Abstract
Background:
Because of high mutation rates, new drug-resistant viruses are rapidly evolving, thus making the necessary control of influenza virus infection difficult. Methods: We screened a constrained cysteine-rich peptide library mimicking μ-conotoxins from Conus geographus and a proline-rich peptide library mimicking lebocin 1 and 2 from Bombyx mori by using influenza virus RNA polymerase (PB1, PB2 and PA) and nucleoprotein (NP) as baits.
Results:
Among the 22 peptides selected from the libraries, we found that the NP-binding proline-rich peptide, PPWCCCSPMKRASPPPAQSDLPATPKCPP, inhibited influenza replicon activity to mean ±SD 40.7% ±15.8% when expressed as a GFP fusion peptide in replicon cells. Moreover, when the GFP fusion peptide was transduced into cells by an HIV-TAT protein transduction domain sequence, the replication of influenza virus A/WSN/33 (WSN) at a multiplicity of infection of 0.01 was inhibited to 20% and 69% at 12 and 24 h post-infection, respectively. In addition, the TAT-GFP fusion peptide was able to slightly protect Balb/c mice from WSN infection when administrated prior to the infection. Conclusions: These results suggest the potential of this peptide as the seed of an anti-influenza drug and reveal the usefulness of the constrained peptide strategy for generating inhibitors of influenza infection. The results also suggest that influenza NP, which is conserved among the influenza A viruses, is a good target for influenza inhibition, despite being the most abundant protein in infected cells.
Introduction
Peptide inhibitors of viral infection have been studied intensively, since enfuvirtide (T20) showed potent inhibitory activity against HIV [1] and was approved for clinical use [2]. In addition to peptide inhibitors that mimic viral protein sequences [3], peptide aptamers that mimic natural auto-constrained peptides have been designed and demonstrated to be potent anti-rabies virus drugs [4]. Among these constrained peptides, which have been generated as a part of peptide coaptamer libraries, those that mimic venom toxins of a predatory cone snail and insect antimicrobial peptides, have been constructed. The former library is rich in cysteine (Cys), whereas the latter one is rich in proline (Pro). The Cys-rich constrained peptides mimic μ-conotoxins from Conus geographus [5], and the Pro-rich constrained peptides mimic lebocin 1 and 2 from Bombyx mori [6]. These libraries can be screened using a number of sequences as baits, thus providing an opportunity for the identification of novel antiviral lead compounds.
Influenza A viruses cause diseases in humans, pigs and other mammals and in birds [7], and can result in pandemics, such as the latest ones caused by the novel swine-origin influenza virus in 2009 [8]. Influenza viruses rapidly undergo random mutation and thereby evolve into new types with diverse antigenic determinants, which pose a significant challenge for the control of influenza infection [9]. Hence, the role of anti-influenza drugs has become more important than ever for preparing for a pandemic influenza [10]. Recently, a new class of small chemicals targeting the viral nucleoprotein (NP) [11] and RNA-dependent RNA polymerase (RdRp) [12] were identified as inhibitors of influenza virus replication. However, anti-influenza drugs invariably encounter the emergence of mutants with drug resistance [13–16]. All the H3N2 isolates are resistant to amantadine [17], and H1N1 isolates have been reported to be resistant to oseltamivir [18,19]. Even swine-origin influenza virus rapidly acquired oseltamivir resistance [20,21].
In order to expand the options for the control of influenza virus infection, we screened the Cys-rich and Pro-rich constrained peptide libraries [4] using influenza viral RdRp subunits (PB2, PB1 and PA) and NP as baits and found that an NP-targeting peptide had the potential to inhibit influenza viral replication and even protect mice from influenza infection.
Materials and methods
Chemistry
Plasmid construction
PR8PB2 sequence was moved from pBSKPB2wt [22] by BamHI and NdeI digestion and cloned into the bait vector pGBKT7, resulting in pGBKPR8PB2. PR8PB1 sequence was moved from pUC19HAPB1wt [22] by NcoI and XhoI digestion and cloned into pGBKT7 at NcoI and SalI sites, yielding pGBKPR8PB1. PR8PA sequence was moved from pUC19HAPAwt [22] by NcoI and XhoI digestion and cloned into pGBKT7, resulting in pGBKPR8PA. PR8 NP was PCR-amplified from pCNP [23] and cloned into pGBKT7, resulting in pGBKPR8NP.
Constrained peptide sequences, which included a haemagglutinin (HA) tag [4], were PCR-amplified and cloned into pGEX-6p-3, pcdna3.1+ and pEGFP-C3, yielding pGEX-, pcdna-, and pEGFP-constrained peptides, respectively. The nuclear localization signal (NLS) of the SV40 T-antigen was inserted into the BamH1 site of the pEGFP-constrained peptide (PB1 1-2, 4-1, NP3-2, and 6-1). The HIV-TAT protein transduction domain (PTD) sequence was inserted into NcoI and XhoI sites of pET28b, resulting in pET28b-TAT. GFP NP9-5 was PCR-amplified from pEGFP-NP9-5 and cloned into pET28-TAT, yielding pET28-TAT-GFP-NP9-5. An N-terminal His tag was added by PCR amplification into pET28-TAT-GFP-NP9-5. The amplified fragment was inserted into pET21b(KM) [24], resulting in pET21(KM)-TAT-GFP-NP9-5. The control His-TAT-GFP expression plasmid was constructed by HindIII digestion of pET21(KM)-TAT-GFP-NP9-5 to remove the constrained peptide sequences, resulting in pET21(KM)-TAT-GFP. The sequence information for the primers is available on request.
Yeast two-hybrid screening of constrained peptides
Two constrained peptide libraries, CNCMRox (Cys-rich peptide; C-X2 -C-X5 -C-X6 -C-X5 -C-X2 -C) and CNCM001 (Pro-rich peptide; PP-X5 -PPP-X5 -P-X5 -PP) [4], were screened using influenza virus PB2, PB1, PA and NP as baits by a sequential transformation method using Matchmaker Two-Hybrid System 3 and Matchmaker Yeast Transformation System 2 (Clontech, Mountain View, CA, USA). The size of the CNCMRox and CNCM001 libraries was ×107 and 3×107, respectively. Screening of the PA-binding peptides was performed using 3 mM 3-amino-1,2,4-triazole (3-AT) to eliminate false-positive clones, since PA is acidic and can auto-activate the two-hybrid system.
GST pull-down assay
Escherichia coli (E. coli) Rosetta was transformed with pGEX-6p-3 carrying the constrained peptide sequences containing HA. The GST-fused constrained peptides were expressed by 1 mM IPTG-induction for 2 h at 37°C. The bacterial cells were disrupted by sonication 3× for 10 s in phosphate buffered saline (PBS) and kept on ice between sonication. The GST-constrained peptides were bound to glutathione-Sepharose by incubation at 4°C for 2 h. The 293T cells in 24-well plates were transfected with pCPB2, pCPB1, pCPA and pCNP using Lipofectamin2000 (Invitrogen, Carlsbad, CA, USA). The transfected cells were lysed in 50 mM Tris-HCl (pH 8.0), 500 mM NaCl, 0.1% Triton X-100, 1 mM DTT and 1 mM phenylmethylsulfonyl fluoride (PMSF; high-salt extraction buffer) at 48 h after trans-fection. After centrifugation at 15,000 rpm at 4°C in an Eppendorf microcentrifuge (Eppendorf), the supernatants were diluted with 50 mM Tris-HCl (pH 8.0), 150 mM NaCl, 0.1% Triton X-100, 1 mM DTT, 1 mM PMSF (GST pull-down buffer) and mixed with the glutathione-Sepharose bound to the GST-constrained peptides for 2 h at 4°C. The glutathione-Sepharose was then washed 5× with the GST pull-down buffer, resuspended in sodium dodecyl sulfate (SDS) sample buffer, and boiled for 5 min. Thereafter, the bound proteins were examined by SDS-PAGE.
Replicon inhibition assay
The replicon inhibition assay was performed by cotransfection of 100 ng of pCPB2, pCPB1, pCPA, pCNP or pPolINSluc [25], 10 ng of pRLSV40, and 50 or 100 ng of pEGFP-C3-constrained peptides in 293T cells in 24-well plates using Lipofectamine2000 (Invitrogen). At 24 h after transfection, luciferase activity was measured using the Dual-Glo luciferase assay kit and a GloMax 96 Microplate Luminometer (Promega, Madison, WI, USA). The inhibition rate was expressed as a percentage of the relative luciferase activity normalized to the control.
Purification of the GFP-fused constrained peptide tagged with the TAT PTD at the N-terminus
The Pro-rich constrained peptide NP9-5 fused with the N-terminal His-TAT-GFP sequences was expressed in E. coli and purified in a denatured condition. Briefly, E. coli BL21(DE3) was transformed with pET21(KM)-TAT-GFP-NP9-5, pET21(KM)-TAT-GFP and pET21(KM)-GFP. TAT-GFP-NP9-5, TAT-GFP and GFP were expressed by 1 mM IPTG induction at 30°C for 3 h after a bacterial growth of optical density600 = 0.58 was achieved. After harvesting, the cells were resuspended in a buffer containing 50 mM NaH2 PO4 (pH 8.0), 500 mM NaCl, 0.1% Triton X-100, 8 M urea, 0.1% 2-mercaptoethanol, 1 mM PMSF and 2 mM imidazole, and then were lysed by sonication. After centrifugation at 11,000 rpm for 30 min at 4°C (Beckman JA rotor, Beckman Coulter, Brea, CA, USA), the supernatant was incubated with Ni-NTA agarose (Qiagen, Valencia, CA, USA), and the bound proteins were eluted with PBS containing 4 M urea and 250 mM imidazole after the column had been washed with 20 mM imidazole. The eluted proteins were dialyzed against 500 ml of PBS containing 10% glycerol by exchanging the buffer every 3 h overnight at 4°C. Aliquots of purified proteins were collected and stocked at −80°C.
Western blot
The replicon cells were lysed with SDS-sample buffer and applied on SDS-PAGE, followed by semi-dry electroblotting (BioRad, Hercules, CA, USA) on nitrocellulose membranes (Millipore, Billerica, MA, USA). After being blocked with 10% skim milk in 20 mM Tris-HCl (pH 7.5), 150 mM NaCl and 0.02% Tween 20, the membranes were incubated with rabbit anti-GFP (1: 1000; SIBS Antibody Center, Shanghai, China), NP (5/1; 1:1000), which was a gift from RG Webster (St Jude Children's Research Hospital, Memphis, TN, USA) [26], and anti-actin antibodies (1:50) as the primary antibodies, respectively. After the membranes were incubated with alkaline phosphatase-bound anti-rabbit immunoglobulin G (IgG) or anti-mouse IgG (1:7500), the position of the bound antibodies was visualized using nitroblue tetrazolium and 5-bromo-4-chloro-3-indolyl phosphate (BCIP).
Reagent
Oligonucleotides, pcdna3.1+ and Lipofectamin2000 were purchased from Invitrogen, Matchmaker Two-Hybrid System 3, Matchmaker Yeast Transformation System 2, and pEGFP-C3 from Clontech, 3-AT and anti-actin antibodies from Sigma-Aldrich (St Louis, MO, USA), pGEX-6p-3 and glutathione-Sepharose from GE Healthcare (Pittsburgh, PA, USA), the Dual-Glo luciferase assay kit, alkaline phosphatase-bound anti-rabbit IgG, anti-mouse IgG, nitroblue tetrazolium and BCIP from Promega and pET28b and E. coli Rosetta/pLysS from Novagen (Merck, Darmstadt, Germany). The 3-[4,5-dimethylthiazol-2-yl]-2,5-diphenyltetrazolium bromide (MTT) was from Shanghai Generay Biotech Co., Ltd (Shanghai, China).
Virology
Viruses and cells
Madin-Darby canine kidney (MDCK) and Vero cells were obtained from the cell bank of Shanghai Institute of Biological Sciences (Shanghai, China). The 293T cells were kindly obtained from Y Kawaoka (University of Tokyo, Tokyo, Japan). MDCK cells were maintained in Dulbecco's modified Eagle medium (DMEM; GIBCO, Invitrogen) supplemented with 5% fetal bovine serum (FBS; GIBCO, Invitrogen). The 293T and Vero cells were maintained in DMEM supplemented with 10% FBS. Influenza virus A/WSN/33 (rWSN) was reconstituted by the 12 plasmid-transfection method in 293T cells, amplified in MDCK cells, and stocked at −80°C [27]. Virus titre was determined by the plaque assay in MDCK cells.
Virus inhibition by immunofluorescence assay
Vero cells were seeded on coverslips in 24-well plates. The cells were transfected with pEGFP-C3-constrained peptide (100 ng). At 12 h after transfection, the cells were infected with rWSN at a multiplicity of infection (MOI) of 5. At 8 h post-infection, the cells were fixed with PBS containing 4% formaldehyde for 5 min and permeabilized with 0.1% Triton in PBS (PBST). The fixed cells were incubated for 1 h with the anti-NP monoclonal antibody (1:1,000), NP (5/1) [26], followed by incubation with Alexa fluor 555-conjugated anti-mouse secondary antibodies (1:500; Molecular Probes, Invitrogen) for 1 h. After being washed with PBST, the cells were mounted in Mowiol (Kuraray, Tokyo, Japan). The expressed constrained peptides and NP were observed under the fluorescence microscope (Leica DM-IRB; Leica, Wetzlar, Germany).
Plaque reduction assay
The 293T cells in a 24-well plate were transfected with 1 μg of pEGFP-C3-constrained peptides. The cells were then infected with the rWSN at an MOI of 0.01 at 12 h after transfection. The virus titre in the culture supernatant was determined by a plaque-forming assay in MDCK cells at 12 h post infection.
Protein transduction and plaque reduction assay of influenza virus WSN
TAT-GFP-NP9-5 and TAT-GFP were transduced into Vero cells [28]. Briefly, Vero cells at 80% confluence in 12-well plates were incubated with 0.04 mg/ml of TAT-GFP-NP9-5 or TAT-GFP in the culture media for 1 h. After incubation with the proteins, the cells were washed 3× with the culture media. Next, the transduced Vero cells were infected with rWSN (MOI=0.01, 0.1 and 1), and the culture supernatants were harvested at 12 and 24 h post-infection. The virus titre in the culture supernatant was measured by a standard plaque assay in MDCK cells. The growth kinetics of rWSN in the Vero cells was analysed as previously described [29].
Cytotoxicity test of TAT-GFP-NP9-5 and TAT-GFP proteins by MTT assay
MDCK and Vero cells were seeded at 104 cells per well and 293T cells were seeded at 3×104 cells per well in 96-well plates at 12 h before protein transduction. For transduction, media were removed and incubated with TAT-GFP-NP9-5 and TAT-GFP diluted in DMEM at 0.08, 0.04 and 0.02 mg/ml, respectively.
After 1 h incubation, the supernatant was replaced with 100 μl DMEM supplemented with FBS. Cytotoxicity of TAT-GFP-NP9-5 and TAT-GFP proteins was tested by a standard MTT assay at 1, 2 or 3 days after protein-transduction [30]. Briefly, after culture media were removed, cells were incubated with 100 ml of 4% MTT in DMEM for 4 h. Then, the supernatant were removed and the cells were dissolved in 150 μl DMSO by pipetting. The colour of formazan produced by viable cells was measured at 490 nm. Viability of cells was expressed as percentage of optical density490 of the control cells without protein-transduction.
Mice protection experiment
Endotoxin was removed from TAT-GFP-NP9-5 and TAT-GFP by the ToxinEraser™ Endotoxin Removal Kit (GenScript, Piscataway, NJ, USA). Groups of four 6-week-old female Balb/c mice (Sino-British Laboratory Animal, Shanghai, China) were anesthetized using ether and inoculated with 50 μl of 105 PFU of rWSN by nasal dropping before and after 4 h of intra-nasal administration of 50 and 5 μg of TAT-GFP-NP9-5 and TAT-GFP in 50 μl of PBS. Their body weight was monitored every day for 10 days after infection. The uninfected mice were sacrificed under anaesthesia and dissected at 1 day after inoculation with TAT-GFP-NP9-5, TAT-GFP and PBS. The lungs and the tracheas were fixed with 10% formalin/PBS, sliced, mounted in 90% glycerol and 100 mM Tris-HCl (pH 8.0), and observed under the fluorescence microscope.
Statistical analysis
Significant differences were evaluated using P-values calculated from the Student's t-test.
Results
Screening of the constrained peptide libraries using influenza virus PB2, PB1, PA and NP as baits
We screened the constrained peptide libraries using influenza virus PB2, PB1, PA, and NP sequences as baits (Additional file 1). The sequences of the constrained peptides are listed in Additional file 2. The Pro-rich constrained peptides bound to NP contained the designed sequences. However, those bound to PA and the Cys-rich constrained peptides showed frame-shift from the designed sequences, even though they remained bound to their bait sequences.
Screening of the constrained peptides by influenza replicon inhibition assay
First, the cloned constrained peptide sequences were tested for their ability to inhibit influenza replicon activity by cotransfection of the expression plasmids of the peptide sequences with or without GFP, or GFP with the SV40 T-antigen NLS, PKKKRKVG (Figure 1). Most of the constrained peptide sequences did not inhibit influenza replicon activity. Only Cys-rich peptide, GFP-NP3-2, and Pro-rich peptide, GFP-NP9-5, significantly inhibited the replicon activity at a plasmid concentration of 100 ng. The replicon activity of pEGFP-NP3-2 and pEGFP-NP9-5 transfected cells was mean ±sd 63.6% ±5.7% (Student's t-test, P<0.05) and 40.7% ±15.8% (P<0.05) of pEGFP-C3 transfected cells (control), respectively. When the NLS was introduced into GFP-NP3-2 and 9-5, the replicon activity was not inhibited. Cotransfection of NP9-2, 9-3, 9-5, 9-6, 3-2 and PA A6 (50 and 100 ng in total) also did not inhibit the replicon activity (Mix), perhaps because the amount of DNA for NP9-5 was too small.
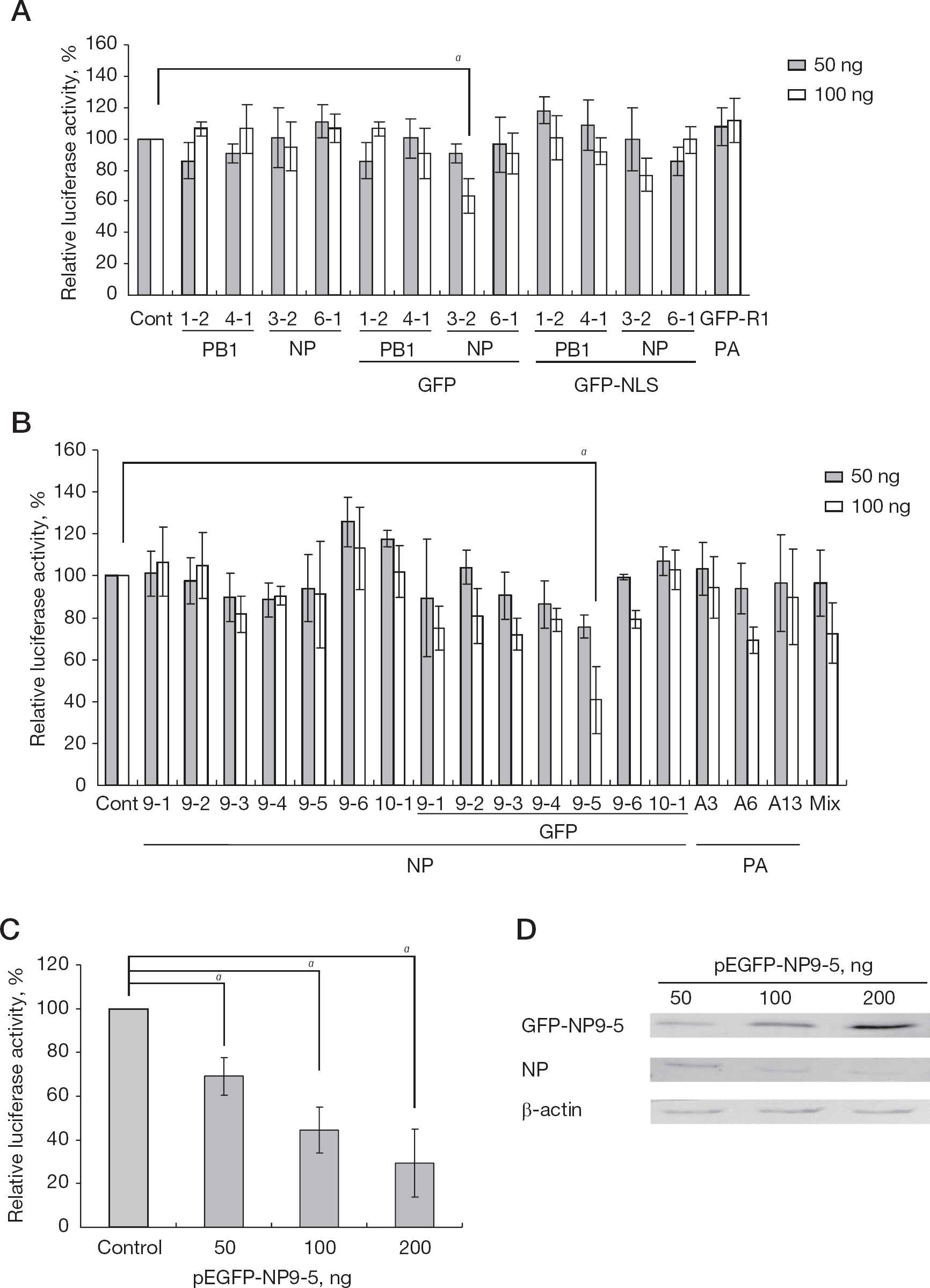
Influenza replicon inhibition assay by coexpression of the constrained peptide
We therefore further examined only the inhibitory effect of GFP-fused NP9-5. The dose-response inhibitory effect of pEGFP-NP9-5 on influenza replicon activity was examined at 50, 100, and 200 ng (Figure 1C). Transfection of pEGFP-NP9-5 inhibited influenza replicon activity significantly at these concentrations (P<0.05). The amount of NP in the replicon cells was then examined by western blot analysis (Figure 1D). The level of NP in the replicon cells decreased according to the amount of the transfected pEGFP-NP9-5, while that of the control β-actin was similar.
Because our screening did not cover the entire scale of the libraries (Additional file 1), peptides efficiently inhibiting the influenza replicon activity might remain to be picked up in the libraries.
Interaction between the constrained peptides and bait influenza proteins by GST pull-down assay
We used GST pull-down assays to investigate the interactions between PB1 and the GST-fused Cys-rich peptides, PB1 1-2 and 4-1, between PA and the Pro-rich peptide, PA A6, and between NP and the Pro-rich peptides, NP9-3, 9-5, and 9-6 (Figure 2). The constrained peptide sequences were fused to GST. PB1, PA and NP were independently expressed in 293T cells. PB1 weakly bound to GST-PB1 1-2 and 4-1. PA bound to GST-PA A6. NP bound to GST-NP9-3, 9-5 and 9-6.
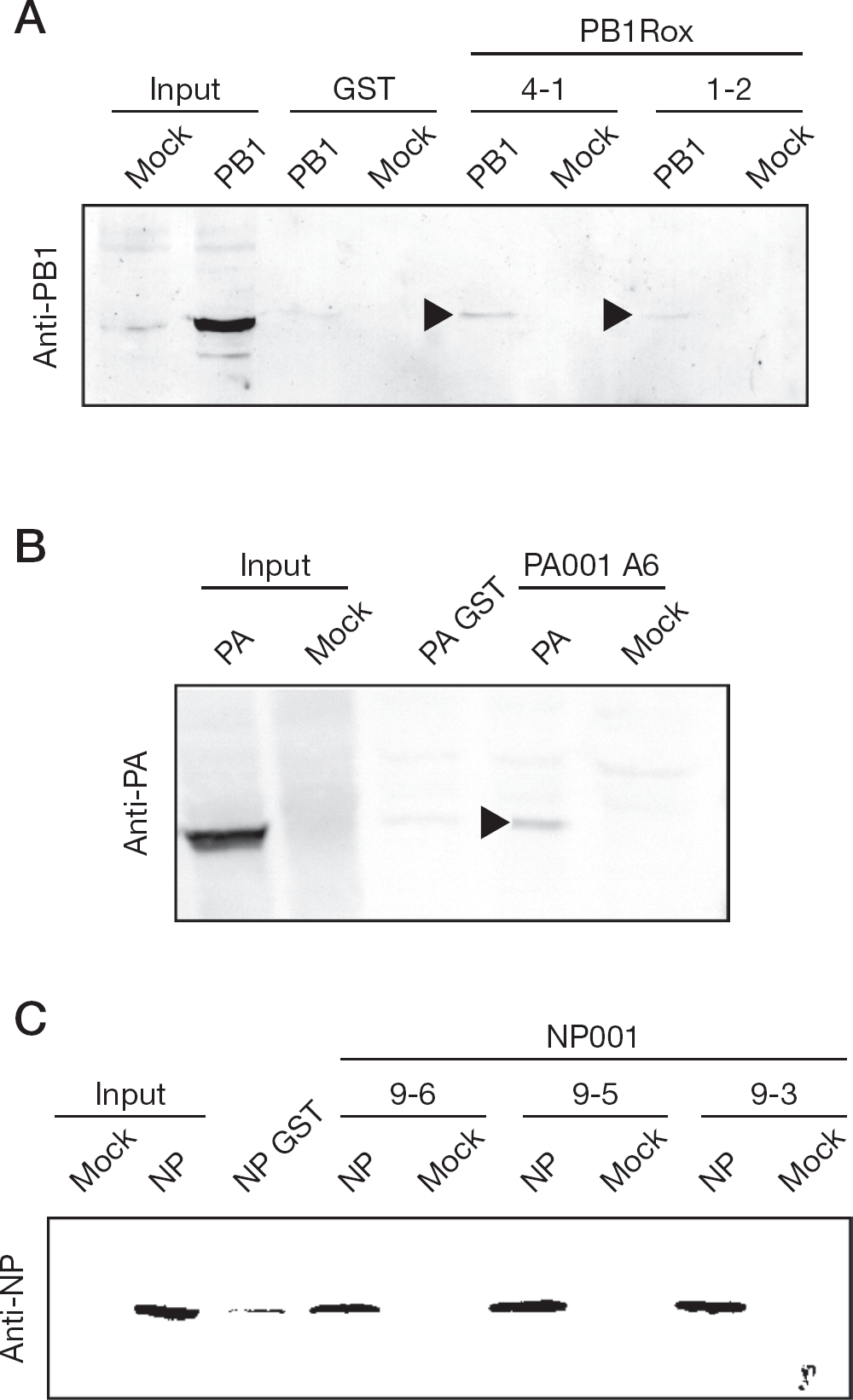
Interaction of PB1, PA and NP with the constrained peptides
Influenza virus plaque reduction assay of pEGFP-constrained peptides
In order to confirm the inhibition activity of the constrained peptides obtained by the replicon inhibition assay, we further examined the inhibition activity of the pEGFP-constrained peptides-transfected cells by a plaque assay of the rWSN strain [27] in 293T cells. The virus titre of pEGFP-NP9-5- and pEGFP-PA A6-transfected cells was mean ±sd 83.1% ±3.1% (P<0.05) and 88.9% ±3.7% (P<0.05) of pEGFP- C3-transfected cells (control), respectively (Figure 3). The titre of pEGFP-NP3-2- and pEGFP-PB1 4-1-transfected cells was similar to that of pEGFP-C3-transfected cells. Although GFP-NP3-2 inhibited influenza replicon activity, rWSN replication was not inhibited. The titre of the cotransfected NP3-2, 9-2, 9-3, 9-5, 9-6 and PA A6 (1 mg in total) was also similar to that of the control cells, perhaps because the amount of the DNA for NP9-5 was too small.
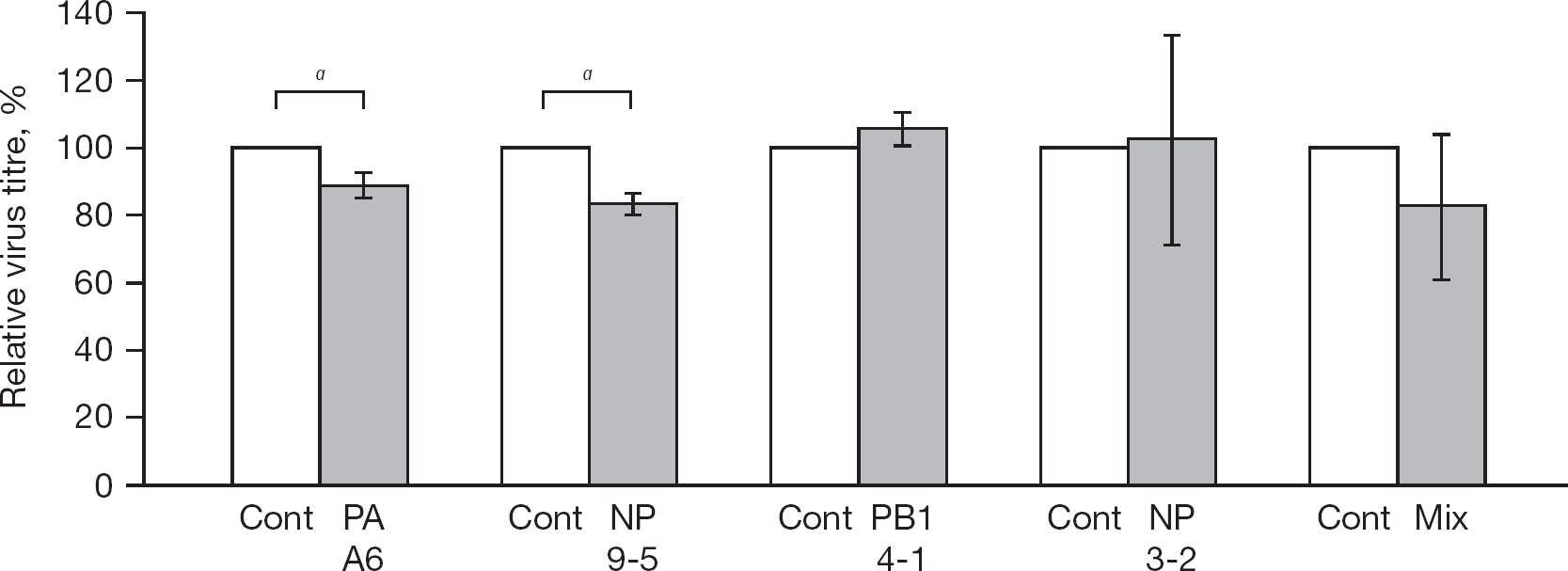
Influenza plaque reduction assay by transfection of GFP-fused PB1 4-1, PA A-6, NP3-2 and NP9-5
Influenza virus inhibition of GFP-NP9-5 detected by immunofluorescence assay
Next, we tested the effect of pEGFP-NP9-5 on rWSN growth in Vero cells by immunofluorescence assay (Figure 4A). Approximately 80% of the pEGFP-NP9-5-transfected cells expressed the peptide, and no apparent cytotoxicity was observed for all transfected cells. NP9-5 likely inhibited viral replication, since some of the cells expressing GFP-NP9-5 (green signal) did not show the NP signal (Figure 4A, panel 6). The number of green and orange cells in one field was counted (Figure 4B), and the inhibition rate was calculated as 1-(number of merged orange cells/number of GFP-expressing cells). The inhibition rate of the pEGFP-NP9-5-transfected cells was 0.84 whereas that of the control pEGFP-C3-transfected cells was 0.46.
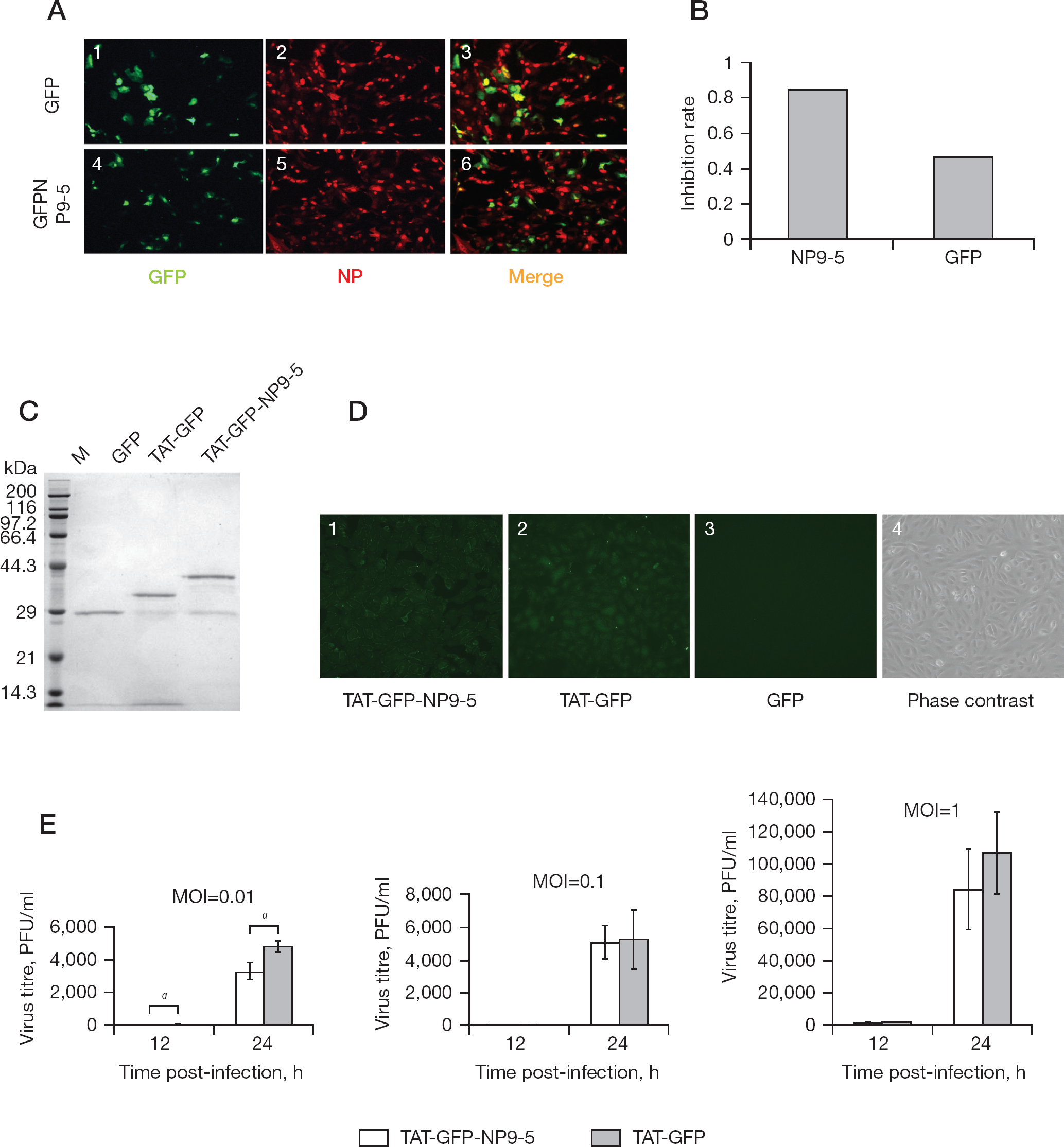
Inhibition of rWSN infected cells by Pro-rich constrained peptide, NP9-5
Influenza virus inhibition by protein transduction of GFP-NP9-5
Although the inhibition of influenza viral replication by transfection of pEGFP-NP9-5 was not efficient, we tested the inhibition by direct protein transduction of GFP-NP9-5 using the HIV-TAT PTD sequence (YGRKKRRQRRR). HIV-TAT PTD and polybasic amino acid sequences can efficiently deliver molecules into cells by endocytosis [28,31]. NP9-5 was fused with GFP (GFP-NP9-5) and TAT PTD (TAT-GFP-NP9-5) at the N-terminus together with the 6×His sequence for Ni-affinity column purification. Both TAT-GFP-NP9-5 and TAT-GFP were purified from bacteria (Figure 4C).
Protein transduction efficiency was checked by using the GFP signal (Figure 4D). We tested Vero, MDCK and 293T cells for protein transduction efficiency and decided to use Vero cells because the protein transduction of Vero cells was the most efficient (Figure 4D, panels 1 and 2). Without TAT-PTD, GFP (Figure 4D, panel 3) were not transduced in the cells.
Vero cells transduced with TAT-GFP-NP9-5 and TAT-GFP for 1 h were infected with rWSN at MOI of 0.01, 0.1 and 1, respectively. The virus titre in the culture supernatant was measured by plaque assay in the MDCK cells (Figure 4E). When the cells were infected at MOI of 0.01, the virus titre of TAT-GFP-NP9-5 was mean ±SD 10.4±2.6 and 3,260 ±510 PFU/ml at 12 and 24 h post-infection, respectively, while that of the control TAT-GFP transduced cells was 51.1 ±25.0, and 4,740 ±340 PFU/ml at 12 and 24 h post-infection, respectively. In the TAT-GFP-NP9-5 transduced cells, the virus titre was significantly lower (P<0.05) than that of the control cells at both time periods. However, when the cells were infected with the virus at MOI of 0.1 and 1, the virus titre at 12 and 24 h post-infection was similar in between TAT-GFP-NP9-5 and TAT-GFP-transduced cells.
Cytotoxicity of TAT-GFP-NP9-5 and TAT-GFP
Cytotoxicity of protein transduction of TAT-GFP-NP9-5 and TAT-GFP into Vero, MDCK and 293T cells were examined by MTT method (Figure 5). Up to 3 days incubation, the viability of Vero and MDCK cells was statistically similar (Student's t-test) among non-transduced, TAT-GFP-NP9-5-transduced and TAT-GFP-transduced cells at 0.02, 0.04 and 0.08 mg/ ml. The viability of 293T cells at 1 day after transduction of 0.04 and 0.08 mg/ml of TAT-GFP-NP9-5 and TAT-GFP was lower than that of the non-transduced cells (P<0.05). The viability of 293T cells 2 days after transduction of 0.04 and 0.08 mg/ml TAT-GFP-NP9-5 and 0.02 and 0.08 mg/ml TAT-GFP was lower than that of the non-transduced cells (P<0.05). The viability of 293T cells 3 days after transduction of 0.02 and 0.08 mg/ml TAT-GFP-NP9-5 and 0.08 mg/ml TAT-GFP was lower than that of the non-transduced cells (P<0.05). The viability of the other 293T cells was similar to that of the non-transduced cells. The MTT assay indicated that the cytotoxicity of these peptides varied and Vero cells were tolerate to protein transduction of the concentration so far examined.
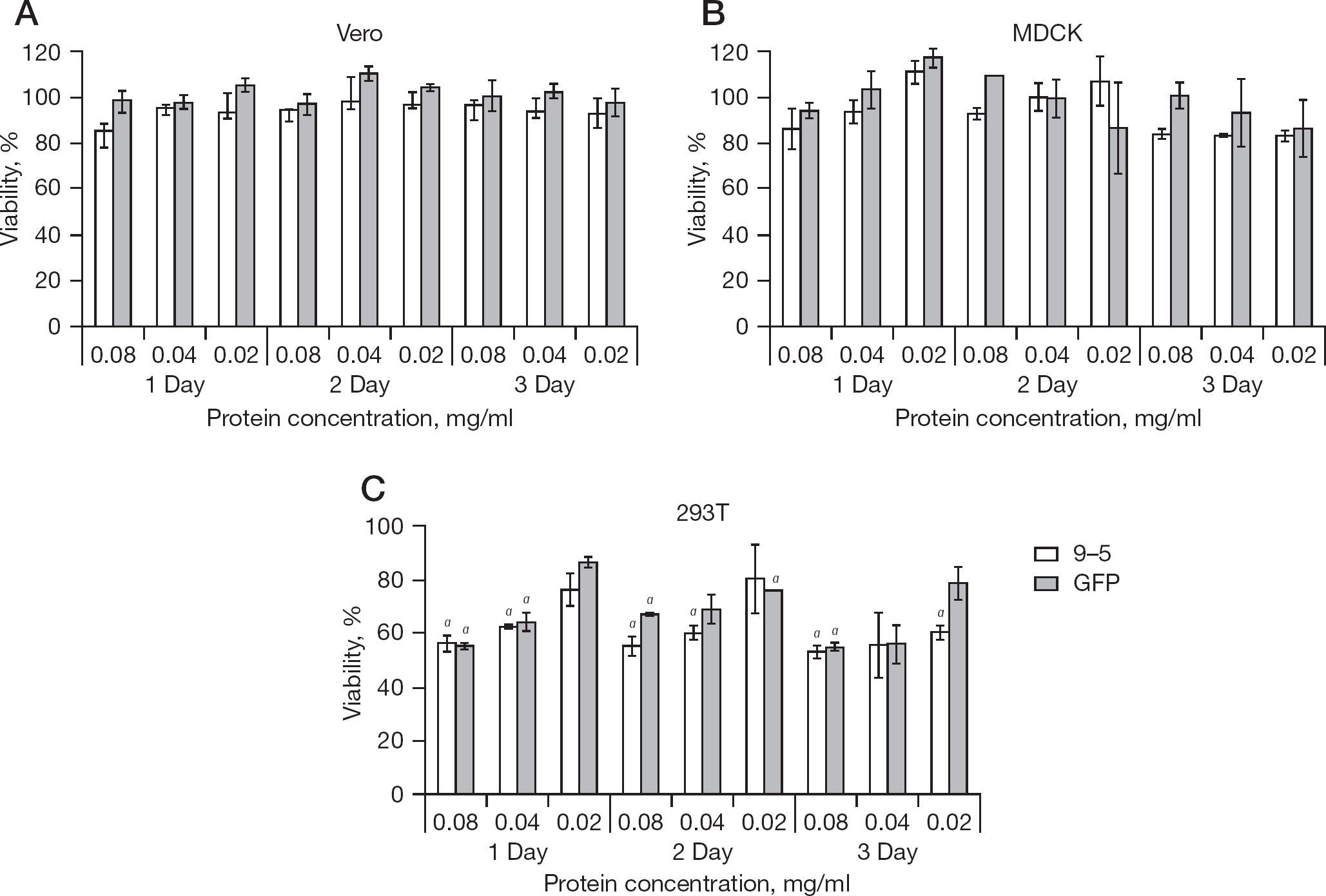
Cytotoxicity of protein-transduction of TAT-GFP-NP9-5 and TAT-GFP into Vero, MDCK and 293T cells
Protection of mice infection by TAT-GFP-NP9-5
Finally, the protective effect of TAT-GFP-NP9-5 was tested in rWSN-infected mice (Figure 6). The lungs and tracheas of the TAT-GFP-NP9-5- and TAT-GFP-administrated mice showed GFP signal even 1 day after transduction (Figure 5A). When the mice were administered 50 μg of TAT-GFP-NP9-5 intranasally at 4 h before infection by rWSN at 105 PFU, their body weight loss at the seventh day of infection was significantly lower (P<0.05) than those administered 50 μg of TAT-GFP, although no significant differences were observed later (Figure 6B). No differences in the body weight loss were observed when the peptides were administrated at 4 h after infection (Figure 6C).
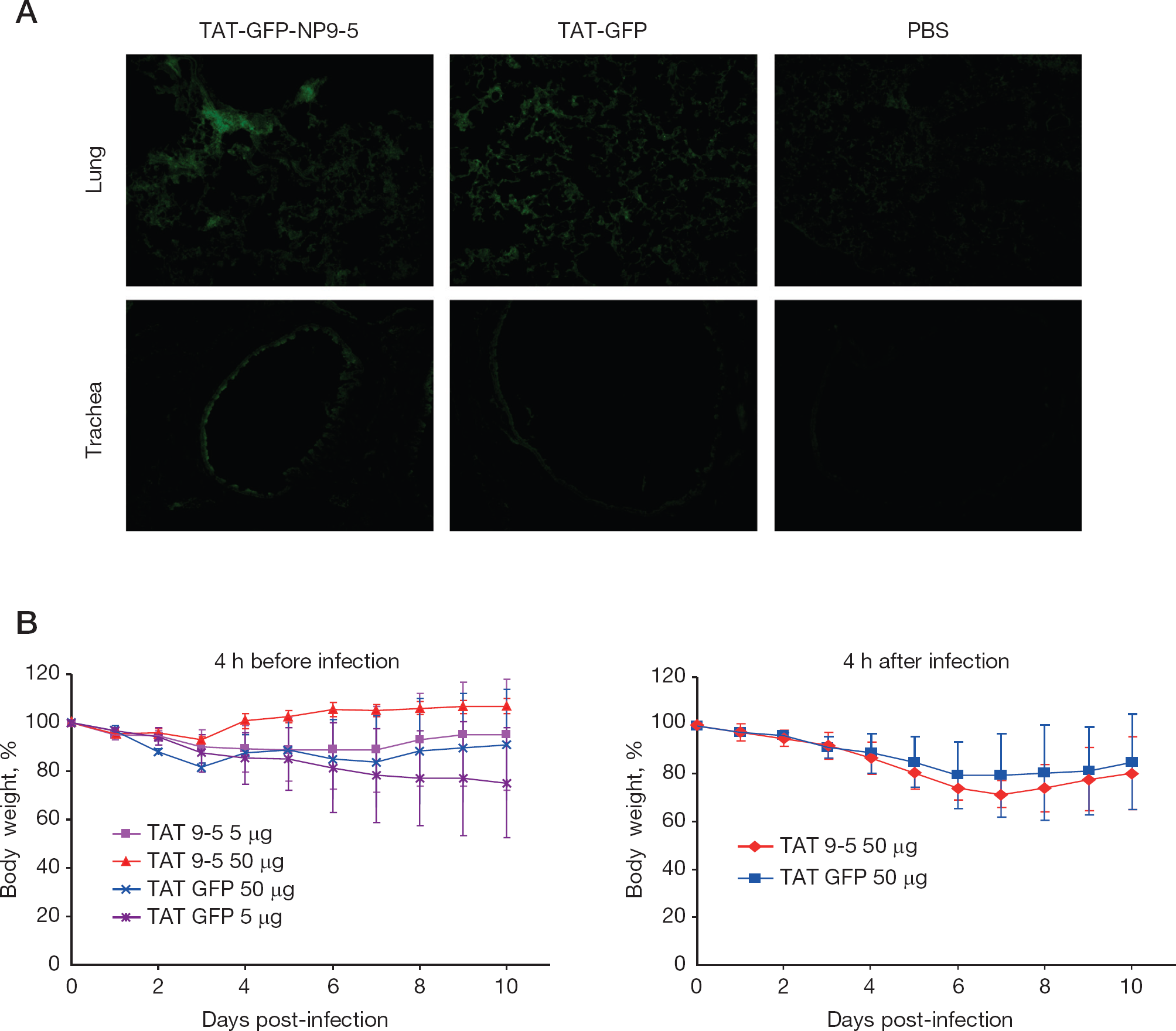
Protection of mice from rWSN infection by TAT-GFP-NP9-5
Discussion
Neuraminidase inhibitors (oseltamivir and zanamivir) and ion channel M2 blockers (amantadine and remantadine) have been widely used to treat influenza virus infection, but the emergence and quick circulation of new, resistant viruses is a serious problem [15,16,32]. Because of the frequent emergence of drug-resistant viruses, it is better to expand the options for influenza virus control. Therefore, we screened constrained peptide libraries, which were successfully used for rabies virus inhibition [4] by using PB2, PB1, PA and NP as baits.
Recently, small chemicals targeting RdRp were identified as inhibiting influenza virus replication [12]. Pyrazinecarboxamide derivatives, such as T-705 (favipiravir) that targets influenza RdRp, have been developed [33,34]. However, the constrained peptides targeting the RdRp subunits did not inhibit influenza replicon activity (Figure 1). Only two Pro-rich constrained peptides targeting NP showed inhibition of replicon activity. NP may not be an effective target for influenza inhibition since the expression of NP in infected cells is abundant [35]. However, NP inhibitors have the potential to control influenza infection because NP is quite conserved among influenza A viruses [36], and recently small chemicals targeting NP were found to inhibit influenza virus replication [11].
The GFP-tagged NP3-2 and NP9-5 inhibited replicon activity, whereas those without the GFP tag did not inhibit it, potentially because of their stability in the cells or because of low affinity due to their conformation. Although NP3-2 and NP9-5 inhibited influenza replicon activity, NP3-2 did not inhibit influenza virus replication in the cells (Figures 1 and 3). Influenza virus replicates its genome RNA in the infected cell nuclei. However, neither NP3-2 nor NP9-5 inhibited the replicon activity when they were tagged with NLS. We further studied only NP9-5 because its replicon inhibition effect was the best among the constrained peptides. The mechanism of NP9-5 inhibition is unclear, but the expression of NP decreased when GFP-NP9-5 was coexpressed (Figure 1D), which is likely due to the targeting of NP to aggresomes (Figure 4A), thus resulting in the degradation of NP [37].
Next, we tested the inhibition of influenza virus replication by GFP-NP9-5 via protein transduction since the GFP-fusion peptide inhibited influenza virus replication only by transfection of the expression plasmid (Figures 3, 4A and 4B). We chose Vero cells because their protein transduction efficiency was almost 100% (Figure 4D). GFP-NP9-5 inhibited rWSN replication to 20% and 69% at 12 and 24 h post-infection, respectively, when cells were infected at a MOI of 0.01. These results were statistically significant (P<0.05; Figure 4E). At the moment, the efficiency of protein transduction and the stability of TAT-GFP-NP9-5 may not be enough to inhibit the virus at higher MOI than 0.1.
The toxicity of protein transduction varied to cell types. At the concentration used to inhibit WSN infection or higher (0.04 and 0.08 mg/ml), no apparent cytotoxicity was observed in Vero cells (Figure 5). Thus, protein transduction introduces large molecules into cells without apparent toxicity. This method will expand the opportunity to develop large molecule inhibitors.
Finally, we tested the protection of influenza-infected mice intranasally, administering GFP-NP9-5 tagged with TAT PTD (Figure 6). GFP signal was detected in the trachea and the lungs of the peptide-administrated mice at 1 day after administration, which indicated that these peptides were transduced in the tissues and remained there for ⩾1 day. NP9-5 partially protected mice from rWSN infection. When these peptides were administrated after 4 h of infection, no differences were observed. The amount and the stability of the NP9-5 in the cell may not be enough to inhibit the virus efficiently.
Peptide inhibitors are successfully used for virus infection. Peptides mimicking HA have been reported to inhibit influenza virus replication [38,39]. These peptides bind to HA and inhibit influenza virus entry [40]. Peptides mimicking influenza virus RdRp binding sites also showed anti-influenza activity [41–43]. However, the results for the zinc finger motif peptide of M1 being used for the inhibition of influenza virus are controversial [44–46]. Zanamivir was developed from the non-inhibitory prototype sialidase inhibitor [47,48]. NP9-5 is considered a candidate for an anti-influenza virus drug once it is modified, especially for stability, although its inhibitory effect is not substantial at the moment. Protein aptamer technology mimicking natural substances was demonstrated to be useful, and the constrained peptide libraries can be used to discover candidate peptides.
Footnotes
Acknowledgements
We thank Y Kawaoka (University of Tokyo, Tokyo, Japan) for the 12-plasmid reconstitution system of influenza virus A/WSN/33 and 293T cells. We also thank RG Webster (St Jude Children's Research Hospital, Memphis, TN, USA) for the monoclonal antibody NP (5/1). This work was supported by Grants-in-aid from the Chinese Academy of Sciences (0514P51131), the National Science Foundation of China (30670090 and 30970153), the Li Ka Shing Foundation (0682P11131), RESPARI (0581P14131) and FLUINNATE (SP5B-CT-2006–044161).
The authors declare no competing interests.
