Abstract
Oseltamivir and zanamivir are currently licensed worldwide for influenza treatment and chemoprophylaxis. Both drugs require twice-daily administration for 5 days for treatment. A new influenza drug, laninamivir (code name R-125489), and its prodrug form, CS-8958 (laninamivir octanoate or laninamivir prodrug), which are long-acting neuraminidase inhibitors, are introduced in this review. Laninamivir potently inhibited the neuraminidase activities of various influenza A and B viruses, including subtypes N1–N9, pandemic (2009) H1N1 virus, highly pathogenic avian influenza (HPAI) H5N1 viruses and oseltamivir-resistant viruses. Because of the long retention of laninamivir in mouse lungs after an intranasal administration of CS-8958, therapeutic administration of a single dose of CS-8958 showed superior efficacy to repeated administrations of zanamivir or oseltamivir in animal infection models for influenza A and B viruses. These include pandemic (2009) H1N1 virus and HPAI H5N1 virus. Prophylactic single administration of CS-8958, as early as 7 days prior to infection, also showed superior efficacy. Finally, the potential of a single inhalation of CS-8958 for influenza patients was demonstrated by clinical studies, and CS-8958 has been approved and is commercially available as Inavir® (Daiichi Sankyo Co., Ltd, Tokyo) in Japan.
Introduction
Influenza is a serious respiratory illness, which can be debilitating, and could cause complications that lead to hospitalization and death, especially in elderly individuals. This respiratory disease is caused by influenza A and B viruses, which are pathogens that are highly contagious among humans. Influenza A viruses are classified into subtypes on the basis of the antigenicities of haemagglutinin (HA) and neuraminidase (NA) molecules. To date, 16 HA subtypes (H1–H16) and 9 NA subtypes (N1–N9) have been reported. Seasonal influenza or influenza epidemics are caused by influenza A viruses H1N1 and H3N2 and influenza B virus [1]. The global burden of influenza epidemics is believed to be 3.5 million cases of severe illness and 300,000–500,000 deaths annually [2]; these numbers were calculated before the new pandemic in 2009. In the past 100 years, humans have experienced three influenza pandemics: the first in 1918 (H1N1), the second in 1957 (H2N2) and the third in 1968 (H3N2) [1]. Since its emergence in the early spring of 2009, pandemic (2009) H1N1 influenza A viruses have been circulating worldwide [3]. Although most infected individuals have exhibited an uncomplicated mild respiratory disorder, the pathogenicity of this virus is clearly higher than that of seasonal influenza viruses in animal models [4–6] and humans, including those who do not have underlying illnesses [7]. The World Health Organization (WHO) [8] has reported >18,000 deaths worldwide as of 2 July 2010 and the US Centers for Disease Control and Prevention [9] estimated a 0.02% mortality rate of midlevel range depending on age. Another possible concern is a pandemic caused by highly pathogenic avian influenza (HPAI) H5N1 viruses. In 1997, human infections with HPAI H5N1 viruses were first documented in Hong Kong [10–12]. Since 2003, these viruses have spread throughout Asia, Europe and Africa with high morbidity and mortality among avian species and with occasional transmission to humans, resulting in high mortality. A total of 507 cases infected with the HPAI H5N1 virus and as many as 302 deaths were reported as of 18 October 2010 [13]. Although human-to-human transmission is rare, once the H5N1 viruses acquire this ability, a devastating pandemic might be inevitable.
Two countermeasures, vaccination and treatment with antivirals, are available to control human influenza. Although vaccination plays a crucial role in influenza prophylaxis, it is insufficient both for prophylaxis and treatment against a pandemic virus; therefore, antivirals are an important tool that might be used to mitigate influenza pandemics. Currently, two types of anti-influenza virus drugs are available: M2 ion channel blockers (adamantanes) [14] and NA inhibitors. In Figure 1, a life cycle of influenza virus and action points of the inhibitors are explained; however, in terms of adamantanes, adamantane-resistant viruses readily emerge and are already prevalent worldwide among the seasonal influenza viruses (both the H1N1 and the H3N2 subtypes) [15,16]. The adamantane drugs have not been recommended for use for the treatment or chemoprophylaxis of influenza in the US since the 2005 influenza season [15,17]. The recently emerged pandemic (2009) H1N1 virus is already adamantane-resistant [3]. Moreover, the emergence of adamantane-resistant HPAI H5N1 viruses in Vietnam, Cambodia and Thailand [18] has prompted the WHO to recommend oseltamivir for the treatment and prophylaxis of human H5N1 influenza virus infections [19]. Accordingly, many countries have stockpiled oseltamivir in anticipation of an H5N1 pandemic. The second and most recently developed class of drugs with activities against influenza A and B viruses are the NA inhibitors, which bind to the NA of newly formed virus particles and prevent their efficient release from the host cell [20]. Two NA inhibitors, zanamivir (inhaled drug, 10 mg/dose; Relenza) and oseltamivir (oral drug, 75 mg/dose; Tamiflu), are currently licensed worldwide. Both drugs require twice-daily administration for treatment. Another NA inhibitor, peramivir (single intravenous drip infusion, 300 mg/dose; Rapiacta), was licensed in 2010 in Japan. Oseltamivir is predominant and is used worldwide for the treatment of influenza, and the generation and circulation of oseltamivir-resistant seasonal influenza viruses have become major concerns [21,–25]. In particular, the worldwide prevalence of oseltamivir-resistant mutants of seasonal H1N1 virus with the H274Y substitution (histidine to tyrosine at position 274, numbering based on the N2 subtype) in NA has been reported. In addition, 95% of H1N1 isolates tested from the fourth quarter of 2008 to January 2009 [26] and almost all the H1N1 isolates of the 2008–2009 season in the US [27] were reported to be oseltamivir-resistant. Additionally, a number of oseltamivir-resistant pandemic 2009 H1N1 viruses [28] and HPAI H5N1 viruses [25] have already appeared, although their appearance is still sporadic. These epidemics of oseltamivir-resistant influenza viruses therefore necessitate the development of alternative antiviral agents.
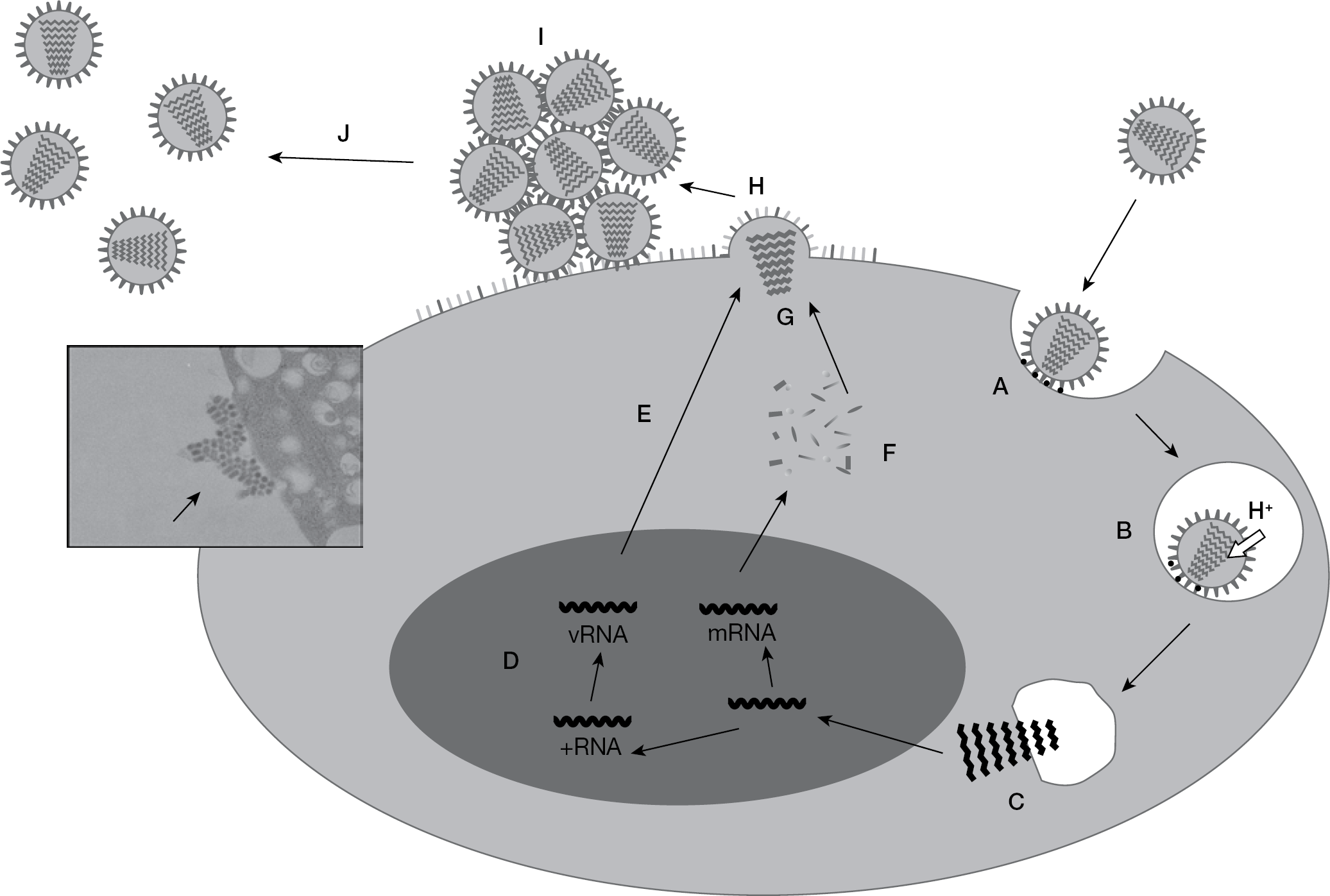
Life cycle of the influenza virus and the inhibition step of neuraminidase inhibitors
Yamashita et al. [29] and Kubo et al. [30] found a new potent NA inhibitor, laninamivir (code name R-125489), and reported that CS-8958 (laninamivir octanoate or laninamivir prodrug) acts as a long-acting NA inhibitor. The results of in vitro and in vivo experiments of laninamivir and CS-8958 [4,29–34] and the clinical studies of CS-8958 [35,36] are introduced in this review. The potential of the single administration of CS-8958 for influenza patients was confirmed by the clinical studies conducted for the 2008–2009 influenza season [36–38]. CS-8958 has been approved and is commercially available as Inavir® (Daiichi Sankyo Co., Ltd, Tokyo) in Japan.
Compound
Laninamivir (R-125489) is a potent NA inhibitor of various influenza viruses. Its chemical structure is (2R,3R,4S)-3-acetamido-2-[(1R,2R)-2,3-dihydroxy-1-methoxypropyl]-4-guanidino-3,4-dihydro-2H-pyran-6-carboxylic acid, as shown in Figure 2 [29]. It demonstrated a prolonged survival effect in a mouse infection model with influenza virus. Moreover, the esterified forms of laninamivir, with acyl chains of various lengths, significantly improved the prolonged survival effect in the same model. Among these forms, 3-(O)-octanoyl laninamivir (CS-8958; Figure 2) is the best compound in terms of its life-prolonging effect [29]. Both compounds were first discovered by Daiichi Sankyo Co., Ltd.
In vitro activities
Neuraminidase inhibitory activity
A summary of NA inhibitory activities is shown in Table 1. Laninamivir had potent inhibitory activity to NAs of various influenza viruses, such as the seasonal H1N1 [29], pandemic (2009) H1N1 [4], H3N2 [29], HPAI H5N1 viruses [33] and influenza B viruses [29]. Laninamivir showed a 50% inhibitory concentration (IC50) of 0.41 nM to pandemic (2009) H1N1, A/California/4/2009, and exhibited a median IC50 of 1.55 nM to nine pandemic (2009) H1N1 strains isolated in Japan. The values were comparable to two other NA inhibitors, zanamivir and oseltamivir carboxylate, an active metabolite of oseltamivir. Also, laninamivir inhibited NA activity of HPAI H5N1 viruses with an IC50 of 0.28–0.32 nM. This IC50 was comparable to that of zanamivir and oseltamivir carboxylate, with the exception of A/Indonesia/UT3006/05 (H5N1), which exhibited reduced susceptibility to oseltamivir carboxylate (IC50=10 nM). Laninamivir showed less potent inhibitory activities against NA of H3N2 and influenza B viruses compared with zanamivir and oseltamivir carboxylate. The IC50 of laninamivir to H3N2 was approximately 10x lower than that of oseltamivir carboxylate.
Inhibitory activities of laninamivir to neuraminidases of various influenza viruses
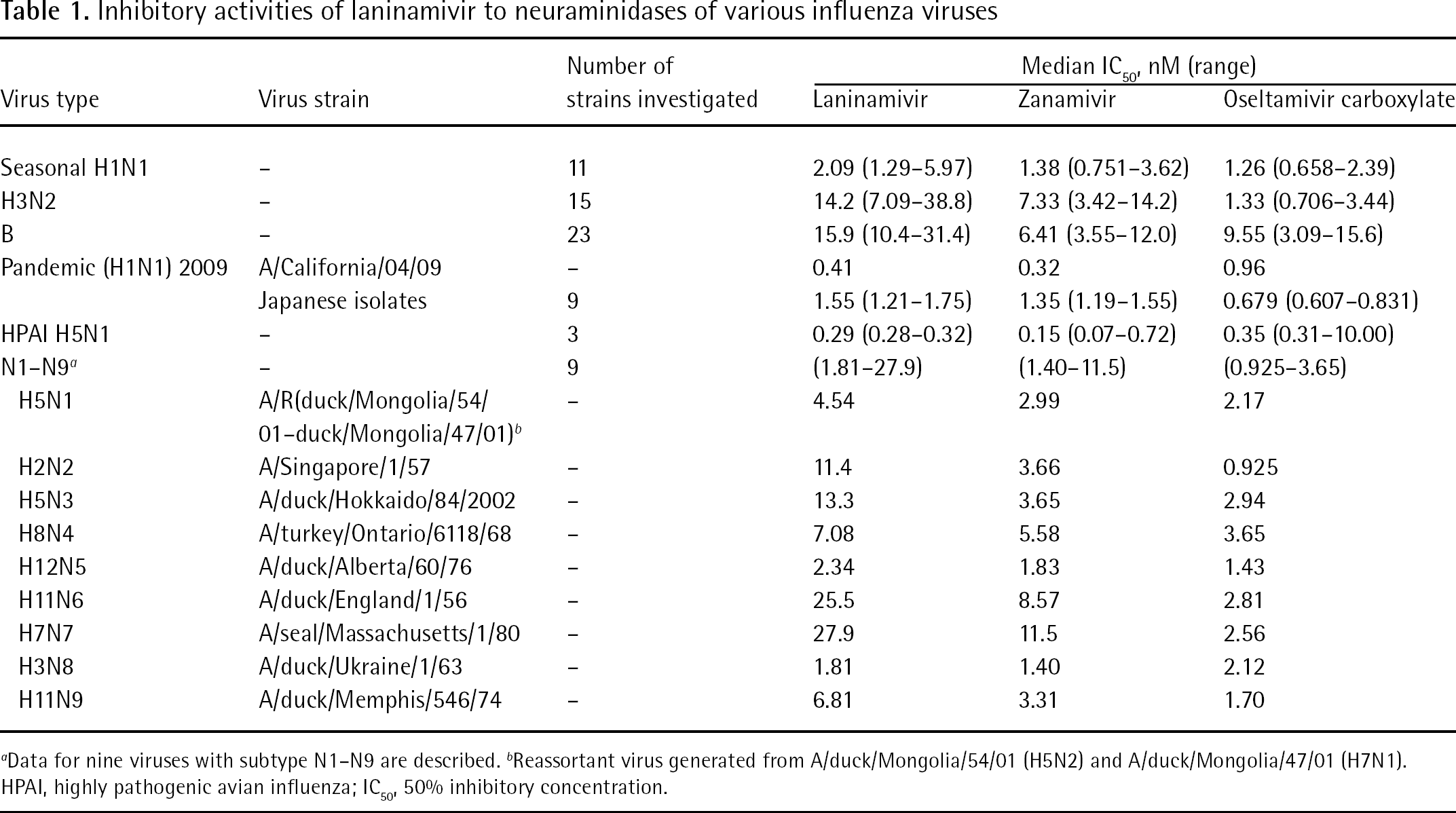
Data for nine viruses with subtype N1–N9 are described.
Reassortant virus generated from A/duck/Mongolia/54/01 (H5N2) and A/duck/Mongolia/47/01 (H7N1). HPAI, highly pathogenic avian influenza; IC50, 50% inhibitory concentration.
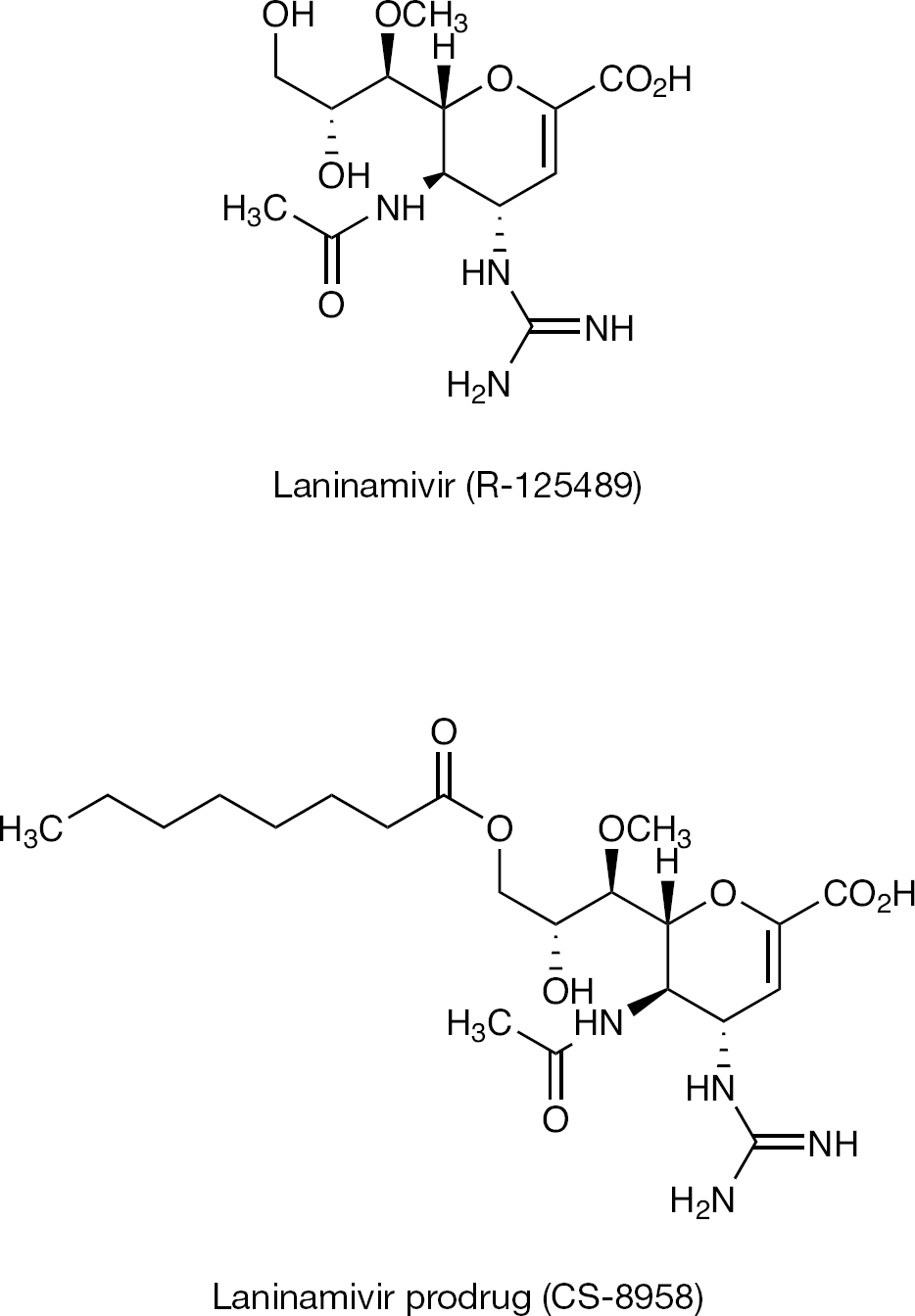
Chemical structures of laninamivir and CS-8958
To date, 16 HA subtypes (H1–H16) and 9 NA subtypes (N1–N9) of influenza A viruses have been described, but only a limited number of subtypes (H1N1, H2N2 and H3N2) have circulated in humans [1]. In the future, new viruses with other NA subtypes will possibly generate and circulate in humans. As shown in Table 1, laninamivir inhibited all NA subtypes and might be effective against emerging viruses with NA subtypes other than N1 and N2 [29]. As shown in Table 2, laninamivir inhibited NA activity of the oseltamivir-resistant viruses of seasonal H1N1, HPAI H5N1 and H3N2, which were isolated from infected patients [29].
Inhibitory activity of laninamivir, zanamivir and oseltamivir carboxylate against oseltamivir-resistant influenza viruses
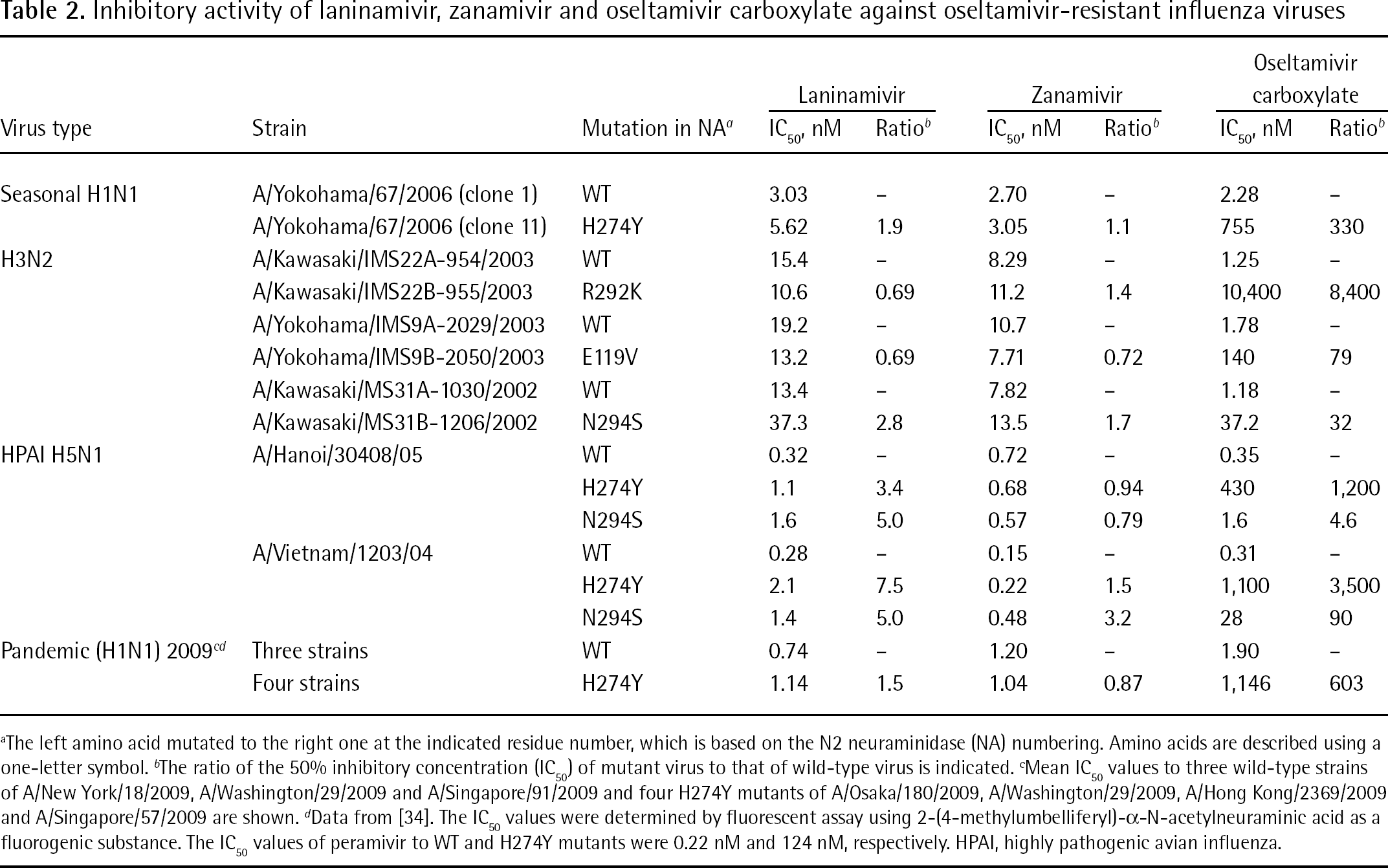
The left amino acid mutated to the right one at the indicated residue number, which is based on the N2 neuraminidase (NA) numbering. Amino acids are described using a one-letter symbol.
The ratio of the 50% inhibitory concentration (IC50) of mutant virus to that of wild-type virus is indicated.
Mean IC50 values to three wild-type strains of A/New York/18/2009, A/Washington/29/2009 and A/Singapore/91/2009 and four H274Y mutants of A/Osaka/180/2009, A/Washington/29/2009, A/Hong Kong/2369/2009 and A/Singapore/57/2009 are shown.
Data from [34]. The IC50 values were determined by fluorescent assay using 2-(4-methylumbelliferyl)-α-N-acetylneuraminic acid as a fluorogenic substance. The IC50 values of peramivir to WT and H274Y mutants were 0.22 nM and 124 nM, respectively. HPAI, highly pathogenic avian influenza.
Also, laninamivir maintained inhibitory activity to oseltamivir-resistant pandemic (2009) H1N1 virus, which possessed the H274Y substitution [34]. According to these results, it should be noted that the degrees of resistance of oseltamivir carboxylate and peramivir to H274Y mutants were comparable to each other, and were 603-fold and 564-fold, respectively (Table 2).
Virus susceptibility in cell culture
The inhibitory effect of laninamivir to viral replication in cultured cells was investigated. Also, the 50% effective concentration (EC50) determined by a plaque assay for seasonal H1N1, H3N2 and influenza B viruses, and the 90% effective concentration on the basis of the median tissue culture infective dose value of virus titre for the pandemic (2009) H1N1 virus are shown in Table 3. Laninamivir strongly inhibited the replication of these viruses. The EC50 values have a very large range. It was reported that the susceptibility of influenza viruses to NA inhibitors was variable, indicating that NA inhibitory activity is currently the only in vitro assay that is predictive of in vivo susceptibility to NA inhibitors [39]. This phenomenon could be partly explained by receptor specificity of the virus. HA is a receptor recognition protein of human influenza virus, which mainly recognizes sialic acid with an α-2–6 linkage as a receptor on the cells [40,41]. Meanwhile, the major sialic acid linkage on the Madine–Darby canine kidney (MDCK) cells, which was used for the plaque assay, has been reported to be an α-2–3 linkage [42,43]. This recognition/receptor mismatch induces a weak binding of viruses to the cells, enabling progeny viruses to be easily released from the infected cells without NA action. In this case, influenza viruses could replicate in the cells in the presence of NA inhibitors.
Inhibitory activities of laninamivir, zanamivir and oseltamivir carboxylate against replication of various influenza viruses in cell cullture
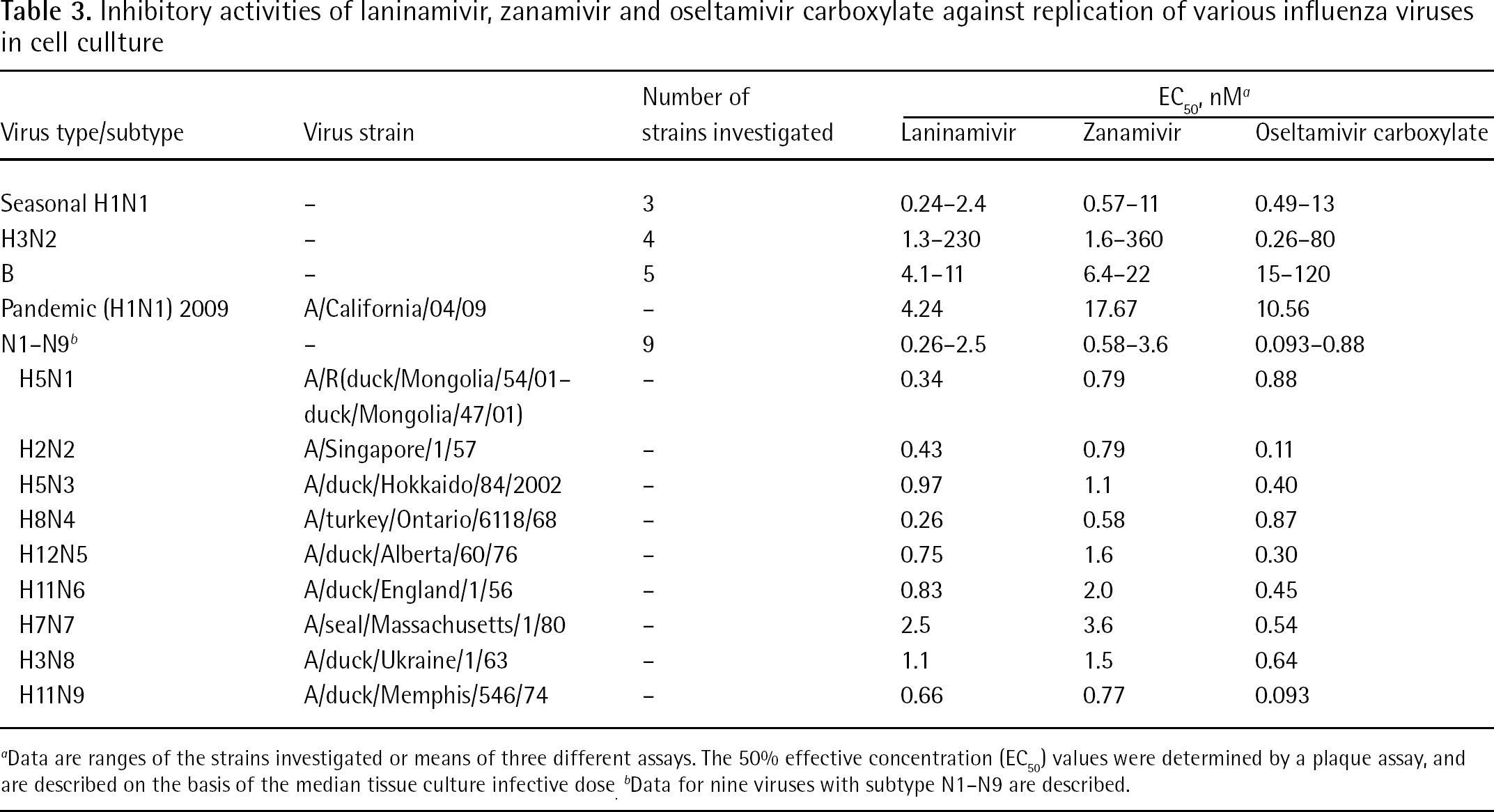
Data are ranges of the strains investigated or means of three different assays. The 50% effective concentration (EC50) values were determined by a plaque assay, and are described on the basis of the median tissue culture infective dose.
Data for nine viruses with subtype N1–N9 are described.
In vivo efficacy
Therapeutic efficacy after a single administration of CS-8958 in animal infection models
In vitro inhibitory activities of laninamivir were not largely different from those of two other NA inhibitors. However, octanoyl laninamivir, CS-8958, showed good efficacy by a single intranasal administration in an animal infection model. The licensed drugs, zanamivir (inhaled drug, 10 mg/dose) and oseltamivir (oral drug, 75 mg/dose), require a twice-daily administration for 5 days for treatment of influenza.
The virus titres in the lungs of mice infected with influenza virus A/PR/8/34 were measured after a single intranasal administration of CS-8958, a twice-daily oral administration of oseltamivir and a twice-daily intranasal administration of zanamivir. Groups treated with CS-8958 at doses of 0.027 and 0.080 mg/kg both showed statistically significant reductions in virus titres compared with the oseltamivir group at a dose of 10 mg/kg (Figure 3A). The CS-8958 group at a dose of 0.24 mg/kg (0.5 μmol/kg) showed a similar reduction in virus titres compared with the zanamivir group at a dose of 0.17 mg/kg (0.5 μmol/kg; Figure 3B). The superior efficacy of the single administration of CS-8958 was confirmed in a lethal mouse infection model [30].
The efficacy of CS-8958 was investigated in ferrets, which are susceptible to influenza virus and also develop an upper respiratory tract infection as a result of influenza virus infection. At 4 h post-infection, ferrets infected with B/Malaysia/2506/2004 received a dose of saline intranasally, a 0.24 mg/kg (0.5 μmol/kg) dose of CS-8958, a 0.17 mg/kg (0.5 μmol/kg) dose of zanamivir or 25 mg/kg of a twice-daily oral administration of oseltamivir from 4 hpi. The virus titres in the nasal washes are shown in Figure 4 [30]. The single intranasal administration of CS-8959 significantly decreased virus titres in the ferret infection model.
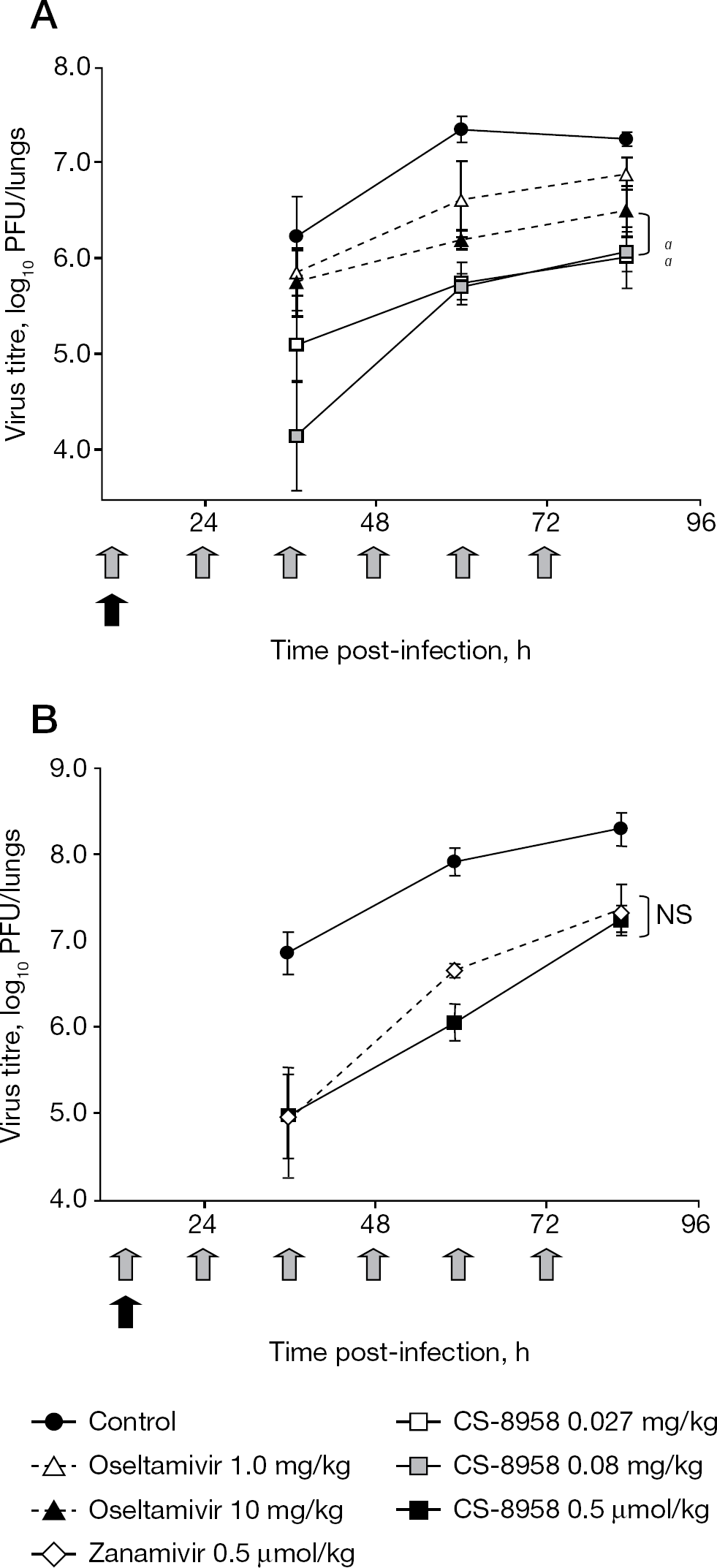
Therapeutic efficacy of a single administration of CS-8958 in the mouse infection model
Therapeutic efficacy after a single administration of CS-8958 in HPAI H5N1 virus and in the pandemic (2009) H1N1 infection mouse model
The therapeutic efficacy of CS-8958 against HPAI H5N1 influenza viruses was evaluated using a mouse lethal infection model. Mice were infected with three different strains of HPAI H5N1 viruses, and were then given a single dose of CS-8958 intranasally or were treated with oseltamivir administered orally twice-daily for 5 days. Survival of the infected mice was monitored for 21 days and survival data of mice infected with A/Vietnam/1203/04 are shown in Figure 5A. A single dose of CS-8958 was efficacious for mice infected with the HPAI H5N1 viruses [33]. This therapeutic efficacy of a single dose of CS-8958 against H5N1 viruses was also confirmed by the determination of virus titres in the lungs and brain of HPAI virus A/Hanoi/30408/05 clone 7- or A/Indonesia/UT3006/05-infected mice. CS-8958 at 0.75 or 1.5 mg/kg dramatically inhibited the replication of both viruses, particularly in the brain, compared with oseltamivir [33]. These results indicate that CS-8958 confers more potent and long-lasting protection to mice against H5N1 influenza viruses than oseltamivir.
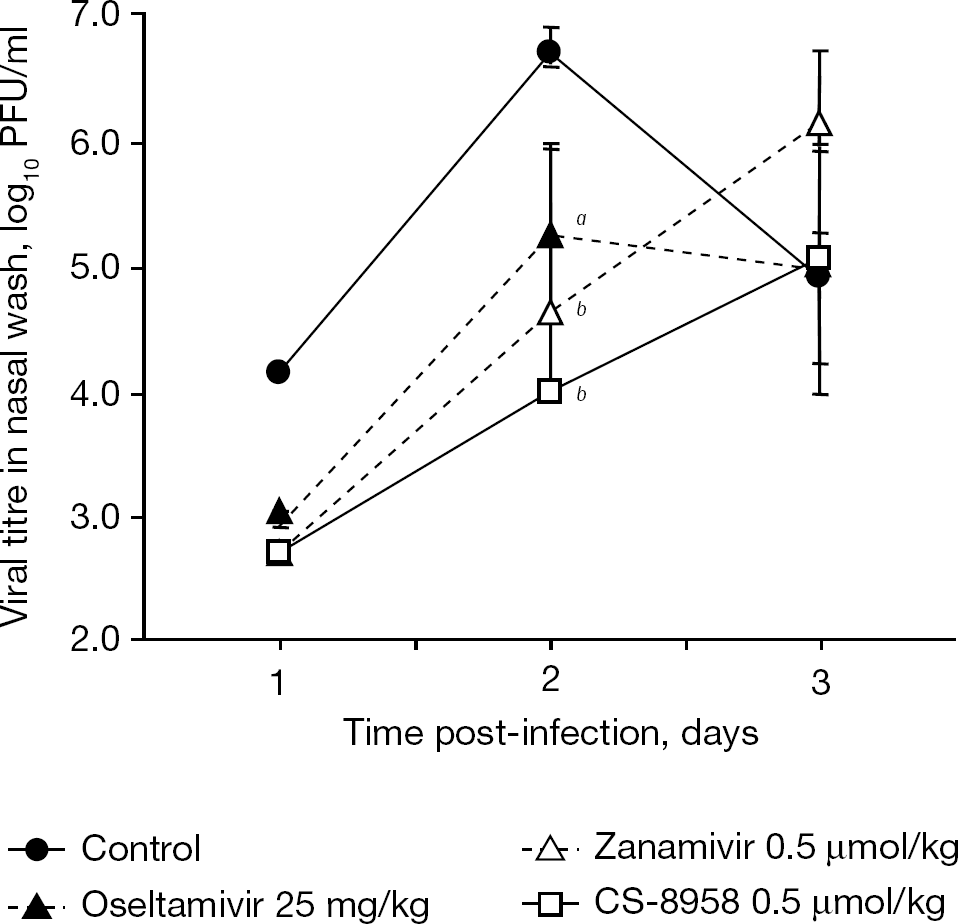
Therapeutic efficacy of a single administration of CS-8958 in the ferret infection model
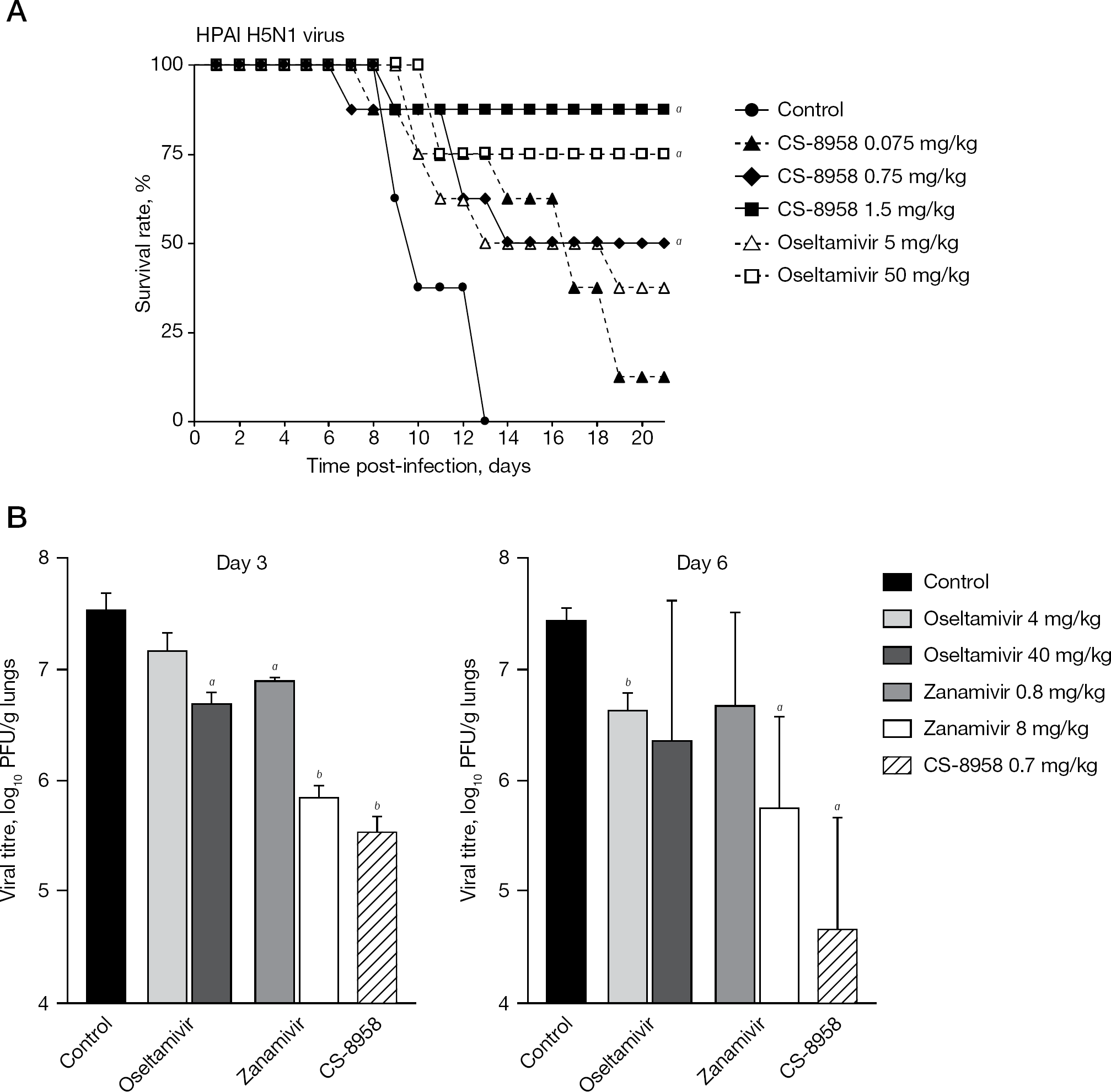
Therapeutic efficacy of CS-8958 against HPAI H5N1 virus and pandemic (2009) H1N1 virus in the mouse infection model
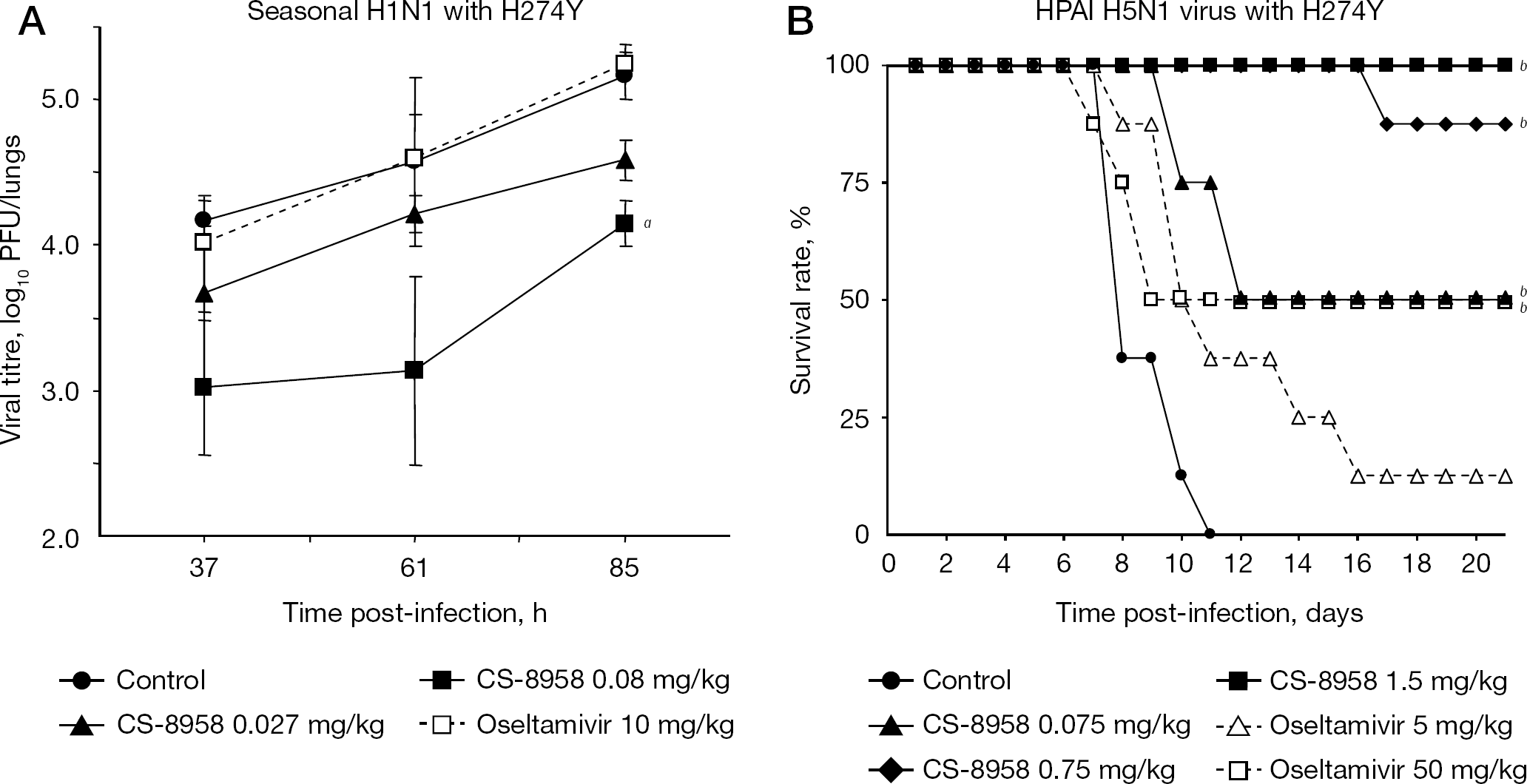
Therapeutic efficacy of CS-8958 against oseltamivir-resistant viruses with the H274Y substitution in the mouse infection model
Next, efficacy against pandemic (2009) H1N1 A/California/04/09 virus was investigated in the mouse infection model. Mice infected with the virus were given a single dose of CS-8958 intranasally, oseltamivir was orally administered twice-daily for 5 days or zanamivir was administered intranasally once-daily for 5 days. The virus titres in the mice lungs were measured on days 3 and 6 after infection (Figure 5B) [4]. Again, a single administration of CS-8958 showed significant efficacy and stronger decrease in viral titres compared with a repeated administration of oseltamivir or zanamivir [4].
Therapeutic efficacy after a single administration of CS-8958 in oseltamivir-resistant H1N1 and HPAI H5N1 virus infection mouse model
The efficacy after a single dose of CS-8958 was confirmed in a mouse model infected with the oseltamivir-resistant seasonal H1N1 virus monitored by virus titres in lungs (Figure 6A) [30] and with the HPAI H5N1 virus monitored by the survival rate (Figure 6B) [33]. Both viruses have the H274Y substitution. In the seasonal A/Yokohama/67/2006 (H1N1) H274Y infection model, statistically significant virus titre reduction was observed in a single dose of CS-8958 at 0.08 mg/kg compared with a control. No reduction in virus titres was observed in a twice-daily administration of oseltamivir at 10 mg/kg. In the HPAI A/Vietnam/1203/04 (H5N1) H274Y virus infection model, only 12.5% and 50% of mice survived at a dose of 5 and 50 mg/kg of oseltamivir (twice daily for 5 days), respectively. By contrast, one-half of the mice survived with a single dose of 0.075 mg/kg of CS-8958. It should be noted that for one-half of the mice to survive, the dose of CS-8958 was comparable to a presumed clinical dose of CS-8958, whereas as much as 50 mg/kg (twice daily for 5 days) of oseltamivir (clinical dose, 75 mg/dose) was required. Thus, CS-8958 also showed good efficacy to oseltamivir-resistant virus.
Prophylactic efficacy after a single administration of CS-8958 in animal models
Once-daily administration of zanamivir (inhaled drug, 10 mg/dose) and of oseltamivir (oral drug, 75 mg/dose) have been licensed as prophylactic drugs. This suggests that the half life (t1/2) of each drug in target tissue is not long; therefore, they require once-daily administration. By comparison, the t1/2 of CS-8958 after intranasal administration to mice is quite long. Prophylactic efficacy of a single dose of CS-8958 was investigated in a mouse infection model. A single administration of CS-8958 prior to infection with A/PR/8/34 showed a life-prolonging effect in the mouse infection model. One-half of the mice administered CS-8958 at 0.17 mg/kg 7 days before infection survived. By contrast, only one-quarter of the mice treated with oseltamivir at as much as 110 mg/kg 1 day before infection survived. The dose of oseltamivir was much higher than the clinical setting (Figure 7) [30], probably because of a short t1/2 of 3 h and time of maximum concentration of 0.5 h of oseltamivir carboxylate in mouse plasma after an intravenous administration of oseltamivir carboxylate and oral administration of oseltamivir, respectively [44]. Prophylactic efficacies against H5N1 virus after the single intranasal administration of CS-8958 were also confirmed in both the lethal mice infection model and in the measurement of virus titre reduction in mice lungs and brain [33].
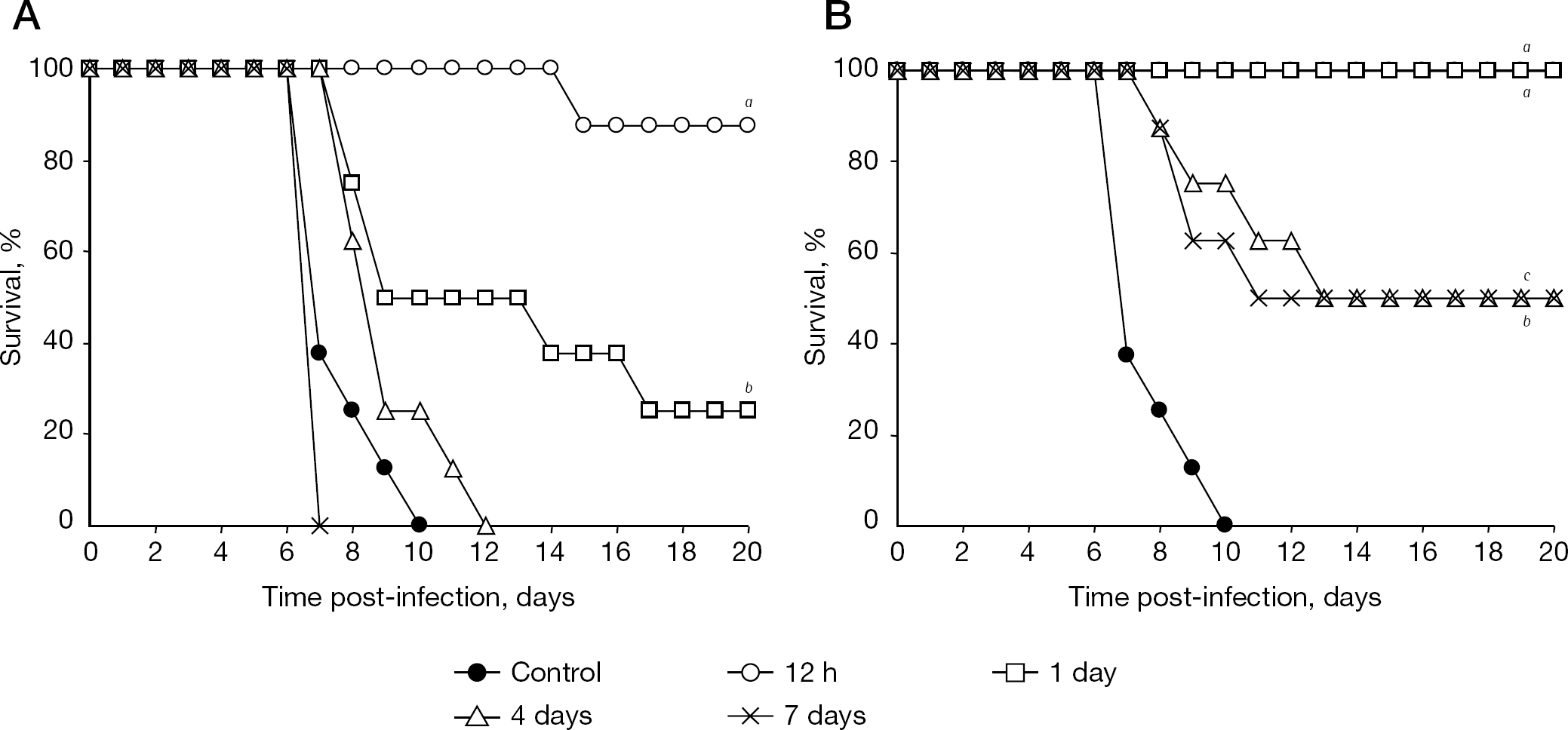
Prophylactic efficacy of CS-8958 in the mouse infection model
Two unique characteristics of CS-8958 and laninamivir as a long-acting drug
The therapeutic and prophylactic efficacies achieved after the single intranasal administration of CS-8958 could be a result of the unique characteristics of the compounds described below.
Long-lasting drug in respiratory organs of animals
The lung concentration–time profiles of CS-8958 and laninamivir after a single intranasal administration of CS-8958 at a dose of 0.5 μmol/kg (0.236 mg/kg) to mice are shown in Figure 8 [31]. After administration, the lung CS-8958 concentrations decreased with a t1/2 of 0.833 h and, at 12 h post-dose or later, the CS-8958 concentrations were below the lower limit of quantification. By contrast, the lung laninamivir concentrations increased soon after administration, and, subsequently, laninamivir retained a t1/2 of as long as 41.4 h. Even at 120 h post-dose, laninamivir remained in lungs at a concentration of 0.915 nmol/g [31], which is considerably higher than the IC50 of laninamivir against the NA activities of various influenza viruses (Table 1) [31]. In addition, similar long retention of laninamivir was observed in the target tissues (trachea and lung) after intratracheal administration of CS-8958 to rats, and approximately 20% of the dose (0.2 mg/kg) was retained in the lungs at 24 h [32]. These results demonstrated that CS-8958 administered intranasally or intratracheally to animals was converted/hydrolysed to laninamivir and retained as the hydrolysed form in the target tissues for a long time. The long-lasting characteristics of laninamivir in the target tissues for influenza virus infection result in superior efficacy by a single dose of CS-8958, whereas repeated doses of other NA inhibitors are required for efficacy.
Binding stability of laninamivir to viruses
Bantia et al. [45] demonstrated that peramivir, a selective NA inhibitor [46,47], binds to NA, with the N9 subtype more tightly than oseltamivir carboxylate or zanamivir. This characteristic is thought to be one of the reasons why peramivir is efficacious by a single intravenous injection. To assess the binding stability of laninamivir to various virus NAs, similar experiments with whole influenza virions were performed, and the results are shown in Figure 9 [33]. In these experiments, virions were treated with excess NA inhibitors, then unbound forms of NA inhibitor were removed and the NA reaction was chased over time after adding the NA substrate. Considerable differences in the dissociation rates of the NA inhibitors among the test virus strains were observed. NA activity of A/Panama/2007/99 (H3N2) was severely inhibited by all test NA inhibitors for at least 5 h. NA activity of B/Mie/1/93 treated with laninamivir was appreciably inhibited by approximately 50% at 6 h, whereas no inhibitory effect was observed at this time point for the other three NA inhibitors. NA activity of A/New Caledonia/20/99 (H1N1) treated with laninamivir and peramivir was suppressed to approximately 30% at 6 h, whereas the NA activity treated with zanamivir and oseltamivir carboxylate recovered to 80–90% at 6 h. These results suggest that laninamivir bound to viral NA more stably than any other NA inhibitors tested. This stable binding of laninamivir might be another reason for the long-acting characteristics of CS-8958.
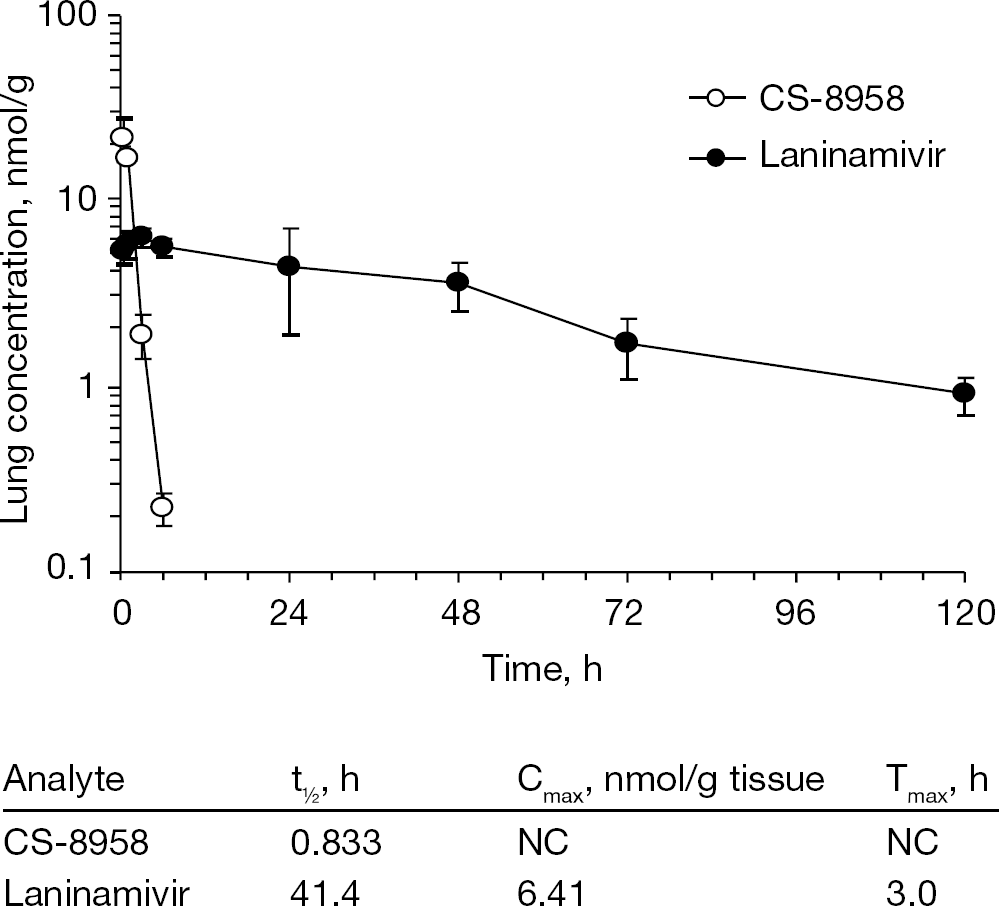
Pharmacokinetics in the mouse lungs after intranasal administration of CS-8958
Clinical studies
Phase I studies
The next important point is to confirm whether the long-lasting characteristic of laninamivir after a single intranasal administration of CS-8958 observed in animals is applicable to humans or not. Healthy male volunteers participated in double-blinded randomized placebo-controlled trials and received a single dose of 5, 10, 20, 40, 80 or 120 mg CS-8958 or a twice-daily dose of 20 or 40 mg CS-8958 for 3 days. The clinical and laboratory parameters and plasma and urinary concentrations of CS-8958 and laninamivir for the 144 h post-dosing were measured. CS-8958 disappeared from plasma with a t1/2 of approximately 2 h. By contrast, laninamivir was slowly eliminated from the body, lasting for up to 144 h after administration with a t1/2 of approximately 3 days. There were no adverse events related to the test drug in all groups, and it was indicated that when CS-8958 was inhaled by healthy volunteers, it was well-tolerated [35].
Although the detection of the active metabolite in plasma for an extended period might be consistent with a long half-life in the target tissues, such as the trachea and lung, it was not possible to demonstrate this directly through sampling of the target tissue because of the limitations of the Phase I study; however, these data suggested that the long-lasting concentration of laninamivir after a single intranasal administration of CS-8958 in animals might also be applicable to humans who inhaled CS-8958.
Clinical studies in influenza patients
Phase II/III studies in paediatric patients were conducted as a double-blinded randomized controlled trial to compare CS-8958 with oseltamivir in the 2008–2009 influenza season in Japan. During this season, almost all H1N1 viruses were oseltamivir-resistant viruses with the H274Y substitution. Eligible patients were children, 9 years of age and under, who had febrile influenza symptoms of no more than 36 h duration. Patients were randomized to one of three treatment groups: a CS-8958 40 mg group, a CS-8958 20 mg group or an oseltamivir group (n=61–62 patients per group). CS-8958 was administered as a single inhalation. Oseltamivir 2 mg/kg was orally administered twice daily for 5 days, which is the regular regimen for paediatric use. CS-8958 markedly reduced the median time to alleviation of influenza illness in comparison with oseltamivir in patients infected with oseltamivir-resistant influenza A (H1N1); the difference was −60.9 h in the 40 mg group and −66.2 h in the 20 mg group (Figure 10) [36]. By contrast, there were no significant differences in the time to alleviation of illness between the CS-8958 groups and the oseltamivir group against influenza A (H3N2) or B infection. CS-8958 was well tolerated [36]. CS-8958 was an effective and well-tolerated treatment for children with oseltamivir-resistant influenza A (H1N1) virus infection. A single inhalation of CS-8958 during the course of illness might be a beneficial regimen in terms of therapeutic compliance.
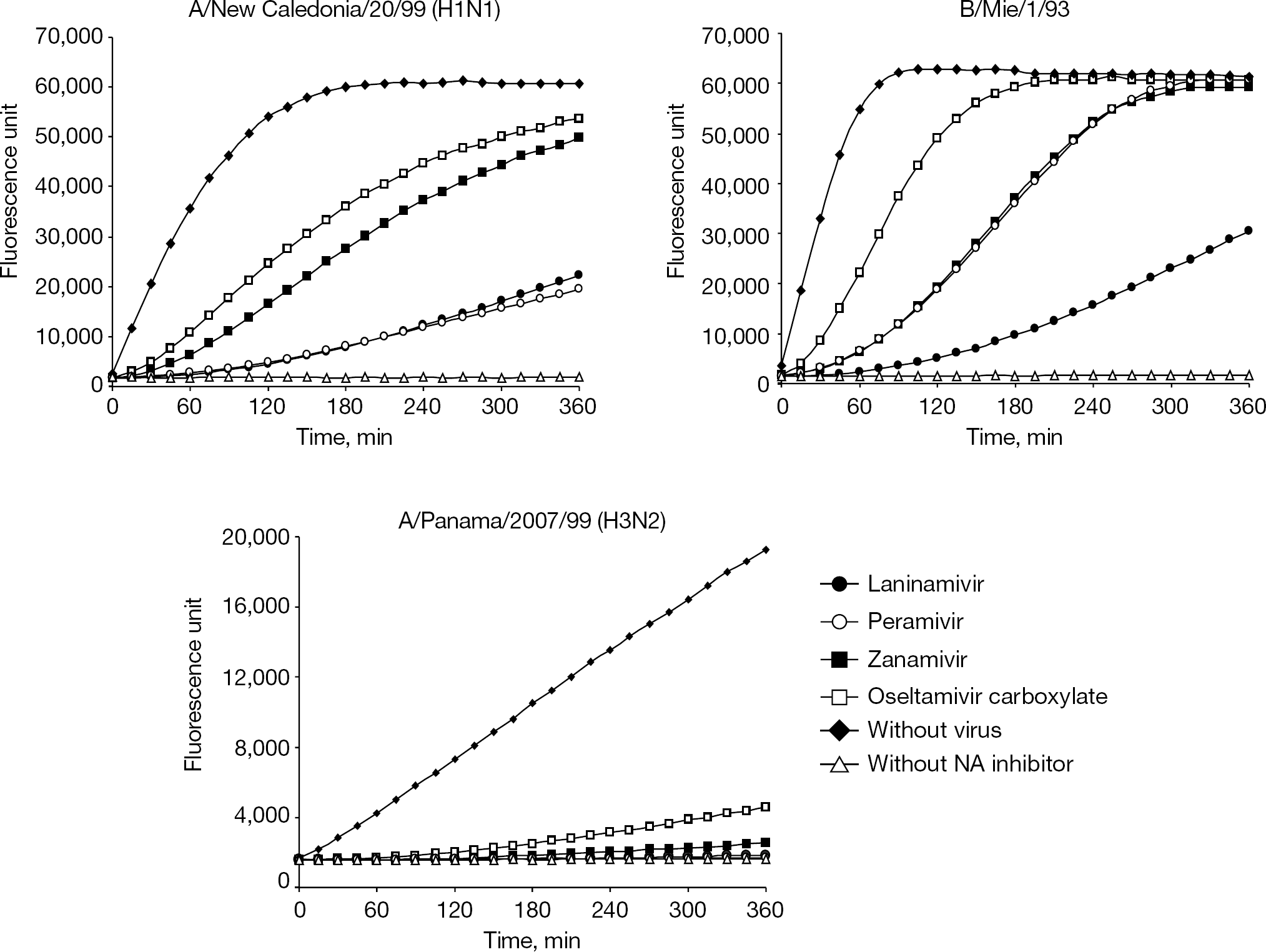
Binding stability of NA inhibitors to viruses
A Phase III study of adults, the Multinational Asian Clinical Research for Influenza Virus Extermination on Long-Acting Neuraminidase-Inhibitor Study (MARVEL), was performed as a randomized double-blinded active-controlled study in order to confirm the efficacy and safety of CS-8958 administered as a single inhaled dose of 20 or 40 mg compared with oseltamivir 75 mg orally administered twice daily for 5 days (a total of 10 times) in adult patients with influenza A or B virus infection [38]. According to the results, non-inferiority to oseltamivir was confirmed in both the 20 mg and 40 mg group of CS-8958 in terms of the primary end point, which was the time to alleviation of influenza illness. Both CS-8958 doses were well-tolerated [37,38].
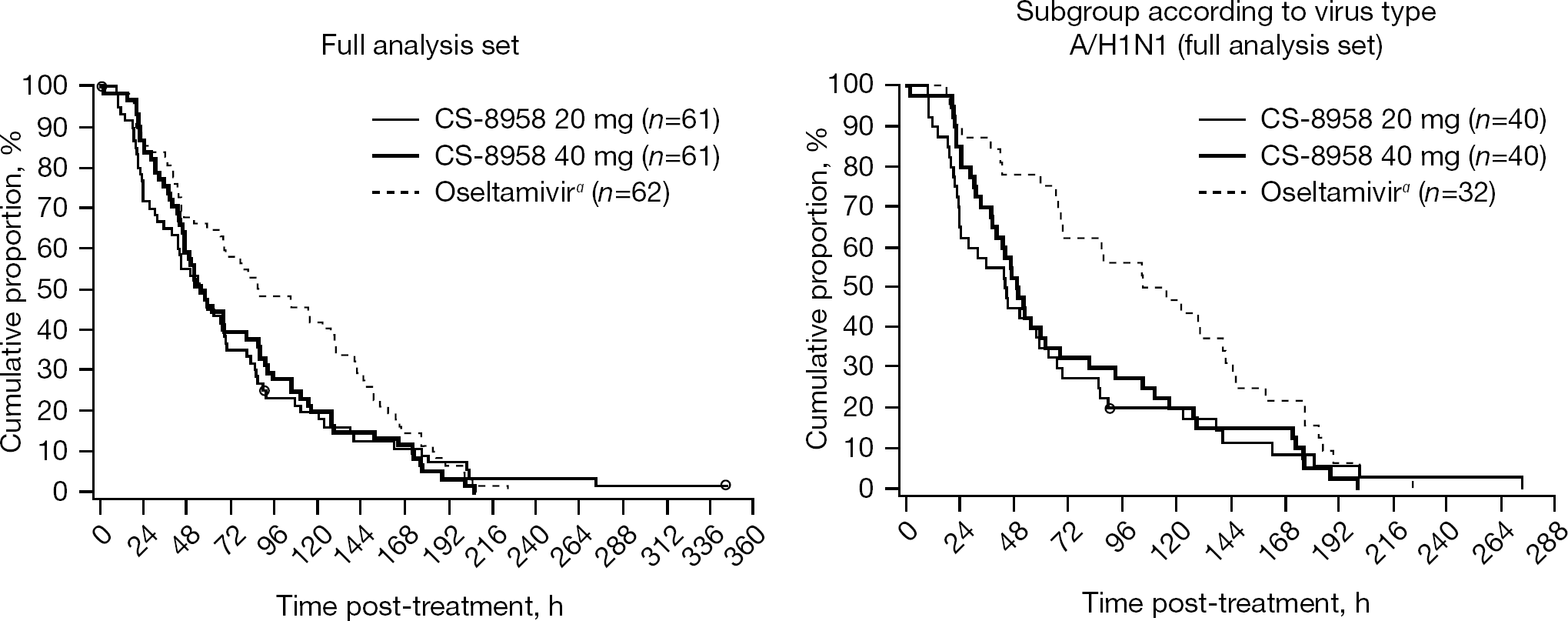
Therapeutic efficacy of CS-8958 in paediatric influenza patients
Conclusions
In contrast to the currently available drugs, which require multiple dosing, for example, twice daily for 5 days, it was confirmed that the single inhalation of CS-8958 is sufficient to treat influenza in humans. This CS-8958 dosing regimen will allow better compliance for patients with influenza. Furthermore, individuals with influenza might spread influenza virus from up to 2 days before to approximately 5 days after the onset of symptoms, and children can spread the virus for 10 days or longer. Therefore, continuous suppression of influenza virus replication in influenza patients is important. The dosing regimen of CS-8958, which is a single administration, has potential benefit compared with that of currently available drugs, which require twice-daily administration for 5 days, particularly because interruption of administration of current drugs after relief or forgetfulness might increase the risk of transmission to others.
We learned from the pandemic in 2009 that anti-influenza drugs are important for treatment and prophylaxis of pandemic influenza. Also, in case of epidemic of influenza, a line-up of drugs with different characteristics will be beneficial for both patients and clinicians. The ease of administration of CS-8958 is especially noteworthy because only a single inhalation is required during the course of illness. In addition to currently available drugs, including zanamivir and oseltamivir, it is expected that CS-8958 will be another option for treatment of influenza.
Footnotes
The author declares no competing interests.
