic dysfunction caused by virus infection, especially where B19V is involved. Further preclinical research is crucial to identify the persons at risk of heart failure in response to specific virus serotypes. International randomized trials based on this improved patient selection will have to test novel treatment options to improve the outcome of patients selected on cardiac inflammation and the presence of virus.
Footnotes
The authors declare no competing interests.
