Abstract
Background:
Nine novel uracil analogues were synthesized and evaluated as inhibitors of HIV-1.
Methods:
Key structural modifications included replacement of the 6-chloro group of 1-benzyl-6-chloro-3-(3,5-dimethylbenzyl)uracil by other functional groups or N1-alkylation of 3-(3,5-dimethylbenzyl)-5-fluorouracil.
Results:
These compounds showed only micromolar potency against HIV-1 in MT-4, though two of them; 6-azido-1-benzyl-3-(3,5-dimethylbenzyl) uracil and 6-amino-1-benzyl-3-(3,5-dimethylbenzyl) uracil were highly potent (half maximal effective concentration =0.067 and 0.069 μM) and selective (selectivity index =685 and 661), respectively. Structure–activity relationships among the newly synthesized uracil analogues suggest the importance of the H-bond formed between 6-amino group of 6-amino-1-benzyl-3-(3,5-dimethylbenzyl) uracil and amide group of HIV-1 reverse transcriptase.
Conclusions:
We discovered two 6-substituted 1-benzyl-3-(3,5-dimethylbenzyl) uracils, (6-azido-1-benzyl-3-(3,5-dimethylbenzyl) uracil and 6-amino-1-benzyl-3-(3,5-dimethylbenzyl) uracil) as novel anti-HIV agents. These compounds should be further pursued for their toxicity and pharmacokinetics in vivo as well as antiviral activity against non-nucleoside reverse transcriptase inhibitor-resistant strains.
Introduction
Non-nucleoside reverse transcriptase inhibitor (NNRTI), which is not the basic structure of nucleoside, binds at an allosteric binding site that is present in the reverse transcriptase (RT) [1–3]. In 1989, Baba et al. [4] first discovered that 1-[(2-hydroxyethoxy) methyl]-6-phenylthiothymine (HEPT) had a strong anti-HIV-1 activity, though its 5′-triphosphate derivative (HEPT-TP) showed no inhibitory effect on HIV-1 transcriptase. At present, there are a variety of pharmaceutical agents that can be classified as NNRTIs, such as nevirapine (NVP), efavirenz (EFV), delavirdine (DLV) and etravirine (ETR). NNRTIs have a high level of drug safety and good adherence, however, they have a disadvantage which is a tendency to evolve drug-resistant viruses caused by mutations of their binding site in HIV-1 RT. NVP, EFV and DLV bind to similar sites of HIV RT, thus HIV-1 resistant to one of these compounds shows cross-resistance to others; by contrast, ETR appears to bind the multiple sites of RT. As a result, ETR retains the activity against NVP, EFV or DLV-resistant mutants.
Since the discovery of HEPT, related uracil derivatives have been subsequently synthesized. Interestingly, one of them, SJ-3366, has potent antiviral activity against HIV-2 as well as HIV-1 [5]. All of these compounds are 6-substituted uracil derivatives, which are comparatively difficult to synthesize in large quantities; therefore, 1,3-disubstituted uracils have been synthesized and examined for their anti-HIV-1 activity [6]. The introduction of 3-methylbenzyl group at N3-position of uracil was highly effective in inhibiting HIV-1 replication in vitro. Moreover, 3-(3,5-dimethybenzyl) uracil derivatives exhibited an excellent anti-HIV-1 activity. As for the substituent of N1 position, cyanomethyl group and benzyl group have brought about good results. Based on the estimation that the hydrogen bond accepting nitrogen of the cyanomethyl group might be related to the strong anti-HIV-1 activity, the introduction of 2-picolyl group or 4-picolyl group at N1-position of the uracil analogue has been investigated and resulted in further elevation of anti-HIV-1 activity. These results were confirmed by a computing science method, in which the docking energy of a substrate on HIV-1 RT has been calculated [7].
An attempt at introducing a methyl or iodo group at C5-position of 1-benzyl-3-(3,5-dimethybenzyl)uracil resulted in significant decrease of anti-HIV-1 activity, suggesting that the bulky group at C5-position of uracil disturbs the binding to HIV-1 RT. However, a fluorine atom is about the same van der Waals radius as a hydrogen atom, and it makes the compound metabolically and chemically stable, thus many pharmaceutical agents possess fluorine atom(s) [8]. These backgrounds prompted us to prepare 1-substituted 3-(3,5-dimethybenzyl)-5-fluorouracils and examine their anti-HIV-1 activity. In addition, we attempt, in this report, to introduce a small group at 6-position of 1-benzyl-3-(3,5-dimethybenzyl)uracil to explore the effect of 6-substitution on the uracil ring.
Methods
Preparation of 6-chloro-1-benzyl-3-(3,5-dimethylbenzyl)uracil (3a )
Synthesis of 6-substituted analogues of 1-benzyl-3-(3,5-dimethylbenzyl)uracil were accomplished from the key compound 6-chloro-1-benzyl-3-(3,5-dimethylbenzyl) uracil (
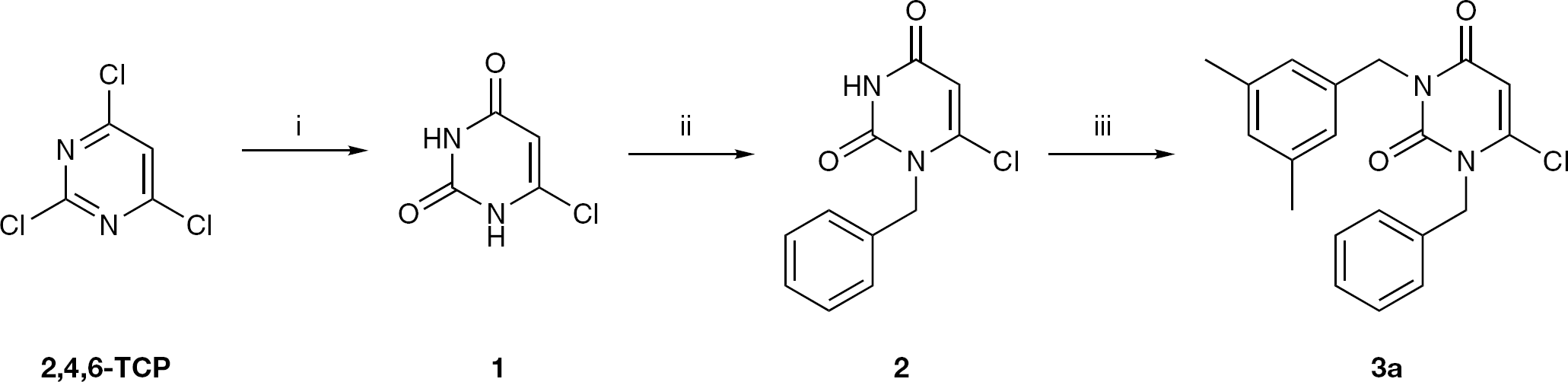
Synthesis of 6-chloro-1-benzyl-3-(3,5-dimethylbenzyl)uracil (
Synthesis of 6-substituted-1-benzyl-3-(3,5-dimethylbenzyl)uracil (3b–3f )
Briefly, displacement of the 6-chloro group of
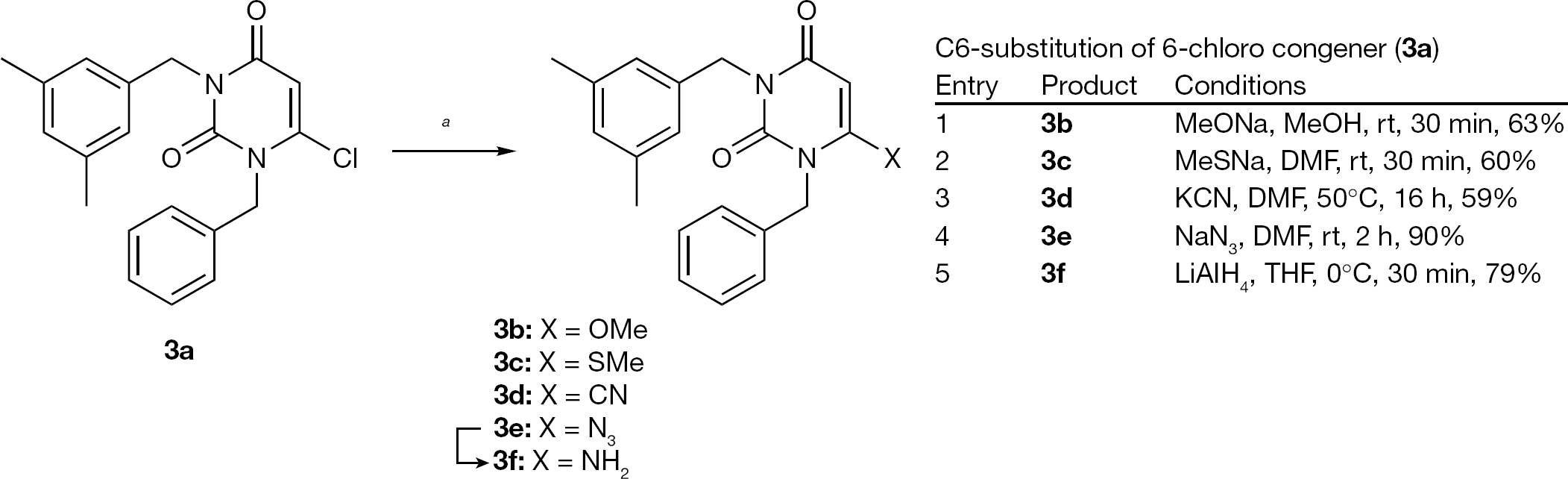
Synthesis of 6-substituted-1-benzyl-3-(3,5-dimethylbenzyl)uracil (
Preparation of 3-(3,5-dimethylbenzyl)-5-fluorouracil (9 )
Next, synthesis of 1-substituted analogues of 3-(3,5-dimethylbenzyl)-5-fluorouracil (
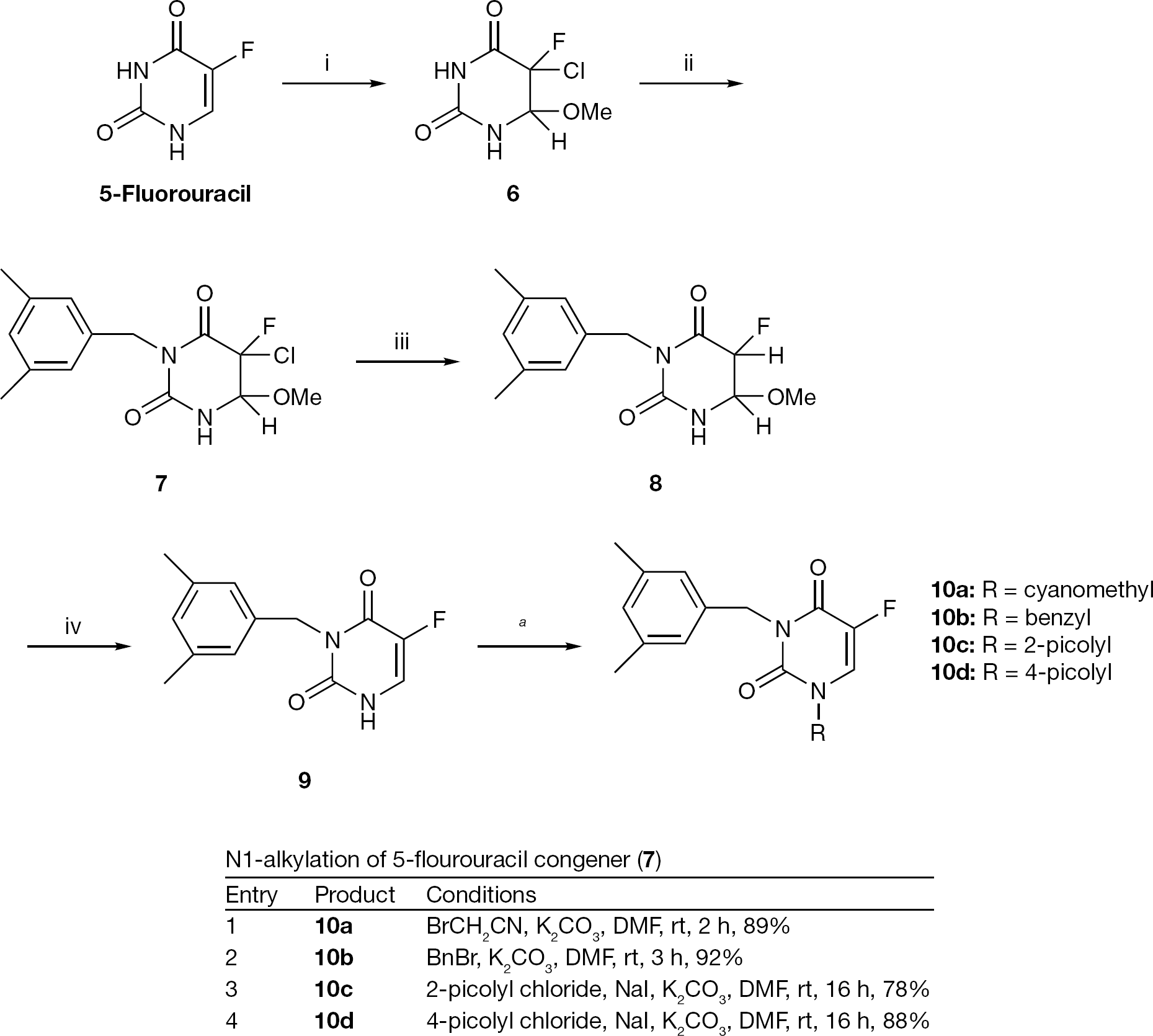
Synthesis of 3-(3,5-dimethylbenzyl)-5-fluorouracil (
N1-alkylation of key compound 9
N1-Alkylation of the key compound
Anti-HIV assay
MT-4 cells were maintained in RPMI 1640 medium supplemented with 10% heat-inactivated fetal bovine serum (FBS), 100 U/ml of penicillin G, and 0.10 g/ml of streptomycin. The IIIB strain of HIV-1 was used throughout the experiment. The virus was propagated and titrated in MT-4 cells. Virus stocks were stored at −80°C until use. The anti-HIV-1 activity of the test compounds was determined by the inhibition of virus-induced cytopathogenicity in MT-4 cells [19]. Briefly, MT-4 cells (1×105 cells/ml) were infected with HIV-1 at a multiplicity of infection (MOI) of 0.1 and were cultured in the presence of various concentrations of the test compounds. After a 4-day incubation at 37°C in 5% CO2, the number of viable cells was monitored by the 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyl-2H-tetrazolium bromide (MTT) method [20]. The cytotoxicity of the compounds was evaluated in parallel with their antiviral activity, based on the viability of mock-infected cells, as determined by the MTT method.
Materials
Instrumentation
1H NMR and 13C NMR spectra were taken with a UltrashieldTM 400 Plus FT NMR System (BRUKER, Yokohama, Japan). Chemical shifts and coupling constants (J) were given in δ and Hz, respectively. Melting points were determined on a Yanaco MP-500D (Yanaco, Tokyo, Japan). Elementary analyses were determined by a Perkin Elmer Series II CHNS/O Analyzer 2400 (Perkin Elmer, Yokohama, Japan). High-resolution mass spectrometry was performed on a APEX IV mass spectrometer (BRUKER) with electrospray ionization mass spectroscopy (ESI-MS).
Compounds
6-Chlorouracil (
1
)
A mixture of 2,4,6-Trichloropyrimidine (TCI, 13 ml, 113 mmol) and NaOH (18.1 g,0.45 mol, 4 eq) in water (185 ml) was refluxed for 1 h, then added concentrated HCl (ca. 40 ml) over a pH range 2–3, and recrystallized at 4°C. The residue was filtered off, and recrystallized from MeOH to give white crystals (12.26 g, 83.67 mmol, 74%).
Mp>300 °C. 1H NMR (DMSO-d6): δ 11.20, 12.05 (1 H, br s, NH), 5.72 (1 H, d, H5, J 1.6).
1-Benzyl-6-chlorouracil (
2
)
6-Chlorouracil (
HRMS (ESI) Calcd for C11H9ClN2NaO2 [M+Na]+: 259.0245. Found 259.0249. Mp 164.6–164.9 °C. 1H-NMR (DMSO-d6): δ 11.75 (1 H, br s, NH), 7.26–7.40 (5 H, m, -CH2C6
1-Benzyl-6-chloro-3-(3,5-dimethylbenzyl) uracil (
3a
)
A solution of compound
1- Benzyl-3-(3,5-dimethylbenzyl)-6-methoxyuracil (3b )
A mixture of compound
HRMS (ESI) Calcd for C21H22N2NaO3 [M+Na]+: 373.1523. Found 373.1535. Mp 93.3–95.7 °C. 1H NMR (CDCl3): δ 6.84–7.37 (8 H, m, -CH2C6
1-Benzyl-3-(3,5-dimethylbenzyl)-6-thiomethoxyuracil (3c )
A compound
HRMS (ESI) Calcd for C20H22N2NaO2S [M+Na]+: 389.1294. Found 389.1305. Mp 119.0–120.4 °C. 1H NMR (CDCl3): δ 6.89–7.38 (8 H, m, -CH2C6
1- Benzyl-6-cyano-3-(3,5-dimethylbenzyl) uracil (
3d
)
Compound
HRMS (ESI) Calcd for C21H19N3NaO2 [M+Na]+: 368.1370. Found 386.1384. IR (Nujol) cm−1: 2231. Mp 158.1–158.6 °C. 1H NMR (CDCl3): δ 7.08–7.68 (8 H, m, -CH2C6H3, -CH2C6
6-Azido-1-benzyl-3-(3,5-dimethylbenzyl) uracil (
3e
)
A mixture of compound
HRMS (ESI) Calcd for C20H19N5NaO2 [M+Na]+: 384.1431. Found 386.1439. IR (Nujol) cm−1: 2133. 1H NMR (CDCl3): δ 6.89–7.37 (8 H, m, -CH2C6
6-Amino-1-benzyl-3-(3,5-dimethylbenzyl) uracil (
3f
)
Compound
HRMS (ESI) Calcd for C20H21N3NaO2 [M+Na]+: 358.1526. Found 358.1526. 1H NMR (CDCl3): δ 6.86–7.43 (8 H, m, -CH2C6
5-Chloro-5,6-dihydro-5-fluoro-6-methoxyuracil (
6
)
5-Fluorouracil (5FU, Sigma-Aldrich, Tokyo, Japan; 6.50 g, 50.0 mmol) was dissolved in MeOH (350.0 ml) and N-Chlorosuccinimide (Sigma-Aldrich, 13.30 g, 100.0 mmol) was added to the solution, and then stirred for overnight at 50°C. The mixture was evaporated, and was recrystallized from 50% EtOH in H2O to give white crystals of
Mp 213.5–215.3 °C. 1H NMR (DMSO-d6): δ 11.19 (1 H, br s, 3-NH), 9.21 (1 H, br s, 1-NH), 5.00 (1 H, d, H6, J 4.8), 3.38 (3 H, d, -OMe J 1.2).
3-(3,5-Dimethylbenzyl)-5-fluorouracil (
9
)
A solution of compound
HRMS (ESI) Calcd for C13H13FN2NaO2 [M+Na]+: 271.0853. Found 271.0844. Mp 163.0–167.1 °C. UV: max 268.0nm (MeOH) max 298.7nm (NaOH) 1H NMR (CDCl3): δ 9.27 (1 H, br s, 1-NH), 7.17 (1 H, s, H6 of uracil), 7.04 (2 H, s, H2+H6 of -C6H3), 6.92 (1 H, s, H4 of -C6H3), 5.04 (2 H, s, -C
1-Cyanomethyl-3-(3,5-dimethylbenzyl)-5-fluorouracil (
10a
)
Compound
HRMS (ESI) Calcd for C15H14FN3NaO2 [M+Na]+: 310.0962. Found 310.0950. Mp 177.1–178.6 °C. 1H NMR (CDCl3): δ 7.30 (1 H, d, H6 of uracil, J 6.4), 7.08 (2 H, s, H2+H6 of -C6H3), 6.93 (1 H, s, H4 of -C6H3), 5.07, (2 H, s, -CH2), 4.66 (2 H, s, -CH2), 2.29 (3 H, s, -CH3), 2.29 (3 H, s, -CH3).
1-Benzyl-3-(3,5-dimethylbenzyl)-5-fluorouracil (
10b
)
Compound
HRMS (ESI) Calcd for C20H19FN2NaO2 [M+Na]+: 361.1323. Found 361.1320. Mp 103.3–104.8 °C. 1H NMR (DMSO-d6): δ 8.36 (1 H, d, H6 of uracil, J 6.4), 7.30–7.38 (5 H, m, -C6H4), 6.86 (1 H, s, H4 of -C6H3), 6.83 (2 H, s, H2+H6 of -C6H3), 4.92, (2 H, s, -CH2), 4.91 (2 H, s, -CH2), 2.20 (6 H, s, -CH3×2).
3-(3,5-Dimethylbenzyl)-1-(2-picolyl)-5-fluorouracil (
10c
)
Compound
HRMS (ESI) Calcd for C19H18FN3NaO2 [M+Na]+: 362.1275. Found 362.1265. Mp 114.3–115.5 °C. 1H NMR (DMSO-d6): δ 8.52 (1 H, ddd, H6 of 2-picolyl, J 4.8, 1.6 and 0.8), 8.34 (1 H, d, H6 of uracil, J 6.4), 7.80 (1 H, ddd, H4 of 2-picolyl, J 7.6, 7.6 and 1.6),7.37 (1H, d, H3 of 2-picolyl, J 7.6) 7.32 (1H, ddd, H5 of 2-picolyl, J 7.6, 4.8 and 0.8) 6.87 (1 H, s, H4 of -C6H3), 6.82 (2 H, s, H2+H6 of -C6H3), 5.04, (2 H, s, -CH2), 4.92 (2 H, s, -CH2), 2.21 (6 H, s, -CH3×2).
3-(3,5-Dimethylbenzyl)-1-(4-picolyl)-5-fluorouracil (
10d
)
Compound
HRMS (ESI) Calcd for C19H18FN3NaO2 [M+Na]+: 362.1275. Found 362.1277. 1H NMR (DMSO-d6): δ 8.60 (1 H, dd, H2+H6 of 4-picolyl, J 4.4 and 1.6), 8.43 (1 H, d, H6 of uracil, J 6.4), 7.36 (2 H, dd, H3+H5 of 4-picolyl, J 4.4 and 1.6), 6.93 (1 H, s, H4 of -C6H3), 6.91 (2 H, s, H2+H6 of -C6H3), 5.02, (2 H, s, -CH2), 4.98 (2 H, s, -CH2), 2.27 (6 H, s, -CH3×2).
Results
Structure-activity relationship
Antiviral activity of the 6-substituted 1-benzyl-3-(3,5-dimethylbenzyl)uracils (
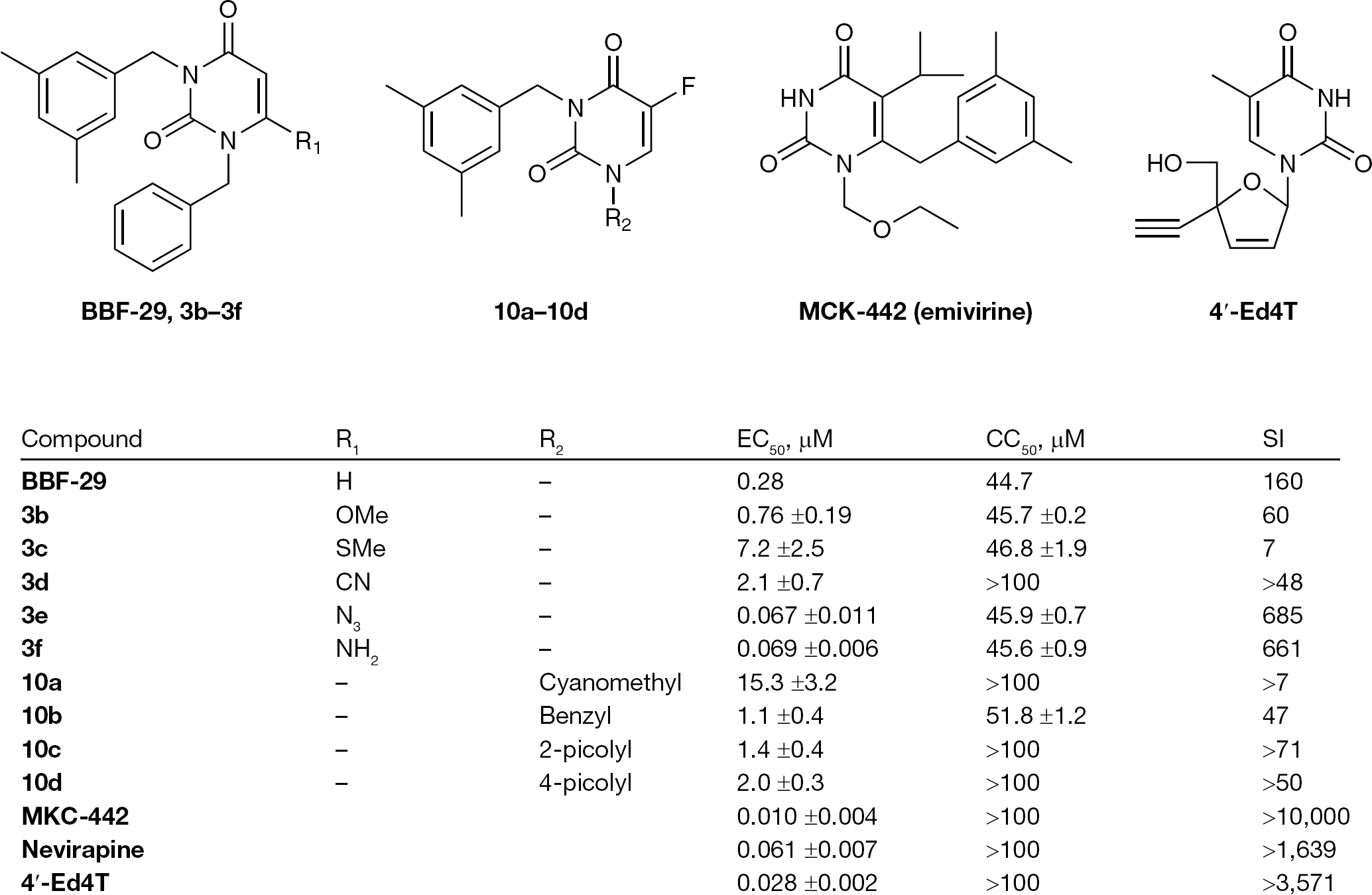
Antiviral activity of 1,3-disubstituted uracils against HIV-1
5-Fluorouracil analogues (
Discussion
We have previously demonstrated that some novel 1,3-disubstituted uracils selectively inhibit HIV-1 replication in cell cultures [6,7]. In this report, hydrogen bonding interaction (H-bond) between ligand and amide group of Lys 101 residue as well as the hydrophobic interaction is important for the binding of uracil derivatives to HIV-1 RT. Strong anti-HIV-1 activity of the 6-amino derivative (
In conclusion, we discovered two 6-substituted 1-benzyl-3-(3,5-dimethylbenzyl) uracils as novel anti-HIV agents. These compounds should be further pursued for their toxicity and pharmacokinetics in vivo as well as antiviral activity against NNRTI-resistant strains.
Footnotes
All authors are inventors of the patent currently submitted to Japan Patent Office.
