Abstract
Background:
Epigallocatechin gallate (EGCG), the most abundant catechin in green tea, has been reported to inhibit HIV-1 replication prior to its integration into host DNA via various proposed mechanisms; however, the specific main target(s) of EGCG remain unclear. In this study, we investigated a number of these proposed detailed mechanism(s) using a cell-based model.
Methods:
Multinuclear activation of galactosidase indicator assays were used for all experiments, including examination of the time of addition and the synergisms with a nucleoside reverse transcriptase inhibitor, 3′-azido-3′-deoxythymidine (AZT).
Results:
The experiments revealed that EGCG suppressed both HIV-1IIIB and HIV-2EHO infection in HeLa-CD4-LTR-β-gal cells, with relatively low 50% effective concentrations of 1.6 and 2.0 μM, respectively. The inhibitory profile of EGCG generated using a time-of-addition assay was identical to that of a non-nucleoside reverse transcriptase inhibitor (NNRTI), MKC-442. Furthermore, synergistic inhibition was observed in EGCG with AZT.
Conclusions:
Based on our findings, EGCG appears to act mainly as an allosteric reverse transcriptase inhibitor with mechanisms different from those of currently approved NNRTIs that directly interact with the NNRTI binding pocket. Thus, EGCG is a good candidate for use as an additional or supportive anti-HIV agent derived from natural plants.
Introduction
Combination antiretroviral therapy has significantly reduced the mortality of patients infected with HIV-1 in recent years and has, importantly, improved their quality of life. In addition to classical inhibitors against reverse transcriptases and proteases, a number of new agents, including CCR5 antagonists [1] and integrase inhibitors [2], strongly suppress the replication of drug-resistant HIV-1 variants when they are combined with optimized background therapy. Therefore, various combinations of such agents with distinct antiviral mechanisms are promising for efficient anti-HIV-1 therapy.
Catechin derivatives from natural food and medicinal plants have been demonstrated to exert various antiviral activities [3–5]. For the inhibition of HIV-1 replication, several possible mechanisms of action of (−)-epigallocatechin gallate (EGCG) have been proposed. For instance, EGCG has been shown to interfere mainly with the HIV-1 viral attachment step by various modes, such as blocking the binding of the HIV-1 glycoprotein 120 to the CD4 molecule on T-cells [6,7], disassembling the HIV-1 virion directly [8] and degrading a peptide fraction termed the semen-derived enhancer of virus infection, which reportedly enhances HIV-1 transmission [9]. Alternatively, Nakane and Ono [10] reported that EGCG inhibits the enzymatic activity of wild-type HIV-1 reverse transcriptase (RT) in an allosteric manner in vitro. Fortunately, these independently performed studies have provided potential mechanisms of the action of EGCG. However, these experiments failed to reveal which inhibitory effects are dominant and/or which major mechanisms of action are at play. Therefore, in the present study, we further investigated the mechanisms of action of both EGCG and the closely related (−)-epigallocatechin (EGC; Figure 1A) on HIV-1 and HIV-2 in a cell-based assay.
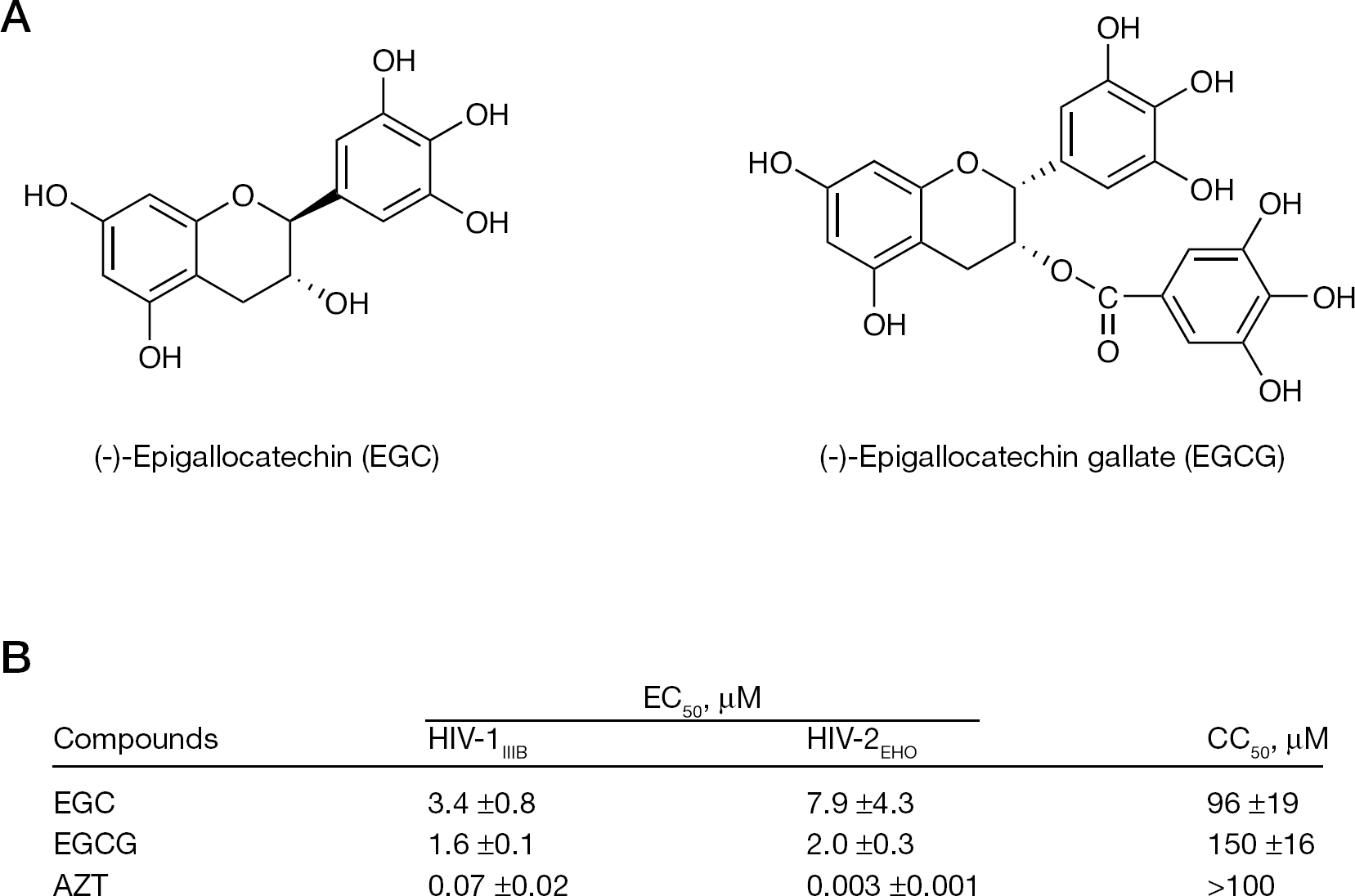
Structures of EGC and EGCG, and the anti-HIV activity of these compounds
Methods
Cells and reagents
HeLa-CD4-LTR-β-gal cells were kindly provided by M Emerman, through the AIDS Research and Reference Reagent Program, Division of AIDS, NIAID, National Institutes of Health (Bethesda, MD, USA). EGC and EGCG were purchased from Funakoshi Co., Ltd. (Tokyo, Japan). Dextran sulfate (DS-5000) and 3′-azido-3′-deoxythymidine (AZT) were purchased from Sigma (St Louis, MO, USA). AMD3100 and MKC-442 were gifts from S Shigata of Fukushima Medical University School of Medicine (Fukushima, Japan). 1-(4,5-Dimethylthiazol-2yl)-3,5-dephenylformazan (MTT) was purchased from Sigma. HIV-1IIIB and HIV-2EHO viruses were used for the anti-HIV activity assays as described previously [11].
Assay for anti-HIV activity
The effects of the antiviral agents were determined with a multinuclear activation of galactosidase indicator (MAGI) assay as described previously [12]. Briefly, HeLa-CD4-LTR-β-gal cells were seeded in 96-well flat tissue culture plates. After overnight incubation of the plates at 37°C, the cells were inoculated with HIV-1IIIB or HIV-2EHO viruses and further cultured in the presence of various concentrations of compounds as indicated. After 48 h of viral exposure, all the blue cells stained with X-gal were counted. The activity of each tested compound was determined as the concentration that blocked HIV replication by 50% (EC50). The cytotoxicity of each compound was measured using an MTT colorimetric assay as described previously [12]; the 50% cytotoxicity concentration (CC50) was defined as the concentration that reduced cell viability by 50%.
Time-of-addition assay
A time-of-addition experiment was carried out with HeLa-CD4-LTR-β-gal cells as described previously [11,12]. Briefly, the cells were infected with HIV-1IIIB virus and the tested compounds were subsequently added onto the cells at different times after viral infection. The blue cell counts were determined microscopically at 48 h after viral infection.
Evaluation of EGCG synergism with AZT
The synergistic anti-HIV activity of EGCG and AZT was evaluated with a MAGI assay involving HIV-1IIIB infection as described above. The EC50 was used to further calculate the fractional inhibitory concentration (FIC), according to the previously reported formula [13]. Specifically, the FIC of drug A is equal to the ratio of the EC50 of drug A in combination with drug B to the EC50 of drug A when it is used alone. Furthermore, the combined FIC index is the sum of the FICs of each drug. The interaction was defined as synergistic if the FIC index was less than 1, additive if equal to 1, and antagonistic if more than 1.
Results
EGC and EGCG suppress both HIV-1 and HIV-2 infection
We evaluated the antiviral activity of the catechins against HIV-1 and HIV-2 with MAGI assays. We found that EGC and EGCG suppressed infection of both HIV-2EHO and HIV-1IIIB. The inhibitory activities of EGC and EGCG were comparable against HIV-1 and HIV-2 infection (Figure 1B). Thus, we further investigated the possible antiviral mechanism with the MAGI cells. In addition, we also evaluated other catechin derivatives, such as (−)-catechin gallate, (−)-epicatechin and (+)-catechin. However, these derivatives did not show any anti-HIV activity up to 100 μM, the maximum concentration tested (SL et al., data not shown). These results are consistent with those in previous reports [6–10] that showed that EGCG inhibits HIV replication.
EGCG inhibition profile is identical to that of MKC-442
To address the mechanism(s) of action of EGCG in a cell-based assay, we performed a time-of-addition experiment. Using this assay, we compared the profile of the antiviral activity of EGCG with those of other representative HIV-1 inhibitors, including DS5000 (an entry inhibitor), AMD3100 (a CXCR4 antagonist), AZT (a nucleoside reverse transcriptase inhibitor [NRTI]) and MKC-442 (a non-nucleoside reverse transcriptase inhibitor [NNRTI]). The profile of the EGCG inhibition curve appeared identical to that of MKC-442, but distinct from those of the other tested inhibitors (Figure 2A). Thus, based on our results from a time-of-addition assay, the major target of EGCG for HIV inhibition appears to be RT function. This finding is consistent with the report by Nakane and Ono [10], which showed that EGCG inhibited HIV-1 RT activity as an allosteric inhibitor in an enzymatic assay in vitro.
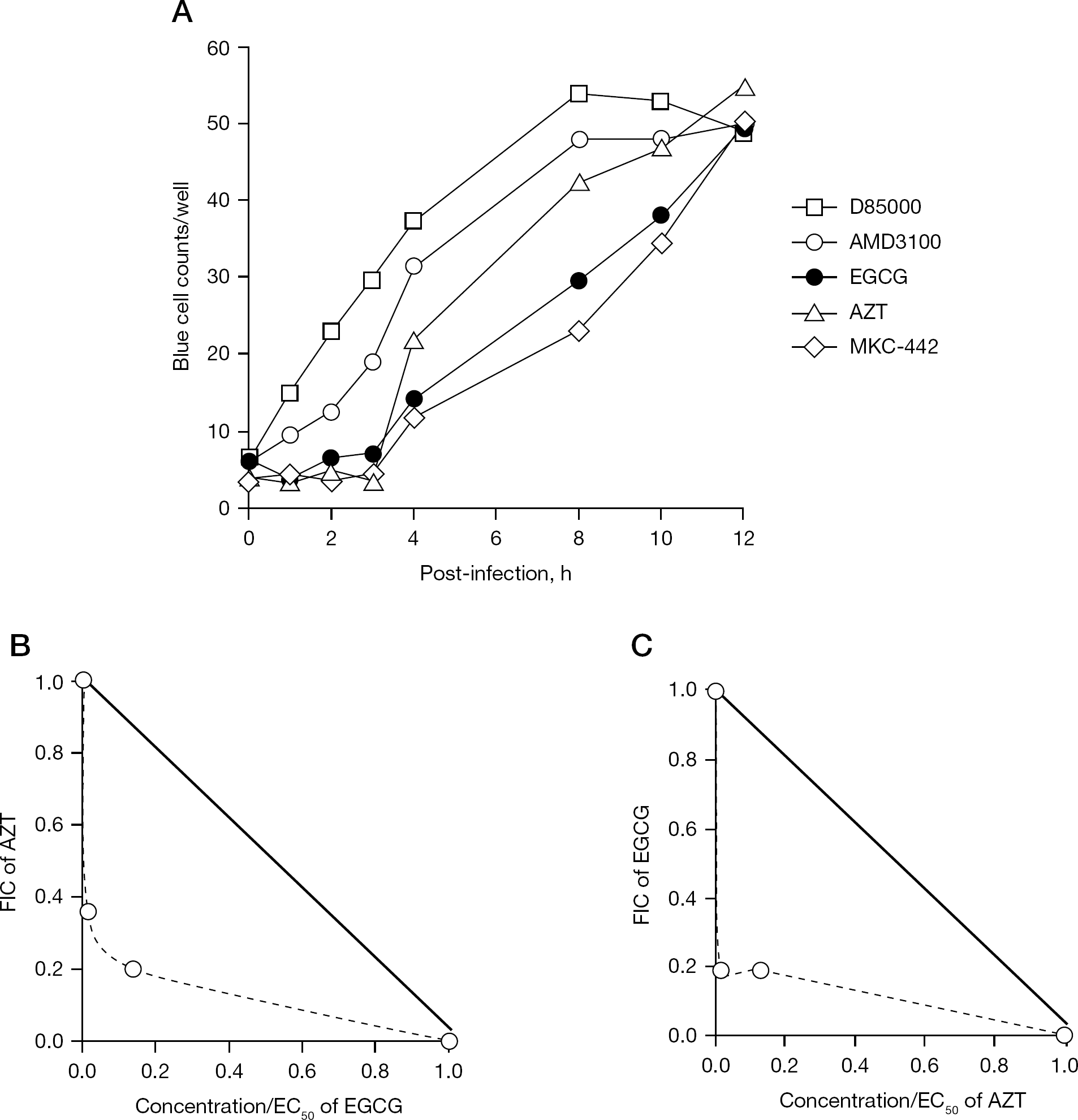
Time-of-addition analysis and synergy analysis
EGCG synergistically acts with AZT
Since the mechanism of action of EGCG is known to be distinct from that of AZT, we further evaluated the interaction between EGCG and AZT in the MAGI assay with HIV-1IIIB infection. The combination of EGCG and AZT inhibited HIV-1IIIB replication at the indicated concentrations (Figure 2B). The actual EC50 values of EGCG in combination with 0.1 or 1 nM of AZT were decreased compared with that of EGCG alone, leading to the FIC of EGCG to be less than 0.2. Furthermore, the FICs of AZT in combination with 0.1 or 1 μM of EGCG were 0.36 and 0.2, respectively. The combined FIC index was in the range of 0.39–0.55, which indicates that the interaction between EGCG and AZT was synergistic. Therefore, EGCG apparently exerts synergistic anti-HIV effects with NRTIs.
Discussion
EGCG is the most abundant catechin in green tea and has been shown to exert various anti-oxidative, anti-inflammatory and anti-tumourigenic effects [14–16]. In our present study, we revealed that EGCG mainly acted as an allosteric inhibitor of RT, and exerted synergistic effects when combined with AZT. The representative allosteric RT inhibitors, NNRTIs, bind to the NNRTI binding pocket of HIV-1 RT and suppress HIV-1 RT, but not HIV-2 RT. This distinction is likely because a certain binding pocket for NNRTIs that is found in HIV-1 is not apparent in HIV-2 RT. However, in our present study, EGCG also inhibited HIV-2 replication, which indicates that EGCG has a distinct mechanism(s) of action from those of currently approved NNRTIs.
Yamaguchi et al. [8] previously found that pre-incubation of HIV-1 particles with EGCG decreased the HIV-1 infectivity in a MAGI assay, which suggests that EGCG may act directly on and/or inactivate HIV-1 virions without the requirement of cellular metabolic activation. However, efavirenz, a tight-binding NNRTI, penetrates through the viral membrane and capsid, binds HIV-1 RT tightly within the virions and inhibits RT activity [17]. Thus, the destructive effects of EGCG observed previously by Yamaguchi et al. [8] might be provided by a tight-binding characteristic of EGCG, rather than by direct destruction of the virions. Therefore, we also investigated the activities of EGCG on several representative NNRTI-resistant HIV-1 variants (HIV-1 with mutations, such as K103N, Y181C and V189I) in MAGI assays. We found that EGCG inhibited the infection of all of these NNRTI-resistant viruses, as well as wild-type HIV-1 viruses (SL et al., data not shown). This finding further strengthens the idea that EGCG and currently approved NNRTIs exhibit distinct mechanisms of action. Although the anti-HIV activity of EGCG may be integrated from an interaction with several steps in HIV replication as described previously [6–10], we propose that it is mainly provided by allosteric inhibition of RT.
It has been reported that, to reach a mean plasma level of 1.0 μM EGCG, which is close to the EC50 of EGCG (1.6–2.0 μM) found in our study, it is necessary to orally consume seven cups of green tea containing 118 mg EGCG per cup [7]. Hence, the antiviral activity of EGCG can be achieved in vivo at the physiological concentrations. Preliminary clinical trials have demonstrated that EGCG exhibits chemoprevention efficacy on human prostate cancer, without causing significant adverse effects (oral administration of 311 mg/day for 1 year) [18]. Collectively, these data indicate that EGCG may be safe and effective for anti-HIV therapy in vivo. However, further enhancement of EGCG oral bioavailability should be needed for complete suppression of HIV replication, which may reduce risk of the resistant variants emergence, by certain chemical modification and/or modification of administration.
In conclusion, our findings indicate that the major mechanism of action of EGCG is the allosteric inhibition of HIV RT. With its distinct mechanisms of action and inhibitory effects on drug-resistant HIV-1 variants, EGCG, which is derived from natural plants, represents a potential alternative and/or supportive medicinal agent, particularly as part of an optimized combination treatment regimen. Also of note, EGCG has the practical advantages of causing less toxicity and having a relatively low cost.
Footnotes
Acknowledgements
This work was supported in part by a Grants-in-Aid for Special Educational grant from the Ministry of Education, Culture, Sports, Science, and Technology, and by the Scientific Research Expenses from the Ministry of Health, Labour and Welfare, Japan. SL was supported by the Japanese AIDS Foundation, Tokyo, Japan.
The authors declare no competing interests.
