Abstract
Background:
Carbohydrate-binding agents are considered as potential therapeutic agents for the inhibition of highly glycosylated enveloped viruses such as HIV type-1. Phenylboronic acids are well-known to bind the cis-diol functionality of carbohydrate structures, thereby identifying themselves as potential lead structures.
Methods:
Bisphenylboronic acids connected via a functionalized linker at variable length (1–13 atoms) bearing the binding boronic acid functionality at the three possible ring geometries relative to the linker have been investigated as probes for selective and non-selective saccharide sensors. Herein, we describe the compilation of a ‘linker-diverse’ compound library of bisphenylboronic acids and the determination of the structure–activity relationship versus a variety of enveloped viruses. Molecular modelling of the gp120 glycans of simian immunodeficiency virus was undertaken to ascertain a theoretical minimum length of the linker unit.
Results:
The compounds demonstrated no pronounced antiviral activity. The general low toxicity of the boronic acids became evident in this study, thereby justifying further studies.
Conclusions:
A higher concentration of phenylboronate functional groups per molecule, resulting in multivalency, might be necessary to bind with sufficient potency to HIV type-1 gp120 and to elicit an antiviral action.
Introduction
The development of carbohydrate-binding agents (CBAs) as potential antiviral therapeutics has recently been advanced [1,2]. Uniquely, these compounds exhibit two potential modes of action: the binding of the cis-diol functionality of the carbohydrate moieties displayed on the glycans of the viral envelope, thereby preventing viral entry and inducement of mutation in the virus (specifically the gp120 glycan) to allow for the escape of CBA pressure (via progressive deletion of the carbohydrate surface of the viral envelope) exposing the mutant virus to an immune response by the host organism [2].
Previous investigations on monophenylboronic acids [3] have shown a general lack of activity of this class of compounds in the inhibition of enveloped virus replication. Evidence is provided for the need for a larger concentration of phenylboronic acids in one molecule to bind to the high number of cis-diols displayed on highly glycosylated enveloped virus surfaces to prevent efficient interaction of such viruses with the host cells. Use of a molecule that possesses multiple binding sites might thus alleviate the need for high concentrations of monomeric compound.
The structure of HIV type-1 (HIV-1) gp120 [4] was considered in this project as a target model. The glycans of the protein are assembled in such a way that a highly diverse array of oligosaccharides comprising the terminal carbohydrate residues of the glycan surface are displayed upon the cell surface of the virus. Such oligosaccharides branch from a peptide backbone and, as a direct result, distances between the extruding oligosaccharide chains, and hence the terminal mannose carbohydrate residues, might vary widely [5]. Thus, any linker unit connecting ≥2 CBA functionalities must be of sufficient length to allow bridging from one functionality of a mannose cis-diol to a second mannose residue. Although the post-attachment structure of HIV-1 gp120, bound to the CD4 receptor, has been known for some time [4], the unattached gp120 structure has yet to be clarified. Because of this situation, it was necessary to utilize simian immunodeficiency virus gp120, whose pre-attachment structure has been crystallized and fully characterized [6]; thus, molecular modelling (Molecular operating environment software; Chemical Computing Group, Montreal, QC, Canada; Figure 1) of the terminal oligosaccharide chains allows for determination of mean distances between mannose cis-diol functionalities. The calculated distances can then be ‘engineered’ into synthesized linkers.
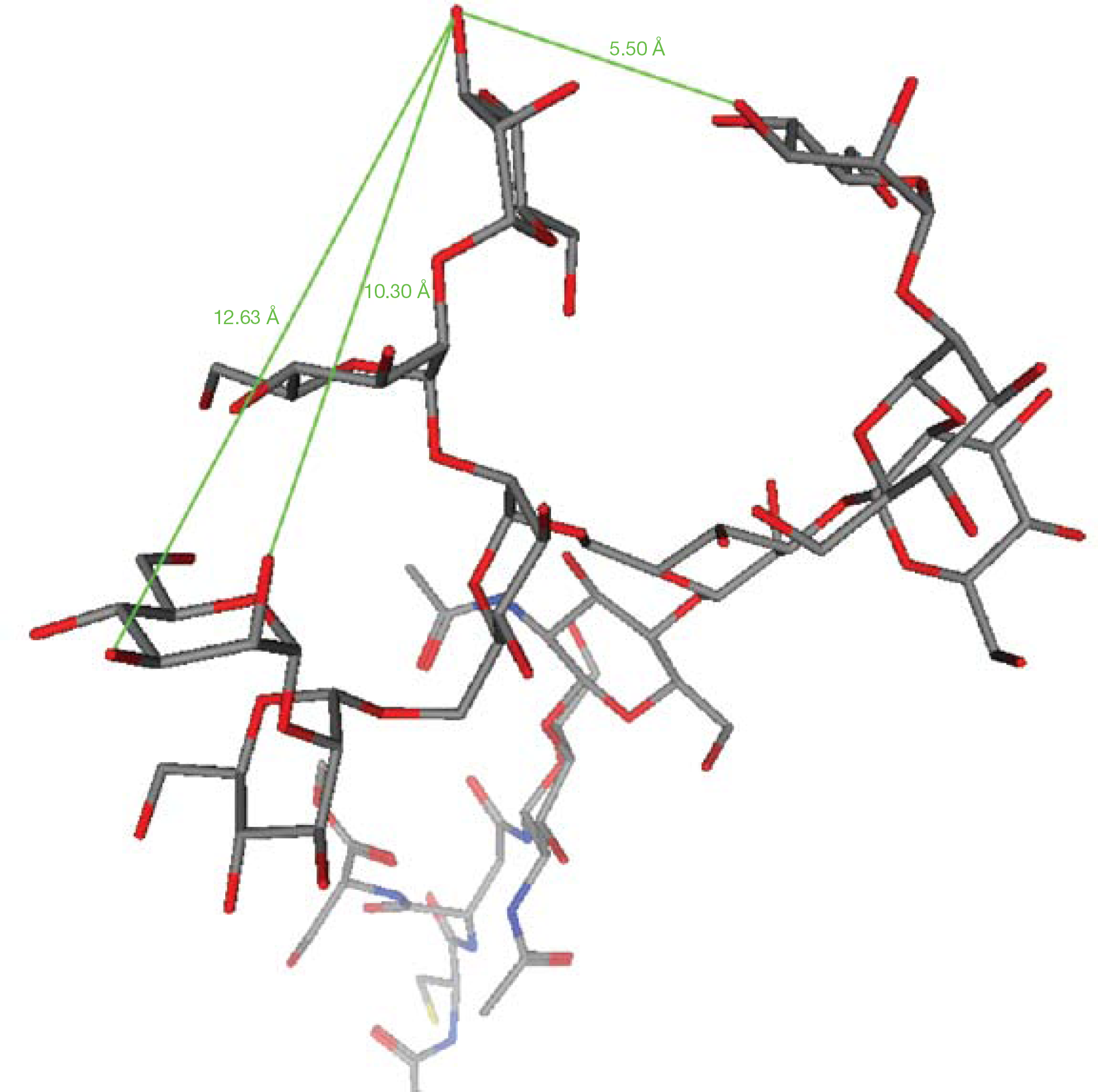
Mean distance approximation between mannose cis-diols
Herein, we report the compilation of a known (Figures 2 and 3) and synthesized (Figures 4 and 5) bisphenylboronic acid library, each constructed around a central linker skeleton of variable length and functionality, ranging from 1 to 13 atom chains. Compounds that contained incremental increases in length of the linker unit were sought. The compound library was screened against HIV-1 and a variety of enveloped viruses to determine antiviral potency.
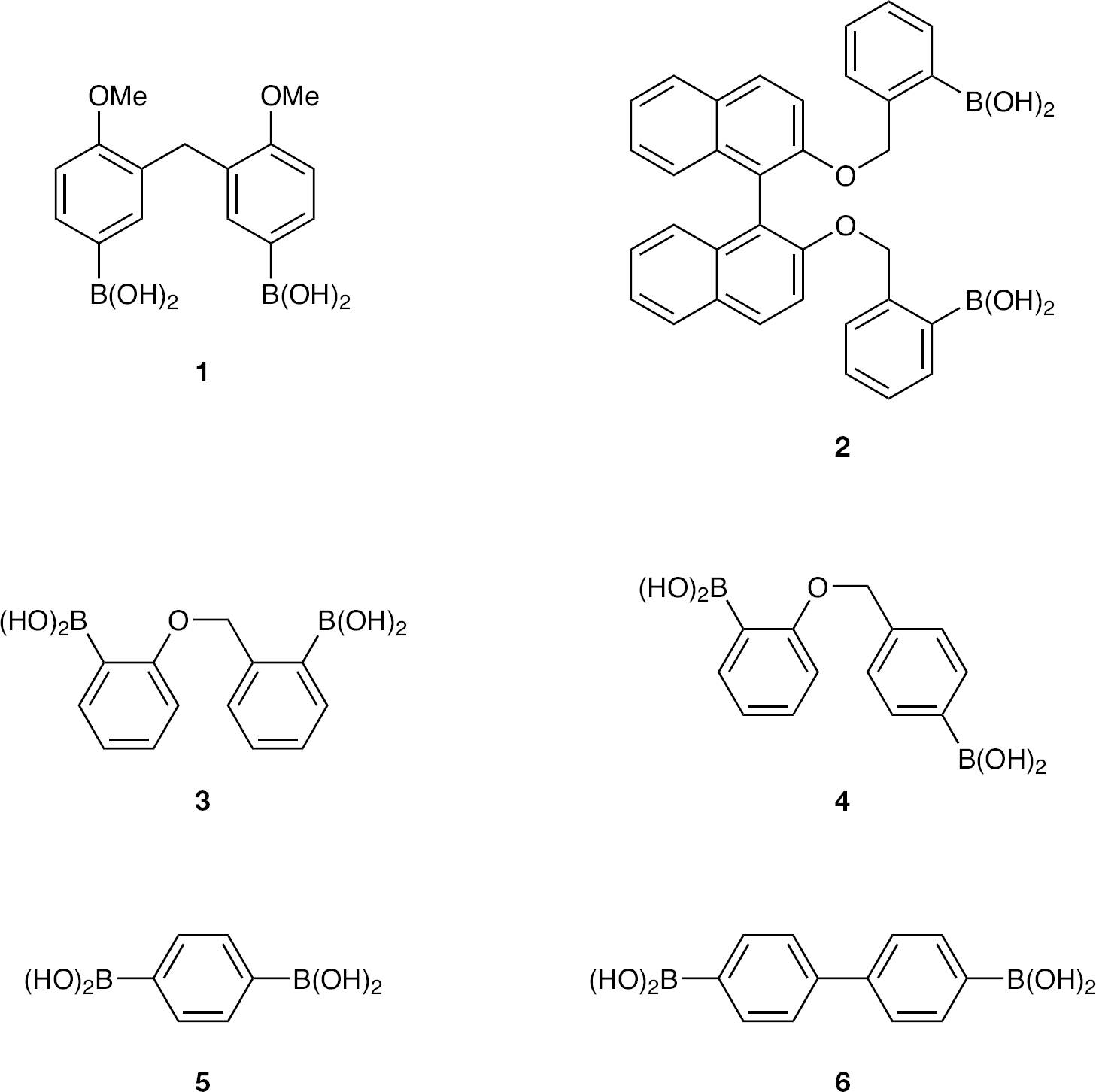
Known and commercially available bisphenylboronic acid library members
Bisphenylboronic acids from known dibromides
Synthesis of bisphenylboronic acids with linkers comprising 2–6 atoms
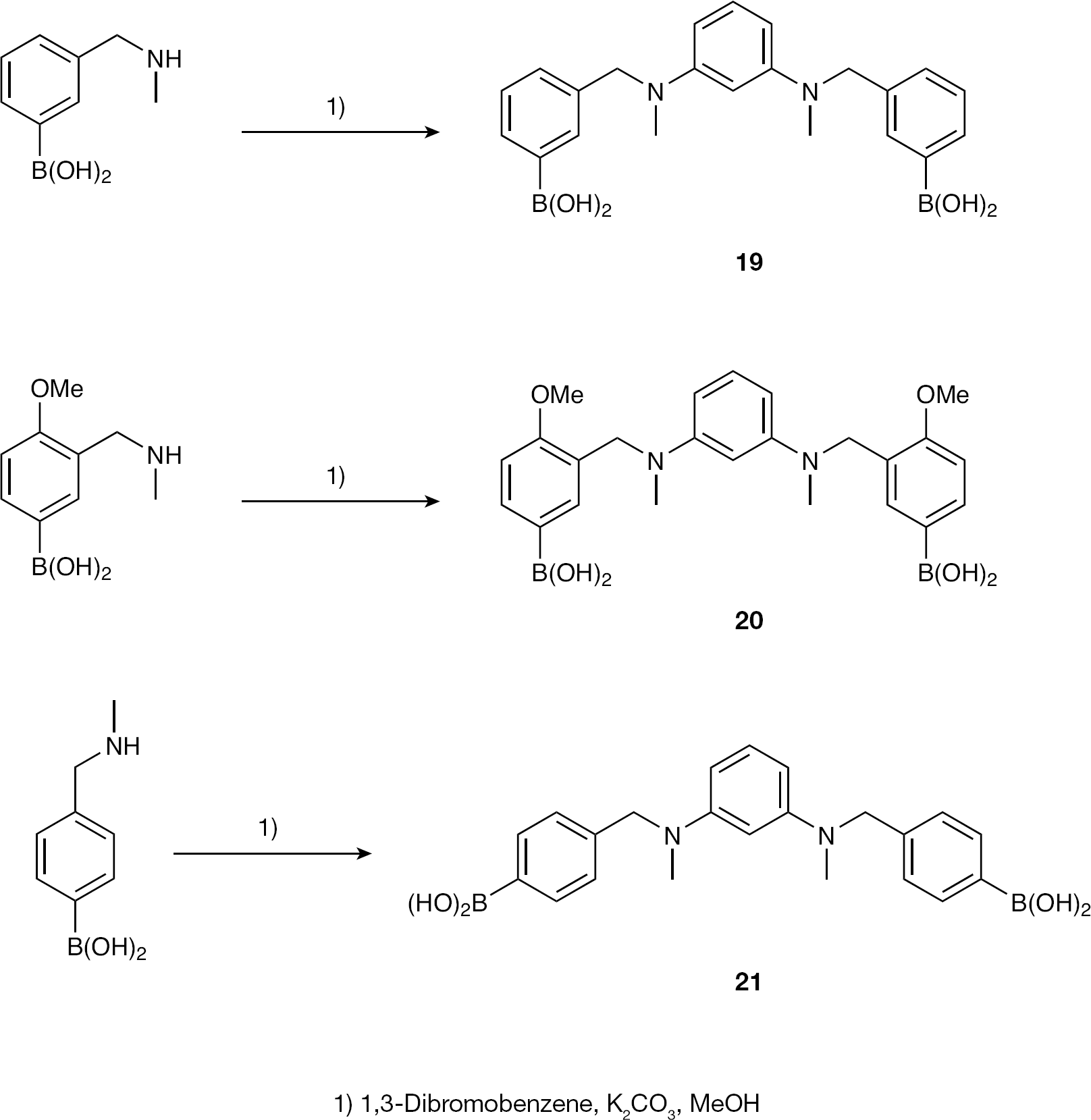
Synthesis of 7-atom-containing linked bisphenylboronic acids
Bisphenylboronic acids comprising boronic acid functionalities at the ortho-, meta- and para- positions relative to the linker substituent were synthesized. The ring geometry of the boronic acid further elongates or contracts the spacing of the binding functionalities, allowing for additional structure–activity relationship investigations upon binding and antiviral activity determination.
The linkers display a variety of constituent atoms (C, N and O) and functionalities (amines, ketones, olefin, furan and amide bonds) to allow for engineered rigidity and synthetic simplicity. Rigidity is introduced through carbonyl groups and amide bonds to reduce conformational freedom and to enable the bridging of ≥2 carbohydrate residues. In particular, carbon chains might adopt many minimum energy configurations, which would be expected to include ‘folded’ conformations with a reduced distance between boronic acid functionalities.
Methods
Chemistry
1H NMR spectra were recorded on a Bruker AMX500 (500 MHz) spectrometer (Bruker Instruments, Inc., Billerica, MA, USA). Chemical shifts (δH) are reported in parts per million (ppm) and are referenced to the residual protonated solvent peak. Abbreviations used in the description of multiplicities are singlet (s), doublet (d), double doublet (dd), double triplet (dt), triplet (t), quartet (q), multiplet (m) and broad (br). Coupling constants (J) are quoted in Hertz (Hz) to the nearest 0.1 Hz.
13C NMR spectra were recorded on a Bruker AMX500 (125.8 MHz) spectrometer. Chemical shifts are quoted in ppm and referenced to the residual protonated solvent peak with proton decoupling, using DEPT, HMQC and APT editing to aid assignment.
11B NMR spectra were recorded on a Bruker AMX500 (160.5 MHz) spectrometer. Chemical shifts are quoted in ppm and referenced to the residual protonated solvent peak with proton decoupling. Samples were performed in a pure quartz 5 mm NMR tube.
Low-resolution and high-resolution mass spectra (m/z) were recorded on a Waters GCT Premier spectrometer (Waters, Elstree, UK) using an electron ionization (EI) source. m/z values of major peaks are reported in Daltons, with intensities quoted as percentages of the base peak.
Thin-layer chromatography (TLC) was performed on Merck and Co. (Darmsted, Germany) aluminum-foil-backed sheets precoated with 0.2 mm Kieselgel 60 F-54. Development of TCL plates was performed by the ascending method. Product spots were visualized by the quenching of ultraviolet fluorescence (λmax 254 nm), or by staining with a solution of 5% (w/v) dodecamolybdophosphoric acid in ethanol.
Flash column chromatography was performed under pressure on a silica gel, 60 Å pore size, 40–60 μm (Phase Separations Ltd, Deeside, UK). Samples were applied as a concentrated solution in the same eluent or preadsorbed on silica gel. Fractions containing the product were identified by TLC, pooled and the solvent removed in vacuo.
All solvents were used, as obtained from Sigma–Aldrich Co. (St Louis, MO, USA). All reactions were performed under an inert argon atmosphere unless otherwise stated.
Compounds
It is well-documented [10–12] that boronic acids poorly fragment in many ionization techniques used in mass spectral analysis because of their low volatility. This is amplified by the facile occurrence of gas-phase dehydration and anhydride formation in the ion source. Derivatization as the ester with ethylene glycol can be beneficial in aiding identification of the molecular species as illustrated in the following experimental section. Many ionization methods were applied to the compounds reported herein, including derivatization with ethylene glycol. Several compounds failed to provide molecular species, those that did are reported below.
General procedure A for boronation of bromides
The corresponding bromide (1.0 eq.) was dissolved in THF or Et2O (40 ml) and cooled to −78°C. n BuLi (1.5 eq. of a 2.5 M solution in hexanes) was added dropwise and stirring continued at −78°C for 1 h. Trimethyl borate (1.5 eq.) was added dropwise and the mixture was allowed to warm gradually to room temperature (RT) and stirred overnight. Water (20 ml) was added and the mixture stirred for 1 h. Brine (25 ml) was added and the product extracted with EtOAc (2×30 ml), dried (MgSO4), filtered and concentrated in vacuo to provide the desired compound.
General procedure B for boronation of bromides
To a stirred suspension of magnesium turnings (2.5 eq.) in Et2O or THF (100 ml), 1,2-dibromoethane (catalyst) and the corresponding bromide precursor (1.0 eq.) was added and stirring was continued at RT overnight. The mixture was cooled to −78°C and trimethyl borate (1.0 eq.) in dry Et2O (1 ml) was added dropwise. The reaction was stirred for a further 1 h at −78°C and allowed to warm to RT overnight. The reaction was cooled to 0°C and 8% HCl (10 ml) was added slowly, the reaction was diluted with water (10 ml) and stirred for 10 min at 0°C. The organic layer was extracted with Et2O (2×25 ml). The organic layers were then combined, dried (MgSO4), filtered and concentrated in vacuo to yield the desired compound.
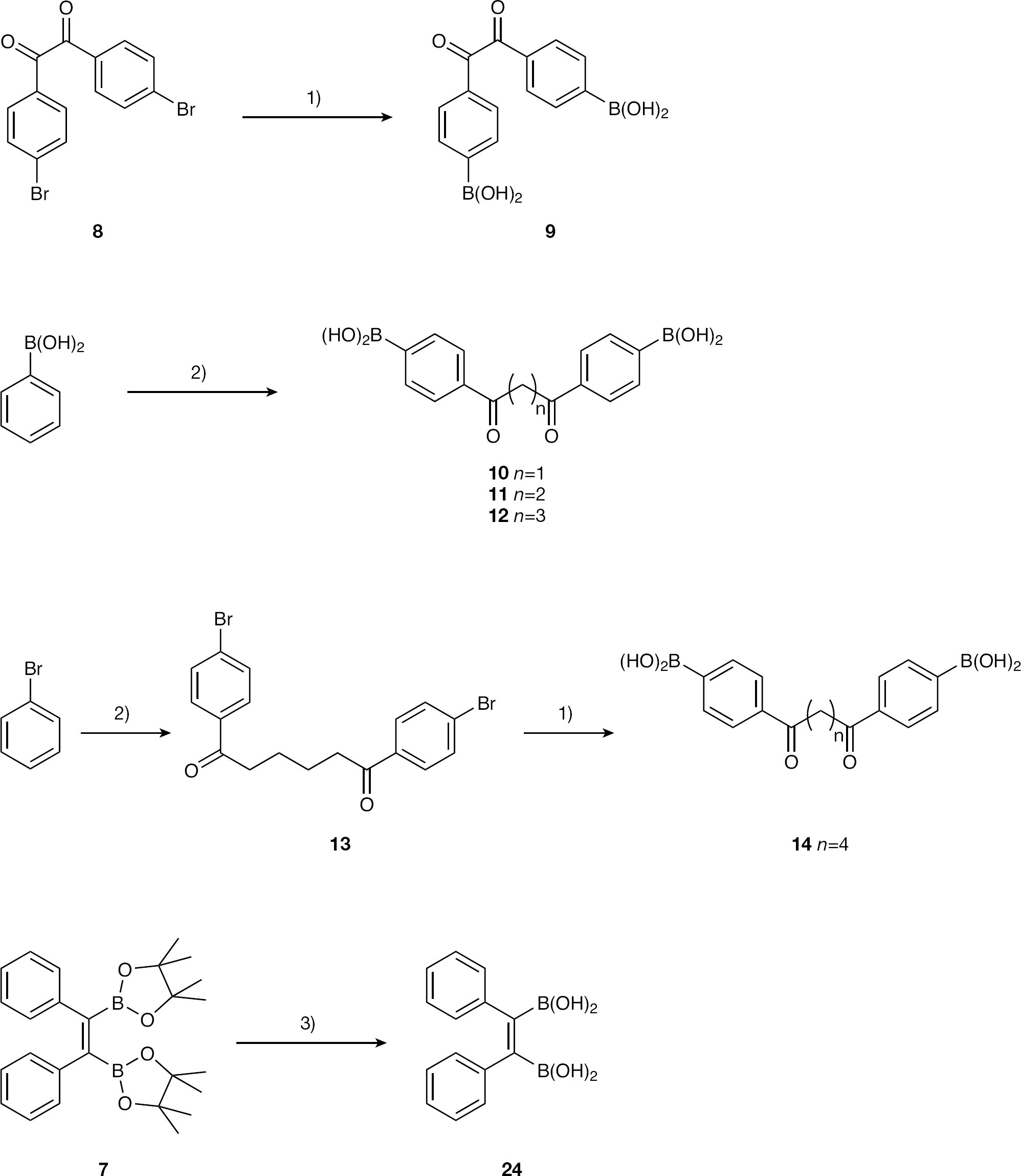
1,2-Bis(4-phenylboronic acid)ethane-1,2-dione (9)
Dibromide dione
1,2-Bis(4-phenylboronic acid)propane-1,3-dione (10)
Phenylboronic acid (750 mg, 6.15 mmol) was dissolved in methanol (30 ml), the solution was cooled to 0°C and AlCl3 (885 mg, 6.65 mmol) was added; stirring was continued until fully solvated. A solution of malonyl chloride (434 mg, 3.08 mmol) in methanol (2 ml) was carefully added dropwise over 1 h at 0°C, stirring was continued for a further 1 h and then allowed to warm to RT overnight. Ice (1.0 g) was added to the resultant orange solution and diluted with methanol (20 ml). The organic layer was washed with 2.5 M NaOH (20 ml) and brine (20 ml), extracted with EtOAc (100 ml), dried (MgSO4), filtered and evaporated in vacuo to yield
1,2-Bis(4-phenylboronic acid)butane-1,4-dione (11)
The procedure for compound
1,2-Bis(4-phenylboronic acid)pentane-1,5-dione (12)
The procedure for compound
1,2-Bis(4-bromophenyl)hexane-1,6-dione (13)
The procedure for compound
1,2-Bis(4-phenylboronic acid)hexane-1,6-dione (14)
Compound
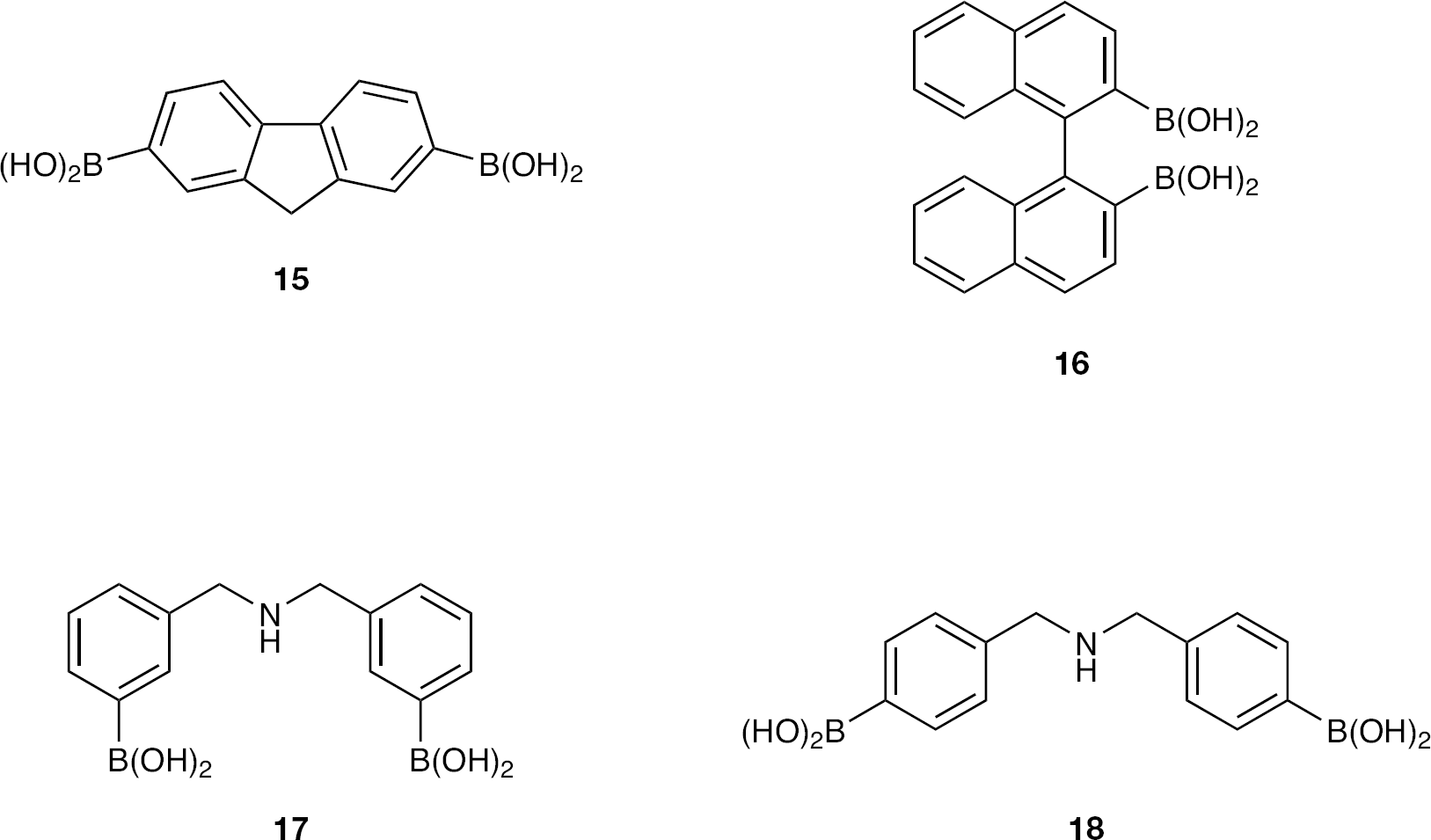
9H-fluorene-2,7-diboronic acid (15)
Commercially (Sigma–Aldrich Co.) available 9H-fluorene-2,7-dibromide underwent general procedure A to provide
1,1′-Binaphthyl-2,2′-diboronic acid (16)
2,2′-Dibromo-1-1′-binaphthyl underwent general procedure A to provide
3,3′-Azanediylbis(methylene)bis(3,1-phenylene)diboronic acid (17)
The corresponding known [14] dibromide underwent general procedure A to provide
4,4′-Azanediylbis(methylene)bis(4,1-phenylene)diboronic acid (18)
The corresponding known [14] dibromide underwent general procedure A to provide
3,3′-(1,3-Phenylenebis(methylazanediyl))bis(methylene)bis(3,1-phenylene)diboronic acid (19)
To a solution of 1,3-dibromobenzene (100 mg, 0.42 mmol) and K2CO3 (580 mg, 4.2 mmol) in anhydrous methanol (20 ml) 3-methylaminomethylphenylboronic acid [15] was added (140 mg, 0.85 mmol) dropwise. The reaction mixture was stirred at RT for 48 h and the solvent evaporated in vacuo to provide a white precipitate contaminated with a red oil. The oil was dissolved in MeOH (4 ml), removed by pipette, concentrated and dried under high vacuum to yield
3,3′-(1,3-Phenylenebis(methylazanediyl))bis(methylene)bis(4-methoxy-3,1-phenylene)diboronic acid (20)
The procedure described previously for compound
4,4′-(1,3-Phenylenebis(methylazanediyl))bis(methylene)bis(4,1-phenylene)diboronic acid (21)
The procedure described previously for compound
4,4′-(3,3′-(Ethane-1,2-diylbis(azanediyl))bis(3-oxopropane-3,1-diyl))bis(4,1-phenylene)diboronic acid (22)
To a solution of known boronic acid [3] (1.0 g, 5.46 mmol), dicyclohexylcarbodiimide (1.2 g, 6.0 mmol) and 4-dimethylaminopyridine (catalyst) in DCM: MeOH (10:1; 80 ml) ethyldiamine was added (0.19 ml, 2.73 mmol) dropwise. The mixture was stirred at RT for 48 h, filtered through a cotton plug to remove precipitated dicyclohexyl urea and evaporated in vacuo to provide
4,4′-(3,3′-(Tetrahydrofuran-2,5-diyl)bis(methylene)bis(azanediyl)bis(3-oxopropane-3,1-diyl))bis(4,1-phenylene)diboronic acid (23)
A solution of known boronic acid [3] (833 mg, 4.3 mmol), dicyclohexylcarbodiimide (976 mg, 4.73 mmol) and 4-dimethylaminopyridine (catalyst) in DCM (40 ml) was cooled to 0°C and a solution of known [16] carboxylic acid (280 mg, 2.15 mmol) in DCM: MeOH (1:1; 6 ml) was added dropwise. The mixture was stirred at 0°C for 15 min and at RT for 12 h (a colour change from opaque to transparent green occurred), was filtered through a cotton plug to remove precipitated dicyclohexyl urea and evaporated in vacuo to provide
(Z)-stillbenediboronic acid (24)
A solution of (Z)-stillbenediboronic acid bis(pinocol) ester (250 mg, 0.58 mmol) in water: MeOH (1:1; 10 ml) was heated at 80°C for 30 min. The mixture was allowed to cool, evaporated in vacuo to approximately 2 ml volume, water was added and allowed to stand at RT overnight. The resultant orange precipitate was filtered to provide
Virological assays
The antiviral assays, other than the anti-HIV assays, were based on inhibition of virus-induced cytopathic effect in human lung fibroblast (herpes simplex virus type-1 [HSV-1; strain KOS], herpes simplex virus type-2 [HSV-2; strain G], vaccinia virus and vesicular stomatitis virus [VSV]), African green monkey kidney (Vero, parainfluenza-3, reovirus-1, Sindbis, Coxsackie virus B4 and Punta Toro virus), human cervix carcinoma (VSV, Coxsackie virus B4 and respiratory syncytial virus) or Madin–Darby canine kidney (influenza A [H1N1 and H3N2] and influenza B) cell cultures. Confluent cell cultures in microtitre 96-well plates were inoculated with 100 50% cell culture infective doses (CCID50) of virus (1 CCID50 being the virus dose to infect 50% of the cell cultures) in the presence of varying concentrations (100, 40, 8, 1.6 and 0.32 μM) of the test compounds. Viral cytopathicity was recorded as soon as it reached completion in the control virus-infected cell cultures that were not treated with the test compounds. For the human cytomegalovirus assays, confluent human embryonic lung (HEL) fibroblasts were grown in 96-well microtitre plates and infected with the human cytomegalovirus strains Davis and AD-169 at 100 plaque-forming units per well. After a 2 h incubation period, residual virus was removed and the infected cells were further incubated with medium containing different concentrations of the test compounds (in duplicate). After incubation for 7 days at 37°C, virus-induced cytopathic effect was monitored microscopically after ethanol fixation and staining with Giemsa. Varicella zoster virus (VZV) drug susceptibility tests were performed on confluent HEL cells in 96-well microtitre plates by the plaque reduction assay. Monolayers were infected with 20 plaque-forming units of cell-associated virus per well. For each assay, virus controls (infected untreated cells) were included. After a 2 h incubation period, the virus inoculum was removed and the media replaced by the different dilutions (in duplicate) of the tested molecules. Serial dilutions of test compounds were incubated with the infected monolayers for 5 days. After a 5-day incubation period, the cells were fixed and stained with Giemsa, and the level of virus-induced cytopathic effect was determined by counting the number of plaques for each dilution. The methodology of the anti-HIV assays was as follows: human T-lymphocyte CEM (approximately 3×105 cells/cm3) cells were exposed to 100 CCID50 of HIV-1(IIIB) or HIV type-2(ROD)/ml and 200 μl/well was seeded into a microtitre plate containing appropriate dilutions of the test compounds. After 4 days of incubation at 37°C, HIV-induced CEM giant cell formation was examined microscopically.
Determination of binding of the test compounds to HIV type-1 gp120 through surface plasmon resonance analysis
Recombinant gp120 protein from HIV-1 strain IIIB (ImmunoDiagnostics Inc., Woburn, MA, USA; produced by Chinese hamster ovary cell cultures) was covalently immobilized on the carboxymethylated dextran matrix of a CM5 sensor chip (GE Healthcare, Uppsala, Sweden) in 10 mM sodium acetate (pH 4.0) using standard amine coupling chemistry to a final density of 122 RU (approximately 1 fmol of gp120). A reference flow cell was used as a control for non-specific binding and refractive index changes. All interaction studies were performed at 25°C on a Biacore T100 instrument (GE Healthcare). The compounds were diluted in HBS-P (10 mM HEPES, 150 mM NaCl and 0.05% surfactant P20; pH 7.4) and with 10 mM CaCl2 (final concentration 50 μM). Samples were injected for 2 min at a flow rate of 45 μl/min and the dissociation was followed for 6 min. Several buffer blanks were used as double referencing. The CM5 sensor chip surface was regenerated with one injection of 50 mM NaOH.
Results
Synthesis of bisphenylboronic acids was based upon three general strategies. Two methods involved the generation of an organolithium (by halogen–lithium exchange) or Grignard intermediate, which was not isolated, from a corresponding dibromide starting material. Subsequent trapping of the pseudo-anion via a boronate species provided access to the desired corresponding bisphenylboronic acid in moderate-to-good yield. A third method was the relatively simple deprotection of commercially available boronic esters, which, upon hydrolysis, yielded the desired boronic acids.
Figure 3 illustrates the compounds accessed through application of either of the organometallic intermediate methods described earlier. The respective dibromide of each bisphenylboronic acid was commercially available or known in the literature.
The family of diones (n=1–4 methylene groups) synthesized as described in Methods (Figure 4) relied upon Friedel–Crafts acylation of either phenylboronic acid or bromobenzene with a suitable length, commercially available acid chloride in moderate (21–76%) yield. Of note was the failure to prepare compound
The synthesis of 7-atom length bisphenylboronic acids based around a central 1,3-disubstituted benzene moiety (Figure 5) proceeded from known boronic acids with methylamine functionality at various ring geometries. Deprotonation followed by nucleophilic attack of the amine anion resulted in compounds
To complete the compound library, 10- and 13-atom length bisphenylboronic acids were synthesized (Figure 6). Dicyclohexylcarbodimide-enabled peptide bond coupling between the known carboxylic-acid-bearing phenylboronic acid and ethyldiamine or known diaminomethylfuran resulted in amide-bond-linked bisphenylboronic acids
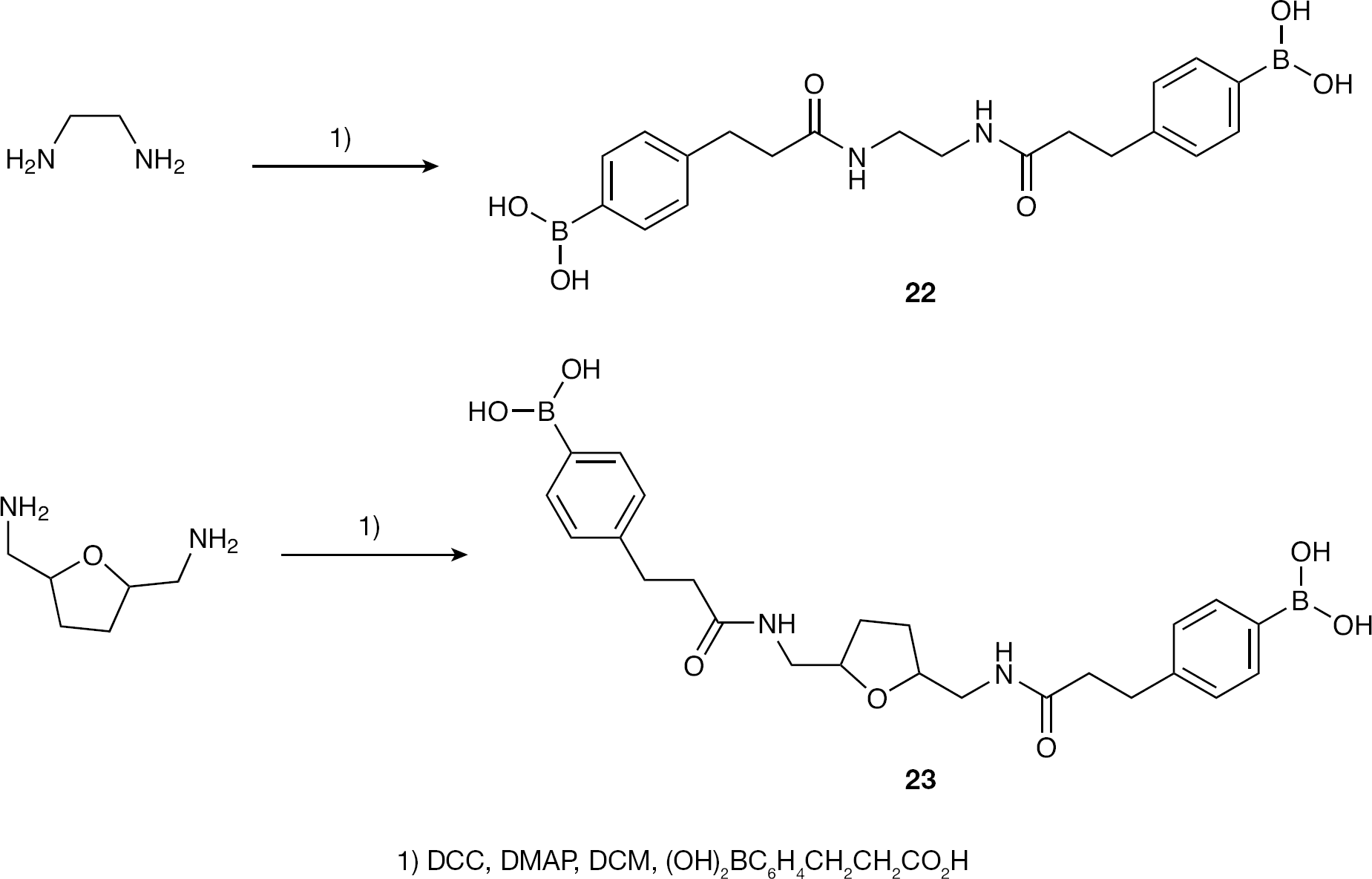
Synthesis of 10- and 13-atom length linked bisphenylboronic acids
Discussion
All compounds were tested for their inhibitory activity against HIV-1 and HIV type-2 (Table 1) in human T-lymphocyte (CEM) cell cultures. Selected compounds were also tested against a variety of enveloped viruses including VZV (Table 2), HSV-1 (KOS strain), acyclovir-resistant thymidine-kinase-deficient HSV-1 KOS strain, HSV-2 (G strain), vaccinia virus and VSV (Table 3) in HEL cells and against influenza A virus (H1N1 and H3N2 subtypes) and influenza B virus (Table 4) in Madin–Darby canine kidney cells. Included in Tables 3 and 4 are data for the current clinical drugs commonly used to treat the virus.
Anti-HIV-1 activity, anti-HIV-2 activity and cytostatic activity in human T-lymphocyte cell cultures
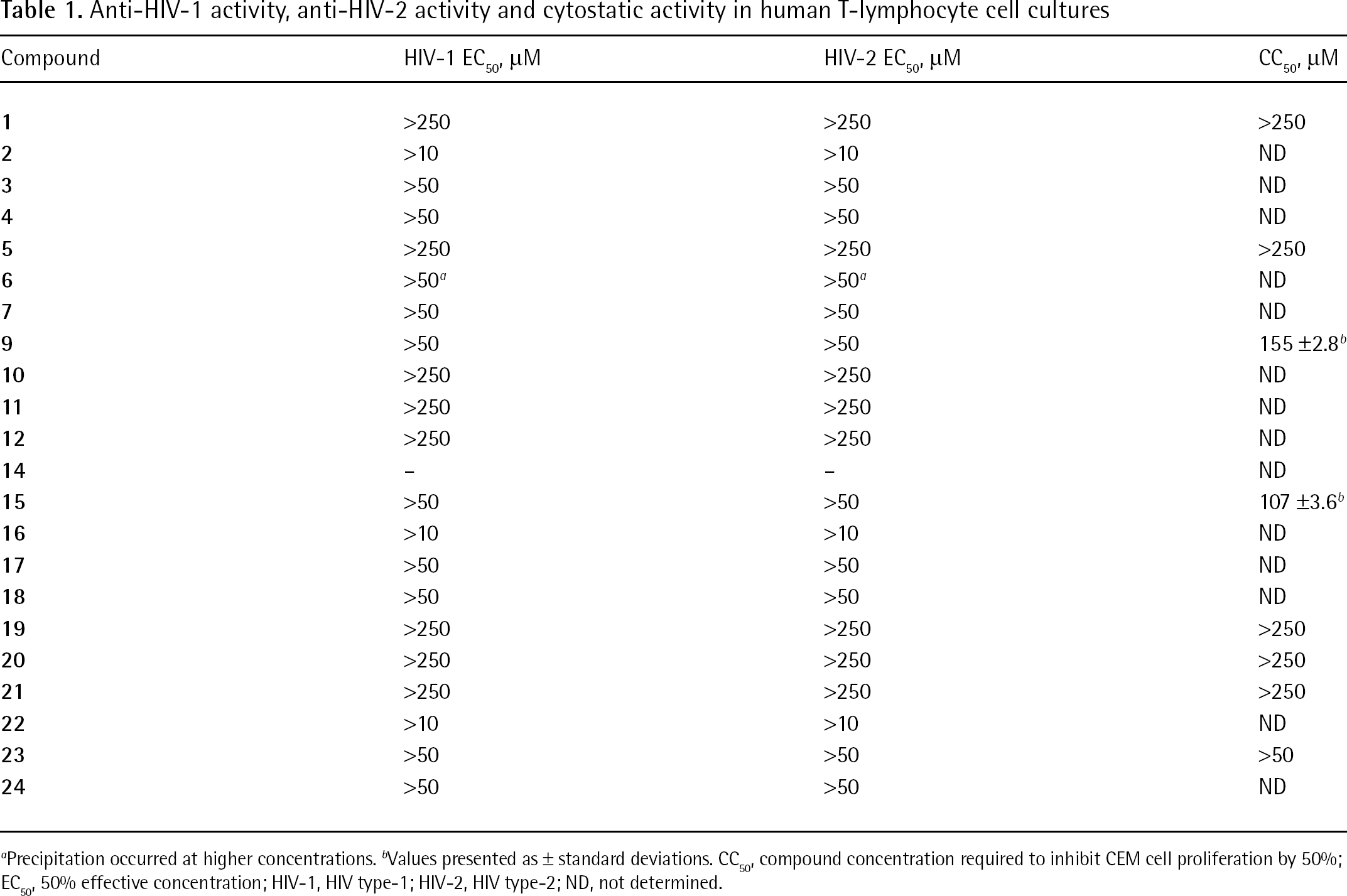
Precipitation occurred at higher concentrations.
Values presented as ± standard deviations.
CC50, compound concentration required to inhibit CEM cell proliferation by 50%
EC50, 50% effective concentration; HIV-1, HIV type-1; HIV-2, HIV type-2; ND, not determined.
Anti-VZV activity and cytotoxic/cytostatic activity in human embryonic lung cells
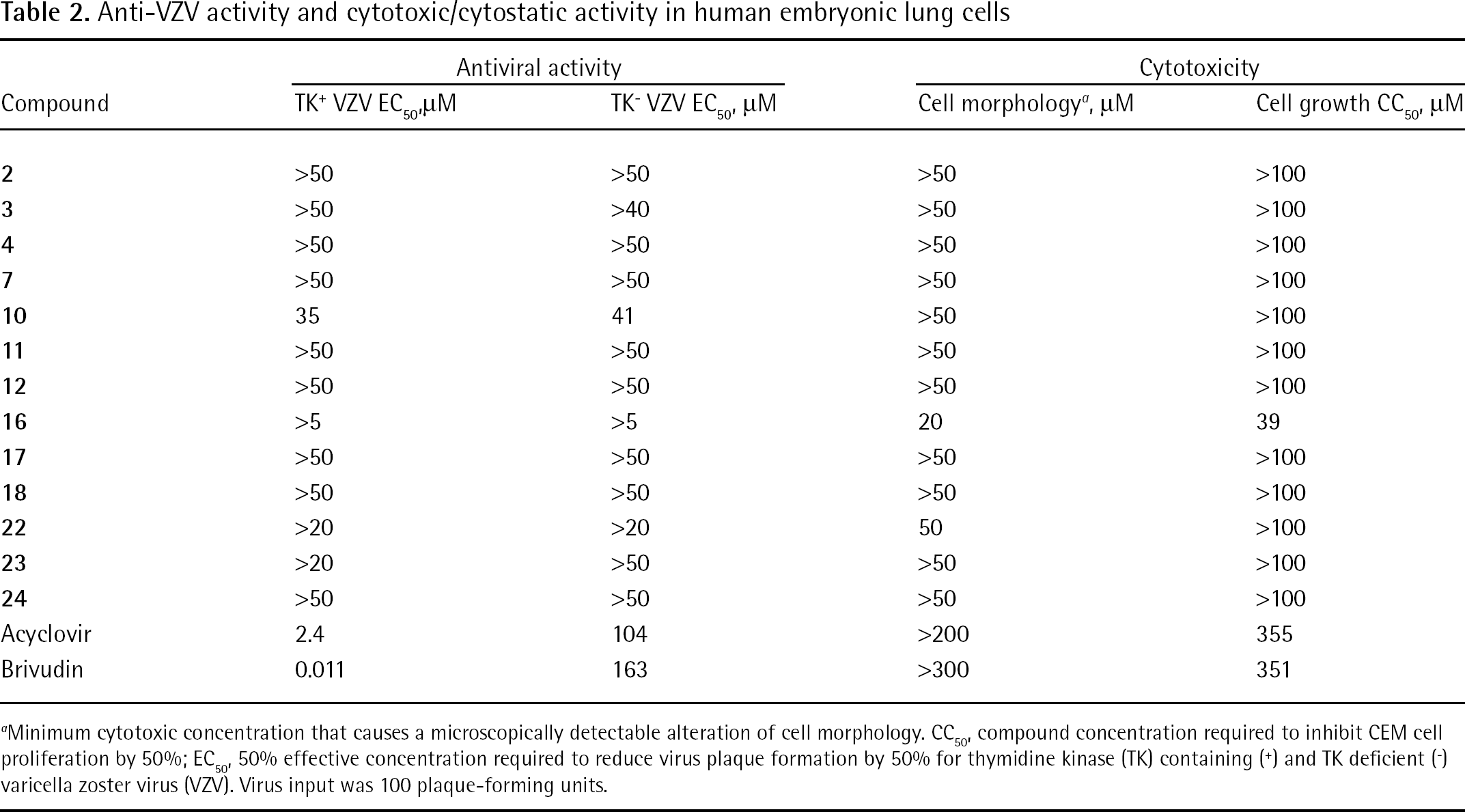
Minimum cytotoxic concentration that causes a microscopically detectable alteration of cell morphology.
CC50, compound concentration required to inhibit CEM cell proliferation by 50%
EC50, 50% effective concentration required to reduce virus plaque formation by 50% for thymidine kinase (TK) containing (+) and TK deficient (−) varicella zoster virus (VZV). Virus input was 100 plaque-forming units.
Antiviral activity and cytotoxicity of compounds in human embryonic lung cell cultures
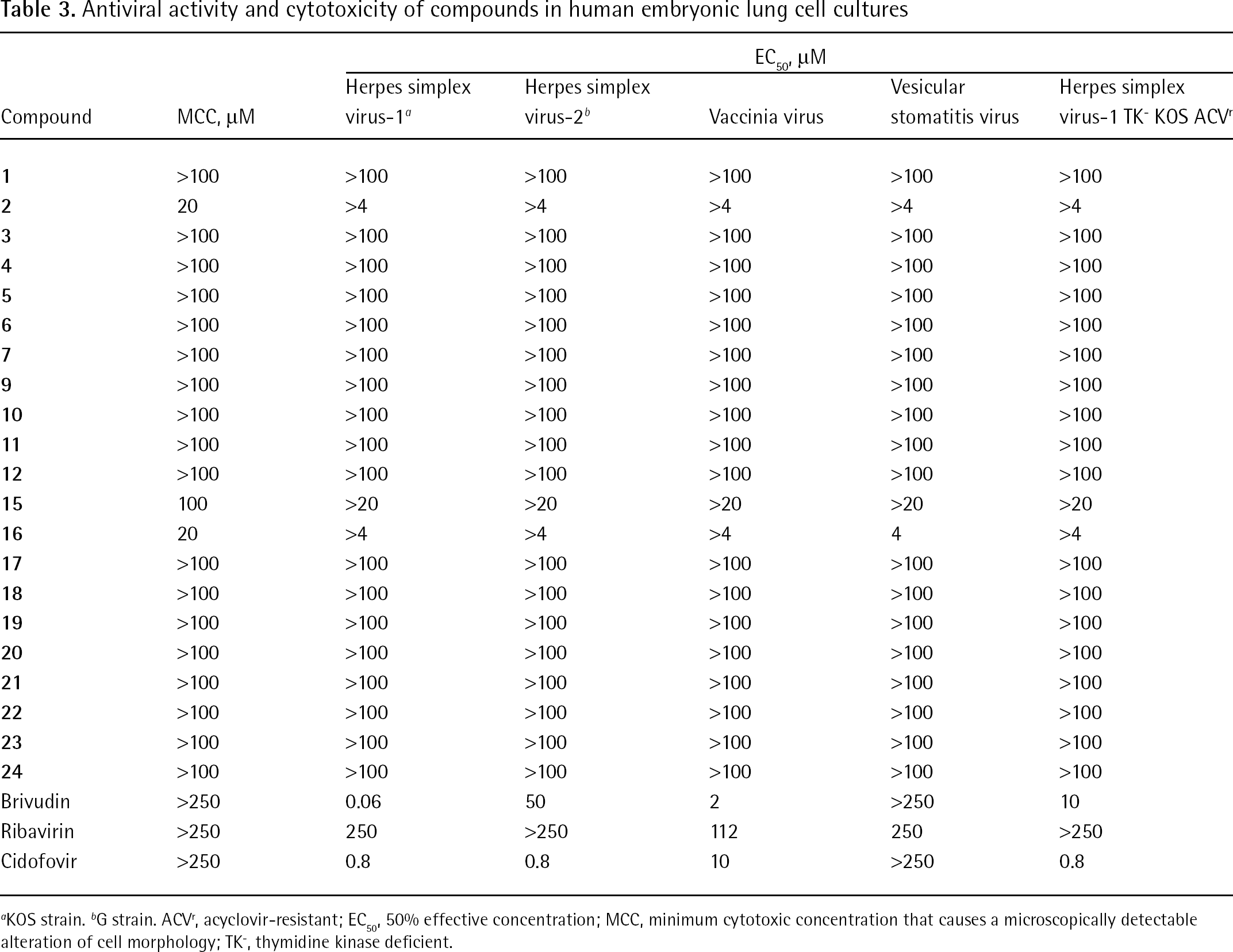
KOS strain.
G strain. ACVr, acyclovir-resistant
EC50, 50% effective concentration; MCC, minimum cytotoxic concentration that causes a microscopically detectable alteration of cell morphology; TK−, thymidine kinase deficient.
Anti-influenza-virus activity and cytostatic activity and cytotoxicity in Madin–Darby canine kidney cell cultures
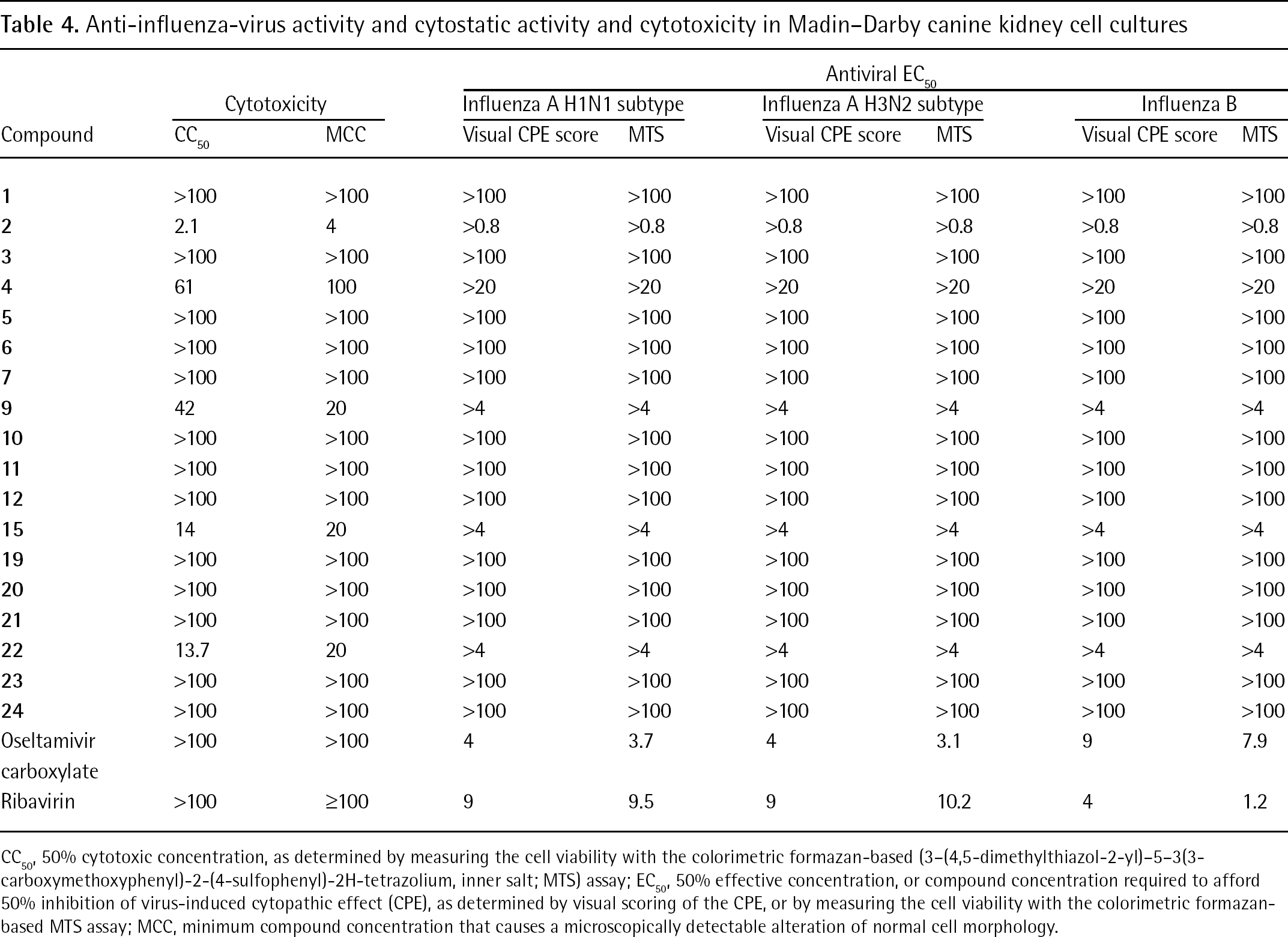
CC50, 50% cytotoxic concentration, as determined by measuring the cell viability with the colorimetric formazan-based (3–(4,5-dimethylthiazol-2-yl)–5–3(3-carboxymethoxyphenyl)-2-(4-sulfophenyl)-2H-tetrazolium, inner salt; MTS) assay
EC50, 50% effective concentration, or compound concentration required to afford 50% inhibition of virus-induced cytopathic effect (CPE), as determined by visual scoring of the CPE, or by measuring the cell viability with the colorimetric formazan-based MTS assay
MCC, minimum compound concentration that causes a microscopically detectable alteration of normal cell morphology.
Table 1 shows that none of the compounds evaluated against HIV showed significant inhibitory activity at subtoxic concentrations. Similar inactivity of the test compounds at the evaluated concentrations was observed for a variety of other enveloped (Tables 2, 3 and 4) and non-enveloped (JB, data not shown) viruses in cell culture. Also, surface plasmon resonance (SPR) studies revealed that the bisphenylboronic acid derivatives do not efficiently bind to HIV-1 gp120 (JB, data not shown) at 50 μM.
To ensure that the bisphenylboronic acids can indeed bind to the cis-diols of carbohydrate residues, high-field (11.74 Tesla) 11B NMR studies were conducted upon the ability of compounds

11B NMR, 160.5 MHz, D2O, binding of
Binaphthylene-based compound
In conclusion, this study demonstrates that bisphenylboronic acids do not inhibit glycosylated enveloped viruses. High-field NMR confirms that binding is possible and suggests it is taking place; however, the data presented herein indicate the binding is not of sufficient strength to inhibit viral entry, as was also observed in the SPR binding studies using HIV-1 gp120 as the ligand (50 μM used as the highest concentration). The low toxicity of almost all compounds presented shows possibilities for further modifications. The synthesis of larger aggregate molecules containing increased numbers of boronic acid functional groups is currently underway in our laboratories (Cardiff University, Cardiff, UK).
Footnotes
Acknowledgements
We thank Rob Jenkins (Chemistry Department, Cardiff University, Cardiff, UK) for use of a 11B probe NMR spectrometer. Helpful discussions and molecular modelling expertise from Andrea Brancale (Cardiff University) are gratefully acknowledged. Financial support was provided by FWO Krediet numbers G.0188.07 and G.485.081, the Geconcerteerde Onderzoeksacties (GOA 10/014) and the Center of Excellence (Krediet number 05/15) of the Katholieke Universiteit, Leuven, Belgium.
The authors declare no competing interests.
