Abstract
Background:
Limited in vivo studies in the scientific literature suggest that components of green tea and elderberry may be beneficial in treating influenza virus infections. They are thought to act by blocking virus adsorption to cells. TheraMax® is a proprietary medication administered by nasal spray that contains both green tea and elderberry extracts that was evaluated for antiviral activity.
Methods:
TheraMax was tested by dilution in Madin–Darby canine kidney cell cultures in standard viral cytopathic effect (CPE) inhibition and virucidal assays against eight influenza A and B strains. It was also administered intranasally to mice to determine protective activity compared to oral oseltamivir against an influenza A/NWS/33 (H1N1) infection.
Results:
In cell culture assays, TheraMax was found to inhibit viral CPE by 50% at a 1:20 dilution for seven of the eight virus strains, with no virucidal activity at 1:2 dilution. The undiluted product was administered to anaesthetized mice twice daily for 4 days starting 2 h before or 12 h after infection. Alternatively, TheraMax and virus were combined for treatment and infection. Oseltamivir was given orally twice daily for 5 days at 10 mg/kg/day starting at −2 h. TheraMax (combined directly with virus) and oseltamivir each prevented death and curtailed weight loss during the infection, and improved lung haemorrhage scores on day 6. TheraMax starting at −2 h or 12 h significantly delayed death by >2 days and reduced lung haemorrhage scores, but most animals died.
Conclusions:
These combined results indicate that TheraMax delayed symptoms during animal infections, likely through blocking of virus adsorption.
Introduction
A novel influenza A (H1N1) virus reached pandemic proportions in the world and became a significant cause of morbidity and mortality in 2009. Four drugs have been licensed for the treatment of influenza infections, namely oseltamivir, zanamivir, amantadine and rimantadine, but oseltamivir is the drug that is used most frequently. The need for new products to treat such infections is apparent, because the novel 2009 influenza virus is already amantadine-resistant, and because oseltamivir-resistant viruses have been isolated from treated patients [1]. Drug resistance to the adamantanes (amantadine and rimantadine) and neuraminidase inhibitors (oseltamivir and zanamivir) is also prevalent to varying degrees in H3N2 and H5N1 viruses [2–5].
Various compounds derived from plant sources have been tested for antiviral activity against influenza and other viruses. Among these, green tea has been the subject of many antiviral investigations. Green tea is derived from the evergreen plant Camellia sinensis and contains polyphenolic compounds, the most prominent of which is a catechin identified as epigallocatechin gallate (EGCG). Catechins comprise about 10% of green tea leaves by weight [6]. The major catechins in green tea extract, when prepared by boiling the leaves, consist of EGCG, epigallocatechin, epicatechin, epicatechin gallate and catechin at approximately 40%, 30%, 10%, 7% and 5% of the total catechins present, respectively [6]. Green tea extract and EGCG have been shown to inhibit some strains of influenza A and B viruses in cell culture [7–9]. Semi-synthetic derivatives of the polyphenolic compounds of green tea have also been shown to possess anti-influenza virus activity [10]. The antiviral action of green tea catechins is due to the blocking of virus adsorption by binding to viral haemagglutinin [7,9]. EGCG may also interfere with viral membrane fusion by inhibiting the acidification of the endosome [8]. These activities affect the ability of influenza viruses to attach to cells and to release viral RNA into the cytoplasm. More recently, green tea catechins were shown to inhibit the endonuclease activity of influenza A RNA polymerase [11]. Besides having antiviral activity against influenza virus, green tea catechins are inhibitory to a number of other unrelated viruses [12–17].
Another plant-derived material inhibiting influenza virus replication is the extract from Sambucus nigra (elderberry). Anthocyanins are the main components in elderberry extract, comprising about 1.4% of the berry by weight, of which about 54% of the anthocyanins is cyanid-3-glucoside and 40% is cyanid-3-sambubioside [18]. Flavonoids in elderberry extract are inhibitory to several strains of influenza A and B viruses [19,20]. These flavonoids were shown to bind directly to H1N1 virions preventing virus adsorption to cells [20]. Various oral formulations of elderberry extract are sold as over-the-counter medications.
Recently, a product called TheraMax® was developed for the treatment of influenza virus infections. TheraMax contains both green tea and elderberry extracts. It was developed for intranasal administration, where direct interaction with virus particles may lead to diminished infectivity based upon the prominent modes of action of the active materials. In this report we evaluated TheraMax against several strains of influenza A and B viruses in cell culture. We also present the efficacy data of TheraMax in treating an influenza A/NWS/33 (H1N1) virus infection in mice, compared to the antiviral drug oseltamivir.
Methods
Compounds
TheraMax was provided by Therabiogen, Inc. (New York, NY, USA) as the 1× formulated (undiluted), saline-based aqueous product that is sold as an over-the-counter medication. TheraMax contains extracts of Camellia sinensis (green tea) at a concentration of 1 mg/ml and Sambucus nigra (elderberry) at 10 mg/ml. The percentages of green tea catechins, elderberry anthocyanins, and other components of these extracts contained in TheraMax will vary slightly with harvesting and manufacturing conditions as different lots are produced. The EGCG concentration is standardized to be ≥60 μg/ml. Other minor components of TheraMax that do not have antiviral activity include extracts from Gelsemium sempervirens and Bryonia alba.
Oseltamivir was purchased as Tamiflu® capsules from a local pharmacy. We recently demonstrated that the efficacy of these capsules is comparable to that of pure oseltamivir phosphate [21]. Because Tamiflu® contains filler material, entire capsules were weighed out and equated to 75 mg of oseltamivir (the active form of oseltamivir phosphate). The amount of oseltamivir phosphate in each capsule is 98.5 mg, based upon the description of the package insert. Oseltamivir was prepared in sterile water for oral gavage administration.
Cell culture antiviral experiments
The formulated product TheraMax was tested by dilution for antiviral activity in Madin–Darby canine kidney (MDCK) cell cultures in 96-well microplates infected with influenza viruses using a 3-day cytopathic effect inhibition assay quantified by neutral red dye uptake [22]. In vitro testing was performed against the following viruses: A/California/07/2009 (H1N1), A/New York/18/2009 (H1N1), A/NWS/33 (H1N1), A/Perth/16/2009 (H3N2), A/Victoria/3/75 (H3N2), A/Duck/MN/1525/81 (H5N1), B/Florida/4/2006 and B/Malaysia/2506/2004. The viruses were obtained from various sources as follows: A/NWS from Kenneth Cochran (University of Michigan, Ann Arbor, MI, USA); A/Victoria from the American Type Culture Collection (Manassas, VA, USA); A/Duck from Robert Webster (St Jude Children's Hospital, Memphis, TN, USA); and the five others from the Centers for Disease Control and Prevention (Atlanta, GA, USA). All of the viruses were used at approximately 50–100 50% cell culture infectious doses (CCID50) per well of cells. Cytotoxicity was assessed in parallel by comparing neutral red dye uptake in stationary monolayers of uninfected MDCK cells after 3 days of incubation with TheraMax in 96-well microplates [22].
Virucidal assays were conducted by incubating each stock virus with TheraMax (resulting in a 1:2 dilution of the product). The virus control consisted of diluting the virus stock with an equal part of cell culture medium. Incubation of these preparations was for 1 h at room temperature, followed by end point dilution titrations in 96-well microplates of MDCK cells using 4 microwells per dilution. Virus titres were calculated by the method of Reed and Muench [23] and were expressed as log10 CCID50/0.1 ml (the amount titrated).
Antiviral testing in mice
For animal studies, BALB/c mice (18–20 g; Charles River Laboratories, Wilmington, MA, USA) were anaesthetized by intraperitoneal injection of ketamine/xylazine (50/5 mg/kg), prior to infection by intranasal route with a 50 μl suspension of influenza A/NWS/33 (H1N1) virus. We have used this mouse-adapted strain of virus for other antiviral studies [21,24–26]. The infectious virus inoculation of 102.5 CCID50 per mouse equated to approximately 4 mouse 50% lethal doses. Because giving intranasal liquid (saline) to mice enhances influenza infection, the virus was pre-titrated in combination with intranasal saline treatments to determine the proper virus challenge dose to administer. To achieve the same lethality in animals not treated with intranasal liquid would have required about 100× more infectious virus. This phenomenon has not been well-studied, and we are not aware of any publications describing it. We believe that intranasal treatment with a physiological liquid helps to spread the virus infection in the lungs. All mice in all groups were treated intranasally twice a day for 4 days with either TheraMax or saline placebo (including the oseltamivir group) to cause the same severity of infection. Intranasal treatments were given in a 50 μl volume to anaesthetized mice twice a day (at 12 h intervals) through day 3 of the infection. In this model it is important to note that treatments beyond day 3 of the infection are risky and may kill the animals during anaesthesia. One group of mice received virus and TheraMax mixed together as the sole treatment. Oral treatments with oseltamivir were given twice a day for 5 days. Infection parameters included death, mean day of death and body weight determinations, and used 10–20 treated animals per group or 20 placebo-treated mice.
Five additional mice per group were treated and sacrificed on day 6 of the infection. Their lungs were removed and given a haemorrhage score from 0 (unaffected) to 4 (entire lung exhibiting a plum colour) in 0.5 unit increments. The lungs were then weighed and frozen at −80°C. Later the lungs were thawed, homogenized in 1 ml of cell culture medium and refrozen. The samples were titrated for virus by end point dilution in 96-well microplates of MDCK cells using four wells per dilution of sample. Tenfold dilutions of samples through eight dilutions (8 log10) were performed. Plates were evaluated for viral cytopathic effect in each well on day 3 and again on day 6 for confirmation. Virus titres were calculated by the end point dilution method of Reed and Muench [23]. Virus titres (log10 CCID50 per 0.1 ml [the amount titrated]) were converted to log10 CCID50/g of lung tissue by taking into account the reciprocal of the amount titrated, the lung weight and the dilution of the lung sample required to prepare the homogenate.
Pairwise comparisons of differences in numbers of animals surviving the infection were made using the two-tailed Fisher's exact test. The two-tailed Mann-Whitney U test was used to make pairwise comparisons of the mean day of death, lung haemorrhage scores and lung virus titres. The analyses were made with InStat® software (GraphPad Inc., San Diego, CA, USA). Comparisons were made between placebo and treated groups.
Results
TheraMax was tested for antiviral activity against several strains of influenza A and B viruses in MDCK cell culture (Figure 1A). A 1:20 dilution of TheraMax inhibited viral cytopathogenicity in MDCK cells by ≥50% for all viruses except influenza A/Victoria (H3N2), which was more resistant to inhibition. The A/Perth (H3N2) virus was the most sensitive virus to the inhibitory effect of TheraMax, followed by A/NWS (H1N1). Virucidal tests were performed with each stock virus combined with TheraMax (1:2 final dilution of TheraMax when combined with virus) for 1 h (Figure 1B). Compared to virus diluted 1:2 in cell culture medium and incubated for 1 h, there were no appreciable differences in resulting virus titres between TheraMax-treated and untreated samples. These data indicate that TheraMax does not possess virucidal activity. TheraMax at a 1:10 dilution caused approximately 50% reduction in neutral red dye uptake into uninfected MDCK cells, although cell monolayers were intact by microscopic inspection. Higher concentrations of TheraMax were not tested because they would dilute out the cell culture medium required for growth of the cells.
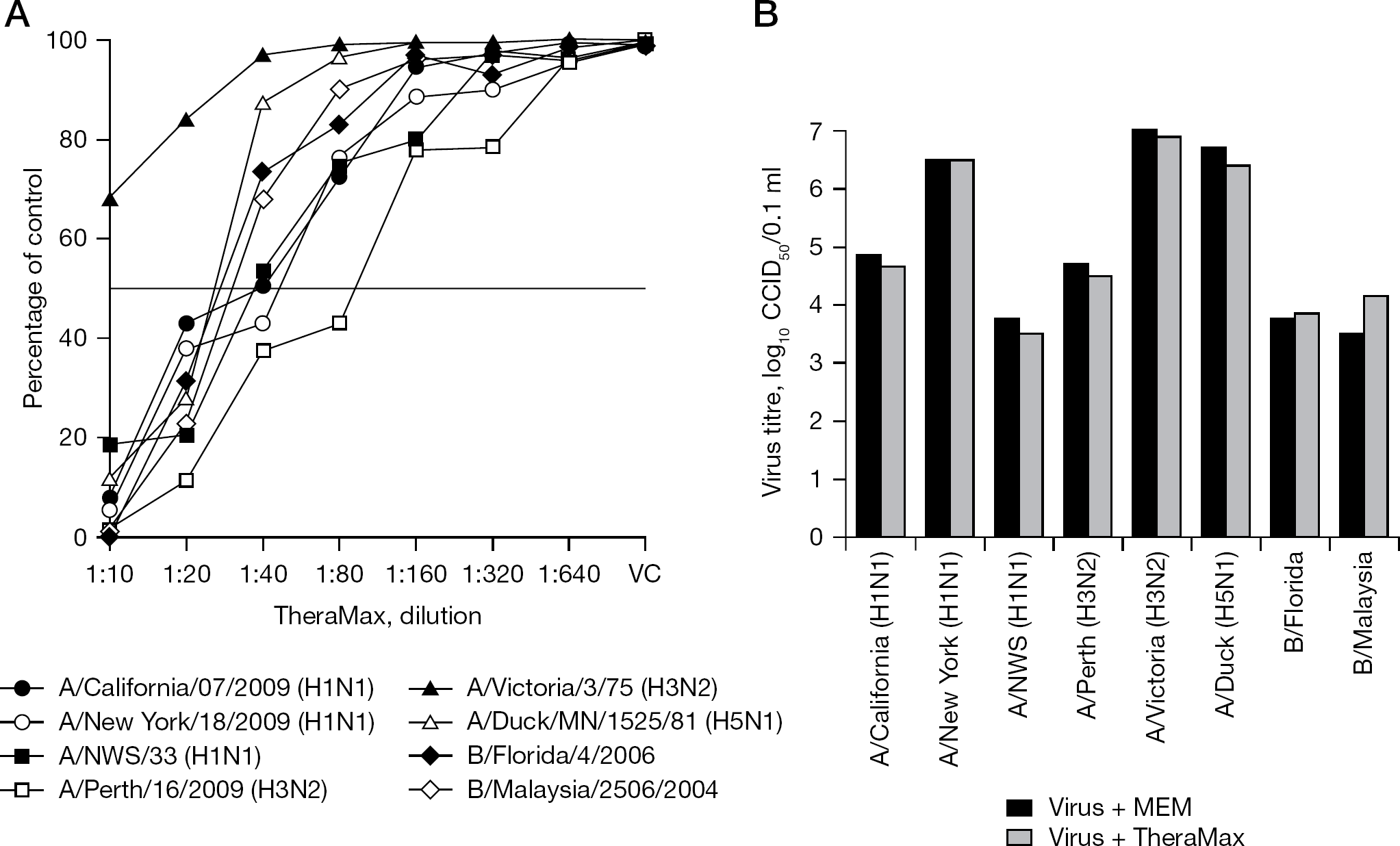
Assessment of antiviral and virucidal activities of TheraMax in cell culture
Prior to conducting antiviral studies with TheraMax in mice, the tolerability of the product by intranasal administration was first determined in uninfected mice (five animals/group). Intranasal treatments with TheraMax in a 50 μl volume were given twice a day at 12 h intervals for 4 days at 1×, 0.5× and 0.25× the concentration of the formulated product, and were compared to saline placebo. Weight loss in these groups, determined on day 5, were −2.3, −2.0, −1.3 and −2.0 g (placebo), respectively. The weight loss in all groups, including placebo, may be attributed to the stress of the intranasal administration.
In addition to assessing toxicity by weight loss and mortality measures, histopathology of lung and nasal tissue were performed on other similarly treated mice (five animals/group). Comparisons were made between mice receiving intranasal TheraMax and those receiving intranasal saline. At 12 h after the final treatment, the animals were euthanized and the lungs and heads were collected and placed in 10% buffered formalin. The tissues were sent to the Utah Veterinary Diagnostic Laboratory (Logan, UT, USA) for standard histological preparation and examination. Mouse heads were sectioned longitudinally, allowing for examination of the entire nasal cavity. There was no significant pathology in tissues from either test group (TheraMax or saline treated). A few animals in both treatment groups had several small clusters of neutrophils and macrophages adjacent to or in the lumen of small bronchioles. This is attributed to aspiration, evidenced by the presence of refractile foreign material associated with many of the cellular aggregates. Sheets of neutrophils and mucus were in the nasal cavity of TheraMax-treated animals, indicating an acute irritative reaction causing a mild suppurative rhinitis. Rhinitis was not evident in the saline-treated group. These toxicity evaluations established that TheraMax, as formulated (undiluted), could be safely administered intranasally to mice, but with accompanying mild irritation.
After completing the toxicity test, undiluted TheraMax was used to treat an influenza A/NWS/33 (H1N1) virus infection in mice (Table 1). TheraMax treatments that began 2 h prior to infection and those starting 12 h after infection could not prevent death. However, these treatments delayed the time to death by over 2 days, which was statistically significant compared to placebo. By comparison, oseltamivir treatments initiated at 2 h prior to infection protected 100% of animals from death. Intranasal infection of mice with a virus solution containing TheraMax resulted in no casualties, likely attributable to the compound blocking the initial infection.
Effects of TheraMax and oseltamivir on survival and lung infection parameters during an influenza A/NWS/33 (H1N1) virus infection in mice
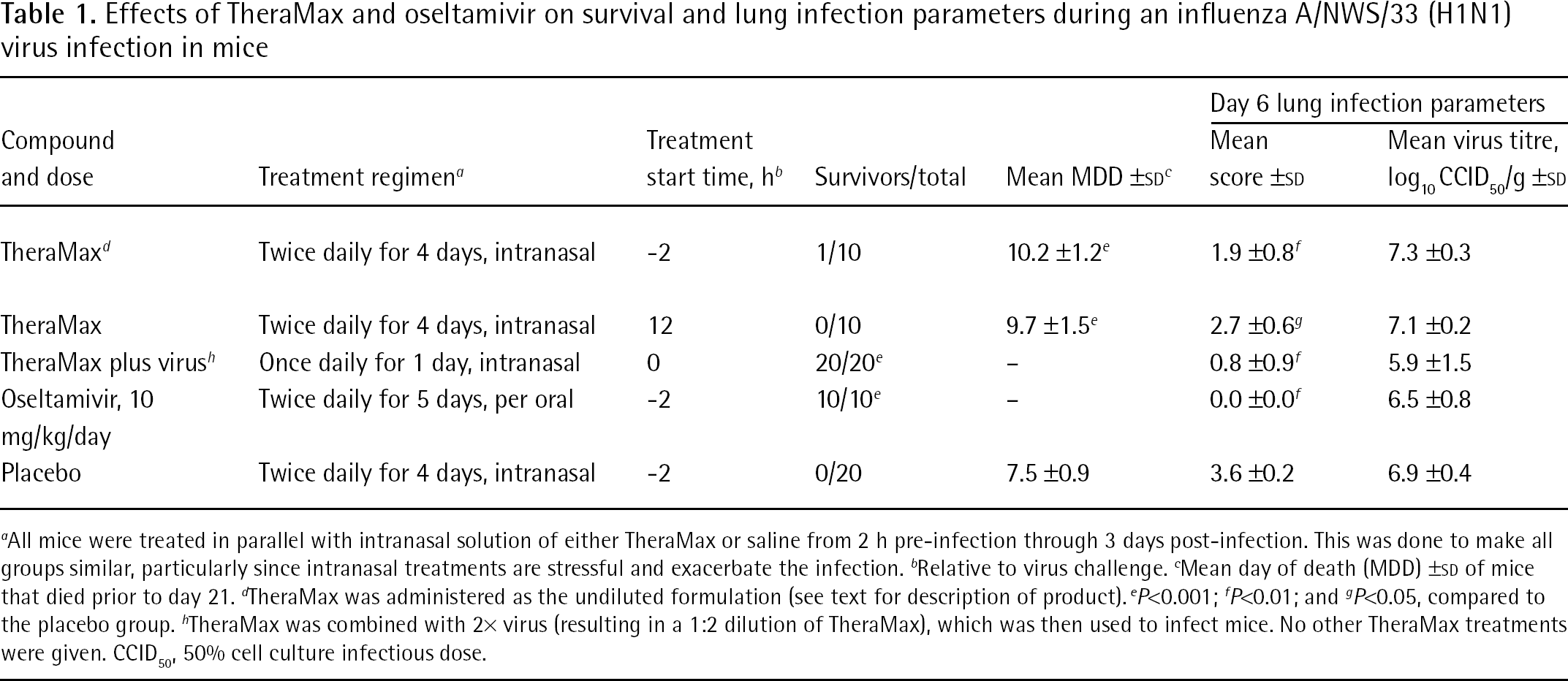
All mice were treated in parallel with intranasal solution of either TheraMax or saline from 2 h pre-infection through 3 days post-infection. This was done to make all groups similar, particularly since intranasal treatments are stressful and exacerbate the infection.
Relative to virus challenge.
Mean day of death (MDD) ±SD of mice that died prior to day 21.
TheraMax was administered as the undiluted formulation (see text for description of product).
P<0.001;
P<0.01; and
P<0.05, compared to the placebo group.
TheraMax was combined with 2× virus (resulting in a 1:2 dilution of TheraMax), which was then used to infect mice. No other TheraMax treatments were given. CCID50, 50% cell culture infectious dose.
Mean body weights of mice on days 0–20 of the infection are shown in Figure 2. Severe weight loss was evident in mice treated daily with TheraMax or placebo. The lone survivor in the −2 h TheraMax group began to gain weight on day 12. Minimal weight loss occurred in the group infected with virus solution containing TheraMax, and in the oseltamivir treatment group.
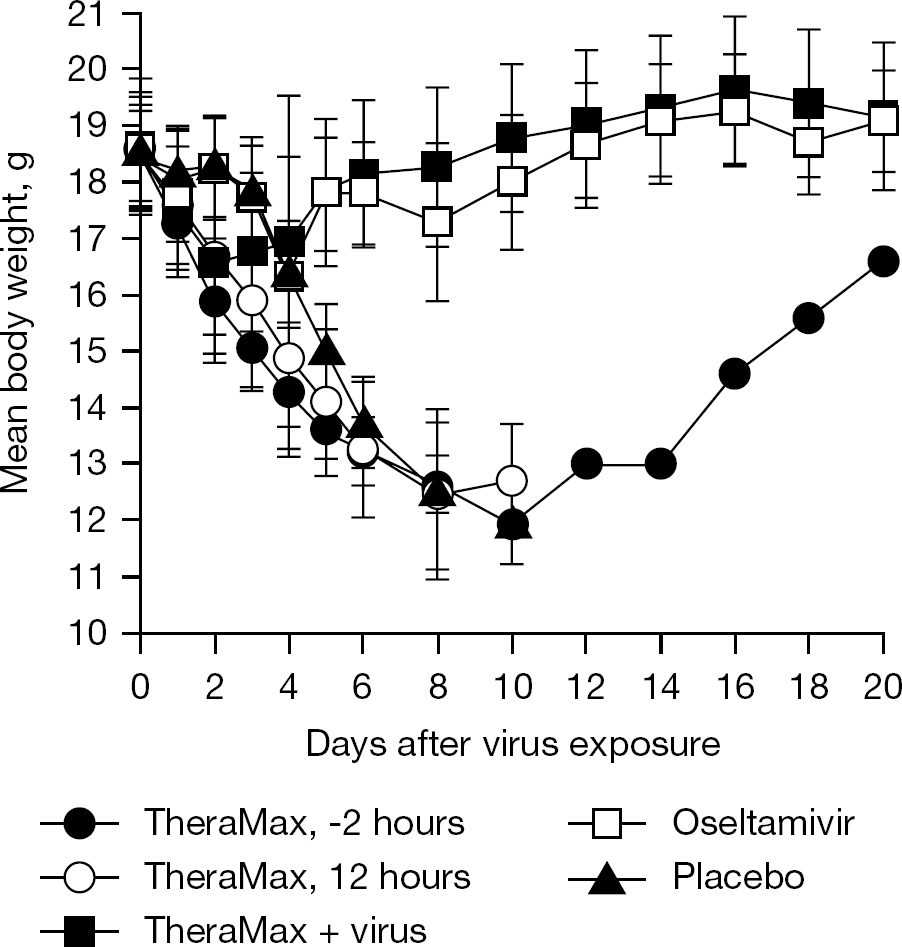
Effect of TheraMax and oseltamivir treatments on body weights during an influenza A/NWS/33 (H1N1) virus infection in mice
Lung infection parameters were determined on day 6 of the infection (Table 1). Lung haemorrhage scores were reduced by all treatments and were the lowest in the oseltamivir treatment group followed by the group infected with the mixture of virus plus TheraMax. Low lung haemorrhage scores appeared to be a good predictor of survival outcome. Virus titres in any treatment group were not appreciably reduced compared to placebo. The least amount of virus was detected in lungs of mice that were initially infected with virus plus TheraMax, but the results were not statistically significant due to variability. The virus titre results show TheraMax combined with virus did not eliminate the infectious virus, but must have reduced its infectivity titre at the time of infection in order to prevent death. Oseltamivir treatments resulted in minimal decreases in lung virus titre on day 6, even though the mice survived the infection. Although these results seem counter-intuitive, previous reports indicated that antiviral compounds such as oseltamivir and ribavirin reduced mortality from influenza infection without causing much of a decrease in lung virus titres on day 6 of the infection [27,28]. However, haemorrhage scores were significantly reduced compared to placebos.
Discussion
The results indicate that TheraMax exhibited antiviral activity in cell culture, which varied slightly from strain to strain of the virus; however, no virucidal activity was evident. The selectivity of TheraMax as an antiviral material in vitro was low. Regardless, TheraMax provided a benefit to mice infected with influenza virus infection by temporarily reducing the symptoms of an influenza virus infection, resulting in delays in the time to death. Because of the stress of intranasal treatment and the risk of death associated with treatment under anaesthesia, the treatments were not given beyond day 3 of the infection. Antiviral treatments with compounds administered orally or parenterally are generally given for 5–7 days, where the medication has a longer period of time to work.
Meaningful comparisons can be made between the results presented here and those of cyanovirin-N. Cyanovirin-N blocks virus entry into the cell as a result of it binding to viral haemagglutinin [29], as do components of green tea and elderberry extracts [7,9,20], although not necessarily by the same mechanism as cyanovirin-N. Intranasally administered cyanovirin-N was effective in preventing death when treatments began 4 h pre- to 6 h after infection, but mice died from infection when the first dose was given at 12 h [28]. TheraMax treatments starting at −2 or 12 h only delayed the time to death, indicating that TheraMax is less potent than cyanovirin-N, and that both materials are limited to prophylaxis or treatment very near the onset of an infection. Similar to the results here, cyanovirin-N did not significantly reduce lung virus titres on day 6 of the infection although lung haemorrhage scores were significantly reduced compared to placebo [28]. Lung virus titres in mice treated with cyanovirin-N were reduced at day 3 of the infection. We did not take a 3-day time point during TheraMax treatment.
The best effect with TheraMax was achieved when the medication was mixed directly with the infecting virus. There is no clinical relevance to this method of treatment; the results merely support the mode of action of TheraMax as an inhibitor of virus adsorption into cells. Because TheraMax induces an irritative reaction characteristic of mild rhinitis, there is also the possibility that some of its antiviral effects may be due to inducing a beneficial immune response. In support of this statement, researchers demonstrated that blood-derived monocytes from human donors that were treated with elderberry extract produced high levels of certain proinflammatory cytokines [30,31]. In addition, the anthocyanins and proanthocyanidins in elderberry extract possess antioxidant properties [32].
The study was not designed to identify which components in TheraMax might be more contributory to the antiviral activity in vivo. Since green tea and elderberry components are active via inhibition of virus adsorption [7,9,20], we hypothesize that they should contribute additively to efficacy.
To keep things in perspective, it is important to realize the difference between intranasal treatment of a mouse and a human. Because of the size of the mouse and the volume of liquid administered, much of the dose goes into the lungs. In contrast, treatment of humans with a small dose of TheraMax will be confined to the upper respiratory tract, and will drain down the esophagus to the stomach if not blown out the nose. Thus, TheraMax should have no effect on lower respiratory infections. It is envisioned that this product could ward off an infection at its very onset, but not have much of an effect once the infection is established. In contrast, oseltamivir, which is clearly superior in efficacy to TheraMax, should provide a benefit against established infections, but with recommendations to start treatment as early as possible after onset of the infection. Because TheraMax is planned to be marketed as an over-the-counter product, it will have greater accessibility to the general populace than oseltamivir, which is only available by prescription. Since clinical trials with TheraMax will likely never be conducted, individuals purchasing the product will ultimately decide if there is a benefit to using it.
Reports of treating humans with either green tea or elderberry extracts suggest their possible efficacy against influenza infections. In a nursing home study, patients who gargled with tea catechin extracts had a reduced incidence of infection compared to those who used a placebo [33]. In other studies, oral treatments with elderberry extract were reported to shorten the duration of influenza virus infections in humans [19,34]. These studies were limited in size and more research needs to be conducted for validation. Our investigation suggests that intranasal TheraMax, which is another formulated method of administering both green tea and elderberry extracts, provides a limited benefit to mice infected with influenza virus and may possibly be beneficial to humans.
Footnotes
Acknowledgements
These experiments were conducted in accordance with the approval of the Institutional Animal Care and Use Committee of Utah State University in the AAALAC-accredited Laboratory Animal Research Center. The investigators and animal technicians followed the National Institutes of Health Guide for the Care and Use of Laboratory Animals.
Therabiogen, Inc. provided the funds to support this research. The presentation and interpretation of the data are those of the authors. The authors declare no competing interests.
