Abstract
Background:
Certain nucleoside, nucleotide and pyrophosphate analogues may be useful for treating severe complications arising as a result of virus dissemination following smallpox (live vaccinia virus) vaccinations, especially in immunocompromised individuals. We used an immunosuppressed hairless mouse model to study the effects of 10 antiviral agents on progressive vaccinia infections.
Methods:
Hairless mice were immunosuppressed by treatment with cyclophosphamide (100 mg/kg) every 4 days starting 1 day prior to vaccinia virus (WR strain) infection of wounded skin. Topical treatments with antiviral agents were applied twice a day for 7 days starting 5 days after virus exposure.
Results:
Topical 1% cidofovir cream treatment was effective in significantly reducing primary lesion severity and decreasing the number of satellite lesions. Topical 1% cyclic HPMPC and 1% phosphonoacetic acid were not quite as active as cidofovir. Ribavirin (5%) treatment reduced lesion severity and diminished the numbers of satellite lesions, but the mice died significantly sooner than placebos. 2-Amino-7-[(1,3,-dihydroxy-2-propoxy)methyl]purine (compound S2242; 1%) moderately reduced primary lesion sizes. Ineffective treatments included 5% arabinosyladenine, 1% arabinosylcytosine, 1% 5-chloro-arabinosylcytosine, 5% arabinosylhypoxanthine 5-monophosphate and 5% viramidine.
Conclusions:
Of the compounds tested, topically applied cidofovir was the most effective treatment of cutaneous vaccinia virus infections in immunosuppressed mice. Topical treatment with cidofovir could be considered as an adjunct to intravenous drug therapy for serious infections.
Introduction
Smallpox vaccination ceased as a general practice around 1980, but has escalated in military personnel and healthcare workers in recent years in response to the perceived threat of deliberate release of smallpox or monkeypox virus by terrorist groups [1,2]. With renewed vaccination, new cases of serious vaccination complications will likely arise. Normally the smallpox vaccination, when given intradermally on the upper arm, remains localized and produces small pustules that become confluent to form a large vesicle. The vesicle eventually scabs over and falls off [3]. Complications arising following smallpox vaccination may include dissemination of the infection to other sites on the body (such as other areas of the skin or the eyes), progressive vaccinia, eczema vaccinatum and/or generalized vaccinia infection [4]. Many of the more serious conditions following vaccination are the result of the individual being in an immunosuppressed state (such as by HIV infection), or due to genetic immunodeficiency, age (young children are more susceptible than adults) or chemotherapy [4]. In 2008 the child of a vaccinated soldier became seriously infected from disseminated vaccinia [5]. Fortunately the infant survived the infection because of effective antiviral treatment.
Mouse models have been developed to study antiviral treatments of severe progressive vaccinia virus infections. Severe combined immunodeficient (SCID) mice infected parenterally or intranasally with vaccinia virus die from infection despite antiviral treatment with cidofovir [6,7], although treated mice live considerably longer than placebos. Mice injected intravenously in the tail develop progressively worse tail lesions before they die [6]. The other routes of infection do not lead to the appearance of external lesions. We developed a cutaneous vaccinia virus infection model where immunosuppression (using cyclophosphamide) of normally immunocompetent hairless mice leads to progressive disease [8]. Primary lesions increase in size and severity, satellite lesions form, and the mice later die from systemic infection. Treatment with cidofovir, either topically or parenterally, can reduce primary and satellite lesions and delay the time to death. Without the use of cyclophosphamide to induce immunosuppression, normal mice treated with cidofovir develop small, non-progressive lesions and the animals do not die from the infection [8,9]. Athymic nude mice have also been used for studying antiviral treatments of vaccinia virus infection [9,10]. In that model the lesions are not quite as severe as with cyclophosphamide immunosuppression, but treated animals die anyway from the infection.
Over the years a number of antiviral substances have been discovered that exhibit anti-poxvirus activity in mouse models [11,12]. The majority of the compounds were tested by oral or parenteral route. We discovered that cidofovir was more effective topically than parenterally in treating vaccinia virus cutaneous infections in immunosuppressed hairless mice [8]. For this reason, we initiated efficacy studies of several other anti-poxvirus agents administered by topical application. They included arabinosyladenine (Ara-A; vidarabine), arabinosylcytosine (Ara-C; cytarabine), 5-chloro-arabinosylcytosine (5-Cl-Ara-C), arabinosylhypoxanthine 5′-monophosphate (Ara-HxMP), cyclic cidofovir (cyclic HPMPC), phosphonoacetic acid (PAA), ribavirin, 2-amino-7-[(1,3,-dihydroxy-2-propoxy)methyl]purine (S2242) and viramidine. Compounds selected were based upon availability and the quantity of each that could be obtained, as well as by prior antiviral activity against orthopoxvirus infections in animals [11,12]. Two of the compounds, 5-Cl-Ara-C and viramidine, failed to meet the last criterion: viramidine had never been tested against poxvirus infections in vivo and 5-Cl-Ara-C had not been found to be active [13]. Viramidine is a prodrug of ribavirin [14] that was expected to perform similarly to ribavirin. We considered testing phosphonoformic acid (foscarnet), an approved drug for human cytomegalovirus infections, which is related to PAA. However, no in vivo orthopoxvirus studies have been published, and the in vitro literature indicated that phosphonoformic acid was less active than PAA against vaccinia virus [15]. The investigation revealed that many of the compounds showed a degree of activity by topical route, but cidofovir emerged as the most effective substance.
Methods
Animals
Female 7–8 week old (approximately 23–25 g) specific pathogen-free SKH-1 hairless mice were obtained from Charles River Laboratories (Wilmington, MA, USA). The animals were quarantined at least 48 h prior to use and maintained on standard rodent chow and tap water. These animals were not available in large numbers from the supplier; thus, the number of mice per group was smaller than what we typically use for studies with other strains of mice.
Virus
Vaccinia virus (WR strain) was purchased from the American Type Culture Collection (Manassas, VA, USA). The virus was propagated in African green monkey kidney (MA-104) cells for use in these studies.
Compounds
Cidofovir and cyclic HPMPC were provided by Gilead Sciences (Foster City, CA, USA). The following were obtained from Sigma–Aldrich (St Louis, MO, USA): Ara-A, Ara-C, 5-Cl-Ara-C and PAA. Aventis Pharma (Frankfurt, Germany) kindly provided S2242. Ara-HxMP, ribavirin and viramidine were acquired from ICN Pharmaceuticals (Costa Mesa, CA, USA) before the company went out of business. Dermovan® (Owen Laboratories, San Antonio, TX, USA) was purchased from a local pharmacy for preparing topical cream containing cidofovir [8,16] or the other antiviral compounds. Each compound was dissolved or suspended (that is, Ara-A) in water at a concentration of 25 mg/ml, then combined with Dermovan to prepare a 1% cream formulation. Dermovan combined with water served as the placebo control.
Experimental design
Mice were anaesthetized with ketamine (100 mg/kg) by intraperitoneal injection. They were scratched, using a 25-gauge needle, sufficiently to penetrate the dermal layer in the hip and shoulder areas on one side of the body. The area of each scratched site was approximately 25 mm2 (5 mm × 5 mm) and was 4–5 scratches in one direction. A 25 μl volume of virus (containing approximately 2.5×105 plaque forming virus units) was placed on each wound area and remained there while the animals rested under the influence of the anaesthesia. Immunosuppression was accomplished by treating the mice intraperitoneally with cyclophosphamide (100 mg/kg/day) once every 4 days until all animals died, starting 1 day before virus exposure. Creams (approximately 50–100 μl) were applied to each lesion site with a spatula twice a day for 7 days starting 5 days after virus challenge. Satellite lesions, as they formed, were treated in a similar manner. Cidofovir (1% cream) was evaluated in each experiment as a positive control compound based upon its previous activity in the mouse model [8].
Three parameters were used to evaluate disease progression or its inhibition. First, deaths were recorded daily for as long as the animals survived, which, in these experiments was <35 days. In prior studies uninfected hairless mice have been treated with cyclophosphamide for up to 45 days, and the animals remained healthy [8]. Second, the primary lesion area was determined in mm2 (length × width). Satellite lesions that merged with the primary lesion were not included in the primary lesion area. Third, the number of satellite lesions per mouse was counted each day. Satellite lesions could occur anywhere on the body. Photographs of infected mice at various stages of disease progression have been published [8].
Mice were tagged by toe clipping and accounted for individually. Their final lesion areas and satellite lesion numbers were maintained for counting purposes through day 21 of the experiment, with the exception of the ribavirin treatment group that died much earlier than placebos. There were 8 mice in each group treated with antiviral compound and 9 or 10 animals in the placebo group. Mice treated with Ara-A, Ara-HxMP, ribavirin and viramidine constituted a separate experiment than the one involving the other compounds (the positive control drug cidofovir was used in each study).
Statistical evaluations
Survival curves for all groups in each experiment were evaluated by the Mantel-Cox log-rank test. Where significance was seen by the above method, pairwise comparisons of survival curves were made between placebo and compound-treated groups by the Gehan-Breslow-Wilcoxon test. The two-tailed Mann-Whitney U test was used to analyse mean primary lesion areas and mean numbers of satellite lesions between placebo and compound-treated groups.
Results
Treatment of infection with cidofovir, cyclic HPMPC and S2242
The effects of topical treatment with cidofovir, cyclic HPMPC and S2242 on survival and vaccinia lesions are presented in Figure 1. Survival curve analysis indicated no significant differences in survival among the groups, although the cidofovir group lived the longest (Figure 1A). Mean ± SD day of death values obtained for treatment with these compounds were 22.4 ±4.8, 21.4 ±2.5 and 20.5 ±4.0 days, respectively, and the value for placebo was 19.8 ±2.9 days. Treatment with cidofovir and cyclic HPMPC significantly reduced primary lesion sizes (Figure 1B). Cidofovir appeared to be more active than cyclic HPMPC in this experiment, although the differences were not statistically significant. S2242 caused minimal reductions in lesion severity compared to placebo, with some significance on days 9, 13, 15 and 17. Treatment with cidofovir and cyclic HPMPC significantly reduced the numbers of satellite lesions on certain days of the infection (Figure 1C). Cidofovir was somewhat more active than cyclic HPMPC, but the differences between these groups were not statistically significant. S2242 caused reductions in numbers of satellite lesions compared to placebo on days 17–25, but the results were not statistically significant.
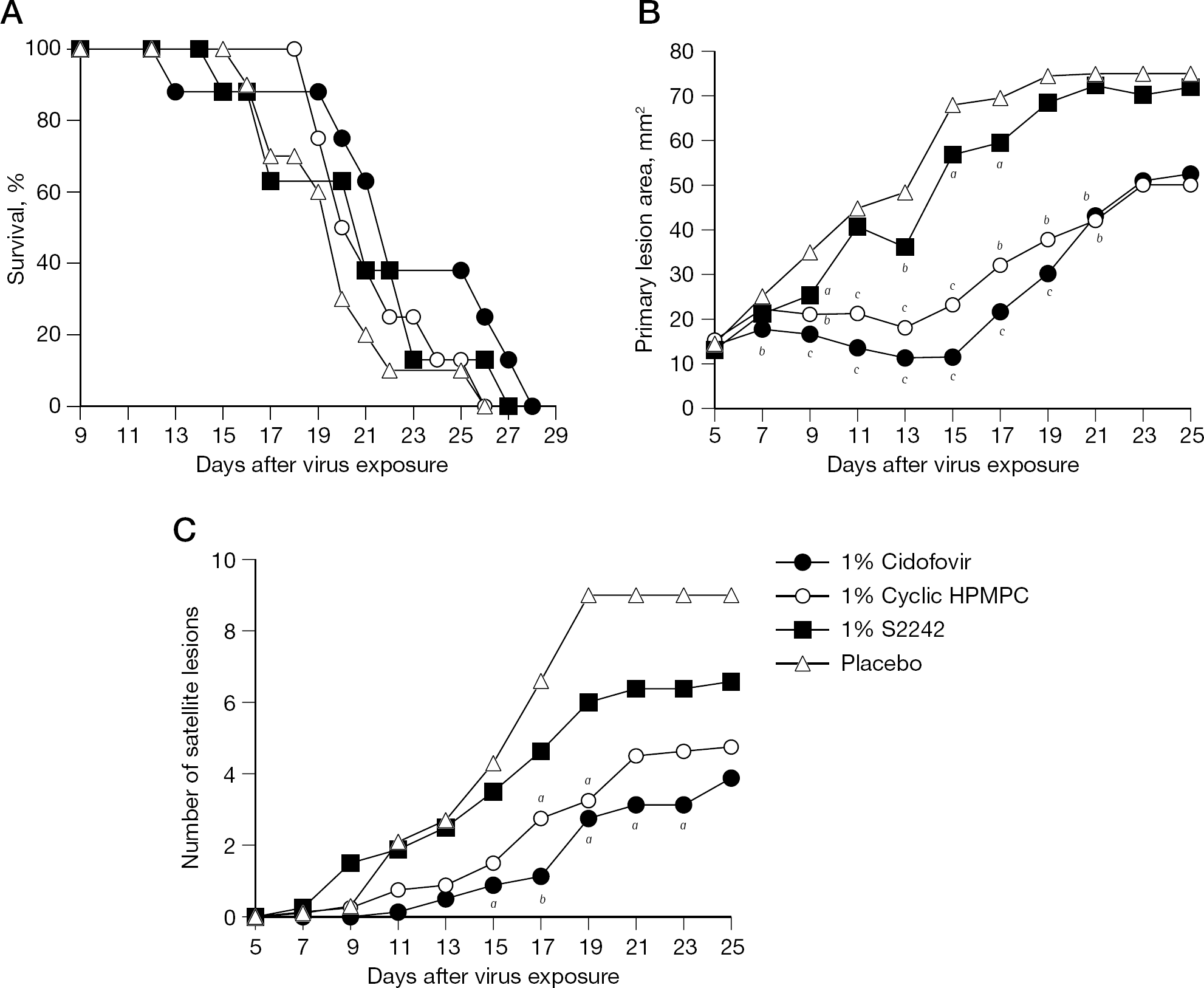
Treatment of a cutaneous vaccinia virus infection in immunosuppressed hairless mice with topically applied cidofovir, cyclic HPMPC or S2242
Treatment of infection with cidofovir, Ara-C, 5-Cl-Ara-C and PAA
The results presented here for cidofovir and placebo are the same as in Figure 1 because they are from the same experiment. The presentation was divided into two parts (Figures 1 and 2) for clarity. Survival curve analysis did not demonstrate statistically significant differences among treatment groups, although cidofovir and PAA appeared to delay the time to death (Figure 2A). Mean ±SD day of death values obtained for treatment with cidofovir, Ara-C, 5-Cl-Ara-C and PAA were 22.4 ±4.8, 20.2 ±2.5, 20.3 ±1.5 and 23.1 ±5.0 days, respectively, and the value for placebo was 19.8 ±2.9 days. Treatment with cidofovir and PAA significantly reduced lesion severity (Figure 2B). Cidofovir was somewhat more active than PAA in this experiment, but differences were not statistically significant through day 15. After day 15, the activities of PAA waned. Treatments with Ara-C and 5-Cl-Ara-C were ineffective. Treatment with cidofovir and PAA significantly reduced the numbers of satellite lesions (Figure 2C). The two compounds had nearly the same effect on reducing satellite lesion formation. Ara-C and 5-Cl-Ara-C did not inhibit satellite lesion formation compared to placebo.
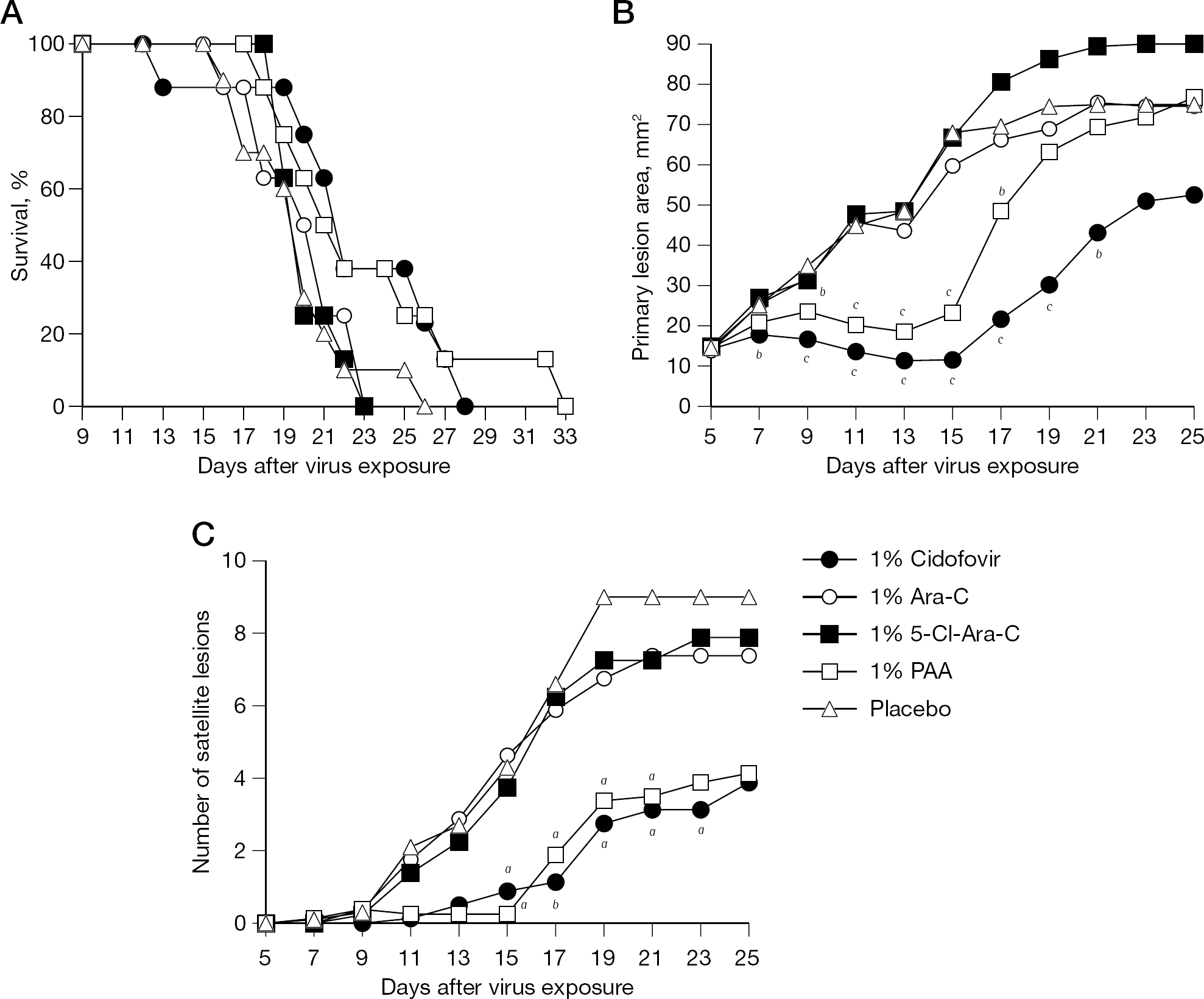
Treatment of a cutaneous vaccinia virus infection in immunosuppressed hairless mice with topically applied cidofovir, Ara-C, 5-Cl-Ara-C or PAA
Treatment of infection with cidofovir, ribavirin, viramidine, Ara-A and Ara-HxMP
The mortality results of this experiment are presented graphically in Figure 3A. Survival curve analysis showed statistical significance among groups, attributed to the ribavirin treatment group. Ribavirin treated mice lived a shorter length of time than did the placebo group (P<0.001), indicating that the treatment was deleterious and enhanced the infection. Treatment with viramidine, which is structurally related to ribavirin, also seemed to cause mice to die sooner than those treated with placebo, but not significantly. The mean ±SD day of death calculations for treatment with cidofovir, ribavirin, viramidine, Ara-A and Ara-HxMP were 19.3 ±2.2, 13.5 ±0.8, 16.6 ±3.4, 18.1 ±2.3 and 17.9 ±2.0 days, respectively, and the value for placebo was 18.1 ±2.4 days. Ribavirin treatment significantly reduced primary lesion areas (Figure 3B); however, after day 15 the ribavirin-treated mice were all dead. Lesions were more severe in the Ara-A group than the placebo group. Ara-HxMP and viramidine treatments were ineffective, as was treatment with cidofovir in this experiment. Cidofovir and ribavirin both suppressed satellite lesion formation, with ribavirin being highly effective up until the treated mice died (Figure 3C). Viramidine's inhibitory effect on satellite lesion numbers was not significant, and Ara-A and Ara-HxMP were not active. The infection in this experiment appeared to be more severe than that reported in Figures 1 and 2. This was based upon the higher burden of satellite lesions and the lesser effect of cidofovir.
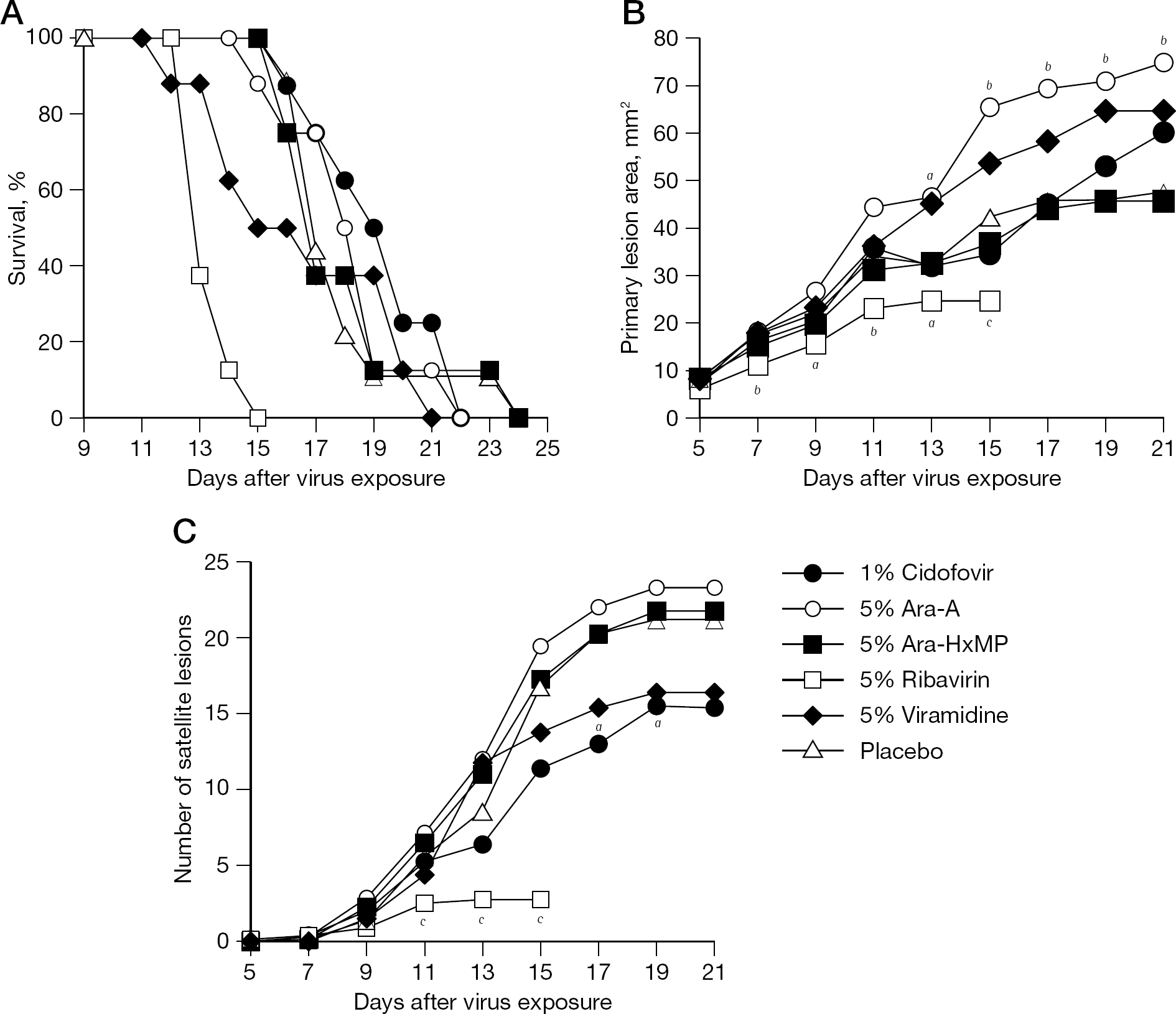
Treatment of a cutaneous vaccinia virus infection in immunosuppressed hairless mice with topically applied cidofovir, Ara-A, Ara-HxMP, ribavirin or viramidine
Discussion
These experiments were conducted with treatments starting 5 days after infection. At that time, high virus titres have been detected in the skin [8] and primary lesions were on the verge of undergoing rapid expansion (Figures 1B, 2B and 3B) [8]. Because of this late time of treatment initiation, cidofovir was not very effective in delaying the time to death. Much better protection was achieved when treatments were initiated 1 day after infection [8]. Cidofovir treatments initiated 5 days after infection were able to reduce primary lesion development and/or numbers of satellite lesions significantly in these studies, and as reported previously [8]. The delayed treatment regimen employed in these experiments has relevance to the treatment of disseminated vaccinia in humans.
Besides cidofovir, compounds exhibiting antiviral activity included cyclic HPMPC, PAA, ribavirin and, to a much lesser extent, S2242. It is not surprising that cyclic HPMPC showed antiviral activity in the model, as it is a prodrug form of cidofovir that converts to cidofovir intracellularly [17]. PAA has been shown to be effective topically in treating vaccinia virus cutaneous lesions in rabbits [18]. The beneficial activity of ribavirin in reducing primary lesion sizes and satellite lesion numbers was overshadowed by the finding that the treatment accelerated the time to death. This might be due to a toxic effect that weakened the animals or was possibly due to immunosuppressive activity of ribavirin at this dose [19]. Parenteral ribavirin was effective against other vaccinia and cowpox virus infections in immunocompetent mice [20,21]. S2242 was much less inhibitory than expected, based upon its high activity when administered parenterally against cowpox and vaccinia virus infections in mice, including SCID mice [22,23].
Compounds that were ineffective in the model included Ara-A, Ara-C, 5-Cl-Ara-C, Ara-HxMP and viramidine. Of these, Ara-C and 5-Cl-Ara-C did not show efficacy by parenteral injection against respiratory cowpox virus infections in mice, whereas Ara-A was only weakly active, and Ara-HxMP and viramidine were not tested [13]. Ara-C was effective parenterally in treating vaccinia tail lesions in mice [20]. Ara-A and Ara-HxMP were active topically in treating vaccinia keratitis in rabbits [24,25], and topical Ara-A was effective in treating vaccinia cutaneous lesions in rabbits [26]. We are not aware of activity seen with 5-Cl-Ara-C or viramidine in other animal models of orthopoxvirus infections.
In the first week of the vaccinia infection in immunosuppressed hairless mice, the majority of the virus burden was in the skin [8]. Reducing this virus burden by topical treatment will slow down disease progression and delay the time to death. Topical treatment of vaccinia complications in humans should be beneficial by providing a high drug concentration at the site of infection. Topically applied cidofovir has already been used to treat molluscum contagiosum in children [16]. More serious vaccinia virus infections that become systemic could be treated by a combination of both topical and parenteral routes.
These studies lay the groundwork for testing cidofovir in combination with some of the other antiviral agents to potentially achieve a greater effect. Ideally, compounds should have different modes of antiviral action to exert synergy. The majority of the compounds that were tested (including cidofovir) are DNA polymerase inhibitors. Thus, two of them in combination may only produce an additive effect. Ribavirin appears to be inhibitory to poxvirus replication due to the inhibition of cellular inosine monophosphate dehydrogenase [27], thus may combine synergistically with cidofovir. However, a lower dose of ribavirin will need to be used to avoid deleterious effects to the animals. An interesting compound to explore in combination with cidofovir is PAA. PAA is a specific inhibitor of virus-induced DNA polymerases by targeting the pyrophosphate binding site [28], which prevents the addition of deoxynucleotides to the elongating DNA chain [29]. Whether its target overlaps or is the same as that of the active form of cidofovir (cidofovir diphosphate) will require additional research.
The vehicle used for topical application of antiviral agents often influences activity [30–32]. Differences in compound size, solubility and ability to diffuse from the topical vehicle and penetrate into the skin are factors that can affect efficacy. Only one vehicle, Dermovan, was used for formulating the compounds used here. Evaluating multiple vehicles with each of these compounds was beyond the scope of this research. By using a common vehicle, we assumed that the results are adequate to identify the compounds that are active and those that are not. The data support the conclusion that cidofovir is the most active of the compounds tested. Topical cidofovir should be considered as a treatment option for cutaneous vaccinia virus infections, and could be used in combination with intravenous administration for more serious progressive infections.
Footnotes
Acknowledgements
This work was supported by Contracts NO1-AI-15435 and NO1-AI-30063 (awarded to the Southern Research Institute, Birmingham, AL, USA) from the Virology Branch, National Institute of Allergy and Infectious Diseases, National Institutes of Health. The contents of this article do not necessarily reflect the position or policy of the government and no official endorsement should be inferred. The investigators adhered to the ‘Guide for the Care and Use of Laboratory Animals’, prepared by the Committee on Care and Use of Laboratory Animals of the Institute of Laboratory Animal Resources, National Research Council, and used facilities fully accredited by the Association for Assessment and Accreditation of Laboratory Animal Care International.
The authors declare no competing interests.
