Abstract
Background:
The recent emergence of pandemic influenza virus H1N1v stresses the need for the development of new anti-influenza drugs.
Methods:
Host proteases responsible for viral haemagglutinin (HA) cleavage are attractive targets for such drugs. Aprotinin, a natural 58-amino-acid polypeptide from bovine lungs, was chosen for this purpose because it is a drug already approved for human use as an antiprotease compound to treat pancreatitis and bleeding, and because it inhibits a wide spectrum of serine proteases, some of which are involved in influenza virus activation.
Results:
First, we show that HA of pandemic H1N1v was intensively cleaved and activated in different host systems (human tracheo-bronchial epithelium, human intestinal Caco-2 cells and chicken embryonated eggs). Second, aprotinin inhibited HA cleavage and replication of pandemic influenza virus H1N1v in all host systems, including human tracheo-bronchial epithelium. Third, aprotinin did not induce any apparent toxic side effects in these hosts.
Conclusions:
Aprotinin can be considered a promising drug against the novel H1N1v pandemic influenza virus.
Introduction
More than 30 years ago, it was found that influenza viruses require activation by specific proteases to maintain multicycle replication [1,2]. Virus activation is accomplished by cleavage of the precursor haemagglutinin (HA) protein HA0 (molecular weight 75 kD) into two disulphide-linked fragments, HA1 (55 kD) and HA2 (20 kD) [1,2]. The activating enzymes are trypsin-like serine proteases that cleave HAs of all subtypes possessing a single arginine at the cleavage site [3] or furin-like proteases that cleave subtypes H5 and H7 at an Arg/Lys-X-Arg motif [4]. The presence of trypsin-like proteases activating human epidemic viruses was demonstrated in many host systems, such as cultured cells [5–8], chicken eggs [9], mouse lungs [10] and human respiratory epithelium [11,12]. In addition, tryptase Clara, plasmin, miniplasmin, clotting factor Xa, human airway trypsin-like protease (HAT), transmembrane protease serine 2 (TMPRSS2) and transmembrane protease serine 4 (TMPRSS4) have been indentified as influenza virus activating proteases (IAPs) [5,6,10,13–16].
Influenza virus HA cleavage is a suitable target for antiviral therapy [17]. HA cleavage and multicycle replication of different laboratory strains of human epidemic influenza A and B viruses in cultured cells [10], chicken embryonated eggs [9], mouse respiratory tract [10] and human respiratory epithelium [12] was shown to be reduced by aprotinin, a natural inhibitor of a wide spectrum of trypsin-like proteases [18]. Whether the novel swine-origin pandemic virus H1N1v is similarly inhibited with aprotinin is not known. HA of pandemic H1N1v virus was derived from a swine virus, and similarly to human epidemic viruses it possessed a monobasic Arg-containing cleavage site [19,20]. At the same time, HA of pandemic influenza H1N1v virus appeared to differ remarkably from an epidemic seasonal one in primary structure and glycosylation pattern, displaying approximately 25% amino acid divergence and more than twofold reduction in the number of glycosylation sites. For these reasons, intracellular pathways and recognition with specific host proteases could be distinct for pandemic and epidemic HAs. These virus-specific HA peculiarities raise the important question of whether pandemic virus has similar sensitivity to host-activating proteases and is susceptible to aprotinin inhibition like human epidemic influenza viruses. To throw light on this issue, we investigated here levels of proteolytic activation of the novel swine-origin pandemic H1N1v influenza virus and its sensitivity to this antiprotease drug in different host systems. First, the HA of pandemic virus was found to be intensively cleaved and activated in all host systems studied, including human tracheo-bronchial epithelium (HTBE). Second, this HA cleavage was remarkably sensitive to aprotinin, and pandemic H1N1v virus multicycle replication was effectively suppressed by this drug.
In the first set, we studied the efficacy of aprotinin in Caco-2 cells infected with pandemic influenza virus A/Hamburg/05/2009 (H1N1v) (A/HB/09) and, for comparison, a laboratory-passaged strain of human epidemic virus A/PR/8/34 (H1N1). Caco-2 cells have been shown earlier to cleave the HA and to maintain multicycle replication of human laboratory viruses of subtypes H1N1 and H3N2 [7,8]. In the present study, the cells were infected at a multiplicity of infection of 1 and incubated in medium with or without aprotinin. Progeny virus was analysed by PAGE to test for HA cleavage and by titration in Madin–Darby canine kidney (MDCK) cells to test for infectivity. The results shown in Figure 1A indicate that viruses grown without aprotinin contained predominantly cleaved HA1/HA2 and possessed maximal infectivity that was not increased after trypsin treatment. By contrast, both viruses grown in Caco-2 cells in the presence of aprotinin had predominantly uncleaved HA0 and possessed low infectivity, which was significantly increased (by a factor of 103) after trypsin treatment (Figure 1A). These observations indicate that proteolytic activation of pandemic H1N1v virus, as well as a human epidemic laboratory strain A/PR/8/34 activation, is effectively suppressed with aprotinin.
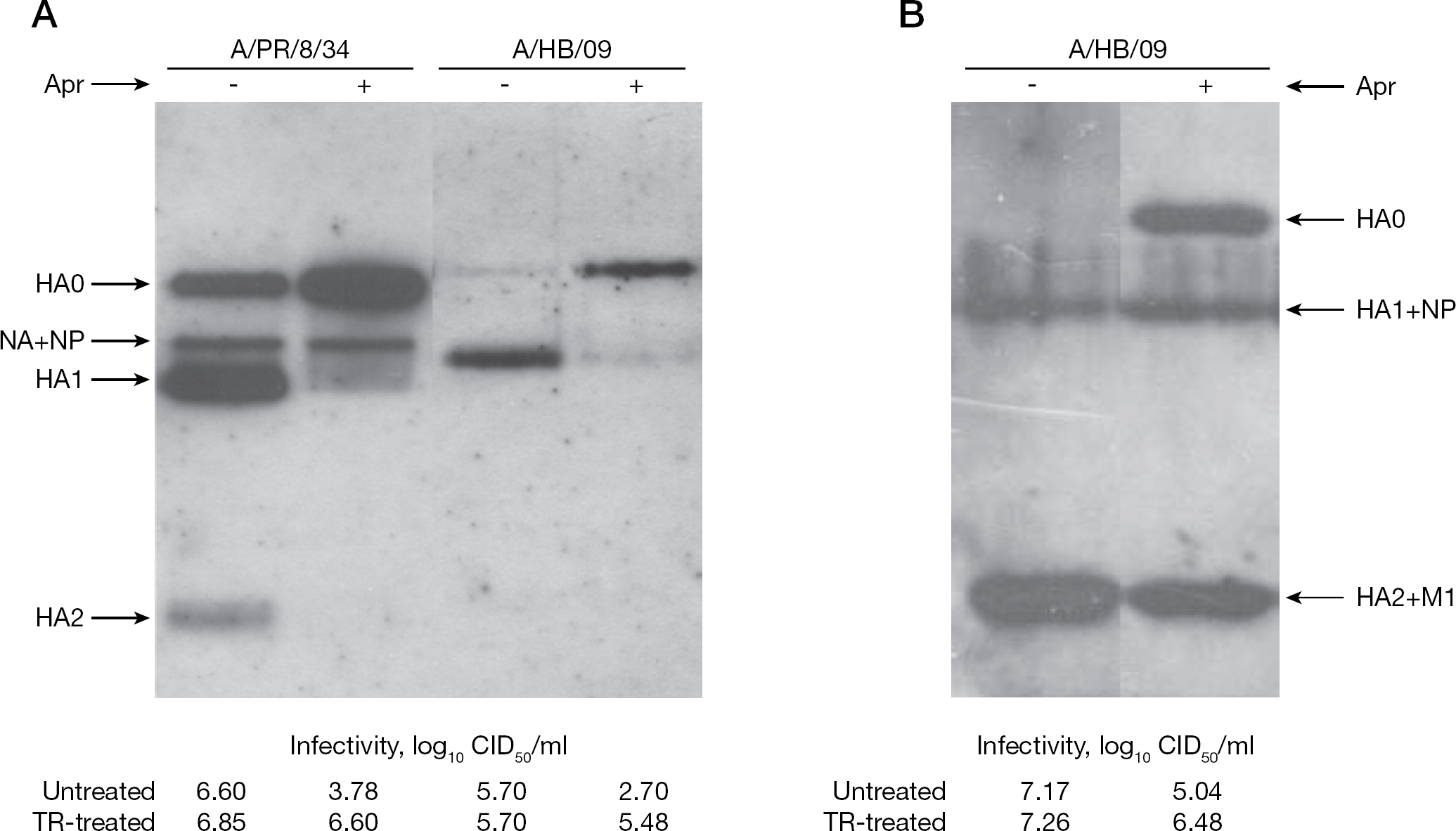
Polypeptide pattern and infectivity of pandemic influenza virus H1N1 grown in Caco-2 cells and chicken eggs in the presence of aprotinin
Tissues of embryonated chicken eggs are well-known to cleave HA0, and both human and avian influenza viruses are effectively activated and, thus, maintain multicycle replication in this host system. It was then of interest to find out whether aprotinin suppresses H1N1v virus activation in embryonated eggs. A/HB/09 virus was injected into the allantoic cavity either alone or together with aprotinin, and 24 h post-infection progeny virus accumulated in allantoic fluid was assayed. Data of a typical experiment are shown in Figure 1B. Virus grown in non-treated eggs lacked the uncleaved HA0 and predominantly contained cleaved HA1/HA2. HA1 and HA2 subunits of the A/HB/09 virus were poorly distinguished in PAGE-western blot because these polypeptides in this virus strain comigrated in the gel with nucleoprotein (NP) and matrix (M1) proteins, respectively, and overlapped in western blot detection procedure. According to the observed polypeptide pattern, the virus was found to display high infectious titres, both without and with trypsin treatment (Figure 1B). In aprotinin-treated eggs, a significant amount of non-cleaved HA0 was detected. The aprotinin-treated virus progeny showed reduced infectivity, which was markedly increased after trypsin treatment in vitro. Importantly, the total virus yield in allantoic fluid, measured by virus infectivity titration after in vitro trypsin treatment, was approximately 25× lower in aprotinin-treated eggs than in non-treated ones. This reduction indicated a depressed virus replication in eggs treated with aprotinin. Similar suppression of the viral HA0→HA1+HA2 cleavage under aprotinin treatment was observed in chicken eggs infected with X-179A reassortant virus containing HA and NA genes of A/California/04/2009 (H1N1) virus and all internal genes from A/PR/8/34 (H1N1) virus (OPZ et al., data not shown).
One of the most interesting medical aspects of this study concerns the level of pandemic virus proteolytic activation in human respiratory epithelium and the effect of aprotinin on virus replication in this host. For this purpose we used differentiated cultures of HTBE. These cultures, which are grown on porous membranes under air–liquid interface conditions, are pseudo-stratified and polarized, contain ciliated, secretory, and basal cells and are covered with the mucus surfactant blanket [21]. Thus, they represent an adequate model for studies on mechanisms of influenza virus infection that take place in human respiratory epithelium in vivo [11,22]. HTBE cells infected with A/HB/09 virus were treated with aprotinin, and virus growth dynamics and polypeptide composition were studied. Typical results are shown in Figure 2A and 2B. Influenza virus grown in HTBE cells without aprotinin contained predominantly cleaved HA1/HA2 and was characterized by high infectivity (Figure 2A). By contrast, virus grown in HTBE cultures in the presence of aprotinin contained predominantly uncleaved HA0 and had low infectivity that increased, however, by a factor of 20 after trypsin treatment (Figure 2A). This reduced factor of trypsin activation of virus grown in HTBE with aprotinin in comparison to analogous virus sample obtained in Caco-2 (Figure 1A) could be explained by a less effective action of trypsin on virus suspended in mucus surfactant of HTBE blanket. Finally, we have compared multicycle growth of A/HB/09 virus in HTBE cultures incubated with and without aprotinin. Addition of aprotinin to culture fluid was found to delay virus growth and reduce virus accumulation (Figure 2B). Taken together these data show that aprotinin suppressed proteolytic activation and multicycle replication of swine-origin pandemic virus H1N1v in HTBE.
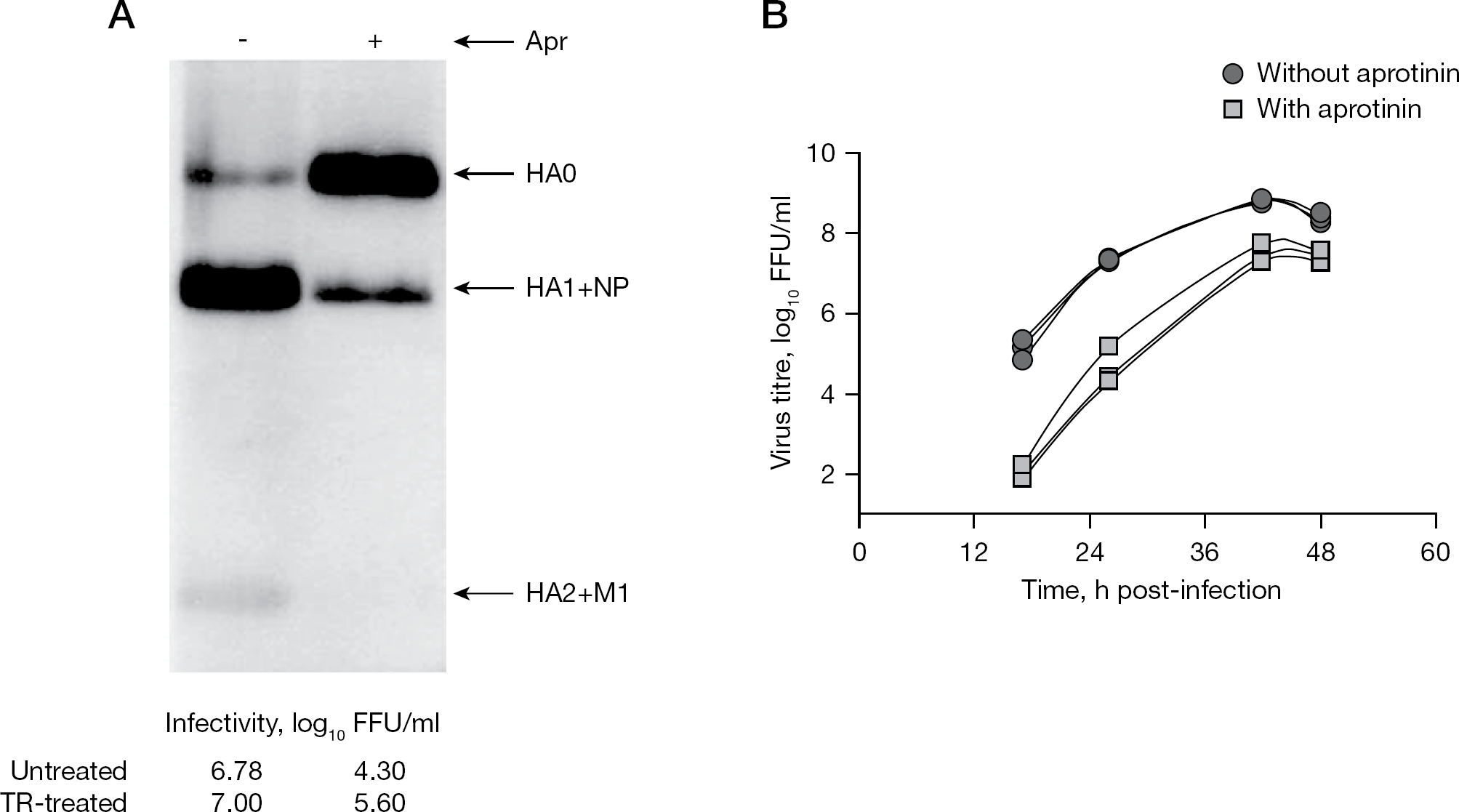
Polypeptide patterns and growth curves of pandemic H1N1v virus in tracheo-bronchial epithelium cultures in the presence of aprotinin
Aprotinin did not display any toxicity for different cells and tissues. We tested several cell cultures, such as Vero, MDCK, human adenoid epithelial (HAEC) cells, HTBE and Caco-2, and they were found to be highly tolerant to aprotinin, which did not cause any apparent toxic side effects in these cells. In order to assess aprotinin toxicity, levels of cell death were monitored in cultured epithelial cells of three different origins using a CytoTox-96 test (tetrazolium-diaphorase color reaction; Promega, Madison, WI, USA) measuring activity of lactate dehydrogenase (LDH) released from dying cells incubated with aprotinin. Confluent monolayers of Caco-2 (human intestinal), Calu-3 (human bronchial) and MDCK-II cells incubated in DMEM containing different concentrations of aprotinin for 45 h at 37°C. Aliquots of culture fluids were clarified and LDH activity was quantitated by tetrazolium colour test and measuring absorbance of the produced red formazan [23]. Culture fluids of cells incubated in medium alone and cells treated with 0.4% of nonionic detergent NP-40 were monitored as viability control and maximal toxicity reference, respectively (Figure 3A). Aprotinin dose 5,500 KIU per ml of culture medium (the highest concentration that could be prepared from the aprotinin stock solution used) did not affect cell morphology and maintained cell toxicity level close to that developed in viability control samples in Caco-2, Calu-3 (Figure 3A) and MDCK-II cells (OPZ et al., data not shown). For example, toxicity levels of 8.5% ±0.9% and 10.1% ±0.7% (P>0.05) were observed in Caco-2 cells untreated and treated with aprotinin, respectively, as compared with the maximal toxicity reference of 100%. Also, injection of 7,500 KIU of aprotinin into allantoic cavity of 12-day-old embryonated egg did not disturb development and delivery of normal chickens. These data are in good agreement with our earlier results about the lack of local irritant, toxic, and allergic side effects in dermal, eye, and respiratory probes and systemic toxicity in other organs of animals [24,25] and humans [26] treated with aerosolized aprotinin.
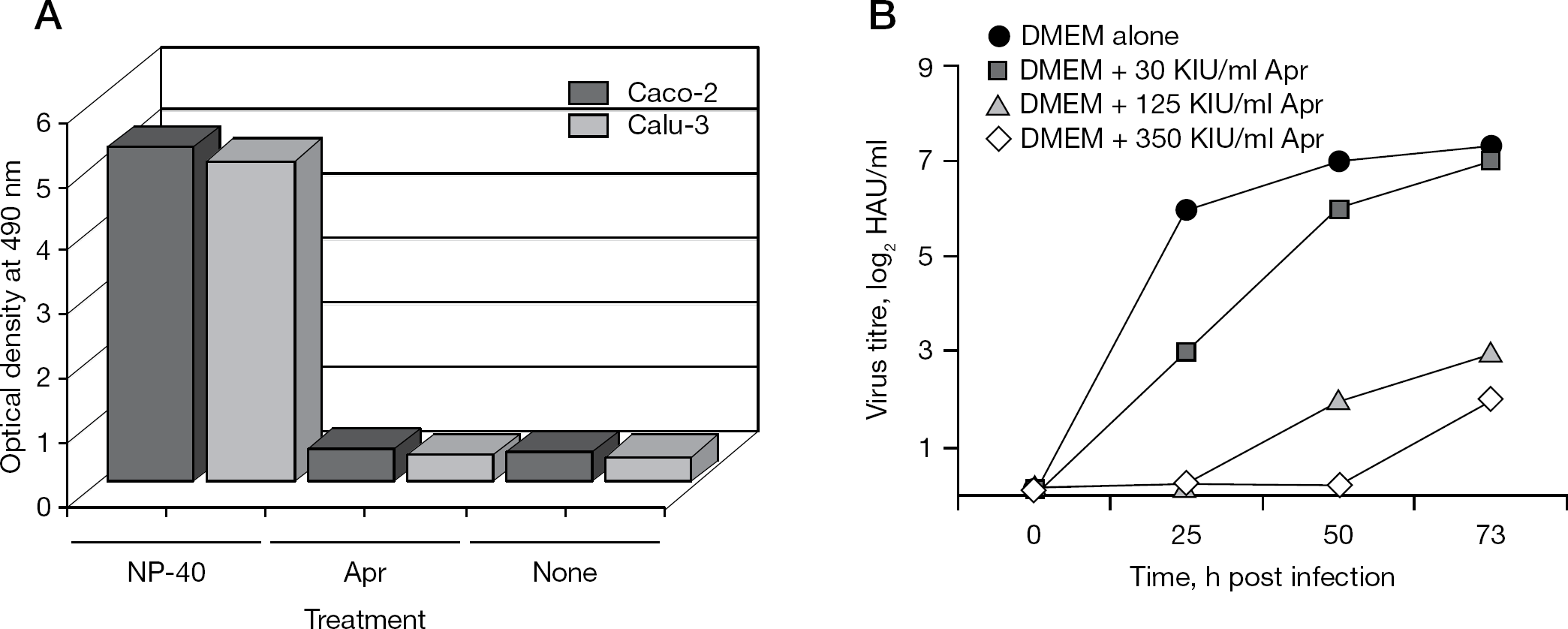
Non-toxic and virus-inhibiting doses of aprotinin in cultured cells
Finally, a selectivity index of aprotinin was assessed by testing in Caco-2 cells. To this end, a range of effective antiviral concentrations of aprotinin was determined. Caco-2 cells infected with HB/09 virus under multicycle conditions were incubated in medium containing different concentrations of aprotinin, and progeny virus production was determined by measuring virus HA titres in culture medium. The apparent decrease of virus production was already observed at aprotinin concentration 20–50 KIU (0.6–1.3 nM) per ml of culture medium (Figure 3B). Thus, the effective antiviral dose of aprotinin was at least 100-fold less than its cytotoxic potential dose.
Our findings presented here are interesting in several respects. First, the HA of pandemic H1N1v was found to be highly sensitive to human respiratory proteases and was intensively cleaved to provide virus activation and multicycle replication in human respiratory epithelium. The observation of the antiviral activity of aprotinin, an inhibitor of trypsin-like serine proteases, in the human epithelium (HTBE) host system indicates that protease(s) activating swine-origin H1N1v virus in human respiratory tract belongs to this group of proteases. Similar results were obtained earlier in primary human adenoid epithelial cells infected with laboratory strains of human epidemic influenza viruses A/WSN/33 (H1N1) and A/Aichi/2/68 (H3N2) [12]. Second, our data imply a possible role of protease–antiprotease balance in influenza pathogenesis. Aprotinin is a natural anti-protease polypeptide isolated from bovine lung, and the respiratory origin suggests that it has high affinity to proteases of the respiratory tract. This inhibitor could therefore play a physiological role as an antagonist of IAP in the human respiratory tract. In the course of influenza infection, the inhibitor–protease balance might be disturbed, and an anti-protease deficit might lead to virus activation and spread, as well as the inflammation process [27]. Indeed, a protease overbalance was found to develop in cultured cells infected with influenza virus [15]. Notably, other physiological respiratory antiproteases, such as lung surfactant [28] and human mucus protease inhibitor [29] are also known to control proteolysis of influenza virus HA in airway epithelium. Respiratory proteases TMPRSS2 and HAT have been identified as IAPs, but little is known about their natural inhibitors in human respiratory tract. Third, aprotinin may be a useful tool for anti-influenza therapy. Previously we have demonstrated that aprotinin suppressed human epidemic influenza viruses by inhibiting HA cleavage and virus activation in cultured cells [16], chicken embryonated eggs [9], mouse lungs [30] and human respiratory epithelium [12], and protects mice from lethal human influenza virus infection when administered through aerosol inhalations [31,32]. Moreover, aprotinin aerosol inhalations administered to humans with seasonal influenza and parainfluenza showed therapeutic efficacy as indicated by marked reduction in duration of fever, headache, throat pain, hoarseness, cough and earlier discharge from the hospital [26]. A metered dose pocket inhaler generating aerosolized aprotinin was approved in Russia (under the trademark AerusTM) as anti-influenza drug. According to our data presented here, aprotinin can be also considered for the rational therapy of humans infected with swine-origin pandemic flu virus.
Footnotes
Acknowledgements
We thank Folker Schwalm from Marburg Institute of Virology for the assistance in virus propagation. Funding was provided by the Russian Foundation of Basic Research (grant 10–04–01824), the German SFB 593 program, the LOEWE Program of the State of Hesse Universities of Giessen and Marburg Lung Center and the Von Behring-Röntgen-Stiftung.
The funders had no role in study design, data analysis, decision to publish or preparation of the manuscript. The authors declare no competing interests.
