Abstract
Pharmacodynamic investigation of antiviral compounds studies the relationship between drug exposure and the virological response. These studies are usually performed in animals and, eventually, in humans and are a very expensive proposition. To find a more efficient and less expensive method for determining pharmacodynamics of antiviral and antimicrobial compounds, the hollow fibre infection model (HFIM) system was developed to perform pharmacodynamic studies in vitro. This review covers the authors' studies on the use of in vitro hollow fibre bioreactor technologies for determining the pharmacodynamics of antiviral compounds for viruses grown in cultured cells, including HIV grown in CD4+ lymphoblastoid cells, vaccinia viruses grown in HeLa-S3 cells and influenza viruses grown in Madin–Darby canine kidney cells. Where possible, correlations between the pharmacodynamic index derived from the in vitro HFIM systems and clinical pharmacodynamic studies are made.
Introduction to hollow fibre technology
Traditionally, mammalian cell culture was performed in stationary cultures in the presence of cell growth medium supplemented with sera from various human or animal sources. When the cells had grown to confluence, they were subdivided several fold (1:2–1:10 or more) and placed into new flasks in the presence of fresh cell growth medium. This procedure expanded the number of cells and replenished the growth factors and other nutrients in the medium required for cell growth. However, this sudden change from a confluent monolayer to a sparse culture is not physiological. Furthermore, due to the accumulation of toxic products of metabolism and depletion of growth factors in the medium, these systems were limited to the production of 105 to 106 cells per ml of growth medium. The need to produce cell densities comparable to that found in tissues (108 to 1010 total cells) prompted Knazek et al., [1] to invent the hollow fibre bioreactor. To that end, they constructed a glass tube containing a bundle of semi-permeable hollow fibres that were attached on the inside to each end of the tube. The tube had an inlet and an outlet port so that medium from a reservoir could be pumped through the hollow fibres in a continuous loop. The cells were added through ports on the top of the glass tube into the extracapillary space (ECS) where cells grew on the outside of the capillaries. The cells were nourished by the medium (Eagle's spinner modified basal essential medium supplemented with fetal bovine serum and antibiotics) flowing through the semipermeable hollow fibre membranes. Using this apparatus, these scientists showed that mouse fibroblasts (L-929 cells) could grow from a total of approximately 2×105 cells to approximately 1.7×107 cells over a 28 day period. They also showed that when 1.5×106 human choriocarcinoma cells (JEG-7 cells) were grown in the hollow fibre bioreactor, the cells produced human chorionic gonadotrophin that concentrated in the ECS and was subsequently collected from the ECS over time. Thus, large quantities of cells could be grown in culture and products made by the growing cells could be concentrated in the ECS of the bioreactor and harvested over time.
Since their inception, hollow fibre bioreactors have been used for the production of monoclonal antibodies [2,3]. More recently these bioreactors have been used to produce genetically engineered proteins in insect cells [4] and mammalian cells [5], viruses for diagnostics and vaccines [6–8] and a malaria parasite [9]. This is just a small sampling of the voluminous literature describing the use of bioreactors for growing cells and producing extracellular products.
There are several advantages to growing cells in hollow fibre bioreactor systems compared to growing cells on glass or plastic flasks [10]. First, a high surface area-to-volume ratio permits extremely rapid exchange between the medium in the central reservoir and the cells growing on the hollow fibres in the cartridge. This leads to rapid equilibration of nutrients, waste products and drugs across the fibres and facilitates the culture of cells at high densities. It is the only culture method that can support cells at physiological cell densities.
Second, cells are bound to a porous support, not a non-porous plastic or glass surface. Since the medium is continuously refreshed without interfering with the cells, adherent cells can be grown for extended periods of time. Non-adherent cells can also grow in the ECS for extended periods of time because of the continuous feeding with fresh medium and constant removal of toxic metabolites. The ability to add medium with or without drugs without disturbing the cells is particularly important when performing dose fractionation pharmacodynamic studies where compounds are added to the system over a short time period and then removed from the system by dilution with drug-free medium without disturbing the environment around the cells. The hollow fibre system is a more in-vivo-like way to grow cells.
Third, the pore size can be controlled to allow small molecules to cross the hollow fibres while retaining larger molecules, virus-infected cells and cell-free viruses in the ECS. This is important for monoclonal antibody and virus production as it permits the monoclonal antibodies and viruses to accumulate in the ECS while the inhibitory substances such as TGF beta can diffuse out of the medium.
Fourth, because of their large size, viruses and virus-infected cells are retained inside of the hollow fibre cartridge. These infectious agents can not pass through the hollow fibres into the medium. This is an added biosafety component of the hollow fibre bioreactor systems which retain all of the dangerous agents (viruses and virus-infected cells) in a tightly closed system protecting laboratory personnel from infection.
Finally, an additional layer of protection for bioreactor studies of viruses is afforded by performing all bioreactor studies under enhanced BSL-2 or BSL-3 conditions.
The hollow fibre perfusion reactor (Figure 1) is a system for growing large quantities of cells at high density. Animal cells or bacteria that are placed into the cartridge through the sampling ports on the top of the cartridge remain outside of the hollow fibres in the ECS. Culture medium is pumped through the lumen of the hollow fibres allowing gases (oxygen and carbon dioxide), low molecular weight nutrients and metabolic products to pass both ways across the hollow fibre membranes. In the system represented in Figure 1, the medium is oxygenated and CO2 is removed as it passes through the semi-permeable tubing connecting the hollow fibre cartridge with the central reservoir. Other systems (Biovest International, Inc., Minneapolis, MN, USA) have a separate cartridge for handling gas exchange and use cell growth medium containing HEPES buffer to maintain the correct pH. Hollow fibres can be constructed from cellulosic, polysulfone, polypropylene or polyethylene materials. The choice of hollow fibre material will depend on the hydrophobicity of the product under study. Cellulosic hollow fibres have lower exchange rates and potentially non-uniform distribution of cells and medium inside the cartridge housing, but have much lower non-specific binding to certain antiviral compounds. Polysulfone hollow fibres have much higher exchange rates, wavy fibres for more uniform distribution of cells and medium inside the cartridge housing, but can have more problems with non-specific binding.
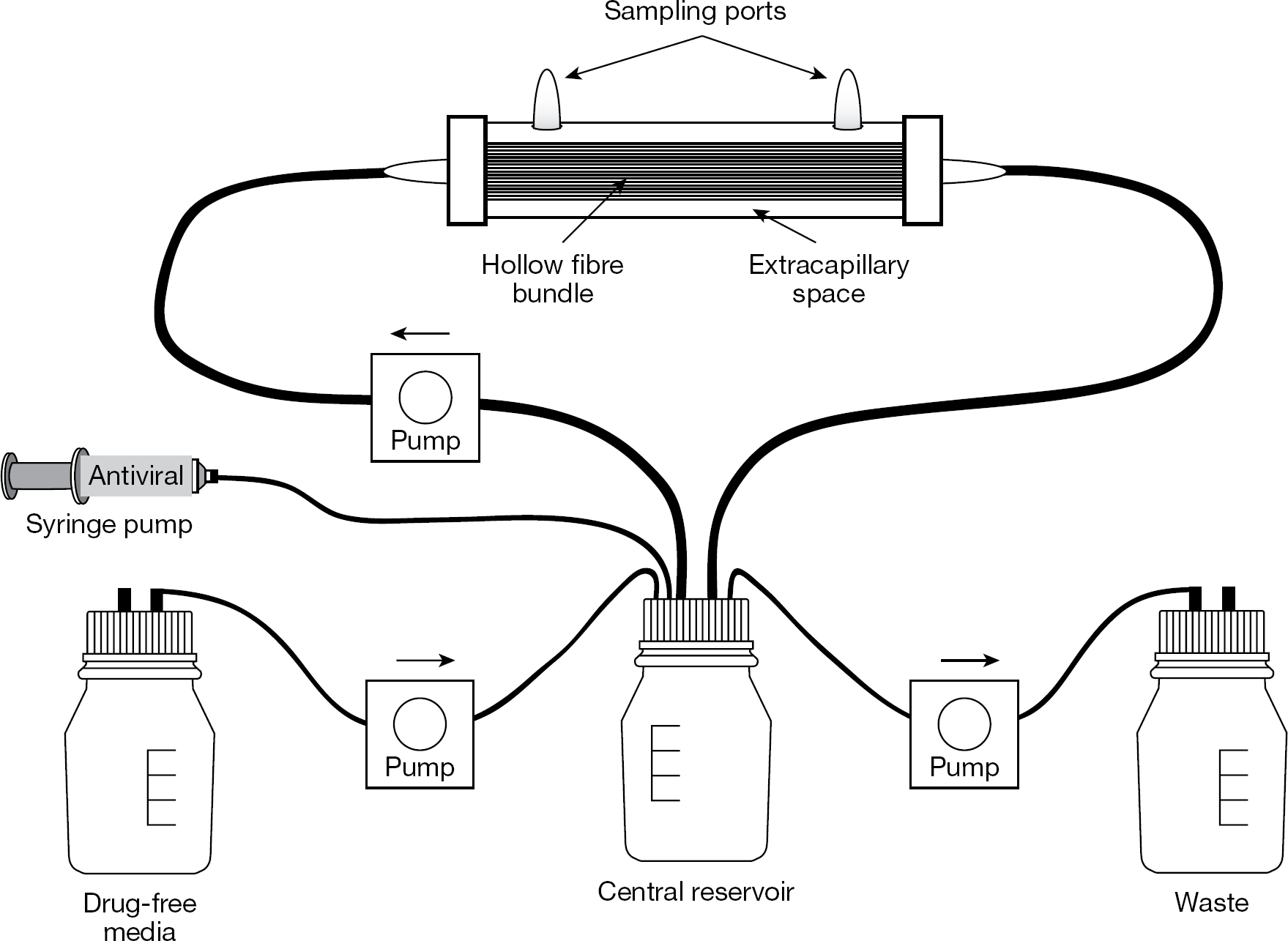
Hollow fibre infection model system
Pore size determines which molecules pass across the membranes. The pore size can range from average molecular weight cutoff values of 10 kd to those containing much larger pores where large molecular weight materials can pass through the fibres. Small pore sizes (50 kd or lower) are used for collection of an excreted product (monoclonal antibodies, proteins of interest or, in our case, viruses). Viruses, antibodies and most proteins are larger than 50 kd and will not enter the pores of hollow fibres with small pore sizes but will accumulate and concentrate in the ECS. The excreted products can then be harvested from the ECS through ports in the top of the cartridge. The capacity of the cartridge can range from 1.5 ml to >1 l. There are several commercial sources for obtaining hollow fibre perfusion bioreactors (FiberCell Systems, Inc., Frederick, MD, USA; Spectrum Laboratories, Inc., Los Angeles, CA, USA; and Biovest International, Inc.). Recent reviews have critiqued the commercial hollow fibre perfusion bioreactor units available for the production of monoclonal antibodies [11,12].
Pharmacodynamic studies for antiviral compounds using hollow fibre perfusion bioreactors
Pharmacodynamics is the science that links drug exposure to response. A key element of pharmacodynamics is the identification of the true pharmacodynamically-linked index. This idea stems from the hypothesis that the shape of the drug concentration–time curve may have an impact on drug effectiveness [13]. For example, the time that free drug concentrations remain above the measure of potency of the drug for the virus in question (that is, the 50% effective concentration [EC50] and 95% effective concentration [EC95]) may be most closely linked to the antiviral effect. In this case, continuous or relatively short dosing intervals lead to maximal effects. Alternatively, peak concentrations relative to the measure of potency (peak/EC50/95 ratio) may be linked to the antiviral effect. Here, infrequent dosing with high peak concentrations result in the best antiviral effect. There are times when the mode of administration does not alter the antiviral effect. Here, the area under the concentration–time curve (AUC) relative to the measure of potency (AUC/EC50/95 ratio) is linked to the antiviral effect. Our laboratory has used the in vitro hollow fibre infection model (HFIM) system to determine the pharmacodynamically-linked index for compounds active against bacteria and viruses [14–16]. This review will attempt to cover the extant literature for the use of hollow fibre technology for determining the pharmacodynamically-linked index of antiviral compounds for a number of viruses. For some of these studies with viruses, the pharmacodynamically-linked index determined in the HFIM system has been confirmed in one or more clinical studies (Table 1).
Correlation between HFIM prediction and clinical usage
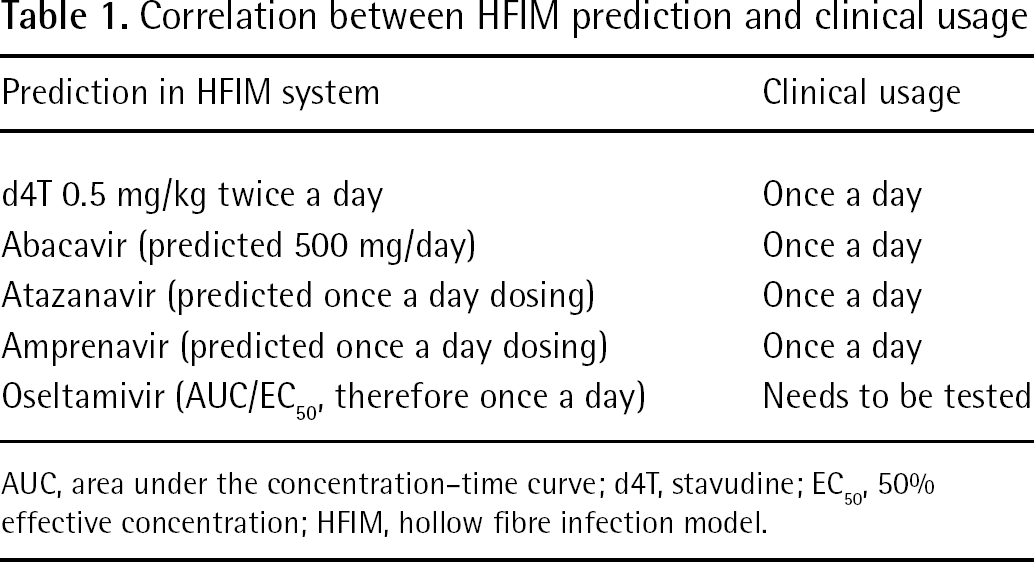
AUC, area under the concentration–time curve; d4T, stavudine; EC50, 50% effective concentration; HFIM, hollow fibre infection model.
There are four things that have to be known to determine the pharmacodynamic index (dose and schedule) of an antiviral compound for a particular virus. First, the range of EC50/EC95 values of the compound for the virus under study needs to be determined in the HFIM system. Second, the binding of the compound to serum proteins has to be determined, as only free drug has antiviral effect. Third, the pharmacokinetics of the drug and the pharmacokinetic variability of the compound within the population of concern must be identified. Fourth, a target exposure and optimal schedule of administration of the compound for the virus in question that will achieve the desired goal of therapy needs to be identified.
For most of the studies reported in this review, 4300-C2011 cartridges (FiberCell Systems, Inc.) containing high molecular weight cutoff (20 kd) polysulfone hollow fibres have been used. Occasionally, cellulosic cartridges with pore sizes of 10 kd average molecular weight cutoff have been used because the compound under study binds to the polysulfone hollow fibre membrane more than to the cellulosic hollow fibre membranes. These cartridges containing hollow fibres with smaller average pore sizes (10 kd) still allow nutrients and gases to pass across the semi-permeable membranes to nourish the cells and remove toxins while preventing the virus-infected cells and cell-free viruses from entering the medium.
The high surface area (2,100 cm2) to volume (15 ml) ratio of the 4300-C2011 cartridge guarantees that the drug exposures in the ECS and the central reservoir rapidly come to equilibrium. The HFIM system allows uninfected and virus-infected cells to grow attached to the hollow fibres where cell-to-cell spread of virus is very efficient. In some cases, non-adherent cell lines such as CD4+ lymphoblastoid cells are used where the cells grow to high densities in the ECS unattached to the hollow fibres. The high cell density also favours very efficient cell-to-cell spread of virus. In either case, released virus and virus-infected cells are excreted into the ECS where they accumulate and concentrate over time. At specific times, drugs are administered by computer controlled pumps into the system through a port in the central reservoir to simulate any schedule of drug delivery (that is, once a day, twice a day or three times a day). The effect of drug on cell-to-cell spread of the virus can be determined by sampling the contents of the ECS through the sampling ports and counting the number of virus-infected cells by FACS analysis of cells treated with a fluorochrome-labelled monoclonal antibody to a specific viral antigen of the virus under study. In addition, the effect of drug on the yield of cell-free virus can be determined by plaque assay of released virus or by ELISA assay of the amount of viral antigen produced in the bioreactor over time. The actual concentration of antiviral drug in the reservoir and the ECS can also be measured by sampling these compartments and determining the amount of drug present by LC/MS/MS (or equivalent chemical methodology) over time. As the virological end point (inhibition of viral replication and prevention of cell-to-cell spread of virus) is measured sequentially and the drug concentration is later validated by direct measurement of achieved drug concentrations by LC/MS/MS analysis, two measured outcomes (antiviral effect and drug exposure) are obtained that allow construction of an exposure–response relationship. Since eight or more drug exposure evaluations are performed simultaneously, a robust exposure-response curve is generated with these experiments.
A procedure for determining the pharmacodynamically-linked index is as follows. First, the EC50 value of the compound for the virus under study is determined in flasks. Flasks are used for this portion of the study for cost concerns (flasks are less expensive than hollow fibre cartridges). Then, a dose range study is performed in the HFIM system to determine the EC50 value for the drug for the virus under study. This analysis uses a continuous infusion profile consisting of a no-drug control hollow fibre unit and several hollow fibre units treated with drug concentrations below and above the EC50 value determined in flasks. This identifies a daily exposure (AUC0−24) that will have a known antiviral effect in the HFIM system. Therefore, the AUC for the chosen degree of suppression (an EC value between 50–95% suppression is usually chosen) is: AUC0−24=X (multiples of the EC50 value) ×EC50×24 h=XEC50×24 nM*h. This is the exposure target.
In a separate experiment, this exposure is administered in a dose fractionated manner as follows. First, one hollow fibre unit receives a continuous infusion of drug at the desired concentration, usually 2× to 8× the EC50 value. Next, in a second hollow fibre unit, the total dose is given as a single administration that is infused over a 1 h period followed by a no drug (drug-free) washout resulting in a peak drug concentration followed by a decrease in the drug concentration with the correct half-life to achieve the same 24 h AUC as the continuous infusion. Then, in a third hollow fibre unit, half the dose is administered every 12 h with a lower peak drug concentration followed by a no-drug washout with the same half-life of decrease and a matching 24-h AUC. Finally, in a fourth hollow fibre unit, the total dose is broken up into three equal parts and administered every 8 h with a still lower peak drug concentration followed by a no-drug washout with the correct half-life and a matching 24 h AUC. A fifth hollow fibre unit receives no drug and serves as the no-drug control. The data derived from the dose fractionation experiment will determine the pharmacodynamically-linked index for that compound for the virus under study. For example, if continuous infusion gives the best antiviral effect, the pharmacodynamically-linked index is time above the EC50/95. If maximal antiviral effect is generated by the least frequent administration schedule, then the peak/EC50/95 ratio is the linked index. Finally, if the continuous infusion and all of the fractionated doses have the same antiviral effect, then the pharmacodynamically-linked index is the AUC/EC50/95 ratio. The pharmacodynamic index is related to how a compound (drug) should be administered to attain the best result. If the index is time above the EC50/95 ratio the drug may have to be administered continuously. If the pharmacodynamically-linked index is the AUC/EC50 ratio, then the drug could be given once a day if toxicity is not concentration-related. The following are specific examples from reports of our laboratory on pharmacodynamic studies for antiviral drugs for viruses.
HIV
Nucleoside analogues
The initial antiviral pharmacodynamic HFIM system study involved HIV-1 and the nucleoside analogue, 2′, 3′-didehydro-3′deoxythymidine (d4T) [17]. In this study, three hollow fibre units containing 104 or 105 HIV-infected CEM cells and 107 uninfected CEM cells were set up. One unit was continuously infused with medium without drug for 9 days and served as a no-drug control. A second unit was continuously infused with an exposure of d4T equivalent to 1 mg/kg/day for 9 days. A third unit received a bolus of d4T equivalent to an exposure of 0.5 mg/kg/day every 12 h followed by a no-drug washout to mimic a 1 h half-life. Each hollow fibre unit was sampled daily and the amount of HIV p24 antigen was determined by ELISA. The results showed that in the absence of d4T the amount of p24 antigen increased from 5,000 pg/ml to >20,000 pg/ml over the 9 days of the experiment. In the presence of 1 mg/kg/day of d4T delivered as a continuous infusion or 0.5 mg/kg delivered twice a day, the amount of p24 antigen did not increase over the 9 days of the experiment. In separate experiments hollow fibre units infected with the same amount of virus and treated with d4T at either 0.25 mg/kg or 0.125 mg/kg every 12 h failed to completely inhibit virus replication. These results indicated that a dose of 1 mg/kg/day delivered as 0.5 mg/kg every 12 h or as a continuous infusion of 1 mg/kg/day inhibited virus replication. The HFIM system predicted that the minimum effective dose of d4T to treat patients infected with HIV was approximately 0.5 mg/kg/day administered twice a day. This prediction was confirmed in a clinical study [18].
The effect of the carbocyclic nucleoside analogue, abacavir, on the replication of HIV in CEM-ss cells was determined in the HFIM system [19]. This compound was licensed for use as a twice-daily drug for the treatment of HIV-infected patients. To determine if abacavir could be a once a day drug, the HFIM system was used to determine the pharmacodynamically-linked index of abacavir for HIV-1. Three hollow fibre units containing 3×105 HIV-1-infected H9IIIB cells and 3×107 uninfected CEM-ss cells were set up. One hollow fibre unit contained medium without drug and served as a no-drug control. A second hollow fibre unit containing the same cell mixture of HIV-infected and uninfected cells was exposed to a 500 mg dose as a continuous infusion. A third hollow fibre unit containing the same cell mixture was exposed to a 500 mg dose given once a day as bolus of drug followed by a drug-free washout with a 1 h half-life. In a separate experiment, three hollow fibre units containing 3×105 HIV-infected cells and 3×107 uninfected CEM-ss cells were set up. One unit was infused with medium only and served as a no-drug control. A second unit was exposed to 250 mg of abacavir twice a day. A third unit was exposed to a simulated 500 mg dose once a day. The results of this study showed that a dose equivalent to 500 mg once a day or doses of 250 mg twice a day gave equivalent results for the suppression of virus replication suggesting that the pharmacodynamic index for abacavir for HIV is the AUC/EC50 ratio. These results suggested that abacavir can be given on a once a day schedule instead of a twice daily regimen as had been recommended for other nucleoside analogues for the treatment of HIV. Several clinical studies have demonstrated that abacavir, as part of a combination of antiretroviral drugs, can be given once a day [20].
Protease inhibitors
The HFIM system was used to determine the minimum concentration of the protease inhibitor, A-77003, that would inhibit the replication of HIV in CEM cells [21]. Four hollow fibre units containing 3.5×105 CEM-H9IIIB-infected cells and 3.5×107 uninfected CEM cells were set up. One hollow fibre unit contained no compound and served as a no-drug control and three hollow fibre units received 0.063 μM, 0.125 μM and 0.25 μM A-77003, respectively, by continuous infusion for 11 days. The media was changed daily to prevent possible decay of the drug. Each hollow fibre unit was sampled daily and the effect of A-77003 on virus replication was determined by p24 ELISA. The results on day 11 showed that the compound had a dose-response but failed to inhibit virus replication completely at the maximum exposure (0.25 μM). In a repeat experiment with continuous exposures of 0.5, 1.0 and 2.0 μM A-77003, virus replication and cell-to-cell spread of virus were completely inhibited by A-77003. These results suggest that the minimum effective exposure to A-77003 by continuous infusion for HIV is 0.5 μM. A Phase 1 clinical trial of A-77003 in HIV-1-infected individuals showed the drug was not effective at the highest concentrations tested (0.28 mg/kg of body weight) and this concentration caused infusion site phlebitis. Since higher doses could not be tested, this compound was not further developed [22].
The HFIM system was used to determine the pharmacodynamically-linked index for another protease inhibitor, BMS-232632 [23]. BMS-232632 is marketed as atazanavir. In this case, four hollow fibre units containing 104 HIV-1 infected CEM-H9IIIB cells and 106 uninfected CEM cells were set up. One hollow fibre unit contained no drug and served as the no-drug control. A second hollow fibre unit received the compound at 4×EC50 by continuous infusion. In a third hollow fibre unit, BMS-232632 was infused into the unit over a 1 h period to achieve a peak exposure of 56.7 nM for 2 h followed by a no-drug washout with a 5.5 h terminal half-life which produced a 24-h concentration of 3.55 nM and an AUC that was the same as the continuous infusion unit. A fourth hollow fibre unit was exposed to compound at 16×EC50 followed by a no-drug washout with a 5.5 h terminal half-life. The results showed that continuous infusion at 4×EC50 completely inhibited virus replication whereas 4×EC50 administered as a bolus followed by a no-drug washout did not suppress virus replication past 9 days of infection in the HFIM system. However, when 16×EC50 was delivered as a bolus followed by a no-drug washout, HIV replication was completely suppressed [23] suggesting that time above the EC50 is the linked variable. Therefore, 16×EC50 value of compound is required to completely suppress virus replication in the HFIM system on a daily administration schedule. This result suggested that once a day dosing at the correct concentration will work for HIV-infected people and this prospective study was confirmed in the clinic [24].
A similar experiment was performed for the protease inhibitor, amprenavir in the presence of ritonavir [25]. The EC50 value for amprenavir for drug susceptible HIV isolates is 0.03 μM and, for less susceptible HIV isolates, the EC50 ranges from 0.05 to 0.08 μM. To determine the effect of amprenavir on the replication of HIVIIIB in the hollow fibre system, four hollow fibre units containing 104 HIV-infected CEM-H9IIIB cells and 106 uninfected CEM cells were set up. One hollow fibre unit received no drug and served as the no-drug control. A second hollow fibre unit received 4×EC50 value of amprenavir (0.12–0.32 μM) delivered by continuous infusion for 12 days. A third hollow fibre unit received the same total exposure to amprenavir delivered as a bolus followed by a no-drug washout twice a day for 12 days. A fourth hollow fibre unit received the same total exposure delivered as a bolus followed by a no-drug washout three times a day for 12 days. The results show that the twice a day drug treatment reduced virus replication by approximately one-half. Three times a day drug treatment and the continuous drug treatments inhibited virus replication by close to 90% (Figure 2). These results indicate that the pharmacodynamically-linked index for amprenavir is time above the EC50/95. The amprenavir/ritonavir combination is part of standard care for HIV-infected patients.
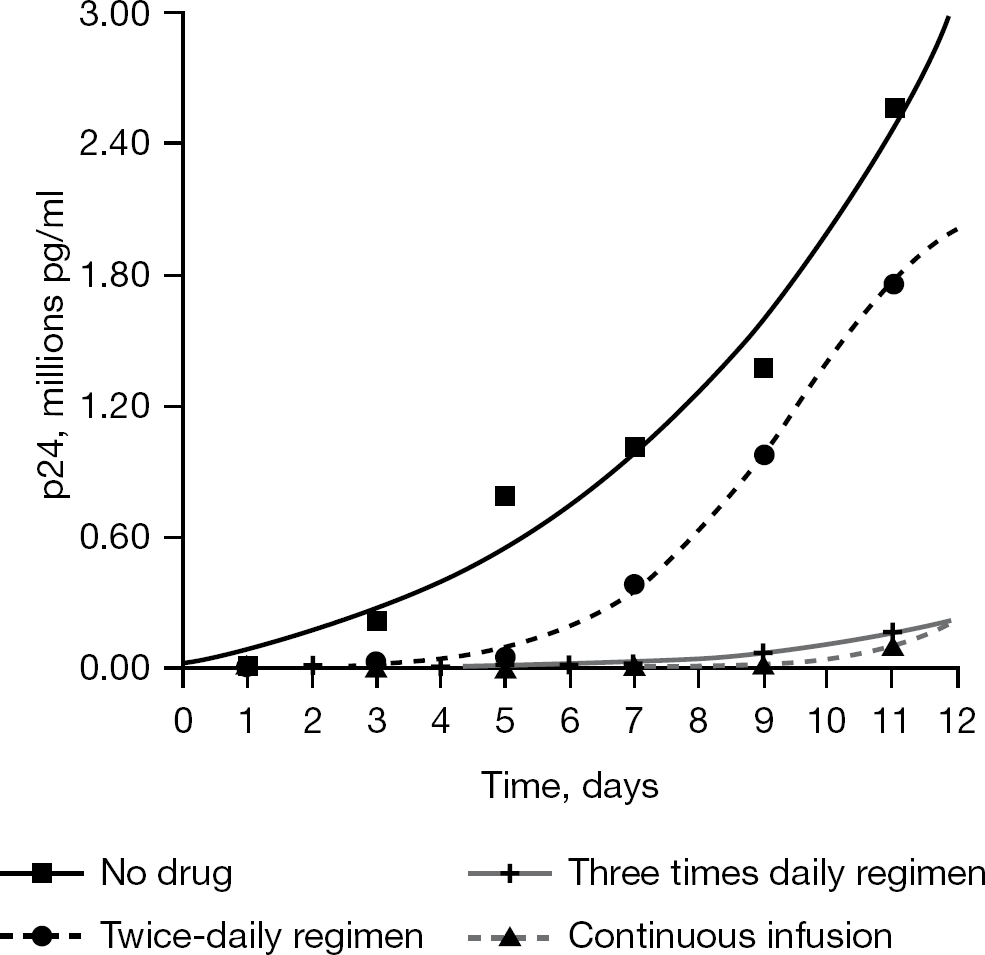
Dose fractionation study of amprenavir for HIVIIIB
Integrase inhibitors
Similar studies have been performed with the integrase inhibitor, raltegravir [26], and several other integrase inhibitors. The results of these studies will be published in the near future.
Vaccinia virus
With concerns about possible bioterror attacks with Variola major virus, governments around the world have initiated research programmes to identify compounds or strategies that will prevent infection with Variola major virus or treat patients already infected with the virus. There is an effective live attenuated vaccine for the prevention of smallpox [27]; however, due to rare but serious side effects, vaccination was stopped in the 1970s in most Western countries and currently it is used only in select populations such as the military and first responders. Thus, the majority of the world's citizens are susceptible to infection with Variola major virus.
There are several antiviral compounds that are effective for the treatment of smallpox [28–30]. One of these is cidofovir which has been approved for the treatment of cytomegalovirus retinitis in immunocompromised patients [31]. The United States government proposed the use of intravenous cidofovir for the treatment of patients exposed to or infected with smallpox virus. The largest dose of cidofovir ever given to people is 10 mg/kg/day. The clinical dose of cidofovir for cytomegalovirus infections is 5 mg/kg of body weight weekly administered with probenecid to prevent nephrotoxicity. To determine if this dose of cidofovir would be effective for curing a person infected with Variola major virus, the HFIM system was used to determine the pharmacodynamically-linked index of cidofovir for vaccinia virus (a surrogate for Variola major virus) grown in Hela-S3 cells [16].
Non-adherent cells are ideal for virus growth in hollow fibres. However, all viruses do not grow well in non-adherent cells. For our pharmacodynamic studies on vaccinia virus, we used HeLa-S3 cells because they have been used to grow large amount of vaccinia virus and they can be grown as adherent cells or as suspension cells [32]. The effect of cidofovir on vaccinia virus replication in the HeLa-S3 cells was monitored by FACS analysis of virus-infected cells and by the production of infectious virus released by the cells by plaque assay. First, the EC50 value of cidofovir for the WR strain of vaccinia virus gown in Hela cells was determined in flasks. HeLa-S3 cell monolayers were infected with the WR strain of vaccinia virus at an multiplicity of infection of 0.01 plaque-forming unit per cell in the presence of various concentrations of cidofovir. Results of three independent assays showed that the EC50 value for cidofovir for this strain of vaccinia virus was 30.85 ±8.78 μM. To determine the EC50 value of cidofovir for the WR strain of vaccinia virus in the HFIM system, six hollow fibre units containing 108 uninfected Hela-S3 cells and 106 vaccinia virus-infected Hela S3 cells were set up and exposed to various concentrations of cidofovir ranging from 0 to 200 μM by continuous infusion. The results in Figure 3 show that 0 (no-drug control), 12.5 and 25 μM cidofovir had little effect on virus replication, whereas 50 and 100 μM cidofovir inhibited virus replication by approximately 90% and 200 μM cidofovir completely inhibited virus replication in the HFIM system. These results are consistent with the EC50 value determined in the flask assay. To determine the pharmacodynamically-linked index of cidofovir for this strain of vaccinia virus, a dose fractionation experiment was performed in which one hollow fibre unit received no cidofovir and served as the no-drug control. A second hollow fibre unit received an exposure of 30 μM cidofovir administered as a continuous infusion. A third hollow fibre unit received an exposure of 100 μM cidofovir administered as a continuous infusion. The fourth and fifth hollow fibre units received doses of 30 and 100 μM cidofovir as boluses given over a 1 h period followed by a no-drug washout with the appropriate half-life. The results in Figure 4 show that vaccinia virus grew well in the absence of cidofovir. The hollow fibre units receiving 30 μM or 100 μM cidofovir as continuous infusions or as bolus doses of 30 μM and 100 μM cidofovir inhibited virus replication. Since the continuous arms and the bolus arms at each drug concentration gave the same inhibition of virus replication, the pharmacodynamically-linked index for cidofovir for vaccinia virus is the AUC/EC50/95 ratio. This is a very large dose of cidofovir, most likely too large to be given safely to humans. The authors concluded that cidofovir would not be useful for treatment of people in the case of a terrorist attack involving smallpox virus.
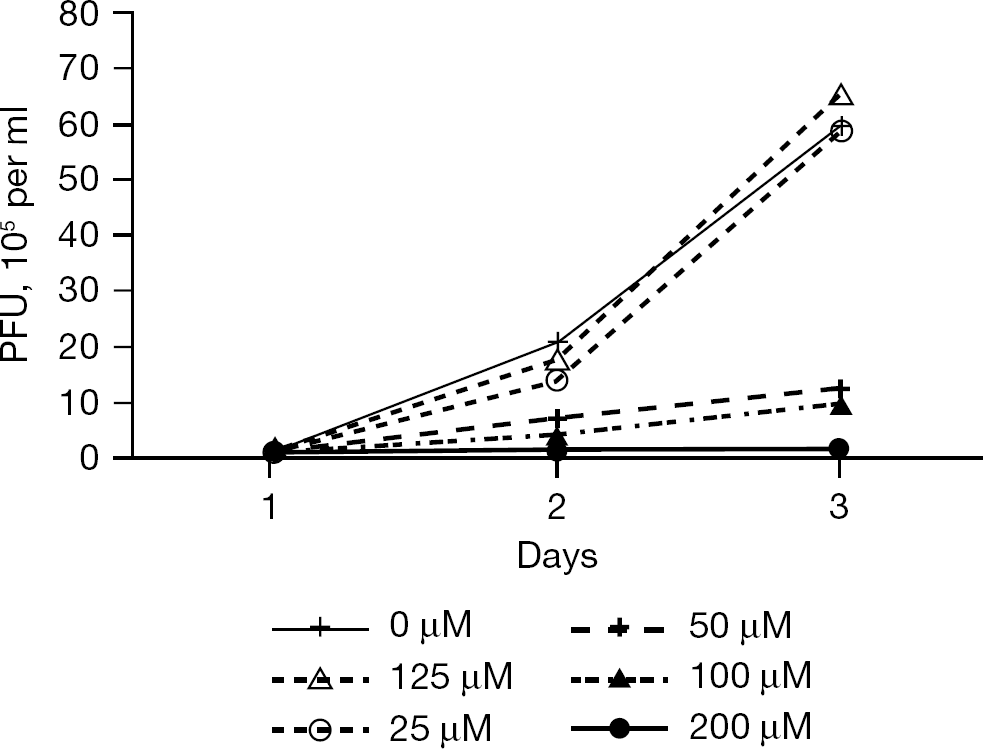
Dose range experiment for vaccinia virus and cidofovir
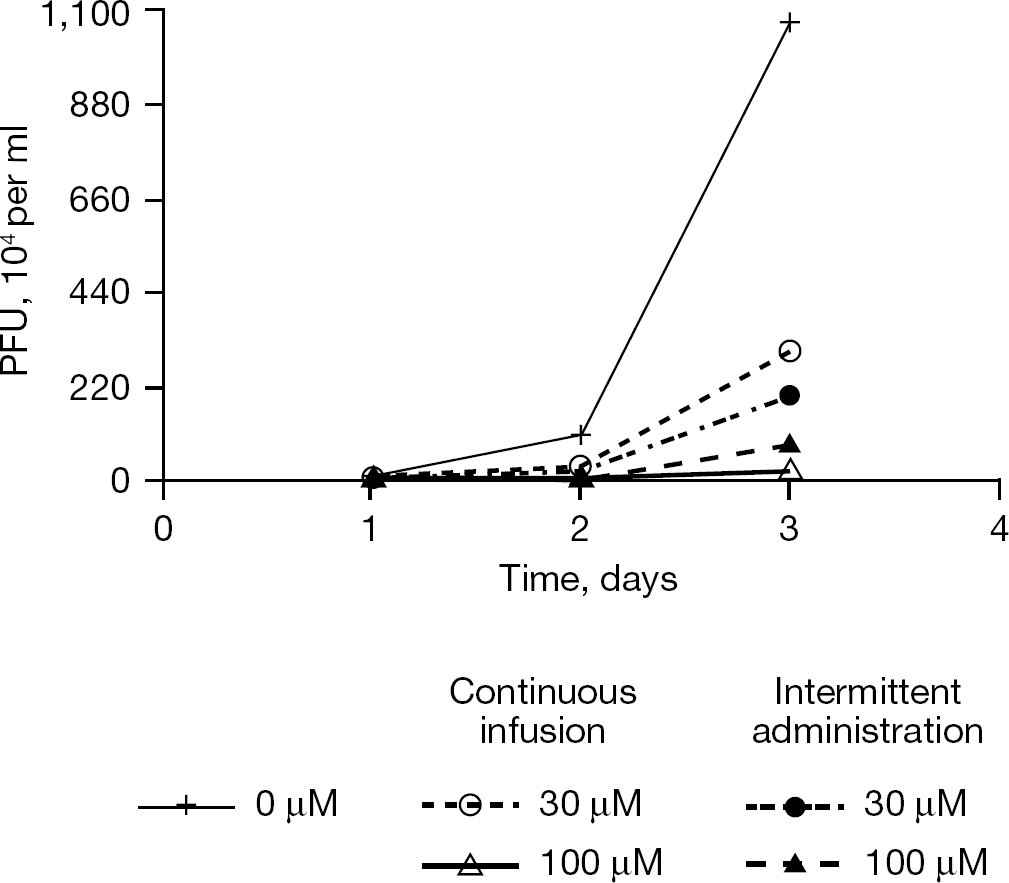
Dose fractionation study for vaccinia virus and cidofovir
Influenza virus
There are several licensed antiviral compounds effective against influenza viruses. These include the adamantanes, amantadine and rimantadine, and the neuraminidase inhibitors, oseltamivir and zanamivir [33]. Amantadine has been used extensively for the treatment of uncomplicated influenza [34]. Recently, almost all circulating influenza viruses that infect humans have become resistant to the adamantanes due to mutations in the M2 gene that encodes an ion channel in the viral envelope [35]. Because of this wide spread resistance to the adamantanes, the US Centers for Disease Control and Prevention has recommended that adamantanes not be used for the prevention and treatment of influenza in humans. Oseltamivir, a neuraminidase inhibitor, is the most commonly used antiviral for uncomplicated influenza [36]. The current recommendation for treatment of uncomplicated influenza with oseltamivir is to take two 75 mg tablets twice a day. We wished to determine if once a day treatment was possible for oseltamivir therapy for uncomplicated influenza. To that end, we performed dose range and dose fractionation experiments in our HFIM system [37].
As was the case for poxviruses, there are no cell lines that continuously produce influenza viruses without cell death. The standard cell line used for antiviral studies on influenza viruses is the Madin-Darby canine kidney (MDCK) cell line. This cell line grows well on glass and plastic surfaces and can be efficiently infected with various strains of influenza virus. To perform pharmacodynamic studies for oseltamivir for influenza viruses, we used a derivative of MDCK cells (AX-4 cells) that expresses higher levels of cell surface glycoprotiens containing α-2,6-linked sialic acid conjugates than the original MDCK cells [38]. The EC50 value for oseltamivir for the A/Sydney/5/97 R292 strain of influenza virus grown in AX-4 cells in flasks was 10.23 ±8.66 ng/ml. It was determined that the R292 strain of influenza virus grows well in AX-4 cells in the hollow fibre units. To perform dose ranging studies in the HFIM system, six hollow fibre units containing 102 influenza A virus-infected AX-4 cells were mixed with 108 uninfected AX-4 cells and continuously infused with various concentrations of oseltamivir. The effect of the drug on virus replication was determined daily by plaque assay and hemagglutination assay of virus released from the infected cells in each hollow fibre unit. The results showed that the EC50 value in the HFIM system for oseltamivir for this isolate of the R292 strain of influenza virus was 0.726 ng/ml with an r2=0.978. To determine the pharmacodynamically-linked index, a dose fractionation study was performed where an exposure equivalent to 1 ng/ml was delivered to one hollow fibre unit by continuous infusion, a second hollow fibre unit received the same exposure once a day followed by a no-drug washout to mimic an 8 h half-life, a third hollow fibre unit received the same exposure delivered twice a day followed by a no-drug washout to mimic an 8 h half-life, and a fourth hollow fibre unit received the same exposure delivered three times a day followed by a no-drug washout to mimic an 8 h half-life. The data showed that in the absence of drug the virus grew well in the HFIM system. The continuous dose and all three fractionated doses gave the same amount of inhibition of virus replication. Therefore, the pharmacodynamically-linked index for oseltamivir for the R292 strain of influenza A virus is the AUC/EC50/95 ratio. This means that, at the appropriate dose, oseltamivir could be given once a day. This observation will have to be verified with a clinical study.
A similar pharmacodynamic study was performed for amantadine [39]. In this study, a wild-type clinical isolate, A/Albany/1/98, was used. The EC50 value determined in MDCK cells in flasks was 0.051 ±0.01 μg/ml (0.337 ±0.06 μM). The dose range experiment used five hollow fibre units containing 102 virus-infected MDCK cells and 108 uninfected MDCK cells each continuously infused with various doses of amantadine (0–6 μg/ml). The hollow fibre units were sampled daily and the effect of drug on virus replication was determined by plaque assay. The data showed a dose response at 24 and 48 h post-infection but by 72 h the antiviral effect was lost. Genetic analysis of viruses produced at 72, 96 and 120 h post-infection in the presence of various concentrations of amantadine showed that these viruses contained mutations in the M2 gene that are associated with resistance to amantadine. Viruses grown in the absence of amantadine did not contain these mutations. Because of the generation of amantadine-resistant mutant viruses, a pharmacodynamic analysis in the HFIM system was not possible.
One of the main uses of the HFIM system is to determine an exposure of a compound that will inhibit virus replication and prevent the emergence of resistance. It is clear from the foregoing that amantadine used as a single drug will not prevent the emergence of resistance. It has been shown by others [40] that double and triple combinations of antiviral drugs for influenza virus may prevent the emergence of resistance. We are in the process of performing HFIM experiments to determine what combination of antiviral drugs will prevent virus replication and the emergence of resistance.
Discussion
The HFIM system is an efficient and relatively inexpensive (compared to a clinical study) in vitro system for determining the pharmacodynamics of antiviral compounds for viruses. Historically, these studies were performed with virus-infected cells (H9IIIB cells) that grow in suspension in the HFIM system. Under these circumstances, cells can be removed from the ECS for analysis of the percentage of virus-infected cells by flow cytometry as a measure of cell-to-cell spread of the virus. Virus yield can be determined after the cells are removed from the sample by low speed centrifugation and the supernatant assayed for the presence of cell-free virus by plaque assay or by ELISA or other similar methodologies. Our initial studies on the pharmacodynamics of antiviral drugs for HIV used mixtures of suspension cultures of CD4+ T-cells chronically-infected with HIV-1 and uninfected CD4+ T-cells growing in the HFIM system. This system was used to determine the pharmacodynamically-linked index for the nucleoside analogues, d4T and abacavir, and for the protease inhibitors A-77003, atazanavir and amprenavir. For the antiretroviral drugs, the pharmacodynamic driver predicted by the HFIM was confirmed in clinical studies (Table 1).
Since most virus infections do not lead to chronically-infected cells, data from papers that showed how the HFIM system could be used to determine the pharmacodynamically-linked index for drugs and virus-infected cells that do not grow as suspension cultures, such as HeLa-S3 cells and MDCK cells, examples of pharmacodynamic studies were also presented. When HeLa S3 cells or MDCK cells are added to the hollow fibre units, the cells adhere tightly to the hollow fibres and do not float free in the ECS. Thus, the cells can not be sampled from the ECS. However, as the cells become infected with either vaccinia virus or influenza A viruses, the actin filaments in the cells are destroyed and the cells are released into the ECS. In addition, when the cells are destroyed by virus infection, as is the case with vaccinia virus infection, or the virus is released from the cell by budding at the plasma membrane, as is the case for influenza virus, the released virus accumulates in the ECS and the cell-free virus can be sampled over time. In this manner, the effect of antiviral compounds on virus replication can be determined either by FACS analysis of virus-infected cells or by plaque assay or ELISA of cell-free virus. A recent development in influenza vaccinology is the ability to grow influenza viruses in non-adherent cells that grow in suspension [41]. These cells may be very useful for determining the pharmacodynamic index for antiviral compounds for influenza viruses in the HFIM system.
The demonstration that adherent cells can be used to grow virus in the HFIM system opens this system up to the pharmacodynamic analysis of antiviral compounds for a wide variety of viruses such as dengue virus and West Nile virus, that grow in adherent cells. As long as the virus-infected cells produce virus that is released into the ECS, then the effect of antiviral compounds can be measured.
In summary, we have presented an in vitro HFIM system for determining the pharmacodynamics of antiviral compounds for viruses. This system is ideal for studying the effects of antiviral compounds on HIV-infected cells because this virus grows in cells in suspension. We have also presented data on the use of the HFIM system for growth of virus in adherent cells. This later demonstration will open this pharmacodynamic system to the vast majority of virus infected cells.
Footnotes
Acknowledgements
This work was supported in part by grant number RO1-AI079729–01 from the National Institutes of Allergy and Infectious Disease to the Virology Therapeutics and Pharmacodynamics Laboratory, Center for Biodefense and Emerging Infections, Ordway Research Institute, Center for Medical Sciences, Albany, NY, USA.
The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Allergy and Infectious Disease or the National Institutes of Health.
The authors declare no competing interests.
