Abstract
Background:
Enfuvirtide (T-20), a C-terminal heptad repeat (C-HR)-derived peptide of HIV-1 glycoprotein, gp41, effectively suppresses HIV-1 replication through a putative mechanism that involves it acting as a decoy and binding to the N-terminal heptad repeat (N-HR) of the virus. In this study, we address whether the anti-HIV-1 activity of T-20 is antagonized by a variety of N-HR-derived peptides.
Methods:
Multinuclear activation of galactosidase indicator assays were used to evaluate T-20 activity in the presence of N-HR-derived peptides. The gp41-derived peptides were chemically synthesized.
Results:
We demonstrate additive anti-HIV activity when T-20 is used in combination with N-HR-derived peptides that do not have a putative binding region for the tryptophan-rich domain in T-20. The presence of a deep pocket-forming region in the N-HR-derived peptides enhanced their anti-HIV-1 activity, but had little effect on the activity of T-20.
Conclusions:
These results indicate that T-20-based antiviral therapies can be combined with N-HR-derived peptides.
Introduction
Two envelope glycoproteins of HIV-1, gp120 and gp41, play an important role in HIV-1 entry into the host cells. The surface subunit gp120 recognizes receptors CD4 and CXCR4/CCR5, which are expressed on the target cell surface. After binding to these receptors, the transmembrane subunit gp41 (Figure 1A) mediates fusion of HIV-1 to the host cells. Viral membrane-anchored gp41 forms a 6-helix bundle composed of an α-helical trimer of an N-terminal heptad repeat (N-HR) with three C-terminal heptad repeats (C-HR) folded onto the N-HR in an anti-parallel orientation [1]. Formation of the 6-helix bundle enables fusion between the viral and the host cell membranes. Therefore, peptides derived from either HIV-1 gp41 N- or C-HR sequences have been thought to inhibit HIV-1 fusion by preventing formation of the 6-helix bundle, and by acting as a dominant negative or a decoy [2,3].
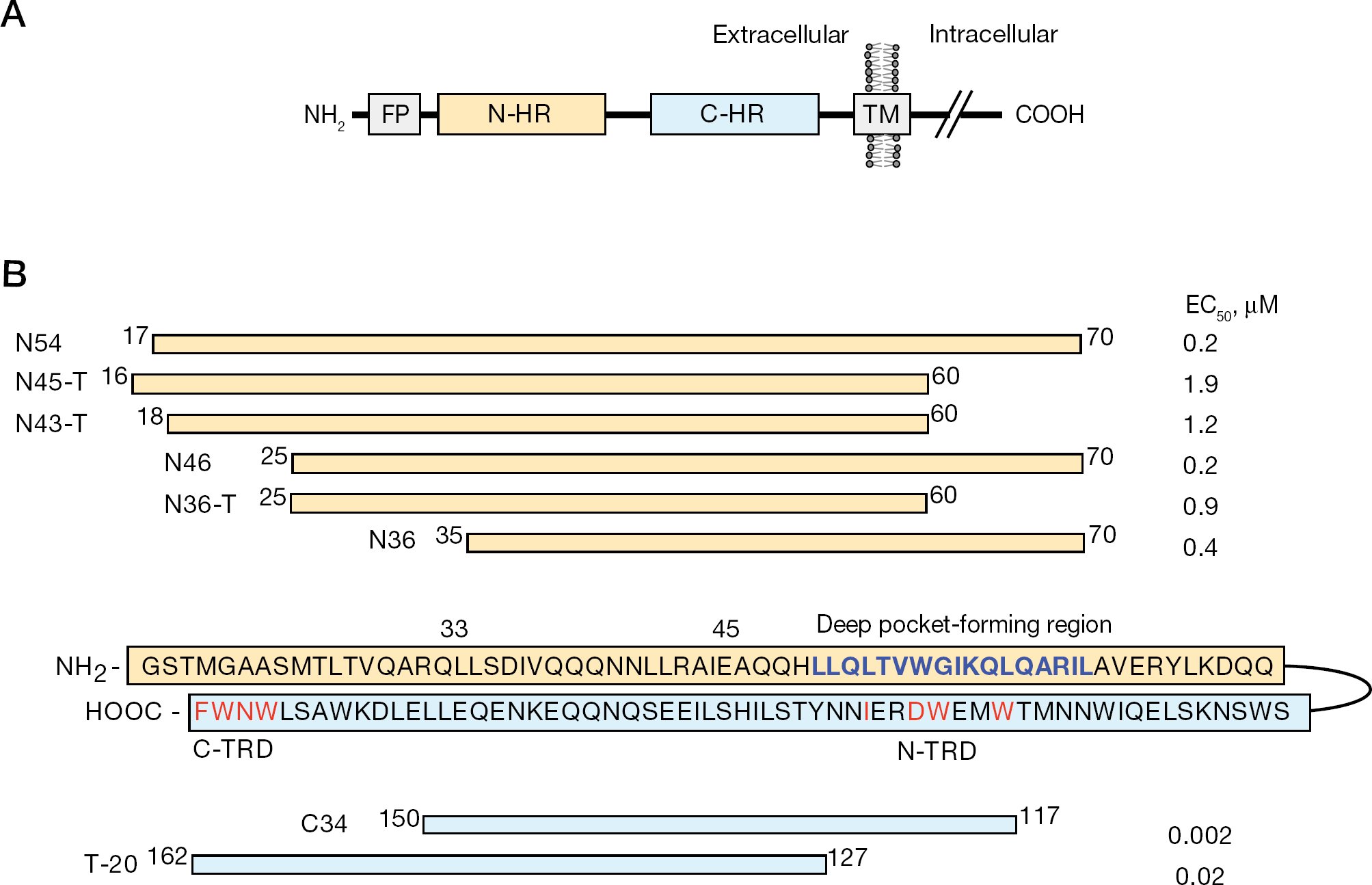
Schematic view of HIV-1 gp41 and the amino acid sequence of gp41-derived peptides, in addition to the inhibitory effect of the N-HR-derived peptides
C34 is a representative C-HR-derived peptide that inhibits HIV-1 fusion to host cells. It contains four key amino acids, W117, W120, D121 and I124, collectively designated as the N-terminal tryptophan-rich domain (N-TRD), which dock into a hydrophobic pocket (termed the ‘deep pocket’) formed by the N-HR trimer (Figure 1B) [4]. Enfuvirtide (T-20), which lacks 10 amino acids in the N-terminus that are present in C34, also contains four key amino acids, W159, N160, W161 and F162, which are collectively designated as the C-terminal tryptophan-rich domain (C-TRD). Both TRDs have been demonstrated to be important for inhibition of HIV-1 fusion [5,–10]. In a previous study, we used enzyme linked immunosorbent assay (ELISA) to demonstrate that T-20 binds weakly to the N-HR, even in the case of N-HR containing the entire T-20 binding region with amino acid positions from 18 to 73, whereas C34 binds strongly [11]. Our previous findings are consistent with other observations that T-20 may only transiently interact with N-HR and, instead, it inhibits HIV-1 fusion by targeting multiple sites in both gp120 and gp41 [12]. However, T20 has been shown to interact with an artificial 5-helix complex comprised of a linker-connected structure containing three N-HRs and two C-HRs, although the interaction was much weaker than that between C34 and the complex [13]. Hence, the C-TRD of T-20 may have a distinct role during the inhibition of HIV-1 fusion, other than binding to the N-HR. A mechanism that involves membrane association of T-20 via C-TRD has been previously proposed for the fusion inhibition [9,12,14].
In contrast to the results from extensive biochemical analyses, the results accumulated from virological experiments support the hypothesis that T-20 interacts with the N-HR region. Hence, the majority of T-20-resistant HIV-1 variants acquire mutations in the N-HR, especially from L33 to L45, which is the putative interactive site of T-20 (Figure 1B) [15,,,–23]. Moreover, the inhibitory effect of T-20 seems to depend on the specific sequence of the N-HR [24].
Therefore, the avidity of T-20 for N-HR remains unclear. We hypothesized that any potential interactions of T-20 with N-HR-derived peptides would attenuate its anti-HIV-1 activity by adsorption into the added N-HR-derived peptides, which form an inactive 6-helix bundle. In this study, using synthesized various N-HR-derived peptides with or without the putative T-20 binding region, we determined the anti-HIV-1 activity of T-20 and C34 in the presence of the synthesized peptides. Our data identify additive combinations of T-20 and N-HR-derived peptides, which may be valuable for treating HIV-1.
Methods
Cells and viruses
293T cells were grown in Dulbecco's modified Eagle medium (DMEM) supplemented with 10% fetal calf serum (FCS), 2 mM l-glutamine, 100 U/ml penicillin, and 50 μg/ml streptomycin. HeLa-CD4/CCR5-LTR/β-gal cells [25] (provided by Julie Overbaugh through the NIH AIDS Research and Reference Reagent Program, Division of AIDS, NIAID, Germantown, MD, USA) were maintained in DMEM supplemented with 10% FCS, 200 μg/ml hygromycin B, 10 μg/ml puromycin and 200 μg/ml geneticin. Wild-type HIV-1 was harvested from the supernatant of an HIV-1 molecular clone, pNL4–3 transfected 293T cells and stored at −80°C.
Determination of drug susceptibility
Anti-HIV activity of inhibitors was determined using multinuclear activation of a galactosidase indicator (MAGI) assay as described previously [19,24,26,–30]. Briefly, HeLa-CD4-LTR-β-gal cells were inoculated with HIV-1NL4–3 (60 MAGI units, generating 60 blue cells after 48 h of incubation) and cultured in the presence of various concentrations of peptides; 48 h after initial viral exposure, we counted all the blue cells that were stained with X-Gal (5-bromo-4-chloro-3-indolyl-β-D-galactopyranoside). We assessed the antiviral activity of peptides based on the effective concentration that blocked HIV-1 infection by 50% (EC50).
Statistical analysis
Dunnett's multiple comparisons test was used to evaluate whether the EC50 values of T-20 were influenced in the presence of N-HR peptides. P-values <0.01 were considered statistically significant.
Results
Antiviral activity of the N-HR-derived peptides
Six N-HR-derived peptides, N36, N36-T, N46, N43-T, N45-T and N54, were synthesized with standard Fmoc-based solid-phase techniques (Figure 1B) [31]. We first examined whether these N-HR-derived peptides exerted anti-HIV-1 activity with the MAGI assay [28]. We found that N36, N46 and N54, which include the entire deep pocket-forming region corresponding to N-TRD, were more active than N36-T, N43-T and N45-T, which include only part of the region (Figure 1B). However, addition of the region of N-HR that is believed to interact with C-TRD to the N-HR derived peptides had little influence on the activity of the N-HR-derived peptides. These results suggest that the addition of a deep pocket-forming region to N-HR-derived peptides provides enhanced antiviral activity of the peptides.
Combination of T20 and N-HR-derived peptides with a deep pocket-forming region
We also tested whether the anti-HIV-1 activity of T20 is attenuated when used in combination with the N-HR-derived peptides (Figure 2). Specifically, we examined the antiviral activity of T-20 or C34 at concentrations ranging from 0.1 to 100 nM, in the presence or absence of each N-HR-derived peptide (concentration at 30–40% inhibition of HIV replication), which provided the apparent effect of the combination among other concentrations tested. We found an additive effect in combination of T-20 and N36, N36-T or N46, which lack the interactive site for C-TRD in T-20 (Figure 2A). By contrast, the activity of C34 was decreased to the level of C34 alone in the presence of N36, N36T or N46, respectively, which contains the N-TRD corresponding region (Figure 2C). Moreover, C34-mediated inhibition of the fusion was attenuated especially by N46 and N54 at 10 nM C34 (P<0.01; Figure 2C and 2D), indicating that a deep pocket region is important for a reduction in the C34 activity, possibly by a direct interaction (adsorption) with N-HR. Although N36-T and N46 include part of the C-TRD binding region, the avidity of the T-20 complex with these peptides seemed to be vulnerable in the fusion between HIV-1 and the host cells. Therefore, we further examined the N-HR-derived peptides that contain an entire interactive region of the C-TRD.
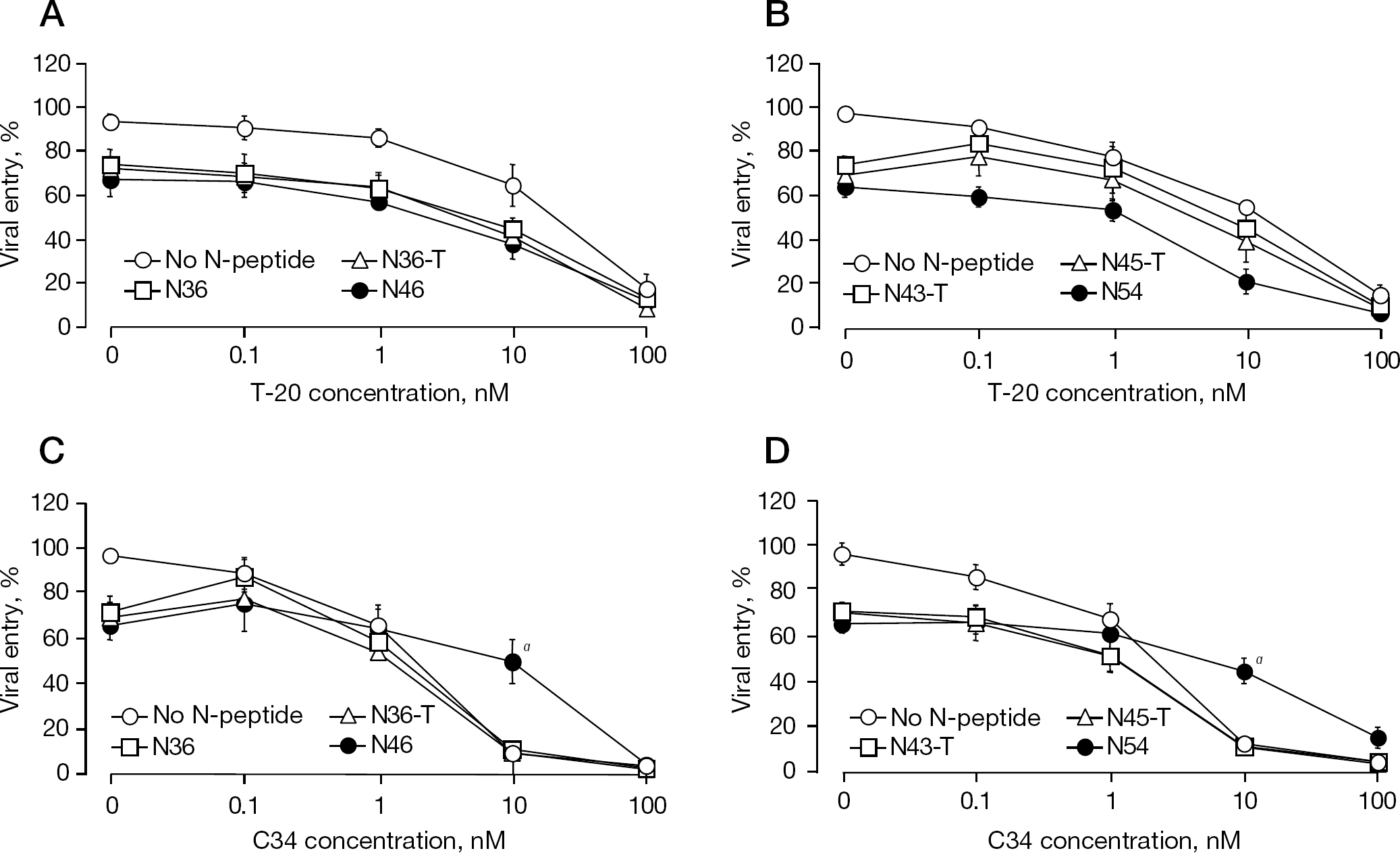
Inhibition curves of the combinations of T-20 or C34 with the individual N-HR-derived peptides determined with the MAGI assay
Role of C-TRD corresponding region in the N-HR-derived peptides
The combination studies of T-20 revealed that the antiviral activity of N43-T or N45-T, but not of N54, which includes both the N- and C-TRD corresponding regions, was reduced to levels comparable to T-20 alone, indicating antagonism (Figures 1B and 2B). However, the antiviral activity of C34 was significantly antagonized by either N46 or N54 at 10 nM (Figure 2C and 2D). These results indicate that putative interactive region in the N-HR-derived peptides for C-TRD plays small role in T-20 binding.
Discussion
In this study, we address possible combination of T-20 with the N-HR-derived peptides through their influence on T-20 activity. To date, the mechanisms of T-20 action have been extensively examined in protein–protein in vitro binding but rarely in actual HIV-1 entry. Previously, Liu et al. [12] also tried to reveal a role of the C-TRD in T-20 activity by T20-ANAA, which is like traditional T-20 but with a substitution from WNWF to ANAA in the C-TRD. T20-ANAA exhibited 450fold less anti-HIV-1 activity than T-20. However, it was shown that the moderate levels of T20-ANAA avidities toward N36-F10 [12] and the 5-helix bundle [13] were maintained, suggesting that the WNWF motif is unnecessary for binding to N-HR, but is important for potent anti-HIV-1 activity of T-20. Indeed, the overall reduction in T-20 activity by the N-HR peptides with the C-TRD corresponding region was moderate. The anti-HIV-1 activity of T-20 was not blocked by combination with N54, which includes both the C-TRD binding region and the deep pocket-forming region. The latter region may enhance the activity of the N-HR peptides and restore the antagonistic effect on T-20.
We clearly demonstrated that unlike the C-TRD, which only weakly interacts with the N-HR, N-TRD, is definitively required for the interaction with the N-HR, thus playing a distinct role in HIV-1 fusion. Recently, it was reported that an N-HR-derived peptide lacking a binding site for the TRD of T-20, (CCIZN17)3, which is covalently stabilized by three interchain disulfides, can be effectively combined with T-20 [32]. To design sustained anti-HIV-1 activity of N-HR-derived peptides in combination with T-20, the amino acid sequence at positions 60–70 that forms a deep pocket should be included in the peptide and the N-terminal sequence of N-HR should be excluded. Our observations should be useful for further development of potent and N-HR-derived peptides that could be effectively combined with T-20.
Footnotes
Acknowledgements
This work was supported in part by a grant for Promotion of AIDS Research from the Ministry of Health, Labour and Welfare of Japan (awarded to ENK and MM), and a grant from the Ministry of Education, Culture, Sports, Science and Technology of Japan (awarded to ENK). SGS was supported in part by National Institutes of Health grants AI076119, AI087489, AI074389 and AI079801.
The authors declare no competing interests.
